Abstract
Background:
Real-world data on chemotherapy-naïve patients with metastatic castration-resistant prostate cancer (mCRPC) treated with abiraterone plus prednisone are limited, largely deriving from small retrospective studies.
Methods:
ABitude is an Italian, observational, prospective, multicenter study of mCRPC patients receiving abiraterone plus prednisone in clinical practice. Chemotherapy-naïve mCRPC patients were consecutively enrolled at abiraterone start (February 2016 to June 2017) and are being followed for 3 years, with evaluation approximately every 6 months. Several clinical and patients reported outcomes were examined.
Results:
In this second interim analysis, among 481 enrolled patients, 453 were evaluable for analyses. At baseline, the median age was 77 years and ~69% of patients had comorbidities (mainly cardiovascular diseases). Metastases were located mainly at bones and lymph nodes; 8.4% of patients had visceral metastases. During a median follow-up of 18 months, 1- and 2-year probability of radiographic progression-free survival were 73.9% and 56.2%, respectively; the corresponding rates for overall survival were 87.3% and 70.4%. In multivariable analyses, the number of bone metastases significantly affected radiographic progression-free survival and overall survival. During abiraterone plus prednisone treatment, 65% of patients had a ⩾50% prostate-specific antigen decline, and quality of life remained appreciably high. Among symptomatic patients according to the Brief Pain Inventory) (32%), scores significantly declined after 6 months of treatment. Overall, eight patients (1.7%) had serious adverse reactions to abiraterone.
Conclusions:
Abiraterone plus prednisone is effective and safe for chemotherapy-naïve mCRPC patients in clinical practice.
Keywords
Introduction
Prostate cancer is the second most frequent cancer and the fifth leading cause of cancer death in men. 1 Most cases are diagnosed in early stages, with localized disease, while some patients (around 4–5 %) had metastatic prostate cancer at first diagnosis. 2 Despite initial treatment, some of the patients with localized disease developed progressively metastatic disease. 3 Androgen deprivation therapy (ADT) is the mainstay of treatment for patients with metastatic disease; however, these patients inevitably progress to metastatic castration-resistant prostate cancer (mCRPC), with an elevated burden of mortality. 4
Until 2010, docetaxel-based chemotherapy was the only therapeutic option improving overall survival (OS) in mCRPC.5,6 Over the last few years, however, the development of new agents with varying mechanisms of actions has dramatically changed the therapeutic landscape of mCRPC.7,8 These included androgen receptor (AR)-directed agents, immunotherapy (i.e. Sipuleucel-T), novel cytotoxic drugs, bone-targeted radiopharmaceuticals, and genetically targeted therapies.
Abiraterone acetate is a prodrug of abiraterone, which is a selective inhibitor of cytochrome P (CYP) 17, a key enzyme in extragonadal and testicular androgen synthesis.
9
Abiraterone acetate plus prednisone demonstrated survival benefits in mCRPC patients, initially in mCRPC patients docetaxel pre-treated10,11 and post ADT12,13 and subsequently in high-risk
Translating results from clinical trials with strict eligibility criteria to unselected patients treated in routine practice remains a concern. 16 Clinical trials tended to exclude patients with pre-existing medical conditions and poor prognostic features, and this may have important implications for the generalizability of their results. This is particularly relevant for mCRPC, as most patients are elderly and have significant comorbidities, namely cardiovascular disease, hypertension and diabetes mellitus. 17 Such vulnerable patients along with those with visceral metastases are under-represented in clinical trials of mCRPC. Real-life data from well-designed prospective studies provide valuable information on the effectiveness and safety of a drug across the full spectrum of patients who are treated in routine practice. However, to date, real-life data on abiraterone in chemotherapy-naïve mCRPC patients are limited18–30 and derived largely from relatively small retrospective studies. To bridge this crucial gap in knowledge in 2016, ABItude, a multicentric real-life observational prospective study, was initiated. With over 450 enrolled patients, it represents one of the largest prospective “real-world” investigations on the clinical effectiveness of abiraterone in the chemotherapy-naïve setting. ABItude is an ongoing study, and in the first planned interim analysis (~9 months median follow-up) abiraterone was active and safe in an elderly population with a high level of comorbidities. 31 The 1-year rPFS was ~74% and adverse reactions occurred in 10% of patients, the vast majority being nonserious. In addition, abiraterone delayed functional decline and improved HRQoL and pain palliation. Here, we present results from the second interim analysis after 18 months of median follow-up.
