Abstract
Background:
The survival status of patients with breast cancer and brain metastasis (BCBM) receiving current treatments is poor.
Method:
We designed a real-world study to investigate using patients’ clinical and genetic aberrations to forecast the prognoses of BCBM patients. We recruited 146 BCBM patients and analyzed their clinical features to evaluate the overall survival (OS). For genetic testing, 30 BCBM and 165 non-brain-metastatic (BM) metastatic breast cancer (MBC) patients from Hunan Cancer Hospital, and 86 BCBM and 1416 non-BM MBC patients from the Geneplus database who received circulating tumor DNA testing, were compared and analyzed.
Results:
Ki67 >14% and >3 metastatic brain tumors were significant risk factors associated with poor OS, while chemotherapy and brain radiotherapy were beneficial factors for better OS. Compared with non-BM MBC patients, BCBM patients had more fibroblast growth factor receptor (
Conclusion:
A group of genetic events, including
Background
Breast cancer (BC) is the most common malignancy in females. Metastases to the brain occurs in 10–16% patients with BC.1,2 Compared with hormone receptor (HR)-positive BCs, which are more likely to recur in bone, triple-negative BCs (TNBC) and HER-2 positive BCs more commonly recur in the brain. 3 BC with brain metastasis (BCBM) is a devastating cause of morbidity and mortality. The mean interval time from primary BC diagnosis to BCBM existence is about 35 months. The two main risk factors for developing BCBM are large primary tumor size and lymph node metastasis. 4 Clinically, brain metastases are treated with surgery, radiation therapy [whole brain radiation therapy (WBRT)] or stereotactic radiosurgery. In practice, no uniformly standard chemotherapy drugs are available for BCBM; in the National Comprehensive Cancer Center guideline, the category 2A recommendations include high-dose methotrexate, capecitabine, temozolomide monotherapy and cisplatin plus etoposide. 5 Despite a lack of consensus, preliminary data suggests that chemotherapy and targeting therapies after WBRT may improve survival outcomes. 6
The majority of BCBM patients receive a multimodality therapy approach, including anti
Magnetic resonance imaging (MRI) is a sensitive imaging tool which is widely used in the detection, treatment planning, and follow-up of BCBM. Circulating tumor DNA (ctDNA) testing is promising regarding to tumor treatment response and progression.
19
Derived from cell-free DNA (cfDNA), ctDNA analysis is a powerful surveillance tool for effective and continuous detection of potential tumor-related gene mutations.20–23 Compared with imaging and serum biomarkers, ctDNA testing provides valuable and sensitive information about gene mutations in tumors following drug-based therapies.
24
ctDNA does provide valuable information, but ctDNA is less likely to be informative about brain disease than extra-cranial disease.
25
However, metastatic BC (MBC) is a systematic disease, and often times BC patients develop brain metastases after metastases have appeared systemically in the lung, liver, and/or bone.
26
Our previous study reports a ctDNA gene mutation profile in MBC patients.
24
Thymidine phosphorylase (
In this study, we recruited 146 BCBM patients in our single center and retrospectively assessed the clinical risk factors. In addition, by using ctDNA testing, we examined the genetic risk factor in 195 single-center cases and in 1501 cases from Geneplus database.
Methods
BCBM patient cohort and data flow
The study was approved by the Ethics Committee at Hunan Cancer Hospital, the Affiliated Cancer Hospital of Xiangya School of Medicine/Central South University. As shown in Online Supplemental Figure S1 data flow, a total of 146 BCBM patients were enrolled in this study. Inclusion criteria were as follows: (a) CT/MRI confirmed BCBM; (b) tolerable to chemotherapy or target therapy with normal heart, liver, and renal function. Basic demographic and clinical information includes age of primary BC diagnosis, age of BCBM diagnosis, time-to-brain metastasis (TTB, from primary BC diagnosis to BCBM diagnosis), primary BC laterality, HR/HER2 status, TNM (T, primary tumor; N, nodal involvement; M, distant metastasis) stage of the primary BC at diagnosis, treatment history before BCBM diagnosis, treatment after BCBM diagnosis, and other metastatic sites.
