Abstract
With the widespread dissemination of robotic surgical platforms, pathology previously deemed insurmountable or challenging has been treated with reliable and replicable outcomes. The advantages of precise articulation for dissection and suturing, tremor reduction, three-dimensional magnified visualization, and minimally invasive trocar sites have allowed for the management of such diverse disease as recurrent or refractory bladder neck stenoses, and radiation-induced ureteral strictures, with excellent perioperative and functional outcomes. Intraoperative adjuncts such as near-infrared imaging aid in identification and preservation of healthy tissue. More recent developments include robotics via the single port platform, gender-affirming surgery, and multidisciplinary approaches to complex pelvic reconstruction. Here, we review the recent literature comprising developments in robotic-assisted genitourinary reconstruction, with a view towards emerging technologies and future trends in techniques.
Current platforms and novel instrumentation
The most widely disseminated robotic-assisted surgical platforms to date are those using multiple instrument arms, and consequently multiple incisions for ports, such as the Intuitive da Vinci Si, X, and Xi devices. While multi-port architecture remains the most accessible and familiar to urologists across a variety of practice settings, the need to triangulate the ports strategically to maximize instrument range of motion and avoid collisions is a limitation, especially in the narrow confines of the deep pelvis. The da Vinci single port (SP) platform addresses some of these concerns by directing three double-jointed, independent arms through a 2.5 cm multichannel trocar from a single incision. 1 Further advantages include the ability to rotate the entire system completely while docked, visual feedback on the surgeon console of the location of each arm, and the ease of performing concurrent procedures (for example, combined transabdominal and perineal dissection) due to the unobtrusiveness of the platform. 2
Apart from the immediate benefits of fewer required incisions, which correspond to less morbidity and improved cosmesis, as well as potentially more rapid convalescence, a variety of pelvic pathology can be approached with the SP system, including posterior urethral or bladder neck stenoses, rectourethral fistula, or vaginoplasty.3,4 The operating distance of 15–25 cm for the instrument arms to articulate is ideal for a transabdominal approach to the deep pelvis. Where the target anatomy is closer to the abdominal wall than this distance, or in a ‘floating dock’ or ‘air dock’ technique, using a GelPoint retractor and AirSeal effectively to create a surrogate for pneumoperitoneum (Figure 1) has been described and is simple to implement. 5 Further limitations of the SP platform include more limited instrumentation relative to its multiport counterparts, in particular lacking a vessel sealer or near-infrared fluorescence (NIRF)-mode camera at the time of this publication. While the double-jointed instruments facilitate dissection and suturing in narrow confines, less force can be applied correspondingly with an individual arm. Due to the need to triangulate the instrument arms from a single trocar, countertraction can be problematic, potentially necessitating the placement of an assistant trocar, or the use of external aids such as magnetic retractors.6,7 Nevertheless, with incremental improvements in the technology from both manufacturers and practitioners, further workarounds or solutions to these issues may be expected in the near future.

Single-port robotic-assisted pyeloplasty with use of a ‘floating dock’ technique in a pediatric patient.
Novel platforms produced by an array of manufacturers and industry-academic collaboratives are on the horizon for clinical deployment and may prompt a diversification of the current offerings, from which surgeons will undoubtedly benefit. 8 Of particular note, the Virtuoso Surgical/Vanderbilt concentric tube technology prototype, allowing for articulation of instruments from a fixed endoscope tip, is an exciting development which possesses striking implications for traditional lower urinary tract reconstruction owing to the potential for endoluminal surgery. 9 The data underpinning these novel techniques are eagerly awaited and herald a bright future for surgical innovation.
Posterior urethra and bladder neck pathology
Robotic-assisted approaches to recalcitrant or refractory posterior urethral stenoses were developed in response to the sheer technical difficulty of visualization and precise suturing in the deep pelvis, as well as the close proximity of critical structures including the external urinary sphincter, cavernous nerves, and rectum. Maneuvers such as pubectomy and combined abdominoperineal dissection may be required to facilitate anastomosis. The functional outcomes of open reconstruction, even when technically successful (as quantified by urethral patency), demonstrate a high rate of de novo stress urinary incontinence. 10 Furthermore, extensive urethral mobilization and bulbar artery transection are independently associated with an increased risk of artificial urinary sphincter (AUS) cuff erosion.11,12 Collectively, the morbidity of these historical approaches may discourage providers from attempting definitive surgical management, and consequently patients may be deemed ‘unreconstructible’, with the end-stage options of repeated endoscopic procedures, chronic catheter drainage, or cystectomy and urinary diversion.