Methods
ABItude is a prospective, observational, multicentric study conducted in Italy aiming at describing the effectiveness and safety of abiraterone acetate plus prednisone when used in clinical practice for the treatment mCRPC in chemotherapy-naïve patients. From February 2016 to June 2017, all eligible patients with mCRPC naïve to chemotherapy were consecutively enrolled in 49 Italian participating centers (urological, radiotherapy and oncological unit) at the time of initiating abiraterone acetate plus prednisone therapy according to clinical practice. Patients eligible were men aged ⩾18 years with histologically or cytologically confirmed metastatic adenocarcinoma of the prostate, asymptomatic or mildly symptomatic according to clinical judgement, naïve to chemotherapy, surgically or medically castrated, who failed ADT and in whom chemotherapy was not clinically indicated, starting treatment with abiraterone acetate within 30 days after the baseline visit according to clinical practice. Patients already treated with chemotherapy for prostate cancer or participating in experimental clinical trials were excluded.
After enrollment, follow-up visits are planned throughout the 36-month observation period according to clinical practice, that is, around every 6 months unless clinically indicated.
The study was approved by the ethic committees of the participating centers (ethics committee approval number of the coordinating center: INT-45/15-29/10/2015) and was conducted according to the principles of the Declaration of Helsinki and the Good Clinical Practice guidelines.
All patients provided written informed consent to participate in the study.
Clinical information was retrieved by medical records. At baseline, the following data were collected: demographic and anthropometric characteristics, relevant medical history (e.g. hepatic, renal, and cardiovascular function), historical data on prostate cancer (tumor–node–metastasis (TNM) stage, Gleason score at diagnosis, date of diagnosis, and date of castrate resistance), and previous treatment for prostate cancer (including ADT and its duration). Data on vital signs, relevant concomitant therapies, Eastern Cooperative Oncology Group (ECOG) performance status (PS), patient-reported pain and HRQoL were collected at baseline and at each follow-up visit. The pain was evaluated through the Brief Pain Inventory (BPI), a validated instrument comprising several individual items measured on a 0–10 scale, with lower scores representing lower levels of pain intensity or less interference of pain with activities of daily living (e.g. sleep, mood, and activity). Patients HRQoL was measured using the Functional Assessment of Cancer Therapy-Prostate (FACT-P) 32 and the EuroQol-5D (EQ-5D) 33 questionnaires. The EQ-5D-3L version includes three levels of severity (no problems, some problems, extreme problems) in each dimension (mobility, self-care, usual activities, pain/discomfort and anxiety/depression) and includes a visual analog scale (VAS), which records the respondent's self-rated general state of health on a scale from 0 (worst imaginable health status) to 100 (best imaginable health status). An appropriate algorithm was used to summarize data in an overall score ranging from −1 (worse-than-death health status) to 1 (best health status).34,35 Only questionnaires filled in while the patient was under treatment with abiraterone were evaluated in the analyses.
All available measurements of prostate-specific antigen (PSA) values were recorded during the course of the study. Data on abiraterone acetate and prednisone treatments (start and stop dates, dose and frequency of administration, reason for start and reason for choice of treatment, dose changes and reason, treatment interruptions and discontinuations) and on therapies subsequent to abiraterone (if any), analgesic and opioid use, clinical and radiographic progression, clinical benefit according to clinician judgement, adherence to treatment with abiraterone, adverse events (AEs) and adverse drug reactions (ADRs), and survival were recorded during the observation period. Adherence to treatment with abiraterone was evaluated using the Morisky 8-Item Self-Report Measure of Medication-taking Behavior (MMAS-8). 36
Details of the statistical analyses are reported as supplementary material.
Results
Study population
From February 2016 to June 2017, a total of 481 patients were recruited in the study; 474 did start abiraterone treatment and were considered in the safety analysis; among them, 453 were evaluable for the analyses (full-analysis set). Analyses on PSA decline were based on a subset of 413 patients with PSA data at baseline and follow-up. Figure S1 (supplementary material) presents the study flowchart showing criteria violations and analysis sets.