Sample collection and plasma ctDNA testing
To investigate the genetic risk factor, plasma ctDNA test was performed. As shown in Figure S1, a total of 195 MBC patients received plasma ctDNA testing at the Department of Breast Cancer Medical Oncology in Hunan Cancer Hospital. Other inclusion criteria included: (a) ⩾1 measurable metastatic lesion; (b) age 18–70 years old; (c) normal liver and renal function (aspartate aminotransferase (AST) and alanine aminotransferase (ALT) > 0.5 upper limit of normal value (ULN), total bilirubin (TBIL) < 1.5 ULN, and creatinin (Cr) < 1.0 ULN); (d) routine-blood test: neutrophil count >2.0 × 109/l, Hb >11 g/l, platelet count >100 × 109/l. The exclusion criteria were: (a) serious heart disease; (b) multiple primary malignancies; (c) immune-deficiency; (d) organ-transplantation history. The plasma ctDNA testing study was also approved by the Ethics Committee of Hunan Cancer Hospital. Of these 195 patients, 30 were BCBM patients as described above, whereas the other 165 were non-brain-metastatic (BM) MBC patients. To compare the different genetic aberrations between BCBM patients and non-BM MBC patients, 1:1 propensity score matching (PSM) was performed to reduce the potential bias between these two subgroups. Propensity scores were calculated through logistic regression with covariates including age, treatment history, and metastatic disease (lung, liver, bone, and lymph node metastases) and recurrent disease. After PSM, 28 BCBM patients and 28 non-BM MBC patients were selected for genetic aberration comparison (heatmap).
From 2016, the Geneplus database pool collected 1501 BC samples; among these BC patients, 86 were BCBM patients. We collected the peripheral blood samples from the patients, and then extracted the DNA content according to the protocol. 24 We sequenced the genomic DNA (gDNA) and used the gDNA sequence as the control.
Target capture, next-generation sequencing, and data analysis
Sequencing libraries of ctDNA were prepared using the DNA Library Preparation Kit for Illumina (New England Biolabs, Ipswich, MA, USA). Custom biotinylated oligonucleotide probes (IDT, Coralville, IA, USA) covering the exons of 1021 genes that are highly mutated in 12 common solid tumors were used for hybrid capture, as described previously. 31 The Illumina HiSeq 3000 Sequencing System (Illumina, San Diego, CA, USA) was used for DNA sequencing with a 2 × 101-bp paired-end strategy, as described previously. 24
Terminal adaptor sequences were removed from the raw sequencing data. Subsequently, reads with more than 50% low-quality bases, or more than 50% undefined bases, were discarded. The remaining reads were mapped to the reference human genome (hg19) using the Burrows-Wheel Aligner with default parameters. Picard’s Mark Duplicates tool (version 1.98) was used to identify duplicate reads. Local realignment and quality recalibration were performed using the Gene Analysis Toolkit. Single-nucleotide variants and small insertions and deletions were called using the MuTect2 algorithm, and further filtration and validation was performed according to established criteria. 31 The Contra algorithm was used to identify somatic copy-number alterations defined using the ratio between the adjusted depths of ctDNA and control gDNA. After automatic calling, candidate variants were manually validated using an online visualization tool.
ctDNA gene aberration frequency
Total cfDNA included ctDNA and other normal cfDNA. Aberrations in ctDNA were identified by comparing the reference genome (hg19) and gDNA. The ctDNA aberration frequency was defined as the proportion of ctDNA gene aberrations in the total cfDNA.
Statistical analyses
Categorical variables were recorded as counts with percentage, and continuous variables were calculated as the mean with standard deviation and median with interquartile range. Chi-square tests were used to compare categorical variables between subgroups. When the sample size was less than five in the comparing subgroup, Fisher’s exact test were applied. Mentel–Haenszel chi-square tests were used when the number of compared subgroups was more than two. To compare continuous variables with symmetrical distributions across subgroups, an analysis of variance test was used. Both the univariate and multivariate Cox proportional hazards regression analyses were used to evaluate the prognostic factors for OS. Log rank tests and drawn Kaplan–Meier curves were also performed to assess the overall survival OS.
To search for significant ctDNA aberrations for BCBM, R package “ComplexHeatmap” was applied to rank the hot genetic aberrations in PSM BCBM and non-BM MBC subgroups. The top aberrations were put into a logistic model for BM prediction. A receiver operating characteristic (ROC) curve was drawn to determine the areas under the ROC curve (AUC) for BM. All statistical analyses were conducted by using SAS 9.4 or R 3.6.0 software. All tests of hypotheses were two-tailed and conducted at a significance level of 0.05 and at a marginal significant level of 0.15.
