Abstract
The field of reconstructive urology has seen a paradigm shift in the surgical approach for intra-abdominal cases, resulting in an increased preference for minimally invasive techniques. The introduction of the surgical robot has led to exponential growth in innovative approaches, reflecting the ongoing efforts to improve patient outcomes and address the limitations of open and laparoscopic surgery. This review article summarizes the knowledge gained in the last 10 years about adult robotic ureteral reconstruction. A non-systematic literature review was conducted on February 18, 2024 using Medline, PubMed, Web of Science, and Embase libraries. Studies published in English since 2014 reporting at least five robotic ureteral reconstructive cases for the management of benign ureteral obstruction in adults were included. A narrative review focusing on robotic ureteral reconstructive techniques, their associated success and complication rates, and how the robotic approach compares to open and laparoscopic reconstruction was performed. The current literature demonstrates increased utilization of the robotic platform in reconstructive urological procedures. Robotic surgery has been shown to be associated with shorter learning curves, lower surgeon fatigue, better visualization and equivalent results to those seen with laparoscopic surgery. While the literature is limited by a lack of comparative data, well-designed randomized controlled trials, and standardized criteria for defining and measuring success, this review demonstrates the safety, feasibility, and efficacy of robotic ureteral reconstruction for managing benign ureteral strictures, confirming it as a viable alternative to open and laparoscopic surgery.
Introduction
Upper urinary tract obstruction can arise from either benign or malignant etiologies, with an array of underlying diagnoses such as kidney stones, retroperitoneal fibrosis, endometriosis, iatrogenic injury, trauma, stricture disease, cancer, or radiation sequelae. 1 Ureteral stricture disease comprises a significant portion of upper urinary tract obstruction in adults. 1 Strictures can be managed endoscopically and with major reconstructive surgery. The exact surgical approach and technique employed for treating ureteral stricture disease often depends on factors such as length, location, and etiology of the stricture, as well as patient and surgeon preference. 1 Until the introduction of laparoscopic surgery to reconstructive urology in the 1990s, the gold standard for treating ureteral stricture disease had been open ureteral reconstruction. 2 Laparoscopic surgery, however, soon became the preferred approach for managing specific urologic conditions. It resulted in lower estimated blood loss (EBL), reduced rates of complications, and shorter hospital length of stay (LOS).3,4 The introduction of the robotic platform with the Da Vinci Surgical System® (Intuitive Surgical, Sunnyvale, CA, USA) in the early 2000s addressed many of the limitations of laparoscopic surgery, namely offering superior image quality and visualization, the option for additional visualization aids like Indocyanine green (ICG) and Firefly technology, and increased degree of motion without sacrificing surgeon ergonomics.2,5
In 20 years, reconstructive urology has trended toward minimally invasive techniques. 4 The introduction of the surgical robot sparked the birth of many novel ureteral reconstructive methods and the adaptation of surgical approaches used during open or laparoscopic surgery to the robotic platform. This review article aims to summarize the knowledge gained in the past ten years about robotic ureteral reconstruction for managing ureteral strictures, focusing on surgical technique, success, complication rates, and how the robotic platform compares to open and laparoscopic ureteral reconstruction.
Anatomy
The ureter is a retroperitoneal tubular structure that drains urine from the kidney into the bladder (Figure 1). In adults, it can measure between 22 and 30 cm. 6 It is anatomically and functionally divided into three segments: abdominal, pelvic, and intramural. The abdominal ureter starts at the ureteropelvic junction (UPJ) and travels anterior to the psoas muscle until it reaches the brim. 6 The pelvic ureter crosses over the bifurcation of the iliac vessels and descends anteriorly to the internal iliac artery along the lateral wall of the pelvis. 6 The intramural ureter is the segment of the ureter that pierces the bladder at the ureterovesical junction (UVJ). 6 The ureter has three areas of physiologic narrowing: the UPJ, as it crosses the iliac vessels, and the UVJ. 6 These are potential sites for obstruction secondary to renal calculi.

Anatomic rendition of the kidney, ureter, and bladder with depiction of reconstructive techniques by stricture location and length: findings from a narrative review.
The blood supply to the abdominal ureter comes medially from branches of the renal artery and posteriorly from the common iliac artery. 6 Branches from the superior vesical artery supply the lateral aspect of the pelvic ureter. 6
Etiology
In adults, obstruction of the upper urinary tract is often a result of ureteral stricture disease, which can be classified as iatrogenic, benign, idiopathic, or malignant. 1 Although it is hard to assess the definitive incidence of ureteral strictures in the general population, it has been suggested that 35% of strictures reported in the literature are due to iatrogenic causes, of which 70% result from gynecologic surgery. 1 Ureteral injury most commonly occurs at the pelvic brim, particularly in women, where the pelvic ureter crosses under the uterine vessels and passes anterior to the vaginal fornix before reaching the bladder. 6 Another 35% of strictures are benign, resulting from stones, infection, retroperitoneal fibrosis, endometriosis, aortic abdominal aneurysm, or trauma. Lastly, in a comprehensive review of the literature, Tyritzis and Wiklund 1 reported that the incidence of idiopathic and malignant obstruction are around 20% and 10%, respectively. While we acknowledge that these figures do not account for 100% of causes of malignant obstruction, this represents an active area of research and more work is needed to definitively examine the causes.
