Abstract
Background:
Prompt identification of actionable genomic alterations (AGAs) is essential in guiding treatment decisions in newly diagnosed advanced non-small-cell lung cancer (NSCLC).
Objectives:
We aimed to determine the optimal testing strategy in terms of proportion of AGAs detected, time-to-treatment decision (TTTD) and average costs/patient in Singapore, a high-frequency AGA population.
Design:
A model was constructed to evaluate AGA testing strategies, comparing the cost-effectiveness and budget impact of each strategy.
Methods:
The 24 strategies studies included (1) upfront tissue/plasma next-generation sequencing (NGS) alone (small/medium/large panel), (2) exclusionary tissue single-gene testing (EGFR, ALK and ROS1) followed by tissue/plasma NGS, (3) exclusionary plasma EGFR followed by tissue or plasma NGS, (4) sequential single-gene testing followed by tissue/plasma NGS, (5) exclusionary or sequential testing without NGS and (6) small panel tissue NGS with fluorescence in situ hybridization (FISH) for ALK, ROS1, RET and MET. Literature review was performed to determine the incidence of NSCLC and the prevalence of each AGA in the population; costs, gene coverage and turnaround times were sourced through market research.
Results:
AGAs and EGFR mutations occurred in 76.7% and 56.0% of patients, respectively. Upfront medium panel tissue NGS detected all AGAs at USD1813/patient, with a TTTD of 7 days—no benefit was seen from using large panel tissue NGS at a higher cost. Exclusionary tissue testing followed by medium panel tissue NGS detected 98.9% of AGAs at USD1506/patient, with a TTTD of 14.9 days. Exclusionary plasma EGFR testing followed by medium panel tissue NGS detected 98.3% of AGAs at USD1047/patient, with a TTTD of 8.4 days. Upfront small panel tissue NGS with FISH also detected 98.3% of AGAs at a cost of USD798/patient, with a TTTD of 14 days.
Conclusion:
In our high EGFR-mutation prevalence population, exclusionary plasma EGFR followed by medium panel tissue NGS was cost-effective. Our analysis provides insight on NGS testing strategies of different gene panel sizes and sample types.
Plain language summary
Lung cancers often carry gene changes that can be targeted using targeted therapy; it is important to identify these gene changes promptly. This study aims to evaluate the cost-effectiveness of the many different panels and strategies available for gene testing in Singapore, in terms of the proportion of gene changes detected and the time to obtain a result for each strategy. Targetable gene changes are present in 76.7% of the population. The strategies of 1) a blood test for the commonly-mutated EGFR gene, and if negative, following with a medium-sized gene panel, or of 2) an upfront small-sized gene panel with additional analysis for fusion genes were found to be cost-effective and detected high proportions of gene changes. These findings will allow healthcare professionals to select optimal testing strategies depending on the needs of each patient, and to study the impact of varying strategies on the national testing expenditure.
Keywords
Introduction
Lung cancer is the leading cause of cancer-related deaths worldwide, of which the majority is comprised of non-small-cell lung cancer (NSCLC). 1 Common sensitizing epidermal growth factor receptor (EGFR) mutations were the first actionable genomic alteration (AGA) identified in NSCLC, with EGFR tyrosine kinase inhibitors established as an effective treatment superior to chemotherapy in these patients.2,3 Since then, the list of AGAs in genes such as anaplastic lymphoma kinase (ALK), C-ros oncogene 1 (ROS1), rearranged during transfection (RET), mesenchymal epithelial transition (MET), V-Raf murine sarcoma viral oncogene homolog B (BRAF), human epidermal growth factor receptor 2 (HER2), Kirsten rat sarcoma viral oncogene homolog (KRAS) and neurotrophic tyrosine receptor kinase (NTRK), for which targeted treatment options are available, has continued to grow. 3 There are now Food and Drug Administration (FDA)-approved treatments available in the first line for alterations in EGFR (gefitinib, erlotinib, afatinib, dacomitinib, osimertinib, amivantamab, lazertinib), ALK (crizotinib, alectinib, brigatinib, ceritinib, lorlatinib), ROS1 (entrectinib, repotrectinib, crizotinib, ceritinib), BRAF (dabrafenib and trametinib, encorafenib and binimetinib), NTRK (larotrectinib, entrectinib, repotrectinib), MET exon 14 skipping (tepotinib, capmatinib, crizotinib) and RET (selpercatinib, pralsetinib). 4
In this era of molecular targeted therapy, it is imperative to identify AGAs promptly, and multiple testing methods have been developed to guide the choice of treatment. 5 Single-gene tumour tissue testing by polymerase chain reaction, fluorescence in situ hybridization (FISH) or immunohistochemistry has been used but is associated with increased time, costs and tissue consumption. Next-generation sequencing (NGS) on tissue allows more comprehensive readouts of molecular alterations.6,7 More recently, performing single-gene testing or NGS on circulating tumour deoxyribonucleic acid (ctDNA) has also been found to be a feasible way of detecting AGAs in a less invasive manner.8–10
The most cost-effective and comprehensive testing strategy for the detection of AGAs has been reported with conflicting results. Previous cost-effectiveness analyses performed in a Western population found that upfront tissue NGS testing was associated with cost savings and shorter time-to-test results, compared to single-gene sequential, exclusionary, or panel testing.11,12 However, in an East Asian NSCLC population with a high frequency of EGFR mutations, exclusionary testing was reported to be more cost-effective. 13 However, it is unknown whether testing strategies using plasma NGS and different-sized NGS gene panels would impact cost-effectiveness.