Results
Clinical risk factors for poor prognosis in BCBM patients
For the 146 recruited BCBM patients, the mean age at BC first diagnosis was 45.30 (±9.80) years old and the mean age at BCBM diagnosis was 48.48 (±9.64) years old. The mean TTB was 3.18 (±2.98) years (Online Supplemental Table S1). A total of 64.83% BCBM patients had bone metastasis and 76.56% BCBM patients had visceral metastasis. Both univariate and multivariate Cox regression analyses were performed to evaluate the potential clinical risk factors for poor OS in BCBM patients. Ki67 > 14% and metastatic brain tumors >3 were significant risk factor for OS in both univariate and multivariate Cox regression analyses (Table 1). Brain radiotherapy and chemotherapy after BM diagnosis were significant beneficial factors for longer OS in both univariate and multivariate Cox regression analyses. In multivariate Cox regression analysis, HER2-positive was a marginally significant favorable factor for OS (
Univariate and multivariate analysis of the clinical risk factors for OS.

Age at diagnosis was evaluated by using univariate Cox regression; but this variable did not exist in the multivariate model because age at brain metastasis and TTB were analyzed in a multivariate model (Age at diagnosis=Age at brain metastasis-TTB).
TTB indicated the time from diagnosis of BC to the diagnosis time of brain metastasis.
Capecitabine usage represents the capecitabine usage history before or after BCBM diagnosis.
Other metastasis indicated the metastatic sites other than brain.
BC, breast cancer; BCBM, breast cancer and brain metastasis; CI, confidence interval; ER, estrogen receptor; HER2, human epidermal growth factor receptor-2; PR, progesterone receptor; TTB, time-to-brain metastasis.
Genetic aberration of BCBM patients
BCBM group and non-BM MBC group had distinct ctDNA aberration patterns (Figure 1). BCBM patients had a significantly higher

Circulating tumor DNA gene mutation profiles for matched 28 brain metastatic breast cancer (BC) patients (a) and 28 non-brain-metastatic metastatic BC patients (b).