Pre-operative planning
Appropriate diagnostic workup is crucial when there are concerns for a ureteral stricture. It allows the surgeon to assess the need for prompt endoscopic intervention to preserve renal function, rule out malignancy, and aid in surgical planning. CT urography and antegrade or retrograde pyelogram are most commonly utilized to define stricture location and length.1,7 Providers can also obtain a nuclear medicine renal scan before intervention to determine baseline renal function and guide management. Traditionally, kidneys contributing less than 15%–20% of total function are not preserved as they are unlikely to have reasonable outcomes from a repair. 8 However, pre-operative assessment should also incorporate renal volume, functional urine output, and patient-reported symptoms to guide decision-making, as functional imaging alone may not reliably determine candidacy for reconstructive intervention. 9
Lastly, an important consideration in pre-operative planning is the use of ureteral rest before robotic ureteral reconstruction. Lee et al. 10 demonstrated that patients who underwent ureteral rest—defined as the absence of internal hardware such as a stent or percutaneous nephroureterostomy tube across the stricture for at least 1–4 weeks prior to surgery—had significantly higher success rates in the repair of proximal or middle ureteral strictures compared to those who did not undergo this practice (90.7% vs. 77.5%, p = 0.027). The authors hypothesized that allowing the ureter to rest can promote the maturation and stabilization of the stricture, leading to clearer delineation of stricture characteristics and potentially a more successful repair. 10 While the exact timing and indications for ureteral rest remain unclear, further research is necessary to understand its biological and radiographic impact and to establish more definitive guidelines regarding its optimal use.
Surgical determination
The choice of management for ureteral strictures and their associated success rates is dependent on the etiology, location, and length of the stricture. Generally, endoscopic treatments are used as first-line management for benign strictures. There are, however, several factors that are considered relative contraindications to endoscopic intervention due to their poor success rates, including long (>2 cm) or ischemic strictures, a poorly functioning ipsilateral kidney, and obstruction due to extrinsic compression. 1 When endoscopic approaches are not an option due to previously failed intervention or contraindication, ureteral reconstruction is recommended via an open, laparoscopic, or robotic approach.
The etiology of strictures is a critical determinant in selecting the optimal treatment strategy. For instance, radiated strictures often require more nuanced management because of their ischemic nature and the surrounding tissue damage. 11 These strictures are less likely to respond to endoscopic treatments and frequently necessitate surgical reconstruction, with ureteral reimplantation or ureteral substitution often preferred. 11 Additionally, strictures resulting from recurrent infections or malignancies may require tailored approaches depending on the broader clinical context.
The choice of surgical technique is often determined by stricture location (Figure 1). Abdominal or upper ureteral strictures are managed with either pyeloplasty, ureterocalycostomy (UC), ureteroplasty, ureteroureterostomy (UU) or transureteroureterostomy (TUU).3,12 Distal strictures are usually managed with ureteral reimplantation (UR) with or without adjunct mobility procedures such as a psoas hitch (PH) or Boari flap (BF).3,12 Complex or pan-ureteral strictures not amenable to other methods of ureteral repair can be managed with intestinal substitution or renal auto-transplantation. 3
Moreover, one of the guiding principles of ureteral reconstruction is the need for a tension-free anastomosis. Therefore, the length of the stricture can limit surgical options, as is the case with UUs, which are usually recommended for strictures measuring ⩽3 cm.1,12 For distal strictures located ⩾5 cm from the ureteral orifice, UR should be performed by adding a PH or BF. 12 Lastly, it is important to remember that bladder function is another critical factor in determining surgical management. Reconstructive techniques, such as UR with a PH or BF, often depend on a well-functioning bladder to achieve successful outcomes. 8 In patients without suspected underlying causes of bladder dysfunction, a uroflow and post void residual assessment is likely sufficient; patients with underlying bladder conditions may benefit from formal urodynamics to assess capacity and compliance. Patients with impaired bladder compliance or capacity, often secondary to prior radiation or neurogenic bladder dysfunction, may not tolerate increased bladder demands imposed by reimplantation techniques. 8 In these cases, more extensive reconstructive strategies may be required.
