Abstract
Background:
The aim of this work was to to evaluate the incidence and risk factors of adverse events (AEs), focusing on cardiovascular events (CVEs) and hypokalemia, in patients treated with abiraterone acetate (AA) and prednisone (PDN) outside clinical trials, and their association with survival outcomes.
Methods:
This was a retrospective cohort study of 105 patients treated from 2011 to 2016. Incidence of AEs was descriptively summarized in the whole cohort and by subgroup (pre-
Results:
Overall, median PFS and OS were 14.9 and 24.6 months, respectively. Prostate-specific antigen (PSA) ⩾ 10 ng/ml (
Conclusions:
Median PFS and OS estimates and incidence of CVEs and hypokalemia in our series are consistent with those of pivotal trials of AA plus PDN, confirming the efficacy and safety of this regimen also in the real-world setting. Elderly patients have higher odds of developing/worsening CVEs. However, regardless of age, CVEs were not associated with worse outcomes. Treatment-related hypokalemia seemed to be associated with longer OS, albeit this finding needs confirmation within larger, prospective series.
Keywords
Introduction
Prostate cancer is the most common malignancy in men in western countries. 1 Androgen deprivation therapy (ADT) represents the cornerstone treatment for metastatic prostate cancer and indeed most patients do benefit from ADT. However, most of them are destined to progress and to become castration-resistant. 2 Median survival of metastatic castration-resistant prostate cancer (mCRPC) is highly variable and expected survival among these patients is associated with tumor bulk and disease spread to distant sites other than the skeleton. 3 Over the past few years, the increasing knowledge about the driving role of the androgen receptor, even in the castration-resistant setting, has dramatically improved survival for patients developing mCRPC. 4 In particular, based on this assumption, new endocrine therapies, such as abiraterone acetate (AA) and enzalutamide, have been developed. 4
Abiraterone acetate is a potent, selective inhibitor of steroidogenesis that interferes with androgen synthesis through the inhibition of the cytochrome P450 17α-hydroxylase/17, 20 lyase (CYP17). This enzyme catalyzes two sequential reactions, namely the conversion of pregnenolone and progesterone to their 17α-hydroxy-derivatives (17α-hydroxylase activity), and the subsequent formation of dehydro-epiandrosterone (DHEA) and androstenedione (17, 20-lyase activity). Androstenedione and DHEA are then converted to testosterone (TST) by 17-beta-hydroxy-steroid-dehydrogenase and eventually to dihydrotestosterone (DHT) by 5α reductase. 5 Thus, the inhibition of CYP17 decreases circulating and tissue levels of androgens, namely DHEA, TST and DHT, preventing the stimulation of androgen-sensitive neoplastic cell clones. The blockade of steroidogenic enzymes by AA leads to the suppression of adrenocorticotropic feedback, which results in an exceeding production of steroid precursors with high mineralocorticoid activity. 5 This effect is responsible for the most frequent adverse events (AEs) occurring during treatment with AA, including hypokalemia, hypertension and fluid overload. Therefore, the concomitant use of low-dose glucocorticoids is required to decrease the frequency and severity of these events and serial monitoring of potassium levels and blood pressure is recommended.6,7
AA plus low-dose prednisone (PDN) was first approved for patients with mCRPC failing prior docetaxel (DX) chemotherapy (CT), based on the COUAA-301 trial results, showing AA plus PDN to be able to improve overall survival (OS) compared with PDN plus placebo [14.8
Despite the widespread use of AA in clinical practice and according to the major guidelines,10–12 few data on the incidence of CV events in ‘real-life’ patients treated with AA and PDN are available, mostly regarding patients failing prior treatment with DX. In the Italian Named Patient Program, the clinical outcomes reported in the AA pivotal trial 7 were reproduced but the incidence of toxicities was lower (hypertension was observed only in 2.6% and hypokalemia only in 1.9% of treated patients). 13 This difference probably reflects lack of systematic monitoring of patients in this clinical practice setting. One single-institution retrospective study included 51 CRPC patients with concomitant CV risk factors, who were also pretreated with DX. Hypertension was observed in 16% while fluid retention in 18% of patients. Moreover, dose reductions due to unacceptable toxicity were necessary in 9.8% of patients. This study confirms that AA plus PDN can be safely delivered, even to patients bearing risk factors for CV diseases. 14 A data update of the same study, with longer follow-up time, confirmed the previously reported safety profile. 15 The results of an additional study, also including mCRPC patients previously treated with DX, were presented at the 2016 American Society of Clinical Oncology Annual Meeting. 16 This was a prospective study specifically aimed at monitoring cardiac functionality during treatment with AA and PDN. A total of 87 patients underwent electrocardiogram (ECG) and echocardiography with evaluation of left ventricular ejection fraction (LVEF) and diastolic function, both at baseline and at every 6 months until treatment discontinuation. Hypertension was observed in 34.6% of patients, without statistically significant variation of LVEF and diastolic function.
