Abstract
Radiation-induced ureteral stricture (RIUS) is a rare but refractory late complication of pelvic radiotherapy, driven by severe fibrosis and microvascular injury that undermines the effectiveness of conventional interventions such as balloon dilation, ureteral stenting, and reconstructive surgery. This review summarizes the pathophysiological basis of treatment resistance in RIUS and critically appraises current management strategies. Beyond its clinical relevance, RIUS is discussed as a paradigmatic model of advanced fibrotic ureteral disease, offering broader insights into the limitations of purely mechanical approaches. We further examine emerging biologically oriented strategies, including mesenchymal stem cell–derived extracellular vesicle–based regenerative therapies, next-generation ureteral stents incorporating advanced biomaterials, and drug-eluting stent platforms for localized antifibrotic and antimicrobial delivery. Although largely investigational, these approaches collectively signal a conceptual shift from palliative drainage toward biologically informed, restorative management of RIUS and related refractory ureteral strictures.
Plain language summary
Radiation therapy for cancers in the pelvis can sometimes cause a blockage in the ureter (the tube that carries urine from the kidney to the bladder). This condition, called radiation-induced ureteral stricture (RIUS), is rare but very difficult to treat. Standard treatments, such as balloon dilation, stents, or surgery, often do not work well because the radiation damage continues to cause scarring and poor blood supply.
This review looked at why current treatments fail and explored new ideas that scientists are testing in the laboratory and in early studies.
Three main areas of innovation were identified: Using tiny particles from stem cells (called extracellular vesicles) to reduce scarring and help healing. Developing new types of stents made from advanced materials, including metals or biodegradable plastics. Designing drug-coated stents that can release medicines directly into the ureter to fight scarring or infection.
These new approaches are still at the experimental stage, but they represent a shift in thinking—from simply relieving symptoms to trying to repair the ureter and restore its function.
Keywords
Introduction
Radiation-induced ureteral stricture (RIUS) is a rare but increasingly recognized late complication of pelvic radiotherapy, particularly following treatment for gynecologic, colorectal, and prostate malignancies. Despite advances in radiation delivery techniques that have reduced off-target toxicity, ureteral injury remains difficult to prevent and often manifests years after treatment.1–3 Progressive obstruction can lead to hydronephrosis, recurrent infection, and irreversible renal dysfunction, posing a substantial clinical challenge. 4
Unlike other benign ureteral strictures, RIUS arises from a uniquely hostile biological environment. Ionizing radiation induces chronic microvascular injury, persistent ischemia, and progressive fibrosis, resulting in obliterated tissue planes and severely impaired regenerative capacity.5,6 These pathophysiological features fundamentally differentiate RIUS from iatrogenic or inflammatory strictures and largely explain its poor response to conventional mechanical interventions. 7 Importantly, this extreme fibrotic phenotype provides a paradigmatic model for understanding the failure of standard approaches and for exploring biologically informed alternatives in ureteral stricture disease. 8
Clinically, RIUS is frequently insidious in onset and may remain asymptomatic until advanced obstruction develops. Diagnosis is often delayed, and bilateral involvement or long-segment disease is not uncommon. Although the overall incidence of RIUS is relatively low, its absolute burden is expected to rise with improved cancer survival and expanding indications for pelvic radiotherapy,2,3 making durable management strategies increasingly important 9 (Table 1).
Risk domains and clinical features of RIUS.

RIUS, radiation-induced ureteral stricture.
Current management of RIUS primarily focuses on restoring urinary drainage through endoscopic or reconstructive approaches, including balloon dilation, endoureterotomy, ureteral stenting, and complex surgical reconstruction. However, long-term success remains limited. High recurrence rates, delayed healing, and procedure-related morbidity are frequently encountered, reflecting the inability of purely mechanical solutions to address the underlying biological dysfunction of irradiated tissue.10–12
These limitations have prompted growing interest in biologically informed and regenerative therapeutic strategies. Rather than focusing solely on luminal patency, emerging approaches aim to modulate the fibrotic microenvironment, enhance angiogenesis, and support tissue repair. In this narrative review, we summarize current management options for radiation-induced ureteral stricture and critically examine emerging biologically oriented and regenerative strategies, using RIUS as a paradigmatic model of severe fibrotic ureteral disease in which conventional mechanical interventions frequently fail. By integrating insights into radiation-induced fibrosis with evolving therapeutic concepts, this review aims to inform future advances not only in RIUS but also in refractory ureteral stricture disease more broadly.
Literature search and review approach
This article is a narrative review focusing on the clinical and translational challenges of radiation-induced ureteral stricture and emerging therapeutic strategies. The literature was primarily identified through searches of the PubMed database using combinations of keywords including “radiation-induced ureteral stricture,” “ureteral stricture,” “radiotherapy,” “ureteral fibrosis,” “drug-eluting ureteral stents,” and “mesenchymal stem cells.”