Methods
A comprehensive narrative review of the literature was performed on February 18, 2024 on MEDline, PubMed, Web of Science, and Embase databases using the following search terms: (“ureteral reconstruction” OR “ureteric reconstruction” OR “pyeloplasty” OR “ureteroureterostomy” OR “ureteroneocystostomy” OR “ureterocalycostomy” OR “ureteral reimplant” OR “ureteral implant” OR “ureteric reimplant” OR “ureteroplasty” OR “transureteroureterostomy” OR “kidney auto-transplantation” OR “kidney auto transplantation” OR “intestinal interposition”) AND (“ureteral obstruction” OR “ureteric obstruction” OR “obstruction” OR “stricture” OR “ureteral stenosis” OR “ureteric stenosis” OR “ureteral stricture” OR “ureteric stricture” OR “upper ureter*” OR “lower ureter*” OR “distal ureter*” OR “mid ureter*” OR “proximal ureter*”) AND (robotic* surgery OR “robotic assisted” OR “robotic” OR “robot” OR “robot assisted” OR “robot-assisted”) AND (“adult*” OR “young adult”) NOT (“children” OR “child” OR “pediatric” OR “paediatric” OR “peds” OR “kid*” OR “infant*” OR “neonate*”).
Study eligibility was defined by the PICOS framework: population (P), intervention (I), comparator (C), outcome (O), and study design (S). Studies that fulfilled the following criteria were included for analysis: (P) adult patients ( ⩾18 years old) who underwent a ureteral reconstructive surgery for the treatment of benign ureteral obstruction; (I) robotic ureteral reconstructive surgery; (C) non-robotic surgical intervention including endoscopic management, open, or laparoscopic surgery (O) peri- and post-operative outcomes, complications, follow-up time and success rates; (S) randomized-controlled trials (RCTs), non-randomized cohort studies, and meta-analyses. Exclusion criteria included publication date before 2014, non-English publications, studies with a follow-up period of less than 12 months, studies with fewer than five robotic cases of any singular ureteral reconstructive technique and studies reporting multiple techniques without providing individual outcomes for each technique. Reviews, conference abstracts, letters, commentaries, and editorials were excluded from the analysis. Articles were reviewed for relevance by the authors (RGJ and RGB), who excluded articles based on the title, followed by the abstract, and finally, the full text. Reference lists of included manuscripts were also screened for eligibility. Each reviewer worked independently. The article selection process is depicted in Figure 2.

Study selection flow chart.
Surgical management
Pyeloplasty
Ureteropelvic junction obstruction (UPJO) in adults can result from congenital anomalies that affect the ureters, including peristaltic ureteric segments, ureteral kinking, and duplex systems, as well as vascular anomalies, as is the case with crossing vessels. 13 More often, UPJO in adults results from acquired causes such as renal calculi, iatrogenic injury, or malignancy. 13
While endoscopic management is often the first-line treatment for patients with UPJO, their associated failure rates are significantly higher than those of definitive surgery, leading to a slight decrease in its utilization in recent decades.14–16 Options for surgical management of UPJO include Anderson-Hynes dismembered pyeloplasty and dismembered pyeloplasty with renal pelvis U-flap, as well as non-dismembering options such as Y-V and Fenger pyeloplasty.13,17 The Anderson-Hynes technique has been the preferred option for the last century, given its adaptability in addressing various reasons for UPJO and its excellent outcomes. Recent comparative studies have consistently shown success rates exceeding 90% across all three surgical modalities (robotic, laparoscopic, and open), further reinforcing its efficacy (Table 1).18–20
Summary of selected studies describing robotic management of ureteral strictures at the level of the UPJ.

Data from this study regarding reconstructive techniques not relevant to this table has been omitted.
Data from this study with less than 12 months FU has not been included in this table.
p ⩽ 0.05.
CD, Clavien Dindo Score; COPD, chronic obstructive pulmonary disease; DV-RAL, da Vinci Si Surgical Robot; EBL, estimated blood loss; KD-RAL, Kangduo Surgical Robot; LAP, laparoscopy; LOS, length of stay; na, not available; OT, operative time; RAL, robotic-assisted laparoscopy; SP, single port; UC, ureterocalicostomy; UPJ, ureteropelvic junction; UTI, urinary tract infection.