More recently, the results of two trials exploring the efficacy of AA and PDN (5 mg daily) in hormone-naïve patients were reported.17,18 In both trials the combination of AA plus PDN with ADT was compared with ADT alone, and both ones reported statistically significant and comparable reductions in the HR of all-cause mortality, the primary endpoint for both studies (LATITUDE trial: HR 0.62, 95% CI 0.51–0.76,
In view of these premises, we report here the results of a retrospective study evaluating patients affected by mCRPC, who were referred to our unit and treated with AA and PDN, outside clinical trials. Defining the incidence of AEs, with a special focus on CV and biochemical events (including hypokalemia) in a ‘real-life’ mono-institutional patient population, looking for the factors putatively predisposing to them, and evaluating the impact, if any, of CV and biochemical events on patients outcome were study aims. The putative association between baseline clinical–pathological characteristics, the incidence of AEs and patient clinical outcome was also investigated, in the attempt to identify
Patients and methods
We reviewed the charts of 105 patients affected by mCRPC and consecutively referred to our unit to receive AA and PDN between June 2011 and 2016. Ethics approval and consequent informed consent were not required for this study, according to ‘Authorization n. 9/2016 – General Authorization for the Processing of Personal Data for Scientific Research Purposes – 15 December 2016’ (Published in
The following information, recorded at the time AA was started, was collected for each patient: age, Eastern Cooperative Oncology Group (ECOG) performance status (PS), body mass index (BMI), presence or absence of pain (evaluated through the Brief Pain Inventory Short Form scale), 21 prostate-specific antigen (PSA) value, type of metastatic sites involved, time on previous ADT, and Gleason score (GS) of primary tumor. Pre-existing cardiac events or CV comorbidities were also recorded. Baseline ECG findings and LVEF were available for all patients, since cardiologic examination before starting treatment with AA and PDN is required by the Italian Drug Agency (AIFA).
All the patients included in the study cohort received AA orally, 1000 mg daily, and PDN, 5 mg twice daily, until disease progression, symptomatic deterioration, or unacceptable toxicity. Pharmacological suppression of gonadal function was maintained in all patients. The incidence of fluid retention, hypertension, hypokalemia, CV disorders, AST-ALT increase, diabetes and hypercholesterolemia were annotated and scored using Common Terminology Criteria for Adverse Events, version 4.0. 22 AE assessment was performed 2 weeks after the beginning of AA plus PDN and at monthly intervals thereafter. In patients at higher risk of developing cardiovascular events (CVEs) and with a baseline LVEF < 50%, assessment was performed every 2 weeks for the first 3 months. Disease progression was defined as per the Response Evaluation Criteria In Solid Tumors (RECIST) criteria, version 1.1 23 and Prostate Cancer Clinical Trials Working Group 2 criteria. 24 Radiographic assessments were performed every 6 months unless required before due to biochemical progression (as defined by an increase in PSA level of 25% or more in respect to nadir value) or clinical deterioration. The date of disease progression was recorded as well as the date of death. The cause of death was recorded whenever we were able to retrieve it. PFS was defined as the time between the initiation of AA plus PDN and the date of disease progression. OS was defined as the time between the initiation of AA plus PDN and the date of death.
Statistical analysis
All the patients treated with AA and PDN at our unit between 2011 and 2016 were included in the analysis.
Patient characteristics, the incidence of AEs (namely of CV and biochemical events) in the overall cohort and by subgroup, the association between patient characteristics, the incidence of AEs and patient outcome were descriptively summarized. The predictive value of AEs on treatment outcome was investigated by comparing outcome indicators in patients who developed CV and biochemical events (namely hypokalemia) and in those who did not.
OS and PFS curves were obtained using the Kaplan–Meier product-limit estimator and compared using the log-rank test.