In this analysis, we aimed to determine the optimal testing strategy in terms of proportion of AGAs detected, time-to-treatment decision (TTTD), as well as annual and average costs per patient in Singapore, a country with patients with a high frequency of AGA NSCLC.14,15
Methods
Model structure
A decision tree was constructed in Microsoft Excel® to capture average costs per patient, TTTD and probability of detecting an AGA from utilizing multiple AGA testing strategies from a Singapore healthcare system perspective. Newly diagnosed advanced NSCLC patients entered the model at the time of diagnosis after completing an initial biopsy and proceeded with AGA testing, as shown in Figure 1. Previous cost-effectiveness studies have used a similar model structure. The reporting of this study conforms to the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) 2022 statement. 16

Decision tree model used to determine costs, proportion of mutations detected and time-to-treatment detected associated with each actionable gene alteration testing strategy.
Three different testing approaches are explored in this model: (1) sequential testing, (2) exclusionary testing and (3) upfront NGS testing. In the sequential testing approach, initial testing of patients employed a pre-specified sequence of individual single-gene tests. Following each test result, patients with a detected AGA proceed with treatment and exit the model. Remaining patients with no detected AGA proceed with the next single gene test in the sequence. Testing is repeated until either an AGA is detected or all tests are completed. Initial testing for the exclusionary testing approach instead employed all pre-specified single-gene tests simultaneously as a panel. Following initial testing, all patients with no AGA detected in both sequential and exclusionary testing approaches would proceed to receive NGS testing as specified in Figure 1 for further detection of AGAs. The upfront NGS testing approach tested all patients with NGS for a panel of pre-specified AGAs. If no AGAs were detected in the initial testing, patients exited the model with no subsequent testing.
To account for the variety of NGS tests available, different NGS panel sizes and sources (plasma or tissue) within each testing approach were explored to investigate the different combinations of initial and subsequent testing approaches. Small panel NGS was defined as one that tested </=20 genes, medium panel NGS tested 21–50 genes and large panel NGS tested >50 genes. Choices of AGA tests included in these initial or subsequent testing phases were decided based on consultation with local clinicians to identify clinically feasible and practiced testing strategies. All AGA tests are available to all patients in Singapore. A total of 24 strategies were explored, as elaborated in Table 1.
Actionable gene alteration tests are used in each testing strategy.
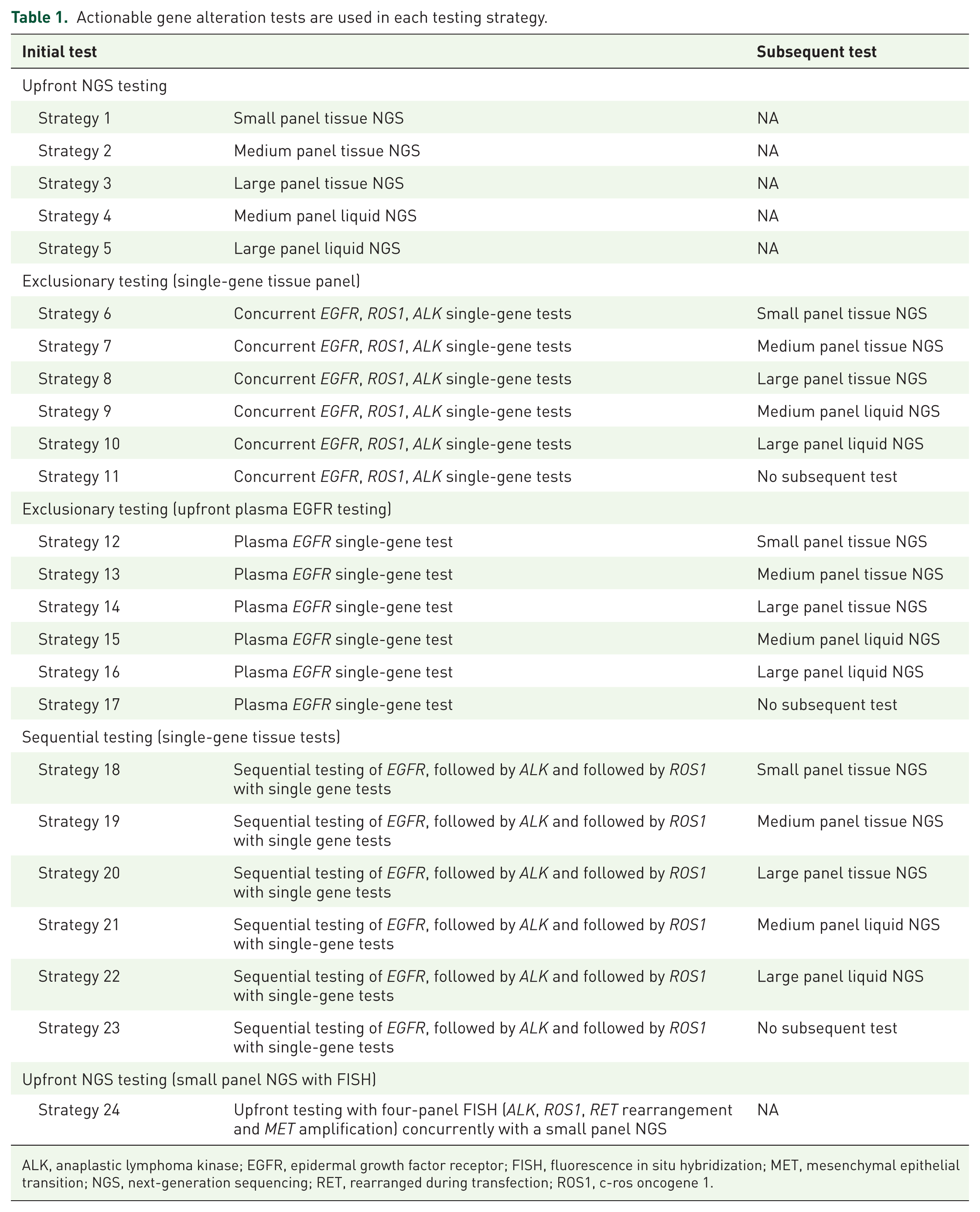