In the past several years, novel techniques to address this pathology have been described. These procedures combine improved articulation and precision, with modalities such as NIRF imaging, to facilitate mucosal anastomosis with excellent short and mid-term results. Table 1 details the published series to date of robotic-assisted reconstruction of bladder neck or posterior urethral stenoses. For non-obliterative disease, a rectal dissection can be avoided by performing anterior dissection only, with scar incision and advancement of well vascularized tissue in the form of a bladder flap or buccal mucosal graft (BMG) onlay.13,14 Where the urethral lumen or vesicourethral orifice is completely obliterated, concomitant transrectal ultrasonography and flexible cystourethroscopy can facilitate circumferential dissection and excision of fibrotic tissue. For stenoses spanning the membranous urethra, combined robotic–perineal dissection can be used in the event distal urethral mobilization is required. 15 The aforementioned techniques are significantly easier to perform with the working space of the SP platform, which facilitates dissection and suturing under the pubic bone, and allows for concurrent endoscopic or transperineal manipulation (Figure 2). In terms of patency and continence, the short and mid-term outcomes reported thus far are highly encouraging. 16 Crucially, if a perineal dissection can be avoided, long-term durability of potential future AUS may be improved.
Review of recent literature encompassing robotic-assisted lower urinary tract reconstruction.
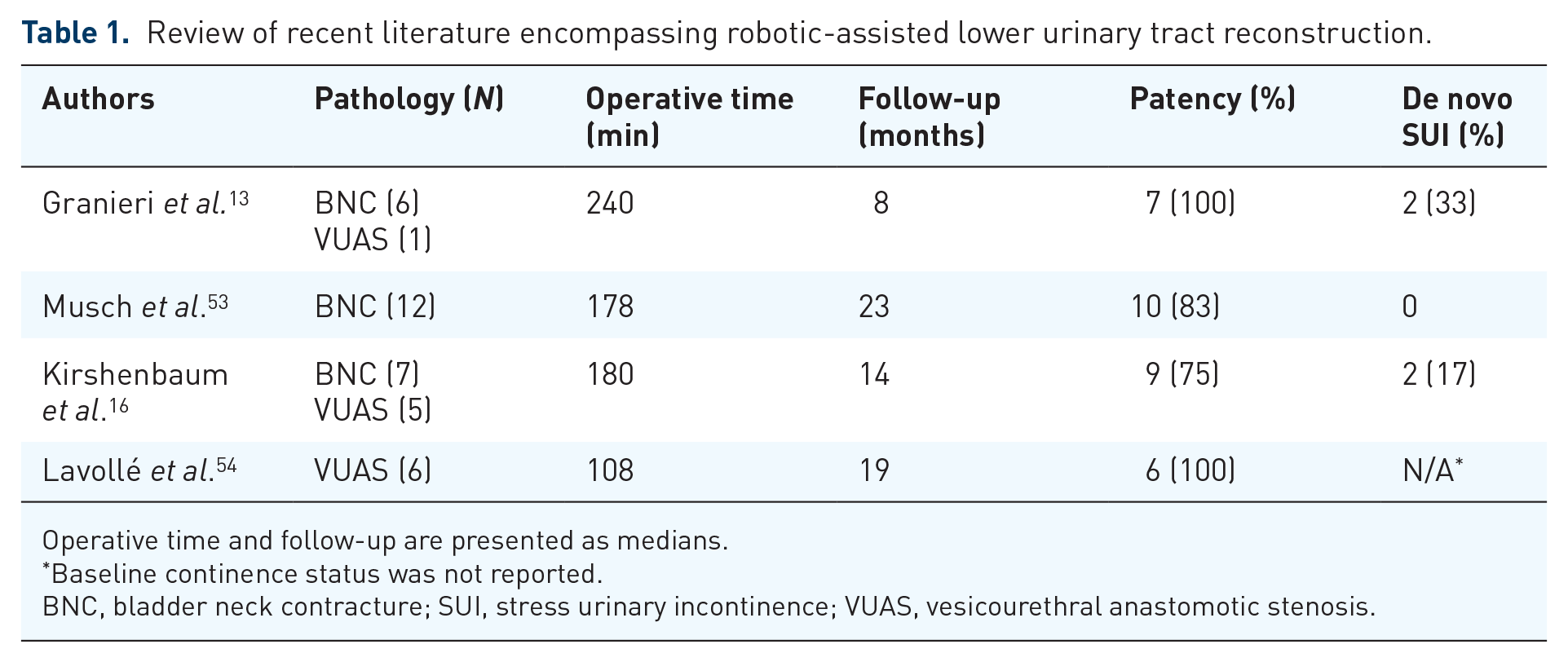
Operative time and follow-up are presented as medians.