25
To evaluate the possible interactions among aforementioned study variables with clinical outcomes, multi-parametric, Cox proportional hazard models were used.
26
We obtained separate models for PFS and OS, adjusting for all the covariates that predicted, after univariate analysis, for PFS or OS, as appropriate. The following covariates were included in the PFS models: baseline PSA level (<10
Sample size calculation
No formal calculation of sample size was performed in this retrospective series, as it would not be possible, anyway, to change cohort size since it included all the patients treated with AA and PDN followed at our unit in the time period mentioned above. Moreover, as we have already pointed out in the premises, we intended to perform an explorative study, just to look at the incidence and different distribution of patient features, outcome indicators and AEs (namely CV and hypokalemia) in our cohort, including real-life patients treated outside of clinical trials.
Results
Cohort characteristics
As already mentioned, 105 consecutive mCRPC patients treated with AA plus PDN were included in the cohort: 75 had been previously treated with DX while 30 had received AA and PDN as front-line treatment. Median follow up was 26.8 (range 1.4–57.4) months for the whole cohort, 20.9 months (range 3.3–28.4) months for CT-naïve patients and 32.6 months (range 1.4–57.4) for patients previously treated with DX. Patient characteristics are summarized in Table 1. Characteristics of pre- and post-DX patients were comparable. Table 2 shows the prevalence of pre-existing CV disorders and of other conditions that might affect patient survival and treatment safety. Before starting treatment with AA and PDN, most of the patients were affected by hypertension (62.9%) and about half of them was overweight or obese (47.6%). However, again there were no major differences between subgroups.
Main characteristic of study patients at AA plus PDN start (disease related).
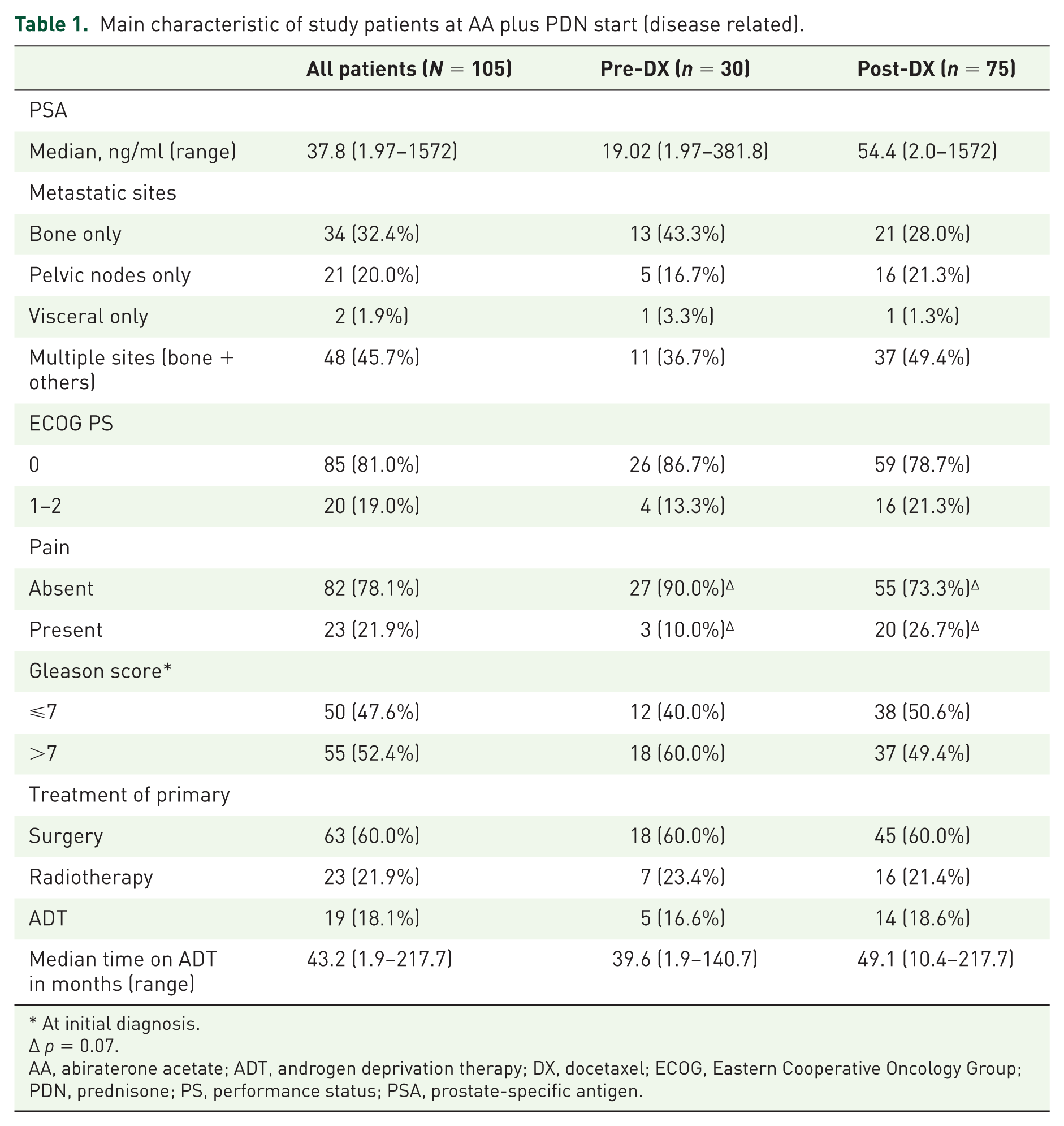
At initial diagnosis.