To capture ongoing translational efforts and emerging clinical applications, publicly available clinical trial registries, including ClinicalTrials.gov, were also reviewed for relevant registered or ongoing studies. The purpose of the search was not to provide an exhaustive or systematic synthesis of all available evidence, but rather to identify representative and relevant studies that inform current clinical practice, underlying pathophysiological mechanisms, and future therapeutic directions. Study selection was guided by relevance to the topic, translational significance, and methodological quality.
Pathophysiology of RIUS
To understand the limited durability of conventional mechanical interventions and the rationale for emerging biologically oriented strategies, it is essential to examine the underlying pathophysiological mechanisms of RIUS. The development of RIUS is a multifactorial and progressive process, primarily initiated by chronic injury to the periureteral microvasculature. Radiation-induced vascular damage disrupts local perfusion, leading to persistent ischemia, low-grade inflammation, and progressive fibrotic remodeling.5,8,13 Over time, these changes result in irreversible ureteral wall thickening, luminal narrowing, and impaired urinary drainage, ultimately predisposing to upper urinary tract deterioration.
Ionizing radiation exerts its biological effects through direct DNA damage and indirect oxidative stress (Figure 1). Endothelial and stromal cell injury, coupled with sustained reactive oxygen species (ROS) generation, activates stress-response pathways such as NF-κB and p53, promoting the production of pro-inflammatory cytokines including interleukin-1β, tumor necrosis factor-α, and interleukin-6.8,13 Rather than resolving, this inflammatory response persists, establishing a chronic profibrotic microenvironment within the irradiated periureteral tissue.

Mechanistic overview of fibrotic remodeling in RIUS.
A key downstream consequence of this milieu is macrophage polarization toward an M2 phenotype. Although classically associated with tissue repair, M2 macrophages in RIUS adopt a predominantly profibrotic role, secreting transforming growth factor-β (TGF-β) and platelet-derived growth factor (PDGF). These mediators drive fibroblast activation and promote the differentiation of mesenchymal progenitors into myofibroblasts, which are the principal source of excessive extracellular matrix (ECM) deposition.6,14,15 Accumulation of collagen and other matrix components within the ureteral adventitia and surrounding connective tissue progressively stiffens the ureteral wall and compromises its compliance.
As fibrosis advances, capillary rarefaction and further microvascular collapse ensue, exacerbating tissue hypoxia and perpetuating oxidative stress. This establishes a self-reinforcing cycle of ischemia, inflammation, and fibrosis, which is central to the progressive nature of RIUS 13 (Figure 1). Importantly, this hostile microenvironment severely limits endogenous repair capacity and undermines the effectiveness of purely mechanical interventions.
Ultimately, these pathological processes culminate in ureteral wall atrophy, fibrotic contraction, and fixed luminal stenosis, often accompanied by dense adhesions to surrounding tissues. 9 Such changes not only impair ureteral peristalsis but also render surgical reconstruction technically challenging and prone to recurrence. 11 Collectively, these mechanisms highlight why RIUS is uniquely refractory to conventional treatment and underscore the need for therapeutic strategies that target fibrosis, restore microvascular integrity, and support tissue regeneration, rather than relying solely on anatomical reconstruction.
Current treatment strategies for ureteral stricture: Limitations and considerations in RIUS
In clinical practice, the overarching goals in managing ureteral stricture are to preserve renal function, alleviate urinary tract obstruction, and restore or maintain ureteral continuity. Treatment selection depends on several factors, including the stricture’s location, length, etiology, and associated comorbidities, as well as renal functional status. Although many approaches are well-established for benign, non-radiation-related strictures, their efficacy becomes markedly diminished in RIUS due to the complex and irreversible tissue changes induced by pelvic radiotherapy.4,9,11
Current treatment options can be broadly categorized into minimally invasive endourological interventions, ureteral stenting, external urinary drainage, and ureteral reconstructive surgery. An overview of current and emerging therapeutic strategies for radiation-induced ureteral stricture, together with their principal indications, evidence base, and key limitations, is summarized in Table 2. Each modality offers distinct advantages and limitations depending on the clinical scenario.
Current and emerging therapeutic strategies for RIUS.

Strength of evidence is descriptive rather than guideline-based and reflects the general level of available data.
RIUS, radiation-induced ureteral stricture.
Balloon dilation
Balloon dilation is commonly considered for short, non-ischemic strictures, as it offers the advantages of minimal invasiveness, short hospital stays, and rapid recovery. However, its utility in RIUS is severely limited. The dense fibrosis, poor vascularity, and often long segment involvement seen in RIUS compromise both the mechanical expansion and subsequent healing of the stricture site.12,16
Richter et al. evaluated 114 patients with benign ureteral strictures treated endoscopically and found that the 2-year recurrence-free rate was 89% in patients with short strictures (<2 cm) and adequate vascular supply, compared to only 40% in those with ischemic strictures. 17 In a separate study, Yam et al. 18 reported a 100% recurrence rate in five patients with RIUS undergoing balloon dilation, underscoring its limited efficacy in this setting. These findings suggest that balloon dilation may be appropriate for select cases of uncomplicated, short-segment strictures, but its utility in RIUS appears to be highly limited and should be approached with caution.