At the time of the second planned interim analysis, the median follow-up time was 18.1 months [interquartile range (IQR) 12.1–24.0]. A total of 331 patients (73%) out of 453 were managed in oncology, 64 (14%) in urology and 58 (13%) in radiotherapy centers. In 24% of cases, abiraterone treatment choice was taken by a multidisciplinary team (73 of 305 patients with available information) (data not shown). At the time of first prostate cancer diagnosis, 14.6% of patients presented distant metastases (information was unknown for 9 patients) and 58.9% of patients had Gleason score ⩾8 (information was unknown for 59 patients).
Demographic and clinical patients’ characteristics at baseline are presented in Table 1. At the initiation of abiraterone treatment, the median age was 77 years (range: 51–93), and 265 patients (58.5%) were ⩾75 years old. The large majority of patients (94.8%) had ECOG-PS of 0 or 1. Over 42% of patients had metastases at bones only, 22% at lymph nodes only, and 23% at bones and lymph nodes; 8% of patients had visceral metastases. Among patients with bone metastases, about 22% had 10 or more lesions. The large majority of patients underwent medical castration (94%) as compared to surgical castration (6%). Over two-third of patients had at least one comorbidity at the start of treatment. In particular, 56% of patients had stable and well-compensated cardiovascular disorders (46% had hypertension) and 23.2% had metabolic disorders (11% had diabetes and 11.5% had hypercholesterolemia).
Demographic and clinical patients’ characteristics at baseline.

CSN, central nervous system; ECOG-PS, Eastern Cooperative Oncology Group performance status
IQR, interquartile range; PSA, prostate-specific antigen.
Including prostatic bed (
One missing value.
A patient could have more than one relevant medical condition/disease.
Hypertension: 48.6%; history of myocardial infarction: 5.5%; arrhythmia: 4.4%; cardiomyopathy: 3.5%; angina pectoris: 0.7%; atherosclerosis: 0.4%; other: 8.8%.
Hypercholesterolemia 11.5%; diabetes 10.8%; hyperglycemia: 0.7%; obesity: 0.7%; other: 5.1%.
Treatment exposure and clinical outcomes
Median duration of abiraterone treatment was 14 months (IQR 7.2–20.5). During follow-up, 285 patients permanently discontinued abiraterone, mainly because of disease progression (184 patients, 65% who discontinued abiraterone). Other reasons for treatment discontinuation included death (
Figure 1 shows rPFS and OS for our cohort of chemotherapy-naïve mCRPC patients. During a median follow-up of 18 months, 307 patients (67.8%) did not experience any radiographical progression or death during the treatment with abiraterone. Median rPFS on abiraterone treatment was not reached and the 1- and 2-year probability of rPFS were 73.9% and 56.2%, respectively. Over the same observation period, 102 patients (22.5%) died. Median OS was not reached and the 1- and 2-year survival rates were 87.3% and 70.4%, respectively.