While open pyeloplasty has historically been considered the gold standard for the definitive management of UPJO, it was noted that from 2002 to 2010, there was a 40% decrease in its use among American urologists. 15 This drop in open pyeloplasties and increased utilization of minimally invasive surgery (MIS) has been driven by the comparative success rates offered by MIS and its superior perioperative outcomes, such as decreased EBL and pain scores, lower risk of postoperative complications and shorter LOS (Table 1).15,16,18,19,21
Compared to the laparoscopic approach, robotic-assisted laparoscopic (RAL) pyeloplasty offers similar success rates and functional outcomes; however, it has been shown to result in shorter operative times, lower surgeon fatigue, and a shorter learning curve of 20–30 cases.18,19,22–24 For these reasons, RAL pyeloplasty has become an attractive and reasonable alternative to treating both primary and recurrent UPJO.17,25–39 RAL pyeloplasty has also been shown to be an effective and safe option for the management of complex cases of UPJO due to concomitant congenital anomalies such as horseshoe kidney, duplex systems and malrotated kidneys. 40
A key advantage of the robotic surgical approach is the ability to employ ICG to assess tissue perfusion and urinary tract patency. ICG can be administered intravenously and will demonstrate perfusion of flap tissue, including tissues being used for intestinal interposition or flaps, such as omental flaps, being used to support the repair. Furthermore, instillation of ICG into the ureter can aid in identification of the patent portions of the urinary tract. One downside of intralumenal ICG use is that spillage after opening the patent portion of the urinary tract can make it difficult to interpret further ICG usage. 32
While RAL pyeloplasty is more commonly performed through transperitoneal access, one author set out to explore the safety and efficacy of performing RAL pyeloplasty through a retroperitoneal approach. In this study, Whiting et al. found retroperitoneal RAL pyeloplasty to have success rates of 94.5%. 41 However, the authors reported that retroperitoneal RAL pyeloplasty has a longer learning curve and operative times. 41 The authors advise that while the retroperitoneal approach offers many benefits, its utilization should be based on a surgeon’s comfort level and training.
It has been estimated that approximately 20% of patients with UPJO will present with concomitant stones. However, the current literature lacks consensus on the superior management mode for stone burden during pyeloplasty. A few recent retrospective single-center studies explored the role of concurrent stone extraction at the time of RAL pyeloplasty to manage concomitant calculi.25,42 Notably, Marien et al. 25 found that patients who underwent pyeloplasty with stone extraction had statistically significant decreased rates of radiographic improvement (87%), as compared to those who underwent pyeloplasty alone and had 99% improvement. These results of this study, while important to consider, are limited by a lack of specification of the technique used to remove stones.
Another area of increased interest in the recent literature has been the adoption of the Da Vinci Single-Port (SP)® system (Intuitive Surgical, Inc.) for the management of UPJO, which is a safe and effective alternative to other more mainstream approaches.43–45 By minimizing the number of entry points into the abdomen, using the SP system could potentially decrease post-operative pain, recovery time, and wound-related complications while providing better cosmetic results.43–45 While all articles cited in our review reported using intraperitoneal access, a major advantage of the SP system lies in its capability for retroperitoneal access. This approach offers significant benefits, particularly for patients with prior abdominal surgeries or a history of abdominopelvic radiation. Retroperitoneal access with the SP system is also advantageous for obese patients, as it avoids the need for complex positioning by allowing them to remain supine during the procedure. This is possible due to the SP’s wide external range of motion, enabling complete 360° anatomical accessibility, which allows surgeons to change the target organ and perform additional procedures without requiring redocking of the robot or repositioning of the patient.
In a multicenter prospective study of SP pyeloplasty, Buffi et al. 43 reported a 93.3% success rate at 13 months of follow-up and minimal complications. The authors noted some potential limitations of the SP system, including instrument crowding and limited triangulation. 43 A single-center retrospective comparative study between SP pyeloplasty and mini-laparoscopic pyeloplasty found both approaches to offer similar results in post-operative narcotic usage and cosmetic satisfaction. 45 In this study, SP pyeloplasty was associated with longer operative times and a significantly higher cost, which could be potential limitations to its adoption. 45
Ureterocalycostomy
Ureterocalycostomy (UC) is another option for treating UPJO, usually reserved for instances of previously failed pyeloplasty or a relatively inaccessible renal pelvis. 46 This technique has yet to be widely adopted to the robotic platform, as many surgeons prefer the open approach due to the complexity of this technique. A small case series reported using RAL UC to manage secondary UPJO with minimal complications and success rates of 100% at 15 months follow-up. 46 Ramanitharan et al. 46 reported using near-infrared fluorescence (NIRF) with Firefly technology during RAL UC to avoid complications related to recurrent obstruction and anastomotic stricture.
Ureteroureterostomy
Ureteroureterostomy (UU) is most commonly used for short strictures (⩽3 cm) located in the upper ureter. RAL UU offers low complication rates and success rates ranging from 83.3% to 100% at short to intermediate-term follow-up (Table 2).25,26,32,47–50 A retrospective single-center comparative study by Sun et al. 47 found significantly lower operative and suturing times, as well as shorter LOS in the robotic group as compared to the laparoscopic group, making this a safe and effective alternative to conventional laparoscopy.