Compared with non-BM MBC patients, more BCBM patients were HER2-positive (40%
Differences of metastases/recurrence between FGFR-aberrant group and wild-type FGFR group.

FGFR aberration in MBC patients
FGFR pathway plays a major role in angiogenesis, metastasis, cell proliferation and drug resistance.28–30 In squamous cell lung cancer and BC, FGFR inhibitors showed promising antitumor activity, especially for patients with FGFR1 and/or FGFR3 amplification.
32
Here, among 195 MBC patients,

Kaplan–Meier curves for progression-free survival (PFS) probabilities (probs.) stratified by circulating tumor DNA
Among 197 MBC patients, 22 patients had
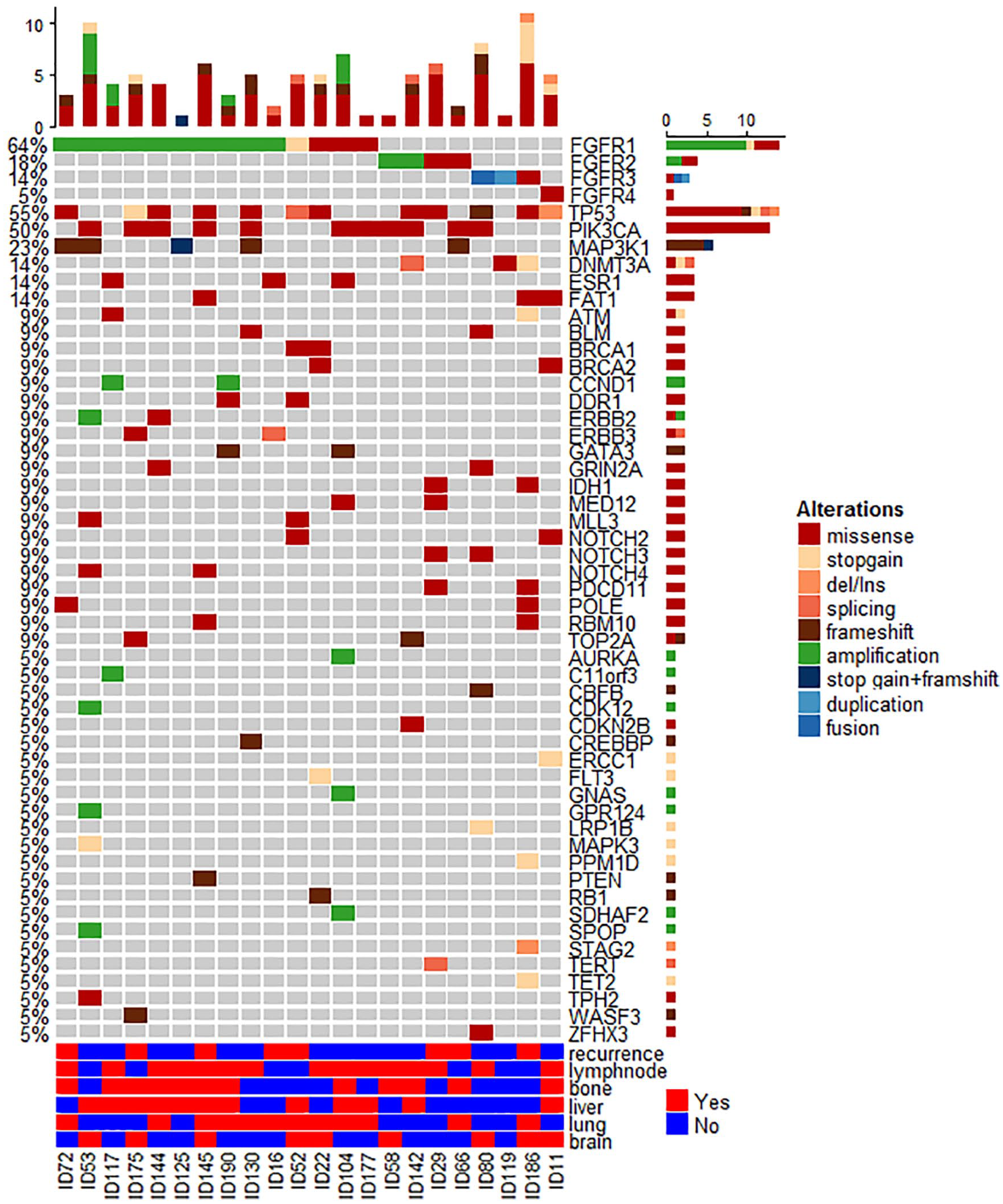
Circulating tumor DNA gene mutation profiles (top) and metastases (bottom) for 22
Differences of ctDNA mutation pattern between FGFR-aberrant group and wild-type group.

FGFR1 aberration increased in MBC patients with brain metastases
FGFR1 aberration rate comparison in bone-, brain-, lung-, and liver-metastatic breast cancer patients in Geneplus database.

Among 57
FGFR aberrations predict poor PFS
Kaplan–Meier curve with lifetest showed that
Treatment response for BC patients with

Patients with PFS > 6months were highlighted in red.
Level of amplification indicated the copy number of
ctDNA surveillance represented the change trend of aberrant levels of
Progression indicated whether patient progressed (1) or not (0) till the date of last follow-up.
PFS represented the progression-free survival (months) for progressed patients (progression = 1), or the duration to the date of last follow-up for censored patients (progression = 0).
COSMIC record represented the pathological effect of
BC, breast cancer; BM, brain metastasis; COSMIC, Catalogue Of Somatic Mutations In Cancer; ctDNA, circulating tumor DNA; HER2, human epidermal growth factor receptor-2; HR+/-, hormone receptor positive or negative; PFS, progression-free survival; TNBC, triple-negative breast cancer.