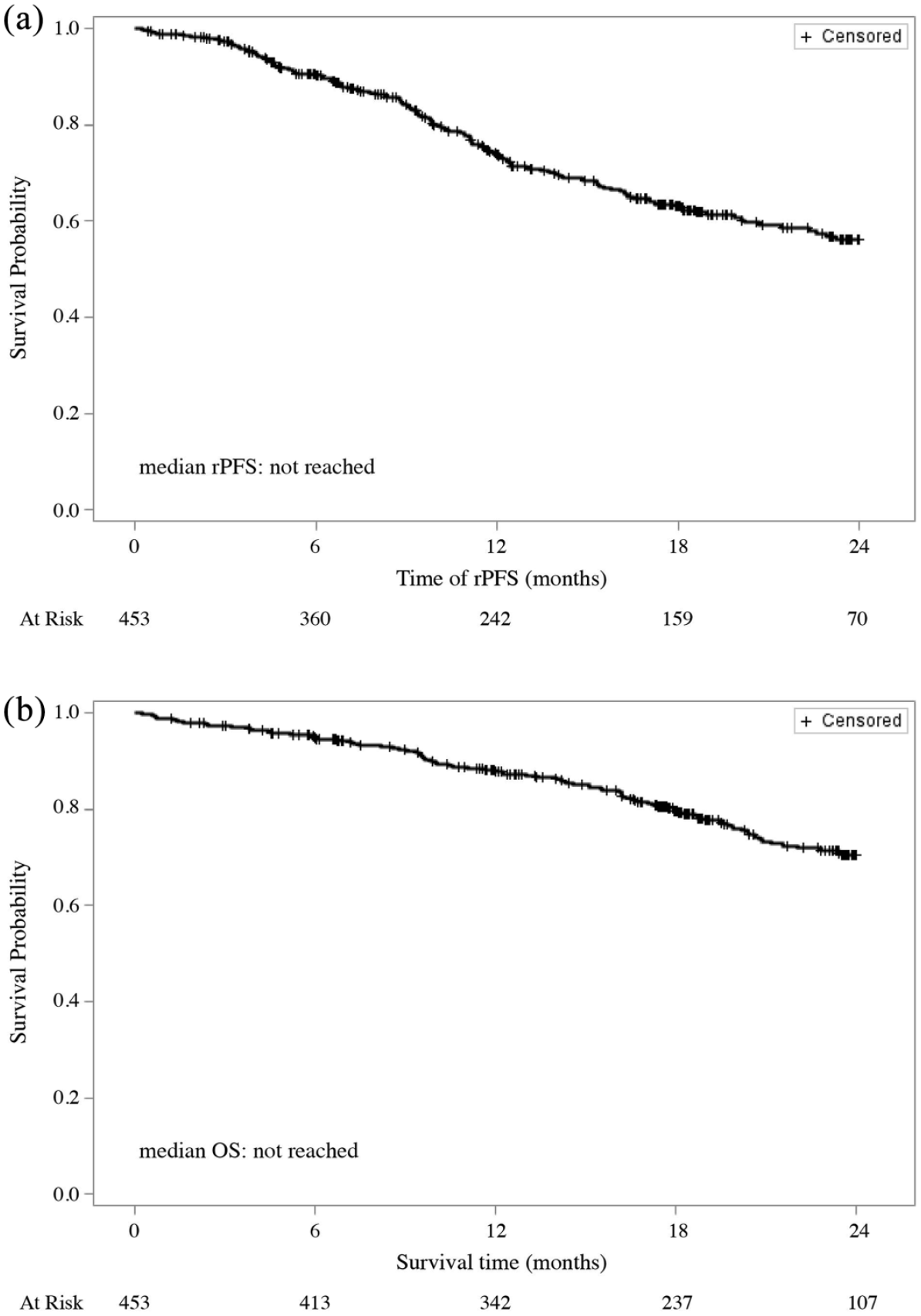
(a) Radiographical progression-free survival (rPFS) and (b) overall survival (OS) during abiraterone treatment.
Table 2 shows results for the association of selected demographic and clinical characteristics with rPFS and OS. Results from univariate and multivariate analyses were consistent. In multivariate analyses, the number of bone metastases was a significant prognostic factor for rPFS [hazard ratio, (HR) 1.76, 95% confidence interval (CI): 1.03–3.03,
Univariate and multivariable analyses a for the association of selected baseline demographic and clinical characteristics with rPFS and OS.

CI, confidence interval; ECOG-PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; PSA, prostate-specific antigen; rPFS, radiographic progression-free survival; OS, overall survival.
Cox proportional hazard models.
Model adjusted for age, PSA at baseline, presence of comorbidities, visceral metastases, bone metastases, and using ECOG-PS as stratification factor.
Reference category
Of the 413 patients evaluated for PSA, 269 (65.1%) achieved a decline in PSA of ⩾50% during abiraterone treatment.
Patient-reported outcomes measures
At baseline, the median VAS score was 70.0 (IQR 50.0–80.0) points. VAS score remained quite stable during abiraterone treatment (

Patient’s health-related quality of life: visual analog scale (VAS) score during abiraterone treatment.
