Abstract
Antibody-drug conjugates (ADCs) have emerged as a promising therapeutic option, combining the specificity of monoclonal antibodies with the potency of cytotoxic payloads. While initially developed for hematologic and select solid malignancies, advances in antigen discovery, linker chemistry, and payload design have expanded their application to metastatic castration-resistant prostate cancer, where treatment options remain limited. Prostate-specific membrane antigen (PSMA), STEAP-1, TROP-2, CD46, Nectin-4, tissue factor, B7-H3, and DLL3 have emerged as clinically relevant targets, with multiple ADCs evaluated in early and late-phase trials. Although early-generation drugs were hindered by modest efficacy and significant toxicity due to linker instability and off-target effects, some novel ADCs have shown improved tolerability and encouraging antitumor activity. Ongoing studies are exploring rational combinations with hormonal, other targeted, and immune-based therapies to enhance efficacy, overcome resistance, and expand the role of ADCs in advanced prostate cancer. Herein, we provide a comprehensive overview of the clinical development of ADCs in advanced prostate cancer.
Plain language summary
Antibody-drug conjugates (ADCs) are a new type of cancer treatment designed to deliver chemotherapy directly to cancer cells. Each ADC has two parts: an antibody that seeks out a marker on cancer cells, and a chemotherapy attached to it. This design allows tumors to be targeted more precisely while limiting damage to healthy cells.
ADCs are now being tested in advanced prostate cancer that no longer responds to hormone therapy, a stage where treatment options are limited. Several tumor markers are being studied as targets for these medicines in clinical trials. Newer ADCs, and combinations with hormone therapy, targeted drugs, and immunotherapy, are also being explored to improve effectiveness and overcome resistance.
Introduction
Prostate cancer remains one of the most prevalent malignancies among men worldwide and continues to pose a significant health burden, particularly in its advanced stages. 1 While androgen deprivation therapy combined with androgen receptor pathway inhibitors (ARPIs) has extended overall survival (OS) in men with metastatic hormone-sensitive prostate cancer (mHSPC), the prognosis remains poor once resistance develops. 2 The median OS for patients with metastatic castration-resistant prostate cancer (mCRPC) still ranges between 15 and 36 months, depending on prior treatment exposure and molecular features. 2 This poor prognosis underscores the urgency to explore and develop novel treatment modalities that can provide durable responses with manageable toxicity profiles.
One such promising class of therapeutics is antibody-drug conjugates (ADCs), which have gained momentum across several solid tumor types. 3 Over the past decade, ADCs have shown transformative clinical activity in malignancies such as triple-negative breast cancer (e.g., sacituzumab govitecan), HER2-positive breast cancer and non-small cell lung cancer (e.g., trastuzumab deruxtecan), and urothelial carcinoma (e.g., enfortumab vedotin). 4 These agents combine the specificity of monoclonal antibodies with the potency of chemotherapeutic payloads, connected through cleavable or non-cleavable linkers that control intracellular drug release. 3 To date, over 10 ADCs have received the US Food and Drug Administration (FDA) approval, and more than 80 others are in various stages of clinical development. 4
In the context of prostate cancer, the application of ADCs is an emerging area of interest driven by the identification of tumor-specific surface antigens such as prostate-specific membrane antigen (PSMA), STEAP-1, TROP-2, CD46, and B7-H3. 5 These targets offer opportunities to leverage ADC technology for mCRPC, particularly in patients who have received standard therapeutic options. Several early-phase clinical trials have tested the safety and efficacy of ADCs targeting these antigens, with mixed but evolving results.6,7 This variability in clinical activity partly reflects prostate cancer’s intrinsic resistance to ADC strategies, driven by heterogeneous and often low antigen expression, limited internalization and lysosomal processing, and active efflux mechanisms that reduce intracellular payload delivery. 8 As technology improves, newer ADCs with enhanced stability, optimized drug-to-antibody ratios, and more potent payloads are entering the clinical space, potentially overcoming previous limitations such as off-target toxicity and narrow therapeutic windows.
In this narrative review, we provide a comprehensive overview of the rationale, current development, and future potential of ADCs in prostate cancer. We highlight key clinical trials, discuss biological targets under investigation, and address the challenges and innovations shaping the next generation of ADCs in this disease setting.