Summary of selected studies describing robotic ureteroureterostomy for the management of ureteral strictures.

Data from this study regarding reconstructive techniques not relevant to this table has been omitted.
p ⩽ 0.05.
CD, Clavien Dindo Score; EBL, estimated blood loss; LAP, laparoscopy; LOS, length of stay; na, not available; OT, operative time; RAL, robotic-assisted laparoscopy; UU, ureteroureterostomy.
RAL UU has also been found to be a suitable option for treating longer strictures in the upper ureter measuring up to 4 cm as well as complex, recurrent, and iatrogenic strictures.25,26,48,49 The use of downward nephropexy to gain 3–5 cm of ureteral mobilization and attain tension-free anastomoses can aid in these cases. 51 Moreover, RAL UU has also been shown to be helpful in distal strictures, contradictory to popular belief.47,49 Yang et al. recently described the use of RAL UU for the management of short distal strictures in 137 young, healthy adults with no history of abdominopelvic radiation. They managed short strictures (1–2 cm) with a 90.1% success rate at a median follow-up time of 54.6 months. 49 The authors used NIRF with intravenous ICG during dissection to identify and spare the tenuous blood supply to the distal ureter. 49
Ureteroplasty
Graft ureteroplasty is often employed for the management of upper ureteral strictures measuring more than 1.5 cm as well as complex and recurrent ureteral strictures that would otherwise require management with ileal ureteral interposition or auto-transplantation, which carry a high degree of morbidity and are much more technically challenging.52,53 Ureteral rest in this context may be beneficial to more accurately define the length and caliber, thereby aiding in graft or flap selection. 10 The most commonly used grafts for the management of ureteral strictures include buccal and lingual grafts.39,51,52,54,55 An essential distinction between grafts and flaps, which can also be used to manage ureteral strictures, is that a graft is made of tissue that does not contain its blood supply and, upon transfer to the recipient site, will develop a new blood supply. In contrast, a flap is a vascularized tissue that maintains blood supply after being transferred to the recipient site. 53
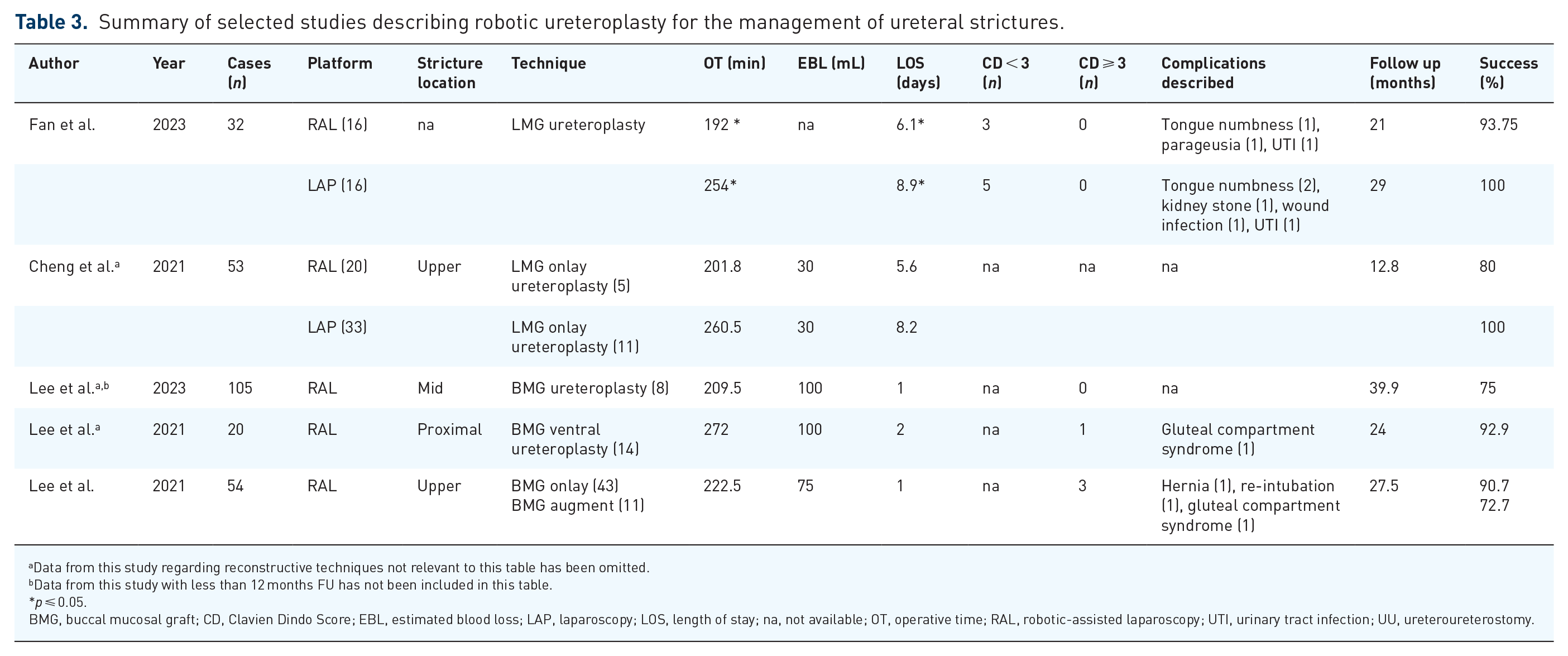
The high success rate associated with the use of buccal mucosal graft (BMG) in urethroplasties led to its adoption for the treatment of ureteral strictures. RAL BMG ureteroplasty is safe and effective for managing upper ureteral strictures and recurrent, complex, or lengthy strictures resulting in low complication rates and success rates ranging between 72.7% and 92.9% at intermediate-term follow-up (Table 3).51,52,55 Moreover, two multi-institutional retrospective studies found RAL BMG ureteroplasty effective for managing recurrent long-segment UPJO or UPJO with significant surrounding fibrosis with success rates over 80%.17,52 Using omental flaps wrapped over the BMG graft has been described to ensure graft survivability and reduce the risk of stricture recurrence. 56
Summary of selected studies describing robotic ureteroplasty for the management of ureteral strictures.