AA, abiraterone acetate; ADT, androgen deprivation therapy; DX, docetaxel; ECOG, Eastern Cooperative Oncology Group; PDN, prednisone; PS, performance status; PSA, prostate-specific antigen.
Main characteristic of study patients at AA plus PDN start (patient related).

AA, abiraterone acetate; BMI, body mass index; CV, cardiovascular; DX, docetaxel; LVEF, left ventricular ejection fraction; PDN, prednisone.
Efficacy data
Median PFS of the whole cohort was 14.9 months (range 1.4–45.7); as expected, PFS was longer in CT-naïve than in post-DX patients: 20.9 (range 1.8–28.4)

PFS and OS curves in all cohort patients and subgroups.
Multivariable analyses of PFS and OS.

Variables not achieving the statistical significance after univariate analysis were not included into the models.
ADT, androgen deprivation therapy; BMI, body mass index; CI, confidence interval; DX, docetaxel; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; OS, overall survival; PFS, progression-free survival; PS, performance status; PSA, prostate-specific antigen.
Presence of pain (
Multivariable analysis of competitive risks of mortality (total patients:

ADT, androgen deprivation therapy; BMI, body mass index; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; PS, performance status; PSA, prostate-specific antigen.
By contrast, all the variables on study, except for the presence of pain, were independently associated with prostate cancer-specific mortality, particularly BMI (
Safety data
Table 5 reports the incidence of AEs developed over the course of treatment in the whole series and in subgroups. As expected, hypertension (17.2%) and hypokalemia (16.2%) were the more commonly observed events, the incidence of both being more elevated in post-DX patients as compared with CT-naïve patients. All the other CV events, including fluid retention and cardiac disorders, were described in <10% of patients (fluid retention in 5/105 patients and cardiac disorders in 9/105 patients). No statistically significant reduction in LVEF compared with baseline was recorded. Worsening of hypertension or of pre-existing cardiac disorders occurred in 16.7% (from G1 to G2 in 9.1% and from G1–G2 to G3–G4 in 7.6%) and 11.1% (from G1 to G2 in 8.3% and from G1–G2 to G3–G4 in 2.8%) of patients, respectively. While 21% of patients affected by diabetes developed further worsening of tolerance to glucose, only 3.6% of patients affected by dyslipidemia showed worsening of this condition.
Patients developing at least one AE during treatment.