Mechanism of action and concept
Antibody-drug conjugates represent a targeted therapeutic approach, designed to deliver cytotoxic agents selectively to tumor cells by utilizing tumor-associated antigens. ADCs are engineered molecules composed of three key parts: a monoclonal antibody targeting a tumor-associated antigen, a potent cytotoxic payload, and a chemical linker that connects them together (Figure 1). 3

Mechanism of action of antibody-drug conjugates 9
Following intravenous administration, the monoclonal antibody component circulates systemically until it binds selectively to its specific antigen on the surface of the cancer cell. This antigen-antibody interaction triggers receptor-mediated endocytosis, internalizing the ADC into lysosomes. 3 Within the lysosomal compartment, the linker is cleaved either chemically or enzymatically, depending on its design, thereby releasing the cytotoxic payload. Released payloads exert their effects by disrupting essential cellular processes. Microtubule inhibitors, such as monomethyl auristatin E (MMAE), arrest mitosis. DNA-damaging agents, like DNA topoisomerase I inhibitors, induce replication stress and apoptosis. 3
Linker chemistry plays a critical role in the ADC mechanism, balancing two opposing requirements: stability in circulation to prevent premature drug release and rapid cleavage upon reaching tumor cells for effective drug delivery. 4 Cleavable linkers use mechanisms such as pH sensitivity or enzyme-mediated cleavage, whereas non-cleavable linkers depend on complete antibody degradation. 10 Recent advancements in linker stability have enabled higher drug-to-antibody ratios, enhancing therapeutic potency without significantly increasing systemic toxicity.
Moreover, ADCs have evolved additional mechanisms to enhance therapeutic efficacy. “Bystander killing” has emerged as an important strategy in ADC development. 10 This approach involves extracellular cleavage, which releases membrane-permeable cytotoxic agents that diffuse into adjacent tumor cells, effectively targeting tumor regions with heterogeneous or low antigen expression. 9 This is particularly relevant in prostate cancer, given the variable expression of surface antigens such as PSMA, STEAP-1, TROP-2, and CD46. The recently developed TMALIN linker employs tumor-selective dual-cleavage, occurring both extracellularly in the tumor microenvironment and intracellularly within lysosomes (Figure 1). 11 This design enhances payload release even in antigen-low tumor cells, thereby improving efficacy against highly heterogeneous solid tumors. 11
Despite their targeted approach, ADCs can still cause off-target toxicity if the antigen is expressed in healthy tissues or if the linker prematurely releases the payload. Overall, the efficacy of ADCs in prostate cancer depends significantly on antigen specificity, linker stability, payload potency, and the capability to mediate bystander effects when direct internalization is insufficient. 4
Immunohistochemical studies demonstrate variable but generally high expression of several ADC target antigens in prostate cancer, with differences in tissue specificity, heterogeneity, and association with disease stage. PSMA is enriched in prostate epithelium compared with most normal tissues; however, its expression becomes increasingly heterogeneous in advanced disease, with reduced or absent membranous expression observed in a subset of mCRPC, particularly in tumors with neuroendocrine differentiation.12,13 STEAP-1 shows greater prostate cancer specificity and relatively consistent expression across disease stages, with high prevalence maintained in advanced and metastatic tumors. 14 TROP-2, while not prostate-specific and broadly expressed across epithelial malignancies, is frequently expressed in mCRPC, with higher expression reported in primary prostate tumors compared with metastatic sites. 15 CD46 is expressed in normal tissues but is overexpressed in prostate cancer and retained in the majority of mCRPC tumors, including neuroendocrine prostate cancer. 16 B7-H3 exhibits limited expression in most normal tissues but is highly prevalent in mCRPC, and appears to be enriched in biologically aggressive subtypes, including tumors with DNA damage repair alterations. 17 These patterns of tissue distribution, heterogeneity, and disease-stage association provide a biologic rationale for ADC development in advanced prostate cancer.
PSMA-targeting ADCs
MLN2704
MLN2704 was the first PSMA-targeting ADC evaluated in clinical trials. PSMA is expressed in different normal tissues, including prostate, kidney, and bladder, but its expression is more important in prostate cancer cells. 18
MLN2704 is comprised of a monoclonal antibody conjugated to the antimicrotubular agent maytansinoid-1 (DM-1) via a disulfide linker. 19 Once internalized by PSMA-expressing cells, the release of DM-1 leads to cell death through apoptosis. MLN2704 was studied in a phase I/II, open-label, single-arm dose-escalation trial enrolling 62 patients with mCRPC with evidence of radiographic, clinical, or PSA progression. 20 The primary objectives of this trial were to evaluate the dose-limiting toxicity (DLT), maximum tolerated dose (MTD), and antitumor activity (Table 1).
PSMA-targeting antibody-drug conjugates in metastatic castration-resistant prostate cancer.

ARPI, androgen receptor pathway inhibitor; CT, chemotherapy; DLT, dose-limiting toxicity; G, grade; mCRPC, metastatic castration-resistant prostate cancer; MTD, maximum tolerated dose; NA, not applicable; ORR, objective response rate; PSA50, ⩾50% decline in prostate-specific antigen; PSMA, prostate-specific membrane antigen; RP2D, recommended phase II dose.