Data from this study regarding reconstructive techniques not relevant to this table has been omitted.
Data from this study with less than 12 months FU has not been included in this table.
p ⩽ 0.05.
BMG, buccal mucosal graft; CD, Clavien Dindo Score; EBL, estimated blood loss; LAP, laparoscopy; LOS, length of stay; na, not available; OT, operative time; RAL, robotic-assisted laparoscopy; UTI, urinary tract infection; UU, ureteroureterostomy.
While the number of studies on using lingual mucosal graft (LMG) during RAL ureteroplasty is limited, recent reports demonstrate excellent outcomes with success rates over 90%.39,54 Consistent with the previous reports comparing robotic and laparoscopic approaches, RAL LMG ureteroplasty resulted in shorter operative times and LOS.39,54 Lastly, while the proposed benefits of LMG include lower donor site morbidity, this review found both BMG and LMG RAL ureteroplasty to have minimal donor site-associated complications.39,51,52,54,55 Currently, there is no consensus on which type of graft is superior or if there are specific scenarios where one type of graft should be picked over another.
Transureteroureterostomy
While transureteroureterostomy (TUU) is not a commonly performed procedure, it is typically used in adults undergoing multi-organ resection for malignancy or treating mid-ureteral strictures not amenable to other reconstructive techniques. 12 TUU is contraindicated in patients with urothelial carcinoma of the ureter, idiopathic retroperitoneal fibrosis, renal stones, and a history of pelvic radiation or ureteral stricture disease in the recipient ureter. 12 No studies were found on RAL TUU that fit our search criteria.
Ureteral reimplantation
Recent comparative studies have demonstrated that managing distal ureteral strictures with RAL UR, also known as ureteroneocystostomy (UNC), has similar success rates to open and laparoscopic UR (Table 4).57–61 Moreover, compared to the open approach, RAL UR has been shown to offer superior results in EBL, operative times, and LOS.57–60 In an analysis of an extensive prospective multicenter national database, Packiam et al. found that in the first 30 days after UR, patients who underwent open surgery were significantly more likely to experience complications related to wounds and infection than patients who underwent MIS. 57 Despite the clear advantages of MIS over open surgery, data from the National Surgical Quality Improvement Program Database showed that between 2007 and 2017, reconstruction of the distal ureter in 926 patients was performed using open surgery in 50.5% of cases. 4
Summary of selected studies describing robotic ureteral reimplantation for the management of ureteral strictures.

Data from this study regarding reconstructive techniques not relevant to this table has been omitted.
p ⩽ 0.05.
BF, boari flap; CD, Clavien Dindo Score; EBL, estimated blood loss; LAP, laparoscopy; LOS, length of stay; na, not available; OT, operative time; PH, Psoas hitch; RAL, robotic-assisted laparoscopy; SP, single port; UNC, ureteroneocystotomy; UTI, urinary tract infection.