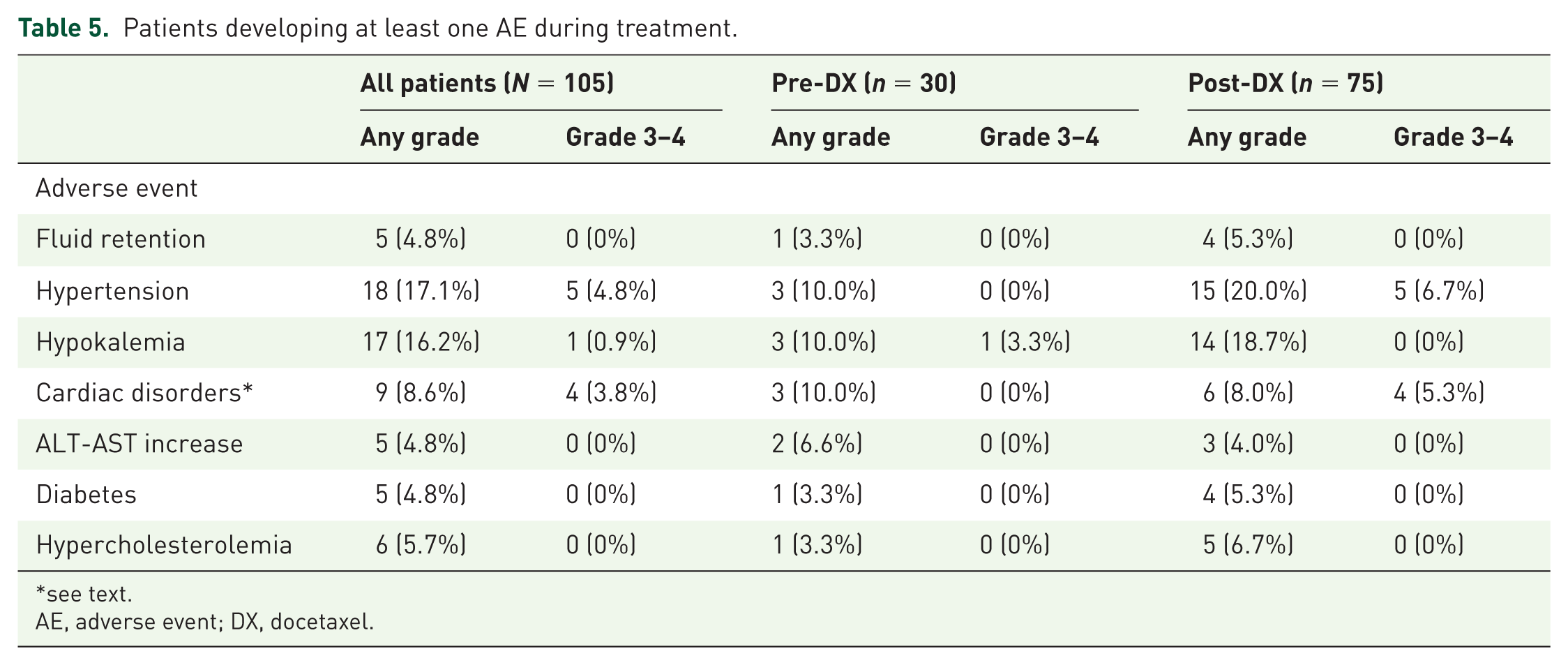
see text.
AE, adverse event; DX, docetaxel.
Presence of baseline hypertension and cardiac disorders was not associated with the development of new hypertensive episodes or cardiac disorders. Conversely, pre-existing diabetes was significantly associated with worsening of glycemic control (
Incidence of CV, biochemical and metabolic events during treatment according to age, BMI, ECOG PS and LVEF.
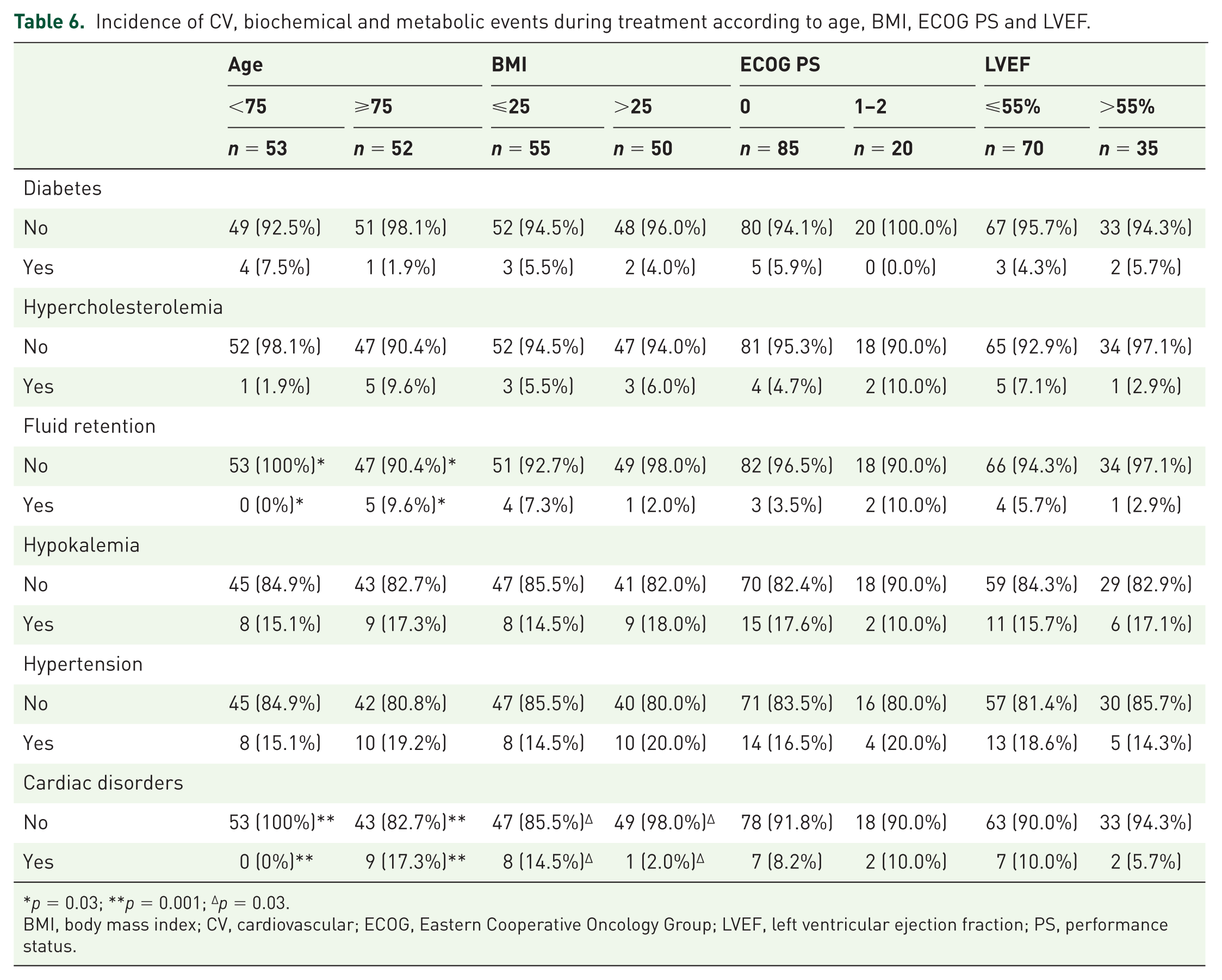
BMI, body mass index; CV, cardiovascular; ECOG, Eastern Cooperative Oncology Group; LVEF, left ventricular ejection fraction; PS, performance status.
No association between the development of cardiac AEs and PFS or OS duration was observed (Table 7). In particular, patients developing one or more of such events did not appear to progress or to die earlier. No association between hypokalemia and PFS was observed, though a trend was found in favor of patients who developed hypokalemia. However, patients developing hypokalemia appeared to live significantly longer (
PFS and OS as a function of the incidence of all AEs and of CV and biochemical events.

AE, adverse event; CI, confidence interval; CV, cardiovascular; HR, hazard ratio; OS, overall survival; PFS, progression-free survival.