Of 62 patients, 56% had received prior chemotherapy. 20 Patients were enrolled into four dosing schedules–weekly, every 2 weeks, every 3 weeks, or on days 1 and 15 of a 6-week cycle–with doses ranging from 60 mg/m2 to 462 mg/m2. Antitumor activity was assessed by PSA response and radiographic response by RECIST v1.1 at 12-week intervals.
Treatment-related adverse events (TRAEs) were frequent. Peripheral neuropathy occurred in 71% of patients, nausea in 61%, and fatigue in 60%. 38% of patients (
Efficacy was modest, with only 8% (
The limited efficacy and substantial neurotoxicity of MLN2704 were likely due to the instability of its disulfide linker, causing off-target toxicity in plasma and resulting in a narrow therapeutic window. 20 Consequently, the development of MLN2704 was discontinued after this phase I/II trial. Nevertheless, this study confirmed PSMA as a promising target, paving the way for further development of PSMA-targeting ADCs.
PSMA-MMAE
Similar to MLN2704, PMSA-MMAE is a PSMA-targeting ADC conjugated to monomethyl auristatin E (MMAE), a microtubule-disrupting payload, via a stable valine-citrulline linker. Unlike disulfide linkers, valine-citrulline linkers are enzymatically cleaved and are less susceptible to premature cleavage in plasma, improving payload stability. 21 In the phase I study, PSMA-MMAE was well tolerated, with no grade ⩾ 3 TRAEs reported. 22
PSMA-MMAE was subsequently evaluated in an open-label, single-arm, phase II clinical trial. 23 A total of 119 patients with mCRPC who experienced progression after treatment with abiraterone and/or enzalutamide were included. The primary objectives were to determine antitumor activity, circulating tumor cell (CTC) response, and radiographic response per RECIST v1.1, as well as to assess safety and tolerability at the MTD. 23
The drug was initially administered at 2.5 mg/kg every 3 weeks for up to eight doses; however, the dose was reduced to 2.3 mg/kg shortly after initiation due to dose-limiting febrile neutropenia and sepsis. Despite this, only 14.3% (
The PSA response was modest, with a PSA50 of 21% in chemotherapy-naïve and 11% in chemotherapy-experienced patients at a dose of 2.3 mg/kg. However, given that PSMA-MMAE does not directly alter PSA expression, PSA response may underestimate its efficacy. Notably, CTC response was more encouraging, with 78% of patients achieving a CTC decline of ⩾ 50% and 63% of patients exhibiting radiographically stable disease. 23 In the chemotherapy-naïve group, 5.7% (2/35) of patients had a partial response. Outcomes tended to favor the chemotherapy-naïve group with a seven-month OS of 97.1% compared to 91.7% in the chemotherapy-experienced group. 23
Like MLN2704, PSMA-MMAE demonstrated limited clinical activity and a considerable side effect profile. The higher toxicity seen in phase II compared to phase I highlights the challenges of dose optimization for ADCs carrying microtubule-disrupting payloads. Development of PSMA-MMAE was discontinued following this trial.
MEDI3726
In contrast to MLN2704 and PSMA-MMAE, which contain microtubule-disrupting payloads, MEDI3726 is a PSMA-targeting ADC conjugated to tesirine, a pyrrolobenzodiazepine (PBD) dimer payload that crosslinks DNA and induces cell death. 24
MEDI3726 was evaluated in a phase I/Ib, open-label, dose-escalation trial enrolling 33 patients with mCRPC who had a progression on abiraterone and/or enzalutamide and taxane-based chemotherapy. 25 The primary objective was to evaluate safety, DLTs, and determine the MTD. Secondary objectives were to evaluate efficacy by radiographic response by RECIST v1.1 and PSA50. MEDI3726 was administered every 3 weeks at doses ranging from 0.015 mg/kg to 0.3 mg/kg, targeting a 30% DLT rate. 25
Like prior PSMA-targeting ADCs, toxicity with MEDI3726 was significant. In total, 11 (33.3%) patients discontinued treatment due to TRAEs, with 10 of these patients receiving doses ⩾ 0.2 mg/kg. Grade ⩾ 3 TRAEs were observed in 69.7% of patients (
Efficacy was limited, with a composite response rate of 12.1%. No RECIST v1.1 responses were confirmed, though 12 patients (36.4%) had stable disease, and one patient had an unconfirmed partial response. Four patients had a CTC response of > 50%, and one patient had a PSA50. The median progression-free survival (PFS) and median OS were 3.8 months. The median PFS and median OS were 3.6 months and 8.9 months, respectively. 25
Despite modest efficacy in a heavily pretreated population, treatment toxicity limited dose escalation and ultimately led to discontinuation of MEDI3726 development after this phase I trial.