Endoureterotomy
Endoureterotomy, performed using cold knife or laser incision, involves full-thickness cutting of the stricture and adjacent periureteral tissue. By extending the incision into surrounding healthy, well-perfused tissue, this technique aims to enhance healing and reduce recurrence. It is considered effective in short, distal or proximal ureteral strictures not caused by ischemia or radiation.
Lojanapiwat et al. 19 reported a 6-month success rate of 75% (9/12) for endoureterotomy in benign strictures, significantly higher than 15% (9/15) for balloon dilation. Wu et al. demonstrated that combining holmium laser endoureterotomy with balloon dilation led to superior outcomes in strictures >2 cm, achieving 1-year and 2-year success rates of 77.2% and 66.6%, respectively, compared to 44.4% and 22.2% for balloon dilation alone. 20 However, in RIUS, the underlying tissue damage and loss of normal vascular architecture reduce the regenerative potential of the ureteral wall, rendering this approach less effective and more prone to recurrence or perforation.
Ureteral stent placement
Ureteral stents are frequently used for temporary relief of obstruction or in patients unfit for definitive surgery. Retrograde or antegrade placement can restore urinary flow, reduce back pressure on the kidneys, and buy time for further intervention. Interestingly, in contrast to extrinsic obstructions caused by tumor compression, intrinsic strictures such as those from RIUS appear to have a higher stenting success rate.21–23 Yossepowitch et al. 21 reported a 94% success rate in intrinsic strictures, compared to 73% in extrinsic ones. However, despite high initial success, stents are not without drawbacks.
Long-term indwelling stents carry significant risks, including biofilm-related infection, encrustation, dislodgement, stent migration, and ureteral fistula formation, especially in irradiated tissue.24–26 Frequent early occlusion due to inflammatory debris or mucosal hyperplasia necessitates shorter exchange intervals than standard practice. 24 Although polymer stents can often remain for 3–6 months and metal stents up to 12 months, RIUS patients frequently require exchanges every 1–2 months, significantly increasing economic burden, infection risk, and overall healthcare utilization.27–29
This reality underscores the urgent need for next-generation stents designed specifically for hostile environments such as RIUS, including self-expanding metallic stents, drug-eluting stents, and biodegradable materials—areas currently under active investigation and discussed later in this review.
Percutaneous nephrostomy
Percutaneous nephrostomy (PCN) provides effective urinary diversion and remains a valuable option in emergent or palliative settings, especially for patients with severe bilateral obstruction, infectious complications, or poor performance status. While technically straightforward, nephrostomy tubes significantly impair quality of life, often requiring frequent maintenance, and are associated with risks of infection, tube dislodgement, and obstruction. In most cases, PCN is viewed as a temporary measure until definitive surgery or as a last resort in patients unfit for other interventions. 30
Ureteral reconstruction
Reconstructive surgery remains the gold standard for definitive correction of complex ureteral strictures. Procedures are tailored to the anatomical site and extent of the lesion. Proximal strictures may be managed with pyeloplasty or end-to-end ureteroureterostomy; distal strictures often require ureteroneocystostomy, sometimes combined with psoas hitch or Boari flap techniques. Longer segments may require tissue substitution using buccal mucosa grafts, appendix, ileum, or even autotransplantation in extreme cases.10,11
For long-segment or complex ureteral strictures, a range of reconstructive substitution and adjunctive techniques has been explored. Buccal mucosa graft ureteroplasty has emerged as a widely adopted tissue substitution approach for selected ureteral defects, owing to its favorable handling characteristics and epithelial robustness; however, in the setting of RIUS, long-term durability may be compromised by the underlying ischemic and fibrotic tissue environment.
For more extensive defects, bowel substitution using ileal or colonic segments—including techniques based on the Yang–Monti principle—has been reported and can restore ureteral continuity in carefully selected patients.31,32 Nevertheless, these approaches are associated with substantial surgical complexity and carry risks of metabolic, infectious, and absorptive complications, particularly in previously irradiated fields. 33 Adjunctive strategies such as omental or peritoneal wrapping have been employed to enhance local vascularization and support healing in hostile tissue environments, although available evidence remains limited to small case series.
In highly selected cases with extensive ureteral damage not amenable to conventional reconstruction, renal autotransplantation has been described as a salvage option; however, its invasiveness and limited feasibility in irradiated patients restrict broader applicability.
The increasing adoption of robot-assisted systems, such as the da Vinci platform, has facilitated greater surgical precision, shorter operative times, and minimized blood loss, particularly in complex reconstructions. Nevertheless, RIUS presents a uniquely hostile surgical field, characterized by obliterated tissue planes, compromised perfusion, adhesions, and multifocal bilateral involvement. Toia et al. 34 reported on 18 patients with RIUS, of whom 78% had bilateral disease, and 89% required complex reconstructive procedures or urinary diversion. Despite surgery, 30% experienced recurrent strictures, often necessitating multiple re-operations.