In patients with normal bladder function and distal strictures located more than 5 cm from the ureteral orifice, the adjunct use of a PH or BF during UR is recommended to achieve tension-free anastomosis. 12 A PH can be beneficial in strictures measuring 6–10 cm, while a BF can be used for strictures measuring up to 15 cm.12,62 Both techniques have been shown to offer high success rates and low rates of complications.11,44,58,62–65 Bladder capacity should be assessed prior to the use of BF to ensure that flap creation does not compromise bladder storage volumes. However, a significant limitation of our current understanding of the efficacy of PH and BF is that most studies report their use as part of large ureteral reconstruction cohorts that don’t often differentiate outcomes by the technique used. One prospective multi-institutional study by Coarse et al. 63 reported using UR with BF in a high-risk cohort of 50 patients with a 90% success rate and minimal significant complications. Moreover, the current data on SP UR is limited to very small cohorts; Heo et al. 44 reported 100% success rates in five patients undergoing SP UR + PH.
A novel ureteral reconstructive technique employing a non-transecting side-to-side anastomosis described by Slawin et al. for managing distal strictures was found to be safe and effective.11,66 This approach seeks to preserve the tenuous blood supply to the distal ureter by eliminating ureteral transection and minimizing dissection.11,66 In a cohort of 16 patients undergoing RAL non-transecting side-to-side anastomosis, 93.8% remained stricture-free at a median follow-up time > 12 months. 66 Increased risk of symptomatic de-novo reflux is something surgeons should keep in mind when performing this technique. 66
Lastly, while managing upper ureteral strictures with UR is not standard of care, Corse et al. described high success rates with RAL UR + BF in 22 patients with proximal/mid strictures. 63 While overall, the study reported excellent outcomes, it offers readers limited insight into the results by stricture location.
Intestinal interposition and onlay
Complex ureteral strictures, including those that are proximal, long, bilateral, obliterative, or radiated, might not always be amenable to management by the techniques above, in which case, intestinal interposition should be considered. 67 Adopting the laparoscopic and robotic platforms for intestinal interposition has been slow, likely due to technical difficulties. Nevertheless, recent studies have reported using intracorporeal and extracorporeal RAL intestinal interposition, with success rates ranging from 86.7% to 100% at a short-term follow-up and minimal significant complications (Table 5).68–70 In a comparative study, Zhu et al. found RAL ileal ureter to offer significantly reduced operative times, minimal EBL, and shorter LOS than the laparoscopic group. 70 Patients in the RAL group also had shorter recovery, as demonstrated by significantly reduced time to oral intake and drain removal.
Summary of studies describing the management of complex strictures.

p ⩽ 0.05.
CD, Clavien Dindo Score; EBL, estimated blood loss; KAT, Kidney auto-transplantation; LAP, laparoscopy; LOS, length of stay; na, not available; OT, operative time; RAL, robotic-assisted laparoscopy; SP, single port; UTI, urinary tract infection.
Although the use of small and large intestinal segments has been described for upper and lower urinary tract reconstructive surgery, the ileum is usually preferred by urologists due to its rich vascular supply, good mobility, and decreased risk of associated metabolic complications compared to other intestinal segments.68–70 In recent years, there has been growing interest in appendiceal interposition, as the smaller surface area of the appendix makes intestinal interposition less likely to be associated with metabolic imbalances. 71 Jun et al. 71 performed RAL appendiceal interposition in five patients with obliterative strictures with no primary bowel-related complications. It has been argued that an appendiceal procedure might be a good option for patients that have an appendix, as it does not carry the donor site morbidity risk that oral mucosal grafts do, nor the metabolic complications associated with the creation of an ileal ureter and offer success rates over 90%; however, it might not be well suited for left-sided ureteral strictures, due to increased risk of anastomotic tension and intestinal volvulus.71–73 The length of ureteral stricture that can be treated with an appendiceal onlay is only limited by the size of the patient’s appendix. In the study by Jun et al. 71 the longest stricture treated with an appendiceal procedure measured 15 cm.
Auto-transplantation
Kidney auto-transplantation (KAT) is considered a last resort in managing complex proximal or pan-ureteric strictures, reserved for renal preservation in patients where other reconstructive procedures are contraindicated. Adapting this technique to MIS has significantly decreased morbidity and complication rates. 74 Breda et al. confirmed the safety and feasibility of RAL KAT in a comparative study of extracorporeal (eRAL KAT) and intracorporeal (iRAL KAT) approaches. 74 In a cohort of 29 patients, 15 underwent eRAL KAT and 14 iRAL KAT, with no statistically significant differences in perioperative outcomes and postoperative complications between the groups. Regarding functional outcomes, the eRAL KAT had significantly better preserved renal function at 90 days post-operatively (93.3% vs 57.1%, p = <0.05), but at 1 year, renal function in both groups was equivalent. 74 While the recent literature on using RAL KAT is promising, the widespread adoption of this technique is limited by its complexity and the need for a highly trained multidisciplinary team of transplant, urology, and vascular surgery.