ARX517
Although PSMA has been shown to be a viable target in mCRPC, systemic toxicity caused by off-target activity has proven to be a significant barrier to the development of ADCs. In response to this challenge, ARX517, a PSMA-targeting ADC, was designed with innovative strategies to minimize toxicity. 26 These included the incorporation of site-specific synthetic amino acids into the antibody to allow for precise attachment of amberstatin-269, a potent tubulin inhibitor. Additionally, ARX517 contains a noncleavable linker, reducing the premature release of amberstatin-269.
ARX517 was investigated in a phase I 3+3 dose-escalation study of 24 patients with mCRPC who had received ⩾ 2 treatments. 27 Key objectives were to assess safety, pharmacokinetics, and clinical efficacy. Patients included in the study had received a median of 4 prior lines of therapy.
In contrast to prior PSMA-targeting ADCs, ARX517 demonstrated favorable tolerability. Four grade 3 adverse effects were reported, with 3/4 occurring at doses 2.4 mg/kg or higher. 27 Notably, no grade 4 TRAEs or serious adverse events were reported. The pharmacokinetic profiles for total antibody and ARX517 were similar, indicating low premature payload release.
The median duration of treatment was 6.3 months. Initial tumor efficacy was promising. Of the 24 patients included in the study, 8 had a PSA50, with 7/8 occurring at doses 2.0 mg/kg or higher.
27
In addition, 63.2% (
Overall, ARX517’s enhanced stability and site-specificity yielded encouraging efficacy in a treatment-refractory population with favorable tolerability in the dose-escalation phase. Investigation of ARX517 is currently ongoing as a phase Ib dose-expansion study (NCT04662580).
STEAP-1-targeting ADCs
DSTP3086S is a humanized monoclonal antibody associated with MMAE and designed to target STEAP-1, which is highly expressed on prostate cancer cells. 7 It includes a protease-labile linker. This agent was assessed in a phase I multicenter open-label 3+3 dose-escalation study in patients with mCRPC (Table 2). The primary endpoints were safety and the recommended phase II dose of the drug.
Antibody-drug conjugates targeting novel tumor antigens in metastatic castration-resistant prostate cancer.
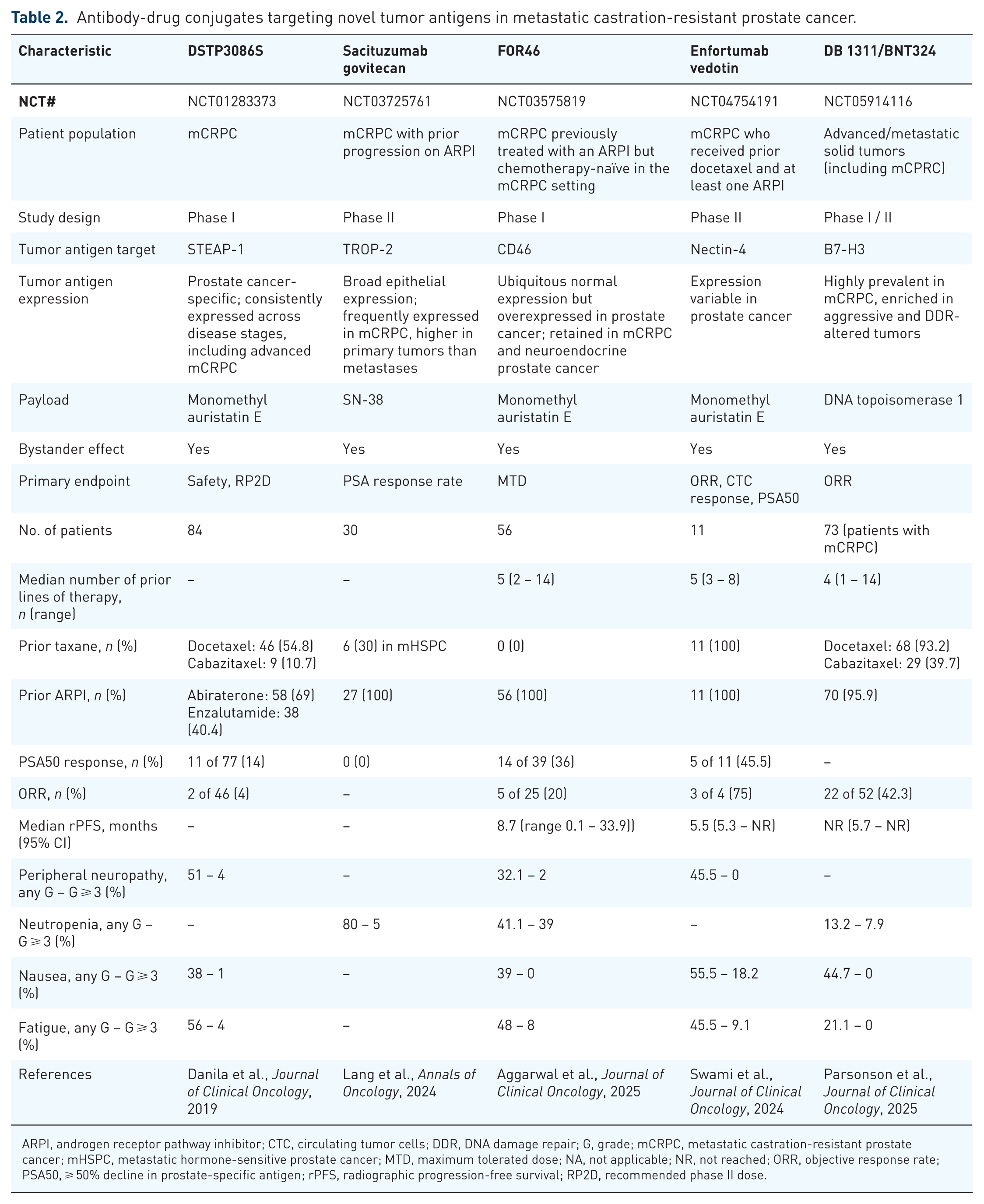
ARPI, androgen receptor pathway inhibitor; CTC, circulating tumor cells; DDR, DNA damage repair; G, grade; mCRPC, metastatic castration-resistant prostate cancer; mHSPC, metastatic hormone-sensitive prostate cancer; MTD, maximum tolerated dose; NA, not applicable; NR, not reached; ORR, objective response rate; PSA50, ⩾ 50% decline in prostate-specific antigen; rPFS, radiographic progression-free survival; RP2D, recommended phase II dose.