Baseline continence status was not reported.
BNC, bladder neck contracture; SUI, stress urinary incontinence; VUAS, vesicourethral anastomotic stenosis.

Robotic-assisted approach to non-obliterative vesicourethral anastomotic stenosis (VUAS) via anterior cystotomy, with cystoscope light and wire visible, and laparoscopic-guided needle used in hydrodissection.
A similar approach may be taken for adjacent disease processes including rectourethral fistulae, or if salvage prostatectomy is clinically indicated. In these situations, the versatility of the robotic platform lies not only in the technical benefits it offers, but also in terms of adjunctive procedures such as the feasibility of minimally invasive flap harvest for vascularized tissue coverage, and the possibility of multiple surgical approaches that are performed concurrently. Further, comparative studies of the long-term durability of these techniques are needed, especially with respect to operating efficiency and potential cost. In addition, stenosis-specific factors lending weight to the selection of one treatment modality versus another need to be better delineated, to aid in the decision-making process (i.e. BMG onlay may not be as efficacious as bladder flap advancement in the setting of radionecrosis or dystrophic calcifications).
Intracorporeal urinary diversions
With the recent publication of the RAZOR randomized trial, in which robotic-assisted radical cystectomy was demonstrated to be non-inferior to open radical cystectomy in terms of oncological outcomes, the safety of the robotic approach was underscored. 17 Urinary diversion, however, was accomplished via an extracorporeal approach in this and other studies. The precision of robotic-assisted ureteroenteric anastomosis, as well as decreased blood loss, lower insensible fluid loss, and more prompt recovery are potential advantages of intracorporeal diversions. 18 Nevertheless, totally intracorporeal approaches have not been adopted widely to date, and ileal conduits constitute the majority of intracorporeal diversions. 19 The learning curve is thought to account for some of the lack of widespread utilization; in a large single-center comparison of open, robotic extracorporeal, and intracorporeal diversion, the anastomotic stricture rate was greatest for patients undergoing intracorporeal diversion, but after 75 cases, this rate declined to significantly less than either extracorporeal or open procedures (4.9%). 20 While operative volume influences operative time and the subsequent development of complications and readmissions, a reasonably high-volume robotic practice may have the appropriate infrastructure and personnel to support routine intracorporeal diversion. 21 This approach may be particularly applicable to the construction of orthotopic neobladder, as performing a tension-free, watertight urethroileal anastomosis may be a challenging step during open surgery. Multiple techniques have been described with the aim of maximizing intraoperative efficiency and teachability. The method first reported and consequently with the longest follow-up is from the Karolinska group, in which the urethroileal anastomosis is performed prior to bowel detubularization and reservoir creation. 22 However, several alternative orders of steps have been reported with similar short and mid-term data with respect to continence and stricture incidence. Larger studies with longer follow-up are warranted in order to define ideal parameters for this technically demanding operation.
The SP platform may also have a role to play in the realm of urinary diversion, not only in terms of reconstruction following radical cystectomy, as has been reported by Kaouk et al., 23 but also in populations with neurogenic or end-stage bladder. Grilo et al. 24 reported a series of 10 patients undergoing totally intracorporeal supratrigonal cystectomy with augmentation cystoplasty, in which the median operative time was 250 minutes, hospital stay was 12 days, and 1 year functional and urodynamic outcomes were acceptable. Conceivably, within a high-volume robotic surgical practice, as refinements of the technique accrue, the intracorporeal approach may fit into a niche that benefits both providers and patients.