The unique pathophysiological milieu of irradiated tissue—marked by dense fibrosis, impaired microcirculation, and limited regenerative capacity—undermines the efficacy of even the most sophisticated surgical techniques. Both endoscopic approaches and open or robot-assisted reconstructive surgeries are associated with elevated risks of recurrence, complications, and delayed healing in this setting.
These observations underscore that surgical correction alone is often insufficient in RIUS. In many cases, anatomical repair fails to address the underlying biological dysfunction, particularly in patients with bilateral involvement, long-segment strictures, or prior high-dose pelvic irradiation. Moreover, the cumulative psychological burden, financial cost, and need for repeated interventions place substantial strain on both patients and healthcare systems.
Viewed longitudinally, renal functional decline in RIUS is not merely a consequence of luminal obstruction, but rather the cumulative result of repeated interventions, persistent ischemia, and progressive fibrosis, highlighting the inherent limitations of drainage- or reconstruction-focused strategies alone.
Renal functional considerations
Preservation of renal function represents a central, yet frequently unmet, objective in the management of radiation-induced ureteral stricture. While conventional approaches—including endoscopic interventions, long-term ureteral stenting, PCN, and reconstructive surgery—primarily aim to restore urinary drainage or anatomical continuity, durable renal functional benefit is often difficult to achieve in RIUS.2,3 Recurrent obstruction, repeated interventions, and ongoing ischemic injury frequently compromise long-term renal preservation, particularly in patients with bilateral involvement or long-segment disease.34,35
In contrast, emerging biologically oriented strategies seek to address the underlying fibrotic and hypovascular microenvironment, with the theoretical potential to support more sustained renal function by modifying disease biology rather than solely correcting luminal narrowing. A conceptual comparison of conventional and emerging treatment strategies with respect to therapeutic focus, clinical applicability, and potential implications for renal functional outcomes is summarized in Table 3.
Conceptual comparison of conventional and emerging treatment strategies for RIUS with respect to renal functional outcomes.

PCN, Percutaneous nephrostomy; RIUS, radiation-induced ureteral stricture.
In this context, emerging treatments such as next-generation ureteral stents and mesenchymal stem cell (MSC)-based regenerative therapies represent leading examples of biologically oriented approaches.36,37 The following sections will explore the current evidence supporting these innovative strategies and their potential to redefine the treatment landscape for RIUS.
Emerging therapeutic strategies for RIUS
Stem cell–based therapeutic approaches for RIUS
Stem cell–based therapies have shown growing promise in addressing RIUS, a condition marked by progressive fibrosis, vascular compromise, and poor tissue regeneration.38,39 Among various cell types, MSCs have attracted particular attention40,41 due to their immunomodulatory, antifibrotic, and pro-regenerative properties, largely mediated by paracrine signaling rather than direct differentiation.41–46
Mechanistic rationale and preclinical validation
MSCs secrete a broad spectrum of bioactive molecules and extracellular vesicles (EVs)—including hepatocyte growth factor (HGF), vascular endothelial growth factor (VEGF), interleukin-10 (IL-10), TNF-stimulated gene-6 (TSG-6), and microRNAs such as miR-29 and miR-21—that modulate key molecular pathways involved in fibrosis and tissue remodeling. 47 These secreted factors primarily target the TGF-β1/Smad signaling axis, which plays a central role in the activation of myofibroblasts and the deposition of collagen-rich extracellular matrix (ECM).42,47 By inhibiting this profibrotic signaling cascade, MSCs reduce α-smooth muscle actin (α-SMA) expression, suppress fibroblast activation, and promote matrix degradation, thereby attenuating fibrosis and facilitating functional recovery.42,47
Across multiple small- and large-animal models, MSC- and EV-based approaches have consistently demonstrated antifibrotic, pro-angiogenic, and tissue-preserving effects in ureteral injury and reconstruction settings.44,48,49
In a rat model of chronic ureteral stricture, periureteral or intra-arterial delivery of bone marrow–derived MSCs attenuated fibrotic remodeling, as evidenced by reduced collagen deposition, downregulation of profibrotic markers such as TGF-β1 and α-SMA, and improved luminal caliber and urinary drainage. 48 Notably, MSC-derived extracellular vesicles produced comparable antifibrotic effects in the same model, with suppression of fibrosis-related gene expression and enhanced matrix-remodeling activity, supporting a predominantly paracrine mechanism of action.41,43,48
Large-animal reconstruction studies further support feasibility, including rabbit ureter repair using urine-derived MSCs seeded on vascular extracellular matrix scaffolds and tissue-engineered grafts constructed on decellularized bladder matrix, both demonstrating maintained patency with evidence of layered tissue regeneration.49,50
Taken together, these studies support paracrine signaling—rather than direct differentiation—as the dominant driver of MSC-mediated repair. 15 EVs, in particular, are now recognized to deliver critical regulatory miRNAs, cytokines, and enzymes that modulate the immune response, remodel fibrotic ECM, and stimulate endogenous stem/progenitor cell recruitment. 43
While these models do not fully replicate the chronic radiation-induced fibrosis seen in human RIUS, they provide strong proof-of-concept evidence that MSC-based interventions can overcome the hostile tissue environment of ureteral strictures. 48 Importantly, the observed efficacy in multiple delivery routes and across species underscores the translational potential of this approach.