A total of 84 patients with mCRPC were enrolled in this study. The most common TRAEs encountered in patients receiving the once-every-3-weeks dosing schedule were fatigue (56%), peripheral neuropathy (51%), and nausea (38%). The most common grade ⩾ 3 TRAEs were fatigue (4%), peripheral neuropathy (4%), and increased alanine aminotransferase (ALT) (3%). 7
Among the 77 patients enrolled in the once-every-3-weeks dosing schedule, a confirmed PSA50 response was encountered in 11 patients (14%). Of the 46 patients with RECIST-evaluable disease, 4% had a partial response (
While DSTP3086S showed evidence of antitumor activity with an acceptable safety profile, further efforts are needed to optimize this agent for further clinical development. These data may inform the design of future STEAP-1-directed ADCs in prostate cancer, as well as other therapeutic approaches targeting this antigen (e.g., T-cell engagers, chimeric antigen receptor T-cells).
ABBV-969 is another ADC designed to bind both PSMA and STEAP-1 expressed on prostate cancer cells and has a topoisomerase 1 inhibitor payload. 28 This drug is currently undergoing assessment in a phase I multicenter open-label dose escalation and dose expansion study in patients with mCRPC with disease progression on at least one prior ARPI and one taxane regimen. The primary objective of this study is to investigate the safety and tolerability of the drug. The trial is currently recruiting and is estimated to enroll around 140 patients (NCT06318273).
TROP-2-targeting ADCs
Sacituzumab govitecan, which is currently approved for the treatment of advanced breast cancer, is a TROP-2 targeting ADC that was tested in a phase II trial in patients with mCRPC previously treated with an ARPI and with or without prior chemotherapy exposure. 29 The primary endpoints of the trial were 6-month radiographic PFS and PSA50 response.
Of 30 patients enrolled in the trial, 27 were treated with single-agent sacituzumab govitecan, and 3 received sacituzumab govitecan with ARPI. 30 Neutropenia was the most common all-grade adverse event (80%) and was easily managed with granulocyte colony-stimulating factor (G-CSF) injections.
At the final analysis, the 6-month PFS rate was 52% in the overall cohort and 68% in chemotherapy-naïve patients, and no PSA50 responses were encountered. 30 Additional analyses of tissue and liquid biopsies are ongoing in an attempt to identify predictive biomarkers for sacituzumab govitecan in prostate cancer (NCT03725761).