Kaouk et al. described the feasibility of SP KAT for the management of complex strictures in a small cohort of eight patients. The mean OT was 520 min and patients had a median LOS of 3 days. The median length of follow up was 13 months, at which time the median change in glomerular filtration rate was 3.5%. The authors point out that an advantage of SP KAT it its unique ability to access multiple quadrants without the need for patient repositioning or re-docking, unlike with the multiport platform. 75
Special populations
Post-kidney transplant ureteral strictures
Ureteral strictures are common complications after kidney transplantation. While endoscopic treatment has become the initial treatment of choice for this condition, success rates are significantly lower than those achieved with open ureteral reconstruction. 76 The literature on the use of the robotic platform for the management of ureteral disease in kidney transplant patients is very limited. No studies were found on the robotic management of post-transplant ureteral strictures that fit our search criteria.
However, this is an area where the robotic platform might have a lot to offer. One of the biggest challenges of working on these types of cases is the difficulty of working through the fibrosis surrounding the graft and failure to identify the graft ureter, which could potentially be ameliorated with the use of ICG and Firefly technology.
Uretero-enteric strictures
In the case of uretero-enteric anastomotic strictures (UAS), a common complication of radical cystectomy, robotic uretero-enteric reimplantation (RAL UER) was shown to offer excellent perioperative outcomes and success rates (Table 6).77–79 Gin et al, reported their outcomes with both open and robotic UER and noted that the robotic groups had shorter LOS and no post-operative complications. 77 A recent meta-analysis reported that the open and robotic approaches were comparable in terms of perioperative outcomes and success rates, with the added caveat of less postoperative complications in the robotic group. 80 While reconstructed anatomy and extensive fibrosis secondary to a complex surgical and medical history in this patient population presents a challenge to the adoption of RAL UER 80 ; the use of intra-ureteral and intra-urinary diversion ICG during RAL UER has been reported to be an advantage of performing UER robotically, as it allows for faster identification and dissection of the UAS.78,79
Summary of selected studies describing the robotic management of uretero-enteric strictures.

CD, Clavien Dindo Score; DVT, deep vein thrombosis; EBL, estimated blood loss; LOS, length of stay; na, not available; OT, operative time; RAL, robotic-assisted laparoscopy; UTI, urinary tract infection.
Limitations and future directions
From the available data, several key insights can be drawn about the role of robotics in upper urinary tract reconstruction. Robotic surgery has proven to be both safe and effective for ureteral reconstruction, with demonstrated benefits such as reduced blood loss, shorter hospital stays, and quicker recovery times compared to traditional open techniques. These advantages, coupled with increased utilization and successful outcomes, suggest that robotic reconstruction has emerged as a new standard of care, challenging the long-held position of open surgery as the gold standard. While open surgery remains an important option for specific cases, the growing body of evidence supports the increasing dominance of robotics in this field.
However, the contemporary literature on robotic ureteral reconstruction, while providing valuable insights, has notable limitations that warrant mentioning. Most of the studies included in this review had a retrospective study design, which inherently introduces biases and hinders the establishment of causal relationships. The small number of RCTs restricts the availability of high-quality evidence, limiting our understanding of how surgical outcomes from robotic ureteral reconstruction truly compare to laparoscopic and open surgery. This is especially true as there is significant variability in the definitions of success employed across different studies. The need for standardized criteria for defining and measuring success in ureteral reconstruction studies across all surgical approaches hampers the ability to compare outcomes consistently, complicating the efforts to draw meaningful conclusions from the collective body of literature. Lastly, the predominance of small, single-institution cohorts within the current literature potentially challenges the generalizability of the results. This limitation underscores the need for more extensive, multi-center studies to bolster the external validity of research findings and enhance their applicability to a more comprehensive patient and surgeon population.
Furthermore, there are several areas within robotic ureteral reconstruction that warrant additional study, particularly newer techniques and technologies. Techniques such as the non-transecting side-to-side anastomosis and ureteroureterostomy (UU) for distal strictures are still in their early stages of adoption. Long-term studies evaluating their safety, efficacy, and success rates are necessary to establish their roles in ureteral reconstruction. Similarly, emerging technologies like the SP robotic platform remain underreported in the literature. While the SP system offers distinct advantages, including retroperitoneal access, the ability to work in confined spaces, and reduced postoperative pain, its role in ureteral reconstruction is uncertain. Currently, the SP system is primarily utilized in large academic centers, and its adoption is limited by a lack of widespread experience and supporting evidence. Future research should focus on evaluating the learning curve associated with the SP platform, comparing its outcomes to multiport robotic systems, and assessing its cost-effectiveness and impact on patient recovery. These efforts will help define the utility of these innovations and guide their integration into clinical practice.
Conclusion
Robotic ureteral reconstruction is a constantly evolving field. This review summarizes the growing body of evidence supporting the efficacy and safety of the robotic platform and highlights its many advantages over open and laparoscopic ureteral reconstructive surgery.