Upper urinary tract reconstruction
Laparoscopic repair of ureteral obstruction, reported first in 1992, has not been widely adopted despite advantages in terms of morbidity and cosmesis compared to open approaches, likely due to a steep learning curve for proficiency and limited instrumentation for precise dissection and suturing. 25 These technical challenges become more apparent when confronting long strictures or when there is an irradiated or reoperative field. Multiple series have now demonstrated that robotic platforms address these concerns satisfactorily, and serve as a viable alternative to open procedures, with demonstrated advantages in terms of intraoperative blood loss, postoperative pain and hospital stay.26,27 The cornerstone of the technical successes reported to date leverage the advantages of enhanced articulation, three-dimensional vision, and the use of adjuncts such as indocyanine green (both intravascular, and for ureteral identification, intraureteral as in Figure 3) and flap interposition to facilitate the tension-free anastomosis of healthy, well vascularized tissues. 28

Identification of the stump of the proximal ureter (U) during robotic reconstruction with the aid of intraluminal indocyanine green (ICG). The ureter had previously been ligated during colectomy and ICG was instilled per nephrostomy tube in order to locate it.
While direct, head-to-head comparison of open versus robotic-assisted ureteral reconstruction may not be logistically feasible, the patency offered by robotic approaches is highly encouraging even in a hostile field. The multi-institutional Collaborative of Reconstructive Robotic Ureteral Surgery (CORRUS) group recently reported outcomes of ureteroplasty specifically for radiation-induced stenoses (32 patients, median length 2.5 cm) with 88% clinical and radiographic success at 13 months. 29 A quarter of the patients in that study were treated with an innovative side-to-side non-transecting anastomosis, with putative advantages of minimal posterior dissection, avoiding transection of the ureteral plate, and preservation of the native ureteral orifice. Slawin et al. 30 elaborated on this technique in a study of 16 patients with distal ureteral obstruction, demonstrating 94% success at 13 months with avoidance of potentially deleterious circumferential ureteral mobilization and dissection over the iliac vessels.
While ureteroureterostomy is ideal for the management of short unifocal stenoses, longer or more complex strictures traditionally required mobilization of either bladder flaps or downward nephropexy, or salvage procedures such as ileal ureteral interposition or renal autotransplant. For the treatment of mid and proximal ureteral strictures, intermediate term outcomes are now available. First described by Zhao et al. 31 in 2015, robotic ureteral reconstruction with BMG has matured as a treatment modality, with the most recent cohort of 54 patients achieving, for median stricture length of 3 cm, 87% success at 28 months with minimal perioperative morbidity. 32 Critically, a third of the patients in this cohort had failed prior ureteral reconstruction. The methodology reported by the CORRUS group incorporates stricture location, length and preoperative luminal patency. A longitudinal anterior ureterotomy preserves the posterior ureteral plate and is treated with a BMG as an onlay repair. For total obliteration, an augmented anastomotic reconstruction is used in which ischemic tissue is excised, healthy ureteral plate is anastomosed posteriorly, and BMG covers the anterior defect. To support the repair, vascularization can be assessed with ICG, and flaps derived from omentum or perinephric fat can be used for coverage. 33
A potential concern of ureteroplasty with BMG is harvest site morbidity, especially when treating long ureteral strictures. In response to this clinical need, Jun et al. 34 reported a series of 13 patients who were treated for long (median length 6.5 cm) right-sided ureteral strictures with robotic appendiceal onlay or interposition. After intraoperative verification of stricture length, the appendix was mobilized with confirmation of its vascular pedicle with intravenous ICG. Success was demonstrated in 92% of renal units at 15 months follow-up. For complete obstruction, appendiceal interposition was utilized; non-obliterative strictures were treated by detubularizing the appendix and performing ventral onlay (Figure 4). Notably, a third of the patients in this series had a history of pelvic radiation. While a long Boari flap could also have bridged similarly long defects, the resultant loss of functional bladder capacity may result in significant morbidity in this population.

Augmented anastomotic appendiceal onlay ureteroplasty for right proximal ureteral stricture. The ureteral plate (U) has been incised anteriorly for the onlay.