Specifically,
FGFR aberrations concentrated in HR-positive BCBM patients
Compared to non-BM MBC patients, more BCBM patients were HER2-positive (40%
Discussion
In practice, we found that some other brain metastases sequentially occurred after bone or other visceral metastases, especially in HR-positive BC patients, suggesting brain metastases as the final deadly stage for this group of patients; some brain metastases appeared earlier than bone and other visceral metastases, especially in HER2-positive BC patients. In this study, we found that
Genetic risk factor
Recently, due to the application of new targeting agents, such as pertuzumab, ado-trastuzumab emtansine, and lapatinib, the survival outcome has been improved for HER2-positive BC patients with brain metastases. Anti-HER2 therapies could extend the median OS from a few months up to 16–20 months.46,47 Lapatinib plus capecitabine regimen is, at present, the most effective option for HER2-postive BCBM patients, with favorable tumor response, disease control, and survival rates.10,48,49 Lapatinib is a lipophilic, small-molecular inhibitor of EGFR and HER2. 50 For patient ID53 in this study, we found that the brain metastatic lesion shrank after capecitabine plus lapatinib and trastuzumab treatment. Besides lapatinib, a recent study suggests neratinib plus capecitabine is active against refractory HER2-positive BCBM 51 ; Tucatinib in combination with capecitabine and trastuzumab also shows preliminary anti-tumor activity in advanced HER2-positive BCBMs 48 (HER2Climb trial to be presented at the San Antonio Breast Cancer Symposium 2019). In addition, pyrotinib plus capecitabine exhibits efficacy in HER2-positive BCBM (Phoenix TRAIL, 2019 American Society of Clinical Oncology).
Besides FGFR, TP overexpression correlates with the pro-angiogenic (VEGF) to promote angiogenesis and metastasis in human tumor tissues.52,53 Since capecitabine is a prodrug which is activated to 5-fluorocytidine by carboxylesterase, cytidine-deaminase and TP, capecitabine is supposed to be more effective in TP-positive tumors, including BC.
52
In this study, capecitabine-based treatment showed activity in
Novelty and impact statement
A group of genetic events could forecast the occurrence of brain metastases in BCs (BCBM), including
Supplemental Material
Table_S1 – Supplemental material for FGFR aberrations increase the risk of brain metastases and predict poor prognosis in metastatic breast cancer patients
Supplemental material, Table_S1 for
Supplemental Material
Table_S2 – Supplemental material for FGFR aberrations increase the risk of brain metastases and predict poor prognosis in metastatic breast cancer patients
Supplemental material, Table_S2 for
Supplemental Material
Table_S3 – Supplemental material for FGFR aberrations increase the risk of brain metastases and predict poor prognosis in metastatic breast cancer patients
Supplemental material, Table_S3 for
Supplemental Material
Table_S4_FGFR – Supplemental material for FGFR aberrations increase the risk of brain metastases and predict poor prognosis in metastatic breast cancer patients
Supplemental material, Table_S4_FGFR for
Supplemental Material
Table_S5-brain – Supplemental material for FGFR aberrations increase the risk of brain metastases and predict poor prognosis in metastatic breast cancer patients
Supplemental material, Table_S5-brain for
Footnotes
Acknowledgements
We would like to thank the researchers from Geneplus Beijing Institute as these individuals sequenced all patients’ ctDNA and gDNA samples for this study.
Author note
Jianbo Yang is also affiliated with Fujian Medical University Union Hospital, Fuzhou, China.
Author contributions
Dr. Quchang Ouyang and Zhe-Yu Hu have the full access to all data in the study and take responsibility for the integrity and accuracy of the data analysis. Study concept and design: Zhe-Yu Hu, Quchang Ouyang. Data acquisition: All authors. Data analysis and interpretation: Zhe-Yu Hu and Quchang Ouyang. Drafting of the manuscript: Zhe-Yu Hu. Critical revision of the manuscript for important intellectual content: All authors
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
The study was approved by the Ethics Committee at Hunan Cancer Hospital, the Affiliated Cancer Hospital of Xiangya School of Medicine/Central South University. The ethical board approval number is 2018119. The authors declare that they have obtained ethical approval and patients consent to participate.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Hunan Provincial Natural Science Youth Foundation Project Grant No.2019JJ50356 (Recipient: Zhe-Yu Hu); Hunan Provincial Health and Sanitation Committee Project Grant No. C2019070 (Recipient: Zhe-Yu Hu); Key Research Program in Hunan Social Development Grant No. 2019SK2032 (Recipient: Zhe-Yu Hu); Changsha Science and Technology Project Program No.30749 (Recipient: Zhe-Yu Hu).
Statement about data availability
We used the data from Geneplus database and from the Hunan Cancer Hospital. Geneplus database is not a public database. Its data will be made available upon reasonable request. ctDNA data from Hunan Cancer Hospital is available in supplemental Table S5 (BM patients) and ![]() (non-BM MBC patients).
(non-BM MBC patients).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