ALK, anaplastic lymphoma kinase; EGFR, epidermal growth factor receptor; FISH, fluorescence in situ hybridization; MET, mesenchymal epithelial transition; NGS, next-generation sequencing; RET, rearranged during transfection; ROS1, c-ros oncogene 1.
Model outcomes
The model sought to identify the average cost per patient, TTTD and the probability of detecting an AGA. The probability of detecting an AGA was defined as the percentage of all newly diagnosed NSCLC patients who had a molecular alteration for which there is an available FDA-approved targeted therapy. TTTD was defined as the time taken for patients to exit the model due to AGA detection, re-biopsy failure or completion of all planned tests with or without a detected AGA. For the exclusionary testing approach, we assumed that single-gene tests used in the initial panel would return concurrently based on the longest test turnover time. As testing is assumed to take place over a short time horizon between diagnosis and TTTD, no discounting or half-cycle correction was applied to costs and outcomes in this model.
Model inputs
A literature review was first conducted to identify appropriate inputs for the model, which were subsequently validated by expert clinical opinion. All relevant costs were provided from a hospital database (in year 2022) in Singapore Dollars (SGD) and reported in US Dollars (USD), utilizing the 2022 average conversion rate from SGD to USD of SGD 1.379 = USD 1. Test turnover time and re-biopsy time were taken from local hospital databases.
AGA prevalence
Prevalence of AGAs among the advanced NSCLC population used in the model was first identified from published literature and validated by practicing clinicians (Supplemental Table 1). In our base case, 76.7% of newly diagnosed advanced NSCLC patients were assumed to have AGAs. Programmed death ligand 1 (PDL1) expression is commonly tested in all patients with advanced NSCLC; cost and outcomes of PDL1 testing were excluded from the analysis, as PDL1 does not directly guide the choice to use a targeted therapy agent.
AGA test inputs
The selection of genes included in each test or test panel was informed by product brochures and consultation with local clinicians (Supplemental Table 2). 15 As the number of NGS tests available in the market falls into a wide range, NGS tests were selected based on the most commonly used NGS tests. This model does not account for the sensitivity or specificity of each test and assumes that each test can perfectly discriminate patients with the specific AGA from patients without. Costs of each test were obtained from market research and inputs from local clinicians (Supplemental Table 2). Turnover times were sourced from product brochures and reflect the time from when a tissue or blood sample is sent for testing. If a panel of tests were employed concurrently, results were assumed to have returned all at once, and turnover time was based on the longest turnover time of any test in each test panel.