Harnessing regenerative potential: MSCs and EVs in RIUS management
The promising results of MSC-based therapies in preclinical ureteral injury models have laid a solid foundation for future translation into the management of RIUS. In particular, MSC-derived EVs have emerged as a compelling therapeutic alternative, offering strong antifibrotic and pro-regenerative effects through paracrine signaling without the risks associated with live cell transplantation.41,47
Local delivery, such as periureteral injection or arterial infusion, has shown efficacy in animal studies by enabling targeted action in fibrotic segments. These methods are especially relevant in RIUS, where radiation-induced ischemia may limit systemic therapeutic access. Moreover, cell-free EV therapy allows for easier handling, storage, and standardization, potentially improving scalability and safety.
Despite these advantages, several challenges remain. Key issues include the standardization of MSC/EV production, optimization of timing and dosage, and the need for long-term safety evaluation in large-animal models. 44 Addressing these will be essential before widespread clinical application can be realized.
Nevertheless, the therapeutic potential of MSC and EV-based strategies in reversing ureteral fibrosis and supporting tissue regeneration is highly encouraging. These biologically active interventions may serve as powerful adjuncts to surgery—or possibly as stand-alone therapies—in carefully selected RIUS patients.
With continued progress in stem cell science, biomaterials, and delivery platforms, MSC-derived EV therapies represent a hopeful frontier in the treatment of radiation-induced urinary tract injuries. Future translational efforts may help redefine the standard of care for patients with complex, fibrosis-driven ureteral strictures.
Next-generation ureteral stents in the management of RIUS
Current research on ureteral stents focuses on two main directions.24,51–53
(1) Optimizing new materials for strength, compatibility, and safety (Figure 2).
(2) Developing drug-eluting stents with antifibrotic or antimicrobial functions.
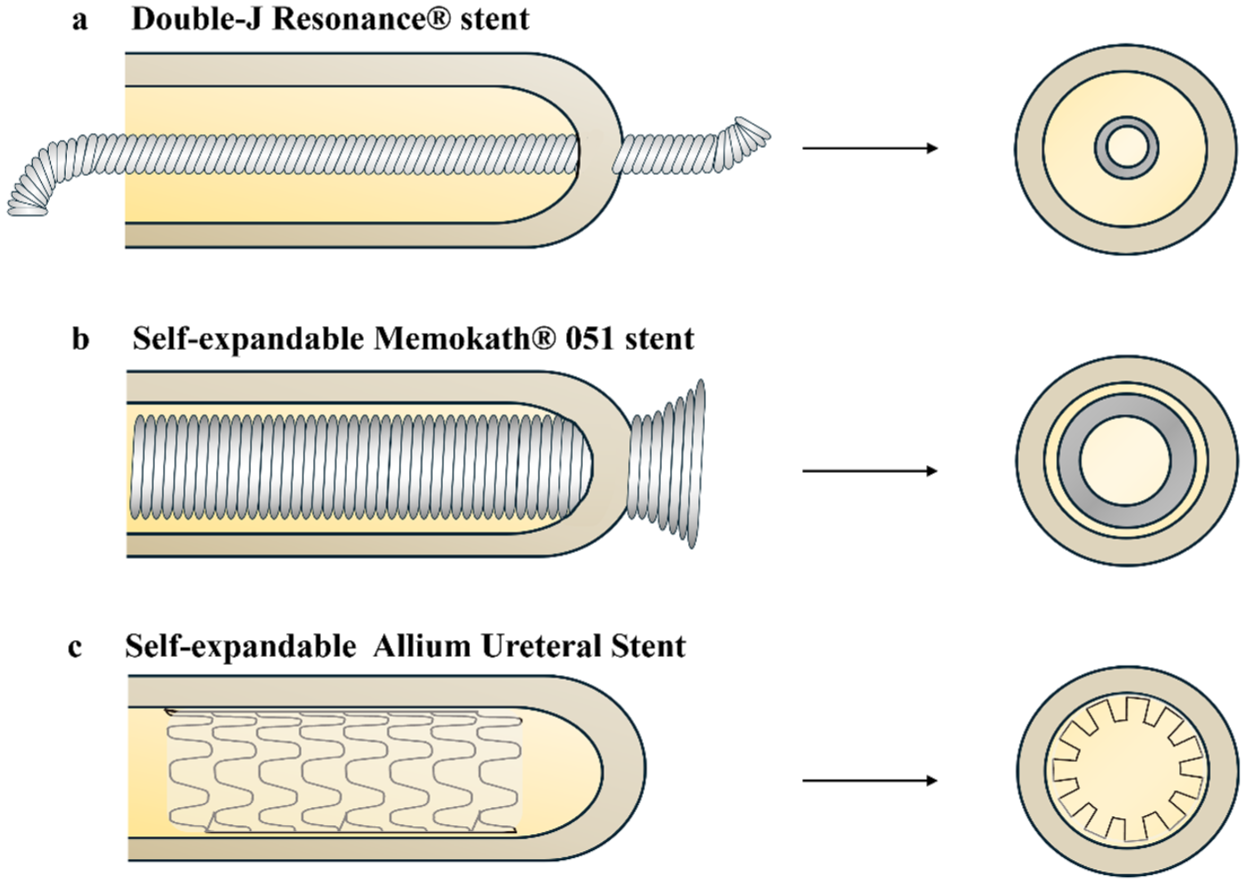
Appearance and insertion of novel metallic stents intended for RIUS. (a) The double-J shaped Resonance stent, made of a nickel–cobalt-based alloy, is inserted with the assistance of a guidewire and remains centrally positioned within the ureteral lumen after deployment. (b) The self-expanding Memokath stent is advanced over a guidewire and expands upon infusion of heated saline (55–60°C), achieving direct contact with the ureteral wall. (c) The self-expanding Allium stent features a metallic framework covered with a polymeric membrane; its large-caliber design facilitates full expansion and close apposition to the lumen.