Datopotamab deruxtecan is another TROP-2-targeting ADC undergoing assessment in a phase II multicenter study as a single agent and in combination with different drugs with a distinct mechanism of action in patients with advanced or metastatic solid tumors, having previously shown efficacy in patients with breast and lung cancer. 31 This drug consists of a humanized anti-TROP-2 monoclonal antibody linked to a topoisomerase-I inhibitor payload. In the substudy 3 of the trial, datopotamab deruxtecan will be assessed in biomarker-unselected patients with mCRPC with disease progression on an ARPI as a single agent in part A and in combination with AZD5305 (a poly[ADP] ribose polymerase [PARP]-1-selective inhibitor) in part B. 31 The primary endpoints are objective response rate (ORR) by RECIST v1.1 and the safety and tolerability of the drug across all the study arms, as well as the PSA50 response in substudy 3 (NCT05489211).
CD46-targeting ADCs
FOR46 (FG-3246) is a fully humanized antibody conjugated to the MMAE payload using a cleavable linker and designed to bind CD46, which is overexpressed in patients with mCRPC. This drug was evaluated in a phase I dose escalation/dose expansion study in patients with mCRPC previously treated with an ARPI but chemotherapy-naïve in the mCRPC setting. The primary objective was to determine the drug’s MTD. 32
Overall, 56 patients were enrolled in this trial, and the median number of lines of previous systemic therapy was 5 (range 2–14). The most frequently encountered any-grade TRAEs were infusion-related reaction (48.2%), followed by neutropenia (41.1%), and peripheral neuropathy (32.1%). The most common grade ⩾3 TRAEs were neutropenia (59%), followed by leukopenia (27%), and lymphopenia and anemia (7%, each). 32
Among 25 patients with RECIST-evaluable disease, the confirmed ORR was 20% (comprising five partial responders). Among 39 patients with PSA-evaluable disease, 36% experienced a PSA50 response (
Given the promising antitumor activity observed with FOR46 and the drug’s manageable safety profile, a randomized study is currently being designed using [89Zr]DFO-YS5 positron emission tomography (PET) as a potential predictive biomarker of response. This imaging approach enables the detection of CD46-expressing prostate adenocarcinoma and neuroendocrine tumors. 33
Nectin-4-targeting ADCs
Enfortumab vedotin (EV) is an ADC targeting Nectin-4, a cell surface adhesion protein, and linked to MMAE, a microtubule-disrupting agent. It was first approved by the FDA in 2019 as monotherapy for adults with locally advanced or metastatic urothelial carcinoma who had previously received both a programmed cell death protein-1 (PD-1)/programmed death-ligand 1 (PD-L1) inhibitor and platinum-based chemotherapy. 34 In 2023, it received expanded approval in combination with pembrolizumab in patients with locally advanced or metastatic urothelial carcinoma regardless of cisplatin eligibility. 35 In prostate cancer, a phase II study is currently investigating EV in a heavily pre-treated patient population with mCRPC. 36 Patients who have received prior docetaxel and at least one ARPI were eligible and enrolled in the study. Primary objectives are ORR in patients with measurable disease, or confirmed conversion of CTC to < 5/7.5 mL blood, or PSA decline of ⩾ 50%, and disease stability for ⩾ 6 months.
EV is given at 1.25 mg/kg IV on days 1, 8, and 15 of a 28-day cycle. In the first stage of this trial, 11 patients were enrolled, with 64% of patients meeting protocol-defined response criteria. The most common grade 3 or 4 TRAEs were fatigue, rash, anemia, and cytopenia, without grade 5 events observed. Stage II of this trial is ongoing, with a target enrollment of 34 patients (NCT04754191). 36
Tissue factor (TF)-targeting ADCs
Tisotumab vedotin is a TF-targeting ADC. Tissue factor is a transmembrane glycoprotein highly expressed in several solid tumors, including prostate cancer. This ADC is a fully human IgG1 monoclonal antibody that targets TF, binds to TF-expressing tumor cells, and releases MMAE intracellularly after internalization. It was approved in 2024 for patients with refractory or recurrent metastatic cervical cancer who progressed on or after chemotherapy. 37
In the context of prostate cancer, TF was explored in the InnovaTV 201 phase I/II trial involving patients with advanced solid tumors. 38 Patients received doses ranging from 0.3 to 2.2 mg/kg intravenously once every 3 weeks, with 2.0 mg/kg being used in the dose expansion phase. Eighteen patients with advanced or metastatic prostate cancer were included in the dose-expansion cohort. The safety profile was similar across tumor types, including epistaxis, fatigue, nausea, alopecia, conjunctivitis, and peripheral neuropathy. None of the patients experienced a radiographic or PSA response. 38 No dedicated prostate cancer-only phase II or III trials investigating tisotumab vedotin have been registered.
B7-H3-targeting ADCs
DB-1311/BNT324 is a B7-H3-targeted ADC that combines a monoclonal antibody that specifically binds to B7-H3 with a potent cytotoxic DNA topoisomerase 1.