Re-biopsy inputs
The model additionally accounted for the need for re-biopsy following a negative test prior to subsequent testing. Re-biopsy rate, re-biopsy costs, and re-biopsy failure rates were sourced from literature and validated against clinician insights (Supplemental Table 3). Upon re-biopsy failure, patients were assumed to exit the model rather than proceed with a second re-biopsy.
Scenario analysis
Scenario analyses were conducted to investigate whether the impact on cost-effectiveness and budget impact would vary under different assumptions. Scenarios tested included differing prevalence of AGAs in the population to match a low EGFR-mutant population. 11 The impact of the uptake of a recently introduced medium panel NGS test (APEX50) was explored as an additional scenario. The impact of varying the price of subsequent NGS tests (75%, 50% and 25% of current costs) was also explored.
Selection of best-performing testing strategies for budget impact analysis
Budget impact analysis (BIA) was conducted on strategies that either lay on the cost-effectiveness frontier or were deemed to be clinically relevant. All strategies were sorted based on cost and proportions of mutations detected. Dominated strategies (strategies that were simultaneously more costly but less effective than any other strategy) were first removed. Incremental cost-effectiveness ratios (ICERs) were determined by dividing the incremental costs associated with each strategy by the incremental proportion of mutations detected. Strategies with higher ICERs than more expensive strategies (extended dominated) were iteratively removed, with ICERs recalculated between each iteration. Once only strategies that were neither dominated nor extended dominated were left, they were plotted into a cost-effectiveness frontier. Additional strategies that did not lie on the cost-effectiveness frontier but were deemed clinically representative of local practice were also included in BIA.
BIA was conducted to estimate the change in total healthcare budget from the national adoption of selected AGA testing strategies. Market share used for the BIA was based on current local practice. The split of testing approaches was based on the distribution of AGA tests used locally, as informed by expert clinical opinion. Market share split of NGS tests was informed by a previously conducted market research (Supplemental Table 4).
Results
Analysis of cost against the percentage of AGAs detected and TTTD
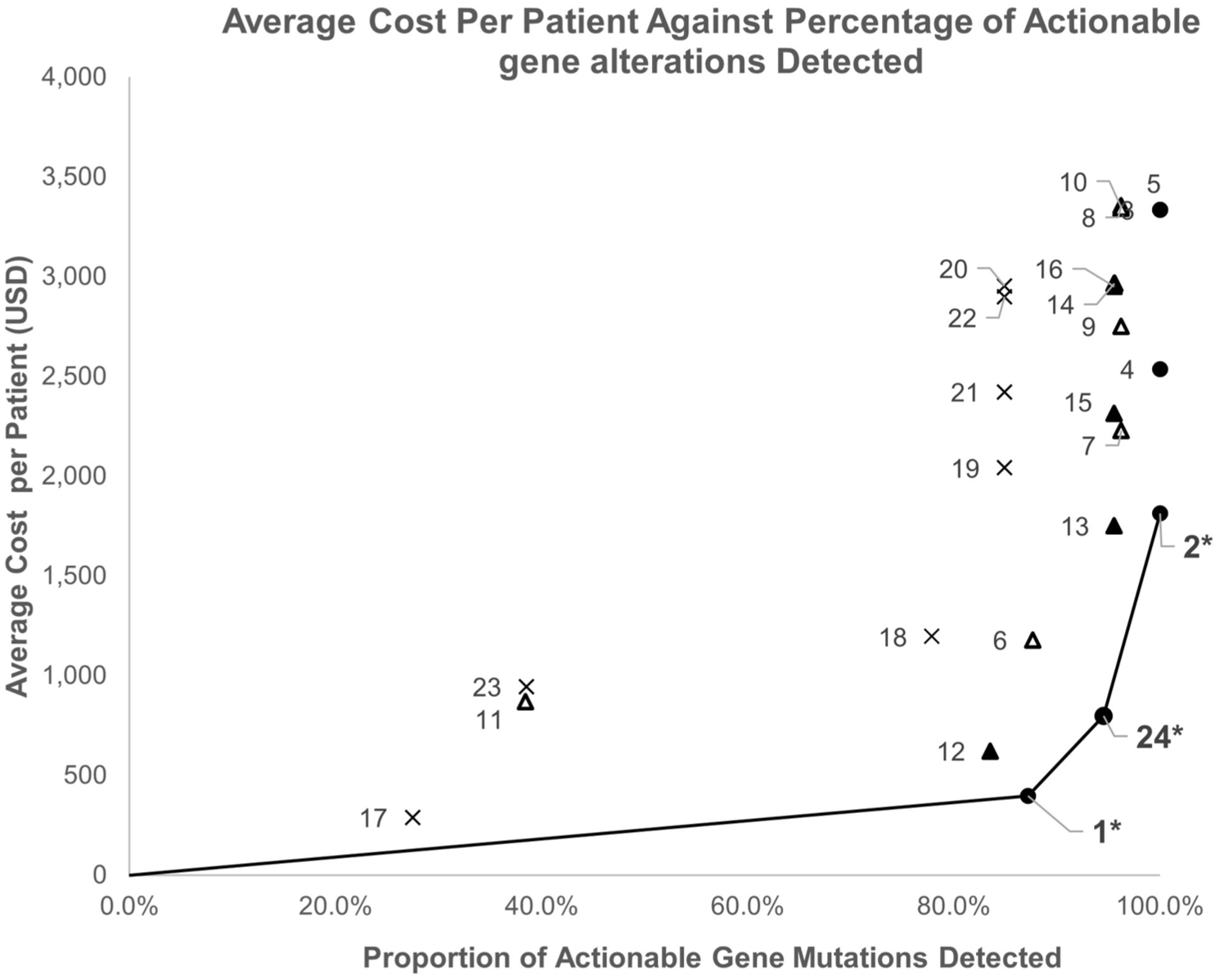