Metal stents
Resonance®
Conventional silicone or polyurethane stents are often insufficient in RIUS because dense fibrosis and impaired vascular supply limit stent compliance and long-term drainage. Metallic ureteral stents, particularly nickel–cobalt–based devices such as Resonance® (American), provide greater radial force and prolonged indwelling potential and have therefore been evaluated in refractory ureteral obstruction.53,54
Across multiple clinical series involving both benign and radiation-associated strictures, Resonance® has demonstrated acceptable short-term patency but consistently inferior outcomes in irradiated ureters compared with non-radiation-related strictures, with prior pelvic radiotherapy repeatedly identified as a risk factor for stent failure and early replacement.27–29 Although reduced exchange frequency may offer economic advantages, these findings collectively indicate that enhanced mechanical strength alone is insufficient to overcome the fibrotic and ischemic microenvironment characteristic of RIUS.
Memokath™
Memokath™ (Denmark) is a thermo-expandable nickel–titanium ureteral stent that expands in situ upon exposure to heated saline, allowing firm anchoring within the stricture segment and atraumatic removal when required.55–57 Clinical reports including patients with radiation-induced ureteral strictures suggest that Memokath™ can maintain medium- to long-term patency in selected cases, although stent migration and infectious complications remain the principal limitations, particularly in complex or long-segment disease. These findings indicate that while Memokath™ may provide durable drainage in carefully selected RIUS patients, its efficacy remains constrained by underlying radiation-related tissue changes.
Allium®
Allium® (Israel) ureteral stents consist of a large-caliber, fully covered nitinol framework designed to resist external compression and prevent tissue ingrowth.58–60 Limited clinical series evaluating ureteral strictures following pelvic surgery or radiotherapy report that Allium® stents can provide sustained luminal patency, particularly as a long-term drainage option in patients unfit for definitive reconstruction. However, variable migration rates and inconsistent durability after stent removal have been reported, suggesting that in RIUS, Allium® stents may function primarily as a drainage solution rather than a means of restoring durable ureteral function.
Although metallic ureteral stents represent an important advancement in maintaining long-term patency, they remain fundamentally mechanical solutions and do not directly address the fibrotic and ischemic microenvironment characteristic of radiation-induced ureteral stricture.
Biodegradable stents
Biodegradable ureteral stents are designed to eliminate the need for secondary removal, reducing procedure-related morbidity, discomfort, and cost. They are typically composed of biodegradable polymers such as polylactic acid, polyglycolic acid, or polycaprolactone, which degrade through hydrolysis into nontoxic metabolites. 61 However, their widespread clinical adoption has been limited by key challenges: maintaining sufficient mechanical strength during the healing phase, ensuring predictable and uniform degradation, and avoiding fragment-related obstruction or inflammation.
In a porcine model, Uriprene, evaluated by Hadaschik et al., began degrading at week 3, with 60% degradation by week 7 and complete dissolution by week 10. 62 Soria et al. 63 studied the BraidStent, a biodegradable antireflux stent in patients undergoing pyeloplasty. It showed performance comparable to traditional double-J stents and degraded within 3–6 weeks without obstructive fragments. However, concerns were raised regarding bacterial colonization and long-term ureteral safety. HydroStent, a hydrogel-based degradable stent, has demonstrated promising anti-encrustation properties, but in vivo data remain scarce.
In the context of RIUS, these limitations become more pronounced. The fibrotic and avascular nature of irradiated tissue often demands prolonged drainage support, beyond the effective lifespan of current biodegradable stents. Moreover, the increased risk of infection and encrustation in RIUS further limits their clinical utility.
Nevertheless, biodegradable stents remain a promising avenue. Research is ongoing into composite or drug-eluting biodegradable platforms that could combine mechanical support with antifibrotic or antimicrobial properties. As materials science and fabrication technologies advance, customized, bioresponsive stents may eventually offer viable solutions for selected RIUS cases, particularly in early-stage or adjunctive settings.