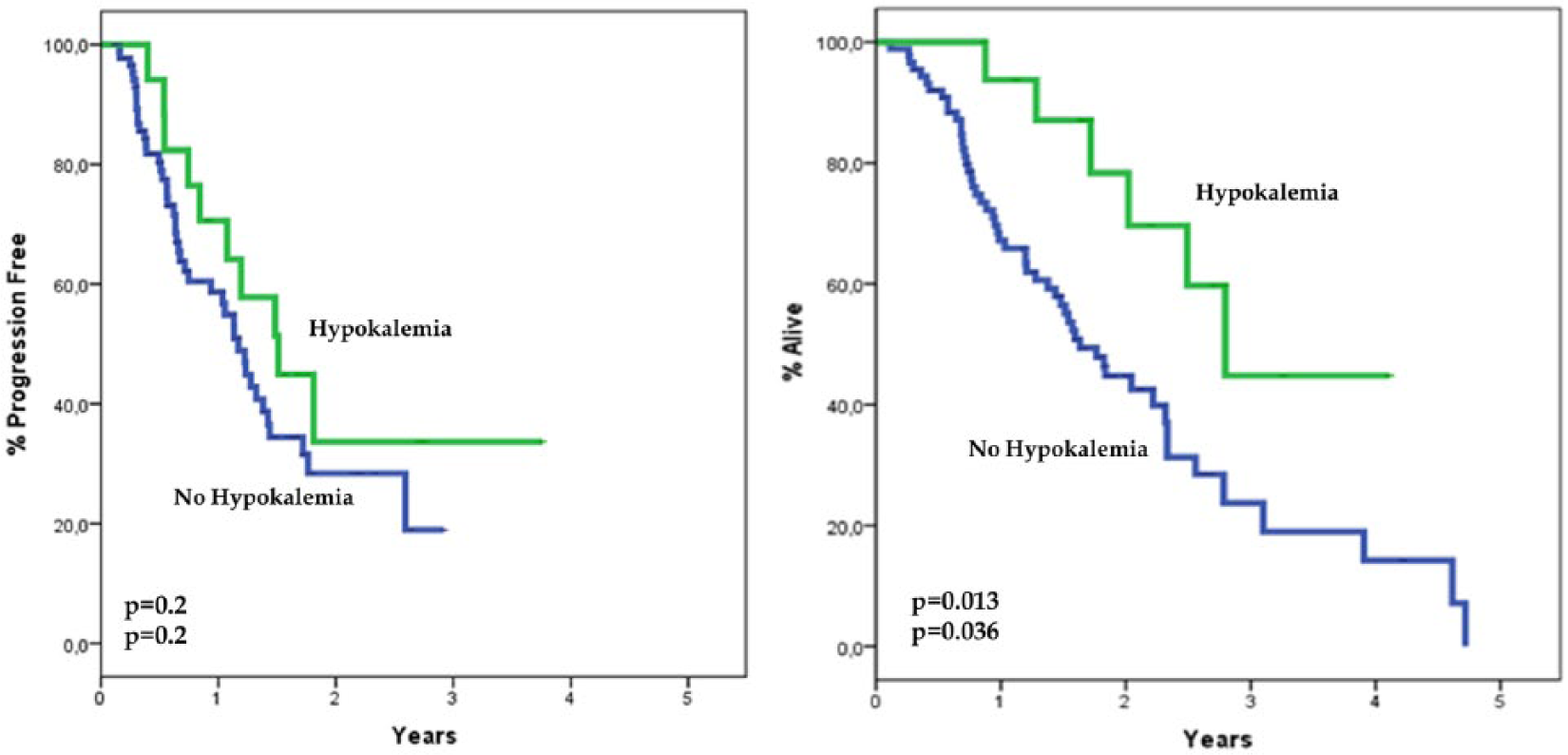
PFS and OS curves in patients who developed hypokalemia and in those who did not.
Discussion
In this retrospective analysis, we report our experience about the efficacy and safety of AA plus PDN in mCRPC patients, either CT-naïve or previously treated with DX, focusing on CV, biochemical (i.e. hypokalemia) and metabolic events. Unfortunately, our study is retrospective, includes a relatively small number of patients, which implies a low statistical power, and is unbalanced relative to the proportion of patients receiving AA plus PDN after DX failure compared with the proportion of those receiving these drugs as their front-line treatment. Moreover, observation times recorded in the two groups also differed. Both of the mentioned disproportions reflect the fact that AA was first licensed for the post-DX setting and that it obtained the approval for the management of CT-naïve patients only subsequently. All these factors might bias findings both in the whole cohort and by subgroup, and limit both direct comparisons between subgroups and indirect comparisons with the COU-AA 301 and 302 trial findings. Nevertheless, we believe that our study provides some interesting information regarding the efficacy and safety of AA plus PDN in ‘real-life’ mCRPC patients, which can prove useful in every day practice.