AGAs and EGFR mutations occurred in 76.7% and 56.0% of patients in our population, respectively. The average cost per patient and percentage of AGAs detected using each of the 24 testing strategies are shown in Figure 2. There was no benefit in AGA detection in using large panel NGS over medium panel NGS, at a higher cost. The strategies on the cost-effectiveness frontier and selected clinically relevant strategies lying close to the cost-effectiveness frontier are summarized in Table 2, with the results for all the strategies summarized in Supplemental Table 5. Of these, upfront medium panel tissue NGS (Strategy 2) detected 100% of AGAs at the highest cost of USD 1813. Exclusionary plasma EGFR followed by medium panel tissue NGS if negative (Strategy 13), and small panel NGS with FISH (Strategy 24) had a slightly lower AGA detection rate (98.3%), but at a lower cost of USD 1047 and USD 798, respectively, with a longer turnaround time for the latter (7 days vs 14 days). Average cost of AGA testing per patient against TTTD for all 24 strategies is shown in Supplemental Figure 1.

Cost-effectiveness plot of the average cost of AGA testing per patient (in USD) against the percentage of AGAs detected in Singapore.
Cost per patient, percentage of AGAs detected and TTTD of strategies on the cost-effectiveness frontier and selected clinically relevant strategies.

AGA, actionable genomic alteration; EGFR, epidermal growth factor receptor; FISH, fluorescence in situ hybridization; NGS, next-generation sequencing; TTTD, time to treatment decision.
Scenario analysis
We performed sensitivity analyses to see how the output of our model would vary depending on the rates of AGA population and on the cost of NGS.
In a low AGA population of the United States, plasma EGFR alone (Strategy 17) was no longer cost-effective; other strategies identified in the high AGA Singapore population remained on the cost-effectiveness frontier (Figure 3). Upfront medium panel tissue NGS (Strategy 2) detected the highest proportion of AGAs (100%) at a cost of USD 1813, while exclusionary plasma EGFR followed by medium panel tissue NGS (Strategy 13) and small panel NGS with FISH (Strategy 24) detected 95.6% and 94.5% of AGAs, respectively, at a cost of USD 1752 and USD 798 respectively. The results of varying the cost of NGS in our model are shown in Supplemental Figure 2 and Supplemental Tables 6 and 7, with upfront medium panel tissue NGS becoming more beneficial as the NGS cost is lowered.
Cost-effectiveness plot of the average cost of AGA testing per patient (in USD) against the percentage of AGAs detected, in a low AGA population.
Impact of reduced cost of medium panel NGS
We had the opportunity to employ our model in a real-world scenario where the cost of NGS testing is reduced. In June 2023, a new medium panel NGS called APEX50 was introduced in Singapore, with a reduced cost of USD 1015 (from USD 1813). When this new price was incorporated into our model, exclusionary tissue testing followed by medium panel tissue NGS (Strategy 7) was no longer cost-effective; the cost differential between upfront medium panel NGS and other strategies on the cost-effectiveness frontier, including Strategy 7 and Strategy 24, became lower (Figure 4).