Coated ureteral stents
In patients with RIUS, prolonged ureteral stenting is frequently complicated by bacterial colonization, biofilm formation, and encrustation, particularly in the context of impaired local immunity and reduced urinary flow. 64 To address these challenges, a variety of surface-modified ureteral stents have been developed, including metal ion–based coatings,64,65 polymeric and hydrogel layers,66,67 peptide-functionalized surfaces, 68 and nanostructured composites,52,69–71 all aiming to reduce bacterial adhesion and mineral deposition.
Preclinical studies have consistently demonstrated reduced biofilm formation and encrustation with these coatings in vitro and in animal models,65,66 and early clinical experiences suggest potential benefits in selected patients requiring long-term drainage.67–70 However, most coating strategies primarily target infection- and encrustation-related complications and do not directly modulate the fibrotic remodeling or ischemic injury that drives RIUS, limiting their capacity to alter disease progression in RIUS.25,26 As such, coated stents should be viewed as adjunctive measures to improve stent tolerance rather than definitive disease-modifying therapies. 72
Drug-eluting ureteral stents
Beyond antimicrobial coatings, drug-eluting ureteral stents (DES) represent a compelling strategy to address the fibrotic and inflammatory milieu underlying RIUS. Given that the pathogenesis of RIUS involves vascular damage, chronic inflammation, and excessive fibrosis, local delivery of therapeutic agents via stents may help delay or even reverse disease progression. Inspired by the clinical success of drug-eluting coronary stents, researchers have begun to explore this concept within the urinary tract, adapting materials and pharmacologic payloads accordingly. 52
In one of the earliest approaches, paclitaxel, a mitotic inhibitor known for its antiproliferative effects, was incorporated into biodegradable polymer coatings on polyurethane stents. In a rat model following ureterostomy, the drug-eluting stent was found to significantly suppress urothelial hyperplasia, reducing the risk of postoperative scar formation and luminal occlusion. 73
Similarly, Huang et al. developed a mitomycin C (MMC)-eluting stent by embedding the agent within a biodegradable polymer layer coated onto a polyurethane substrate. In vitro assays revealed that this platform achieved a controlled release of 10%–50% of MMC over a 28-day period, effectively inhibiting the proliferation of human bladder stromal fibroblasts. These findings underscore the feasibility of local chemotherapeutic modulation of ureteral fibrosis.
In more recent studies, pirfenidone, a small-molecule antifibrotic approved for idiopathic pulmonary fibrosis, has shown promise in urologic applications. Li et al. used pirfenidone in a rat model of urethral injury, showing that it attenuated fibrosis by inhibiting the TGF-β1 pathway and associated inflammatory responses. 15 Building on this, Jiang et al. engineered a novel pirfenidone-eluting ureteral stent by coating nanoparticles onto dopamine-modified polyurethane. 74 In vitro cell culture and rabbit ureteral stricture models demonstrated downregulation of TGF-β1 and reduced collagen deposition in the fibrotic ureteral wall, suggesting potential for slowing or preventing stricture development in RIUS.
The mTOR inhibitor sirolimus (rapamycin), widely used in transplant medicine and vascular stents, has also been evaluated. Ho et al., in a rabbit model of thermal ureteral injury, showed that sirolimus-eluting stents decreased luminal obstruction, suppressed type III collagen deposition, and downregulated TGF-β1 and EMT-related proteins over 4 weeks. 75 Similar results were reported in a YAG laser-induced ureteral injury model in rats, where sirolimus-coated stents effectively reduced fibrotic remodeling. 76 Further validation was provided by Chen et al. in a miniature pig model, where the use of a slow-release sirolimus-eluting stent led to measurable reductions in ureteral wall fibrosis after 4 weeks of placement.
Zhao et al. took this approach further by embedding sirolimus into poly(D,L-lactide) nanoparticles (PDLLA-NPs) and grafting them onto poly(L-lactide) (PLLA) stents using low-temperature plasma polymerization. 77 In vitro studies demonstrated that this formulation selectively inhibited smooth muscle cell proliferation while preserving the growth of endothelial cells, a desirable balance for maintaining patency while minimizing fibrosis.
These studies collectively suggest that drug-eluting ureteral stents can do more than simply drain urine—they may actively modify the local tissue environment to reduce inflammation, limit fibrotic deposition, and ultimately improve ureteral healing in challenging cases such as RIUS. While all current evidence remains preclinical, the integration of antifibrotic, antiproliferative, and anti-inflammatory agents onto stent platforms presents a promising avenue for next-generation stricture management.
Further clinical translation will require detailed investigations into optimal drug-release kinetics, biocompatibility under radiation-induced tissue stress, and long-term safety. Nonetheless, these experimental platforms offer renewed hope for improving outcomes in patients with recurrent or high-risk ureteral strictures where traditional stents or reconstructive surgery may fall short.