The overall EQ-5D-3L score remained stable over time during abiraterone treatment [median (IQR) equal to 0.9 (0.8–1.0) points both at baseline and at 24 months of follow-up,
In the overall population, BPI scores improved while on treatment with abiraterone. Median at baseline and at 24 months of follow-up were 1 (IQR 0–3) and 0 (IQR 0–2.5) for the mean pain intensity, 2 (IQR 0–4) and 0 (IQR 0–3) for the worst pain intensity, and 0.4 (IQR 0–3) and 0.3 (IQR 0–2.1) for interference of pain, respectively.
Among symptomatic patients according to BPI (32%), BPI scores improved over time, with significant declines in all the three BPI domains at 6 months of treatment (Figure 3). Median values at baseline were 4.0 (IQR 3.0–5.0) for mean pain intensity, 5.0 (IQR 4.0–7.0) for worst pain intensity and 3.8 (IQR 1.9–6.0) for the interference of pain; corresponding median changes at 24 months of follow-up for the three variables were −1.4 (IQR −2.9, +0.8), −2.0 (IQR −4.0, +1.0), and −0.3 (IQR −2.0, +0.6), respectively. In this subgroup of patients, median rPFS and OS were not reached. The rPFS and OS at 1 year were 66.4% and 77.2%, respectively.

Pain assessment (Brief Pain Inventory; BPI) during abiraterone treatment in symptomatic patients.
Safety
Among the 474 patients analyzed for safety, 231 (48.7) had at least one AE during abiraterone treatment and 84 patients (17.7%) had at least one serious AE (Table S1, supplementary material). Overall, 61 patients (12.9%) had AEs related to abiraterone per clinician’s judgement, but only 1.7% had a serious AE. The details of the AEs are presented in Table 3.
Adverse events occurred in >3% of patients during abiraterone treatment.

Discussion
In the present second interim analysis of the ABItude study, chemotherapy-naïve mCRPC patients treated with abiraterone in routine clinical practice showed good clinical outcomes, preserved HRQoL, and achieved good pain control over a median of 18 months of follow-up.
In agreement with previous real-world studies,18,20,22,24–26,28,37, including Italian ones,21,27 our findings confirm, in a real-life setting, the efficacy and safety of abiraterone demonstrated in the registration trial (COU-AA-302), despite the poorer clinical features of the enrolled patients, including the advanced age (60% of our patients were ⩾75 years old) and the high prevalence of comorbidity.
In the COU-AA-302 trial, only one-third of patients were ⩾75 years old and those with relevant comorbidities were excluded, as well as those with visceral metastases. Conversely, about 8% of our cohort of patients had visceral metastases at treatment initiation. Although a negative prognostic role of visceral disease has been identified, a few evidences confirmed the efficacy of abiraterone in this population. The post-chemo study on mCRPC patients, COU-AA-301, 38 and the Latitude study in mHSPC 39 showed that patients with visceral disease had a benefit from abiraterone treatment both in rPFS and in OS, similar to the remaining population and irrespective of the organ involved, lung or liver. Furthermore, a recent Italian real-life study found that abiraterone was effective and safe in a small series of patients with visceral metastases, both in the pre-chemotherapy and post-docetaxel settings. 40
In our prospective study of chemotherapy-naïve mCRPC patients, age, presence of comorbidities and visceral disease did not significantly affect rPFS and OS in multivariate analyses, thus indicating good response of patients treated with abiraterone with such baseline frail conditions. Presence of at least 10 bone metastases was directly associated to worst clinical outcomes, in agreement with a prognostic model based on COU-AA-302 data. 41
In mCRPC patients, the progression of the disease usually leads to worsening symptomatology due to complications of metastases and treatment-related toxicities.
42
Delaying symptoms, preserving HRQoL and pain palliation are important therapeutic objectives in the management of mCRPC.