Cost-effectiveness plot of the average cost of AGA testing per patient (in USD) against the percentage of AGAs detected, when the cost of the medium panel NGS is reduced.
Budget impact analysis
Singapore currently adopts a mix of testing strategies (Supplemental Table 4). A BIA was performed to evaluate the impact of switching all testing to one of the four cost-effective strategies with AGA detection rate of >/=95%, on annual national expenditure on testing. Employing medium panel tissue NGS (Strategy 2) or exclusionary tissue testing followed by medium panel tissue NGS (Strategy 7) if negative would increase annual national expenditure (by USD 546,624 and USD 305,870 respectively), while utilizing exclusionary plasma EGFR followed by medium panel tissue NGS (Strategy 13) if negative or small panel NGS with FISH (Strategy 24) led to savings of $55,410 and $251,178 respectively (Figure 5; Supplemental Table 8).

Budget impact analysis following the adoption of alternative testing strategies. The budget impact of each testing strategy compared to the base case is represented to the left of annual costs. Increased budget impact is represented in red, while reduced negative budget impact (cost savings) is represented in green.
Discussion
In this cost-effectiveness analysis, we compare the relative costs of different AGA testing strategies with the outcomes of the percentage of AGAs detected and TTTD. In Singapore, a population with a high AGA prevalence of 76.7%,17–20 we identified upfront medium panel tissue NGS (Strategy 2), exclusionary tissue testing followed by medium panel tissue NGS if negative (Strategy 7), exclusionary plasma EGFR testing followed by medium tissue panel NGS if negative (Strategy 13) and small panel tissue NGS with FISH (Strategy 24) as strategies which detected >/=95% of AGAs, without any lower cost strategy with a higher AGA detection rate. Selection of strategies to focus on was performed by identifying those on the cost-effectiveness frontier, as well as those close to the cost-effectiveness frontier, which were clinically relevant, as costs and AGA detection of each of the strategies are estimated, and some variability in these numbers is expected. Of these, Strategy 24 had the lowest cost (USD 798), with an AGA detection rate of 98.3%, and a TTTD of 14 days. Compared to this, Strategy 2, which detected 1.7% more AGAs, cost more than double at USD 1813, with a cost per percentage additional AGA detected of USD 597; Strategy 7 had a cost per percentage additional AGA detected of USD 1180. Strategy 13 detected the same percentage of AGAs as Strategy 24, at a higher cost of USD 1047, however, at half the turnaround time of 7 days versus 14 days.
Given that this model had three variables: cost, AGA detection and TTTD, the purpose of this analysis, rather than to select a single most cost-effective testing strategy to be employed in all patients, was to crystallize in a model the trade-offs in these three outcomes between the strategies. The reason for this is that priorities need to be individualized for each patient—for instance, in some patients with a heavy burden of disease or impending crisis, it is important for patients and physicians to choose a testing modality with a faster TTTD, perhaps at a higher cost (Strategy 13 rather than Strategy 24). Likewise, the added cost of a small chance of detecting additional AGAs might be considered worthwhile to some patients, such as those who are keen to avoid chemotherapy at all costs. That being said, based on this analysis, exclusionary plasma EGFR followed by medium panel tissue NGS if negative, and small panel NGS with FISH, would be cost-effective selections in the majority of patients.
Cost-effectiveness analyses have previously been reported in other regions. In a Hong Kong study, sequential, exclusionary, hotspot panel and NGS tissue testing were compared, and it was concluded that exclusionary testing of EGFR mutations and ALK rearrangements, followed by NGS if negative, was the most cost-effective approach. 13 A Taiwanese study analysed exclusionary single-gene tissue EGFR mutation testing followed by NGS (either tissue or liquid) versus NGS and found significant savings from the exclusionary strategy. 21 By contrast, Pennell et al from the United States and Arriola et al from Spain reported cost savings from upfront tumour NGS over other strategies.11,22 This is largely due to the higher prevalence of AGAs in East Asia compared to the West—EGFR alterations were present in 55.7% and 26.2% in the Taiwanese and Hong Kong models, respectively, compared to 15% in the United States model. This is illustrated in our model—when AGA prevalence from Pennell et al. was inserted into our model, medium panel NGS also appeared to be more cost-effective, with a significant drop-off in AGAs detected (100% vs 95.6%) when exclusionary plasma EGFR is used, at a very similar cost (USD 1813 vs USD 1752). A Singaporean study has previously compared small panel NGS to sequential EGFR testing and found it to detect an additional 1% of AGAs, but at a higher cost. 17
The novelty of our study is in the wide range of different strategies tested (24 strategies compared to 2–4 strategies in previous studies), allowing us to characterize the costs and outcomes of each strategy in more granular detail compared to previous work. Firstly, this is the first study that incorporates different sizes of NGS panels into the model—this has not been done in previous analyses. This is especially relevant given how rapidly the field of AGA testing has expanded over the last few years, since the first analysis by Pennell et al. in 2019, with varying sizes of NGS panels now available. Secondly, with the improved accuracy and accessibility of plasma AGA testing, we included liquid NGS panels of different sizes as alternative strategies, with the benefits of being less invasive and having the potential to pick up AGAs missed on tissue sampling due to tumour heterogeneity. 23 In the Taiwanese study, the costs of both tissue and liquid NGS were combined rather than analysed and reported separately. 21 Dividing NGS testing to reflect these options instead of approaching it as a single entity allows our model to better inform patient and physician decision-making. Thirdly, we explored strategies using exclusionary plasma EGFR mutation testing. Exclusionary tissue EGFR mutation testing has been reported to be more cost-effective than upfront tissue or plasma NGS in a high EGFR-mutated NSCLC patient population 21 ; we specifically analysed plasma EGFR mutation testing in our local context as it has a rapid turnaround time and can be used to make quick treatment decisions. This is particularly relevant in patients in whom there is an urgency to start treatment—incorporating liquid testing and particularly plasma EGFR can expedite the start of treatment by precious days.