39
B7-H3 is a cell surface protein highly expressed in mCRPC and associated with poor prognosis.
40
DB-1311/BNT324 received a Fast Track designation by the FDA. It was explored in a phase I/II trial in patients with advanced solid tumors, including patients with mCRPC. The primary endpoint was investigator-assessed ORR. Among 465 patients with advanced solid tumors, 73 patients had mCRPC and treated with DB-1311/BNT324. In this trial, among 52 patients with mCRPC post-docetaxel and hormonal therapy with measurable disease, the ORR was 42.3%, with 90.4% achieving disease control (partial response or stable disease). Median radiographic PFS (
Vobramitamab duocarmazine (vobra duo) is another ADC targeting B7-H3 combined with a duocarmycin-based DNA-alkylating payload. The TAMARACK phase II trial evaluated vobra duo in patients previously treated with at least one ARPI, up to three lines of therapy for mCRPC, and prior docetaxel was allowed. Patients were randomized to receive 2.0 mg/kg or 2.7 mg/kg every 4 weeks. 41 The primary endpoint was radiographic PFS. At data cutoff, the 6-month radiographic PFS landmark rate was 69% (2.0 mg/kg) and 70% (2.7 mg/kg), with median radiographic PFS estimates of 8.5 months (2.0 mg/kg) and 7.5 months (2.7 mg/kg). Confirmed objective response rates by RECIST were 20% and 41% in the respective arms. The safety profile was manageable with grade ⩾ 3 adverse events occurring in 46.7% of patients receiving 2.0 mg/kg versus 52.3% in patients receiving 2.7 mg/kg, including asthenia, edema, neutropenia, and stomatiti. 41
Ifinatamab deruxtecan (DS-7300), also known as I DXd, is a B7-H3 targeting ADC comprising a humanized anti-B7-H3 monoclonal antibody linked via a cleavable peptide to a potent DXd payload (a topoisomerase I inhibitor).
42
DS-7300 was explored in a phase I/II dose escalation study for advanced solid tumors, with patients receiving doses ranging from 0.8 to 16 mg/kg every 3 weeks, with 12 mg/kg selected for the expansion phase. A total of 55 patients with mCRPC were included. Among 29 patients with RECIST-evaluable disease, there were 9 partial responses and 12 stable disease cases, corresponding to a disease response rate of 72.4%. In addition, improvements in PSA and bone metastases were observed, including a PSA50 response rate of 21.1% (
Ifinatamab deruxtecan will be explored in the phase III IDeate Prostate01 trial (NCT06925737). It is a global, randomized, open-label phase III study now enrolling around 1440 patients with mCRPC whose disease has progressed after 1–2 ARPIs. Participants are randomized to receive either ifinatamab deruxtecan (12 mg/kg every 3 weeks) or standard docetaxel (75 mg/m² every 3 weeks) plus prednisone. The co-primary endpoints are OS and radiographic PFS, while secondary outcomes include response rates, time to PSA or pain progression, skeletal events, and safety.
DLL3-targeting ADCs
Rovalpituzumab tesirine is an ADC that targets delta-like protein 3 (DLL3), a Notch pathway ligand highly expressed on the surface of castration-resistant neuroendocrine prostate cancer cells. 43 Anti-DLL3 monoclonal antibody binds to DLL-3-expressing tumor cells, leading to internalization of the ADC. Once intracellular, the linker is cleaved by intracellular proteases, releasing cytotoxic PBD dimer payload, which induces DNA cross-linking and subsequent cell death. 43
Rovalpituzumab tesirine was previously investigated in multiple phase III trials in the setting of small cell lung cancer, but did not demonstrate a survival benefit. 44 Consequently, its clinical development has been discontinued and the drug did not receive regulatory approval.
In prostate cancer, a phase I/II trial enrolled patients with advanced DLL3-expressing solid tumors, including 21 patients with neuroendocrine prostate cancer. In this trial, patients received escalating doses, with the recommended phase dose of 0.3 mg/kg IV every 6 weeks for two cycles. Although the small cohort limited formal efficacy assessment in neuroendocrine prostate cancer, the agent showed a manageable toxicity profile. However, these limited early data have not translated into continued clinical investigation of DLL3-targeting ADCs in prostate cancer. 45
Limitations and future directions
Despite encouraging activity in prostate cancer, ADCs face several challenges that limit their clinical efficacy and broader applicability. One important challenge is the narrow therapeutic window observed in many ADCs targeting antigens like PSMA and TROP-2.22,30 Early ADCs, such as MLN2704 and PSMA-MMAE, showed significant off-target toxicities due to linker instability and premature payload release, which limited dose escalation and clinical outcomes.20,21 Even novel ADCs, while improved, often demonstrate dose-limiting toxicities such as peripheral neuropathy and neutropenia, restricting broader clinical adoption.