43
In clinical trials definition of symptomatic or asymptomatic patients was done through BPI, a validated scale to be filled in by the patient, whereas in clinical practice it is usually defined by clinicians’ judgement. Interestingly, in our real-life study we observed a discrepancy between patient and physician assessment. Indeed, 32% of patients reported symptoms per self-assessment at abiraterone initiation whereas judged asymptomatic or mildly symptomatic by physicians. This is the first time that effectiveness and quality of life is prospectively evaluated in first-line symptomatic mCRPC patients, demonstrating that patients with pain at baseline experienced a significant improvement in pain control in the early months of treatment and achieved good clinical outcomes, similar to the overall population enrolled in our study (66.4%
To our knowledge, this is the largest prospective study assessing the effectiveness and safety of abiraterone acetate plus prednisone in mCRPC patients naïve to chemotherapy in a real-world setting. Our study includes a broad range of patients with a high level of comorbidities from several major oncological, urological and radiotherapy centers across the country, reflecting the mCRPC population encountered in real-world settings. This, together with the inclusion of all consecutive patients meeting the inclusion criteria over a 12-month period, is likely to have minimized selection bias and ensured the generalizability of our results to the mCRPC Italian patient population. Another major strength is the
In conclusion, the present large prospective data confirm that treatment with abiraterone plus prednisone is effective and well tolerated in mCRPC patients naïve to chemotherapy, even though in the real-life they are more elderly, vulnerable and have a high burden of disease such as visceral metastases and pain.
Supplemental Material
Supplementary_material_3 – Supplemental material for Effectiveness of abiraterone acetate plus prednisone in chemotherapy-naïve patients with metastatic castration-resistant prostate cancer in a large prospective real-world cohort: the ABItude study
Supplemental material, Supplementary_material_3 for Effectiveness of abiraterone acetate plus prednisone in chemotherapy-naïve patients with metastatic castration-resistant prostate cancer in a large prospective real-world cohort: the ABItude study by Giuseppe Procopio, Vincenzo Emanuele Chiuri, Monica Giordano, Giovanna Mantini, Roberto Maisano, Roberto Bordonaro, Nicola Calvani, Gaetano Facchini, Sabino De Placido, Mario Airoldi, Andrea Sbrana, Donatello Gasparro, Giuseppe Mario Ludovico, Pamela Guglielmini, Emanuele Naglieri, Daniele Fagnani, Massimo Aglietta, Luigi Schips, Patrizia Beccaglia, Alessandro Sciarra, Lorenzo Livi and Daniele Santini in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors acknowledge the contribution of all medical professionals who supported the data collection phase. In particular, they are grateful to: Saverio Cinieri (Brindisi), Luca Galli (Pisa), Rodolfo Passalacqua (Cremona), Lucio Trodella (Roma), Riccardo Santoni (Roma), Giovanni Luca Ceresoli (Bergamo), Stefano Magrini (Brescia), Alessandra Mosca (Novara), Vincenzo Mirone (Napoli), Michele Gallucci (Roma), Mirko Acquati (Monza), Francesco Boccardo (Genova), Giorgio Vittorio Scagliotti (Orbassano), Manlio Mencoboni (Genova), Ugo De Giorgi (Meldola), Virgilio Cicalese (Avellino), Gaetano Lanzetta (Grottaferrata), Donata Sartori (Mirano), Paolo Carlini (Roma), Hector Josè Soto Parra (Catania), Michele Battaglia (Bari), Francesco Uricchio (Napoli), Antonio Bernardo (Pavia), Antonello De Lisa (Cagliari), Giuseppe Carrieri (Foggia), Antonio Ardizzoia (Lecco), Michele Aieta (Rionero in Vulture), Salvatore Pisconti (Taranto), Paolo Marchetti (Roma), and Fabiola Paiar (Pisa).
The authors also thank Carlotta Galeone, who provided medical writing services on behalf of Statinfo srl.
Conflict of interest statement
G. Procopio has an honoraria/consulting or advisory role for AZ, Bayer, BMS, Ipsen, Janssen, Merk, MSD, Pfizer and Novartis. V. Chiuri is an Advisory Board member and speaker for BMS, Ipsen, Janssen and Pfizer. R. Bordonaro has an honoraria/consulting or advisory role/speaker’s bureau for Bayer, AstraZeneca, Sanofi, Novartis, Amgen, Roche, Pfizer, Janssen Cilag and Bristol Mayer Squibb. S. De Placido is an Advisory Board member and invited speaker for Novartis, Roche, Celgene, AstraZeneca, Amgen, Eisai, Lilly, Pfizer and Gentili. P. Beccaglia is an employee of Janssen.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by Janssen-Cilag SpA.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