Our analysis also highlights the fact that the cost-effectiveness of AGA testing strategies is a dynamic outcome, based on the costs, turnaround time and AGA detection of the tests at that snapshot in time, and will continue to change with any change in these parameters. When the cost of medium panel NGS was lowered from USD 1813 to USD 1015, medium panel NGS became a much more feasible and cost-effective option. In parallel, the BIA can inform modifications and consensus on national testing strategies—at this time, adopting small panel tissue NGS with FISH (Strategy 24) would have the largest reduction in overall expenditure on AGA testing. If effective targeted therapy is developed against new driver molecular alterations, the balance between strategies might shift further. With all the options available, there is a need for an online tool to be developed, to allow inputs into this model to be easily accessed and varied. While this study and its conclusions are directly applicable to the Singapore population, this would allow the model to be applied to other countries by inputting different costs of tests and AGA prevalence. There is also potential for the use of artificial intelligence-based models in cost-effectiveness analyses. In this study, the cost of AGA testing was converted to USD through common currency exchange rates for ease of interpretation, but it is worth noting that an alternative conversion would be via purchasing power parity (0.89 SGD = 1USD) to reflect the relative costs.
This study has several limitations. Firstly, as the construction of the decision tree and inputs given are based on market research and estimates by clinicians, there may be uncertainty in these estimates. In addition, while we have attempted to include a wide range of clinically practiced strategies in our decision tree, it may still not be reflective of every potential strategy used by clinicians, including combination approaches such as sending plasma EGFR mutation testing concurrently with NGS. While it is not practical or feasible to cover every possible testing strategy, this analysis does include the largest number of testing strategies compared to previous studies. We also did not account for possible permutations of re-biopsies or repeat liquid NGS in the situation of insufficient tissue or insufficient ctDNA to return a result. Secondly, our model is limited to the costs of tests, and was not intended to look at costs beyond testing, hence it does not extend to downstream costs of managing complications of testing or treatment and costs of treatment. 24 Although our model may detect an AGA, the drug may not necessarily be accessible to the patient due to cost and lack of coverage. Detecting an AGA can also identify patients for suitable clinical trials and may benefit patients even if they do not receive an FDA-approved drug. Finally, the impact of the testing strategy employed on survival outcomes is important in providing a direct and quantifiable health outcome to interpret cost-effectiveness, but it was outside the scope of our study. Survival outcomes have been reported in other studies. In a Korean study, plasma-first EGFR testing compared with tissue EGFR testing increased progression-free survival by 0.5 months and overall survival by about 1 month. 25 A Brazilian study similarly found that utilizing liquid NGS improved progression-free survival (PFS) by 0.75 months and overall survival (OS) by 1.24 months over standard of care. 26 Recently, a Singaporean study looked at the long-term cost-effectiveness of tissue and liquid biopsy-based strategies, taking into account the subsequent costs and efficacy of treatment. They found that taking a sequential tissue followed by plasma NGS approach was most cost-effective, with an ICER of S$31,318/Quality-Adjusted Life Year. 27 This looked mainly at NGS approaches, whereas our study shows that there remains a role for plasma EGFR testing in our population.