Another key limitation is the inherent heterogeneity of antigen expression in prostate tumors. Variability in antigen expression levels between patients and within individual tumor lesions can significantly impact the efficacy of ADCs. Drugs relying solely on antigen internalization may have reduced efficacy in tumors with low or variable antigen density. Although strategies such as “bystander killing” have been developed to mitigate antigen heterogeneity, the efficacy of this approach varies widely among different ADCs. 10 The development of resistance mechanisms further complicates the use of ADCs. Tumors may acquire resistance through alterations in antigen expression, mutations affecting internalization or lysosomal processing of ADCs, or increased activity of efflux pumps expelling cytotoxic payloads. 8
Ongoing studies are exploring strategies to overcome these limitations and optimize the role of ADCs in prostate cancer. These include the development of ADCs with highly potent and novel payloads, such as immune-stimulating agents, natural toxins, and radionuclides, to improve efficacy and specificity. 46 In addition, novel dual-cleavable linkers that preferentially release payloads within tumor tissue have shown reduced off-target toxicity and improved efficacy in early-phase trials, as demonstrated by agents such as FOR46. 32
Innovative delivery mechanisms are being explored to overcome the inherent limitations of single-antigen targeting. Bispecific ADCs, which simultaneously target two different antigens, represent a promising approach to address antigen heterogeneity and enhance tumor selectivity. 47 For instance, dual PSMA/STEAP-1 targeting ADCs, such as ABBV-969, are designed to maintain efficacy even when individual antigen expression varies within the tumor microenvironment. 28 In addition, the incorporation of Proteolysis Targeting Chimeras (PROTACs) into ADC technology represents an emerging therapeutic modality that leverages targeted protein degradation rather than traditional cytotoxic mechanisms. 46 These drug conjugates facilitate the selective elimination of oncogenic proteins crucial for cancer cell viability. These next-generation approaches may address current limitations of single-target ADCs and provide more robust therapeutic responses in the heterogeneous landscape of advanced prostate cancer.
Beyond improving ADC design, combination strategies are also being evaluated to enhance efficacy and delay the development of resistance. For example, a phase Ib/II study is currently investigating the combination of sacituzumab govitecan with two immune checkpoint modulators, etrumadenant (A2A adenosine receptor antagonist) and zimberelimab (PD-1 inhibitor), in patients with mCRPC (NCT04381832). Another phase I/Ib dose escalation and cohort expansion study is exploring the B7-H3-targeted ADC vobramitamab duocarmazine (MGC018) combined with lorigerlimab (dual PD-1 and cytotoxic T-lymphocyte-associated protein 4 [CTLA-4] inhibitor) in patients with advanced solid tumors (NCT05293496).
Although prostate cancer was not included in the DESTINY PanTumor trials, HER2-targeted ADCs may represent a future therapeutic option in this disease. 48 HER2 expression has been reported in prostate cancer, with higher prevalence in advanced and metastatic castration-resistant disease.49,50 Trastuzumab deruxtecan (T-DXd), a tumor-agnostic HER2-directed ADC, has demonstrated broad antitumor activity, and recent case-level reports described responses in patients with mCRPC. 51 An ongoing phase II trial (NCT06610825) is currently enrolling patients to evaluate the efficacy and safety of HER2-directed ADC in HER-2 positive mCRPC.
Moving forward, biomarker-driven patient selection will be essential for optimizing ADC therapy in prostate cancer. Integration of molecular profiling with advanced imaging modalities, such as PET using antigen-specific radiotracers, can significantly refine patient selection strategies. For instance, [89Zr]DFO-YS5 PET imaging can serve as a predictive biomarker, enabling detection of CD46-expressing tumors and monitoring of treatment resistance. 33 Collectively, these integrated strategies have the potential to maximize therapeutic efficacy, minimize toxicity, and establish ADCs as a standard therapeutic option in the management of prostate cancer.
Conclusion
Antibody-drug conjugates are emerging as a promising addition to the therapeutic armamentarium of advanced prostate cancer. Although early development was limited by toxicity and modest efficacy, improvements in target selection, linker stability, and payload potency have produced more effective and better-tolerated agents. Recent clinical data show encouraging activity, particularly with novel agents such as FOR46 and DB-1311/BNT324. The integration of biomarker-driven patient selection and advanced imaging approaches promises to further optimize therapeutic outcomes. Ongoing trials combining ADCs with ARPIs, PARP inhibitors, or immune-targeted therapies may help overcome resistance and expand the clinical utility of ADCs.
Footnotes
Acknowledgements
None