Discussion
RIUS represents a complex and underrecognized sequela of pelvic malignancy treatment, with clinical implications extending far beyond impaired urinary drainage. In addition to progressive renal dysfunction and associated metabolic disturbances, RIUS imposes a substantial psychological burden, particularly in patients undergoing multimodal oncologic therapy. A multicenter study by Li et al. demonstrated significantly reduced quality-of-life scores across physical, mental, and general health domains in patients with ureteral strictures, with anxiety and depressive symptoms being especially prevalent. 78 Recurrent urinary tract infections, nephrostomy dependence, declining renal function, and the psychosocial stress associated with malignancy likely contribute to this burden.
RIUS is also closely linked to oncologic outcomes. Progressive ureteral obstruction and hydronephrosis not only complicate urologic management but have consistently been identified as adverse prognostic factors in pelvic malignancies, particularly cervical cancer.79,80 Hydronephrosis has been shown to independently predict reduced survival, underscoring the importance of timely recognition and effective management of ureteral obstruction to preserve renal function and potentially improve cancer-related outcomes.
Beyond its mechanical consequences, RIUS can directly influence systemic cancer therapy. Renal dysfunction resulting from ureteral obstruction may preclude the use of nephrotoxic agents such as cisplatin, a cornerstone of concurrent chemoradiation in cervical cancer.81,82 Consequently, alternative strategies, including gemcitabine-based regimens or accelerated radiotherapy protocols, have been explored in selected patients to maintain oncologic efficacy while mitigating renal risk.
Although endoscopic and reconstructive urologic surgery has advanced considerably, conventional management strategies often provide only transient benefit in RIUS.10,34 The profoundly fibrotic, hypovascular, and poorly regenerative tissue environment characteristic of irradiated ureters severely limits tissue healing and contributes to high rates of restenosis, particularly in cases involving long-segment or bilateral disease. Importantly, these features render RIUS a paradigmatic model of severe fibrotic ureteral stricture, offering critical insights into why purely mechanical approaches frequently fail in advanced stricture disease.
Accordingly, increasing attention has been directed toward biologically oriented and regenerative strategies that seek to address underlying disease biology rather than luminal narrowing alone. MSC-based therapies and next-generation ureteral stents incorporating novel biomaterials or drug-eluting properties exemplify emerging approaches inspired by lessons learned from radiation-induced fibrosis. Although largely investigational, these strategies may inform therapeutic advances not only for RIUS but also for refractory ureteral strictures of other etiologies.
From a clinical perspective, these emerging strategies are also likely to occupy distinct therapeutic niches rather than serve as interchangeable solutions. Drug-eluting ureteral stents represent a pragmatic, immediately deployable platform that integrates mechanical support with localized antifibrotic or anti-inflammatory delivery, making them particularly attractive for patients requiring prompt restoration of urinary drainage. In contrast, biologically active therapies, including MSC-derived extracellular vesicle–based approaches, may be better suited as adjunctive or early-stage interventions aimed at modulating the fibrotic and hypovascular microenvironment, especially in selected patients with preserved renal function or recurrent disease. 52 Viewed through a translational lens, these approaches should be regarded as complementary rather than competing strategies, with their optimal integration likely to depend on disease stage, stricture extent, and patient-specific oncologic and renal considerations. Importantly, such distinctions remain conceptual at present and should be refined through prospective translational studies and clinical trials.
Despite their promise, several limitations currently constrain the clinical translation of biologically oriented and regenerative therapies for RIUS. Most available evidence derives from preclinical studies or small, early-phase clinical series, with limited long-term data on durability, safety, and renal functional outcomes. Critical questions regarding optimal delivery routes, dosing strategies, treatment timing, and patient selection remain unresolved. 44 In addition, challenges related to biomaterial biocompatibility, regulatory approval, and cost-effectiveness may further limit widespread adoption. Collectively, these considerations highlight the need for rigorous translational research and well-designed clinical trials to define the precise role of regenerative therapies within the broader management framework of ureteral stricture disease.
Regardless of the therapeutic approach employed, the delayed onset and progressive nature of radiation-induced injury necessitate long-term management beyond the initial intervention. Contemporary urological guidelines emphasize the importance of longitudinal surveillance, incorporating periodic imaging and renal functional assessment to detect recurrent obstruction or late deterioration, with follow-up strategies individualized according to disease severity, laterality, and baseline renal function.
Ultimately, therapeutic advances in ureteral stricture disease will likely depend on integrating anatomical reconstruction with biological modulation, informed by insights gained from radiation-induced fibrosis and guided by individualized, long-term management strategies aimed at achieving durable renal preservation.
Conclusion
Radiation-induced ureteral stricture represents a biologically hostile and treatment-refractory form of benign ureteral stricture disease. Accumulating evidence suggests that durable management is unlikely to be achieved through mechanical reconstruction or stenting alone, particularly in patients with long-segment or bilateral disease. Future therapeutic strategies will likely require an integrated approach that combines effective urinary drainage with targeted modulation of fibrosis and microvascular injury. Advances in drug-eluting stent platforms and MSC-derived extracellular vesicle therapies offer promising translational pathways and may, with further validation, redefine long-term management paradigms for RIUS and other severe fibrotic ureteral strictures.