Beyond cost-effectiveness and the detection of AGAs, there are many variabilities in testing strategies that play an important role in the selection of NGS over single-gene testing and the source material used (liquid or tissue biopsy), which are not accounted for in our model. Clinical and disease characteristics should be taken into account, including patient age, smoking status, disease burden, sites of disease and disease stage. Studies have shown that younger, non-smoking patients are more likely to harbour AGAs, including rarer alterations like HER2 and RET—these patients might benefit more from upfront NGS.28,29 In these patients, small panel tissue NGS with FISH (USD 789, TTTD 14 days) might provide a balance between cost-effectiveness and clinical need. In patients who need urgent initiation of treatment, exclusionary testing of plasma EGFR followed by medium panel tissue NGS if negative (USD 1047, TTTD 8.4 days) would allow EGFR mutations, a majority of AGAs, to be detected within 1–2 days by plasma EGFR; if this is negative, chemotherapy could be initiated while awaiting NGS results. This study focuses on testing strategies in advanced disease, but in patients with earlier-stage disease, where only EGFR and ALK currently have management implications, less comprehensive testing strategies may also be considered.
Characteristics of the tests also need to be considered. Our model, similar to others, assumes that all tests are 100% specific and sensitive, when in reality, there are false negatives with each test, with generally lower sensitivity with liquid compared to tissue tests and DNA-only panels compared to DNA and RNA panels, which do need to be incorporated into making a testing decision.11,13 Evidence shows that up to 50% of AGAs detected on tissue NGS might be missed on liquid NGS, particularly in patients with lower disease burden.30,31 Conversely, liquid tests carry the benefits of better representing tumour heterogeneity from multiple tumour sites, faster turnaround time, as well as avoiding the risks of invasive tumour biopsies. 32 Additionally, even in cases with known AGA, the occurrence of certain co-mutations, that might only be detected on more comprehensive sequencing strategies like NGS, affects prognosis, for instance, in patients with sensitizing EGFR mutations, where a concurrent TP53 mutation indicates a poorer prognosis. 33 This can have implications on treatment, with TP53 one of the factors considered when deciding whether for osimertinib alone or in combination with chemotherapy in EGFR-mutant lung cancer. 34 Through NGS, patients may also be identified for clinical trials of agents directed at experimental targets. In general, due to better sensitivity, tissue molecular testing remains the gold standard if sufficient tissue is available; in our analysis, it was also more cost-effective and had a shorter TTTD (USD 1813 vs USD 2126, TTTD 7 days vs 14 days). In scenarios where information on co-mutations would be helpful in determining treatment, medium tissue panel NGS upfront (USD 1813, TTTD 7 days) or even large panel tissue NGS if looking for rare or experimental targets (USD 3336, TTTD 14 days) can be considered. For patients in whom insufficient tumour is available, and re-biopsy is not feasible, medium panel liquid NGS upfront (USD 2126, TTTD 14 days) is the most cost-effective option.
Conclusion
In conclusion, in the high AGA, high EGFR mutation prevalence population of Singapore, exclusionary testing of plasma EGFR mutation testing followed by medium panel NGS was found to be cost-effective, with a high proportion of AGAs detected and short TTTD. Upfront small panel NGS + FISH had similar AGA detection at a lower cost but higher turnaround time. Our analysis provides insight into NGS testing strategies, taking into account different gene panel sizes, sample types and timing.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251405616 – Supplemental material for A cost-effectiveness analysis model of actionable genomic alteration testing strategies incorporating different modalities and sizes of next-generation sequencing panels in non-small-cell lung cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359251405616 for A cost-effectiveness analysis model of actionable genomic alteration testing strategies incorporating different modalities and sizes of next-generation sequencing panels in non-small-cell lung cancer by Yvonne L. E. Ang, Jian Chun Matthew Ong, Yiqing Huang, Jia Li Low, Kenneth Sooi, Jorn Nützinger, Xiao Jun Wang, Meaghan Gibbs, Boon Cher Goh and Ross A. Soo in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251405616 – Supplemental material for A cost-effectiveness analysis model of actionable genomic alteration testing strategies incorporating different modalities and sizes of next-generation sequencing panels in non-small-cell lung cancer
Supplemental material, sj-docx-2-tam-10.1177_17588359251405616 for A cost-effectiveness analysis model of actionable genomic alteration testing strategies incorporating different modalities and sizes of next-generation sequencing panels in non-small-cell lung cancer by Yvonne L. E. Ang, Jian Chun Matthew Ong, Yiqing Huang, Jia Li Low, Kenneth Sooi, Jorn Nützinger, Xiao Jun Wang, Meaghan Gibbs, Boon Cher Goh and Ross A. Soo in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to acknowledge Jenny Zhou and Hong Hao Fang from Analysis Group for their support with model development. This work was previously presented in abstract form at the World Lung Cancer Congress in 2023.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