Efficacy data
Overall, the efficacy results obtained in our series are comparable with those achieved in the two pivotal trials, which contributed to drug approval. Indeed, in post-DX patients, we observed a median PFS of 13.8 months and a median OS of 19.9 months. In the COUAA-301 trial, which comparably accrued post-DX patients, the median duration of PFS and of OS were 5.6 and 15.8 months respectively. The better outcome recorded in our series is probably due to the different selection criteria [patients enrolled in the COUAA-301 trial were younger, had a higher tumor burden (median PSA level was 128.8
In the COUAA-301 and COUAA-302 trials, clinical benefits achieved by AA and PDN compared with PDN and placebo were obtained in all patient subgroups. However, these trials were not designed
In order to answer to this question, we analyzed our PFS and OS results as a function of a number of variables which have been previously shown to significantly predict for patient clinical outcome in mCRPC. 31 In our analysis, we also included two additional variables: BMI and duration of prior ADT. BMI was included because it was shown that an elevated BMI is associated both with an increased risk of cancer-specific mortality in healthy people and with a higher risk of biochemical recurrence in prostate cancer patients. 32 A trend for an increased risk of progression to CRPC was also observed in patients with a BMI > 25. 33 An association between obesity and the aggressiveness of the disease, as well as a higher incidence of complications following ADT, was observed in another study. 34 Different mechanisms, involving the insulin/IGF-1 axis, sex hormones and adipokine signaling, have been proposed to explain the association between obesity and aggressiveness of prostate cancer. 35 Addressing this issue in detail was not the scope of the present study. However, two additional considerations support our choice of including BMI among the selected covariates: (1) the metabolic dysfunction caused by ADT can accelerate CRPC and increase the risk of CV events; (2) human adipose tissue has been shown to be capable of active androgen synthesis and this, in principle, might interfere with the therapeutic activity of AA. 36
Duration of prior ADT has been associated with longer survival in patients receiving AA in a previous study. 37 In our study both BMI and the duration of previous ADT were confirmed to be associated with a longer PFS and OS after multivariable analysis. In particular, overweight patients showed a significantly increased risk both to progress and to die, which was even higher for CT-naïve patients. These findings should be interpreted with caution, due to the small numbers and the retrospective nature of our analysis. However, multivariable competitive risks analysis showed that a BMI > 25 was specifically associated with a higher risk of dying for prostate cancer, but it showed no association with prostate cancer-unrelated mortality, independently of the other covariates included in the model. Of course, these findings might deserve confirmation in larger series. As shown in Table 3, we also confirmed that a longer duration of prior ADT almost halved the risk of progression and of death. In contrast to what we expected, visceral metastases were not significantly associated with prognosis in our cohort, but this finding might be due to the limited number of patients with baseline visceral involvement. Duration of response to previous ADT did also predict for prostate cancer mortality in the competing risk model. This and the borderline predictive value of PSA level, confirm that tumor burden and biology are specific determinants of AA efficacy in CRPC. Taken all together, our data suggest that patients with a lower PSA level, an initial GS ⩽ 7, an ECOG PS 0, no pain, a duration of ADT > 43.2 months and a BMI ⩽ 25 are likely to benefit the most from AA plus PDN treatment, independently of their age, the presence of visceral metastasis and of having been previously treated or not with DX.
Safety data
AA plus PDN proved to be a well tolerated regimen among our cohort. In particular, focusing on CV and biochemical events, in CT-naïve patients we observed a lower incidence of AEs as compared with that reported in the COUAA-302 trial8,9 and only one episode of grade 3–4 hypokalemia. In the post-DX setting, we observed a higher incidence of hypertension (20%
Conclusion
Our findings confirm that AA and PDN is an effective and well tolerated regimen also in ‘real-life’ patients, including elderly ones. Though the incidence of AEs, and especially CVEs, was lower with respect to the figures initially reported in pivotal trials, and though no evidence emerged that AEs might imply a worse clinical outcome, appropriate patient selection and monitoring is recommended. Lower PSA levels, an initial GS ⩽ 7, ECOG PS 0, absence of pain, longer duration of previous ADT manipulations and a BMI ⩽ 25 appear to be associated with a greater benefit from AA treatment, independently of patient age, presence of visceral metastasis and of patients having been previously treated or not with DX. Interestingly, patients developing treatment-related hypokalemia seem to have better outcomes as compared with those who did not. However, this finding requires confirmation in larger, possibly prospective trials. Studies focused on more specific markers able to predict for the effectiveness and safety of AA and PDN are needed in order to improve decision-making and patterns of care among patients with mCRPC. Similar studies are also warranted in hormone-naïve patients in view of the results achieved by adding upfront AA and PDN to standard ADT.
Footnotes
Acknowledgements
The authors are extremely grateful to Dr Claudia Casella (Descriptive Epidemiology Unit: Tumor Registry, San Martino Polyclinic Hospital, Institute for Cancer Research and Treatment, Genoa, Italy) for helping in data collection.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