The potential for innovation in the space of robotic-assisted upper urinary tract reconstruction is high, owing to advances in instrumentation and familiarity with surgical approaches, as knowledge of the outcomes of these techniques is disseminated further. The SP platform has hitherto not been applied widely to ureteral reconstruction, likely due to less availability of the system and the lack of directly available NIRF imaging. However, recent reports have demonstrated promise with respect to the management of adult and pediatric ureteropelvic junction obstruction with same-day discharge. 35 An application of robotic technology which will likely become more accepted in the near future is the management of transplant ureteric strictures. Most recently, Kim et al. 36 described robotic-assisted treatment of distal stenoses in five patients (mean length 2.5 cm). While only short-term data are reported, all patients had successful outcomes, underscoring the reliability of the technique. While more follow-up is needed for this and other cohorts, robotic-assisted ureteral reconstruction will undoubtedly continue to evolve as a useful and facile addition to the reconstructive armamentarium.
Gender-affirming surgery
As reconstructive urologists play an increasingly more prominent role in the multidisciplinary care of patients seeking gender-affirming surgery, robotic-assisted approaches have come to the fore, prompted by the goals of minimizing morbidity while boosting the likelihood of successful reconstruction. Peritoneal Davydov flap augmentation of traditional penile inversion vaginoplasty, first reported via a robotic assisted approach by Jacoby et al. 37 in 2019, provides the advantages of creating a well vascularized neovaginal apex, additional length and depth of the neovaginal canal, anchoring to structures in the pelvis to mitigate the risk of neovaginal prolapse, and robotic-assisted rectoprostatic dissection. At mid-term follow-up, depth appears to be preserved, with negligible risk of rectoneovaginal fistula; the use of a SP platform renders a simultaneous robotic–perineal approach feasible and markedly reduces operative time as compared to multiport. 4 The technique has been applied to revision vaginoplasty as well since the potential hazards of revision surgery center on the rectal dissection. 38 Adjunctive measures such as intraoperative transectal ultrasound may be useful in this scenario.
For patients seeking phalloplasty, robotic-assisted vaginectomy results in reliable closure of the vaginal canal and can be combined with staged urethral reconstruction. Cohen et al. 39 described a technique for simultaneous vaginectomy and gracilis flap harvest. Via a transabdominal vaginotomy, the vaginal mucosa is dissected (Figure 5) and anteriorly is passed transperineally as a flap for tubularization. A gracilis flap buttresses this repair and is used to inset into the vaginal cavity to fill it. For the 16 patients in this cohort, at nearly 1 year follow-up, there was no development of urethrocutaneous fistula. The importance of complete removal of vaginal mucosa is underscored by a report from the same group on robotic-assisted excision of vaginal remnant in patients presenting with lower urinary tract symptoms following phalloplasty with urethral lengthening. 40 Especially for patients with distal urethral obstruction, pressurized urine can be forced into an incompletely closed vaginal canal, which presents as a vaginal remnant acting as a urethral diverticulum. Together with urethroplasty of the distal stenosis as warranted, the robotic-assisted approach demonstrates complete resolution of urinary symptoms at medium-term follow-up.

Robotic-assisted vaginectomy via transverse vaginotomy during gender-affirming staged phallic construction. The anterior vaginal mucosa has been delivered intraperitoneally to complete the distal dissection.
Vesicovaginal fistula repair
Specialists in urological reconstruction may be involved in the repair of vesicovaginal fistula (VVF), which can result from obstructed labor, iatrogenic injury, or as a complication of abdominopelvic radiation at rates reported between 0.3% and 2%. 41 When the fistula is located distally, a transvaginal approach with coverage by vascularized tissue is usually successful. 42 However, for supratrigonal VVF, or those involving the ureters, a robotic-assisted approach may have advantages over traditional open repair in terms of precise identification of unhealthy tissue, ability to treat synchronous ureteral pathology in the manners described above, and the ability to mobilize the bladder wall as well as the peritoneal or omental flap for interposition. First described in 2005, robotic-assisted VVF repair is now well established in the literature, with a comprehensive review by Randazzo et al. 43 of 19 studies demonstrating nearly a 100% resolution rate especially with more recent publications. Further delineation of the ideal flap for interposition, as well as the timing of robotic-assisted fistula repair, is needed.
Novel applications of robotic-assisted reconstruction
Multidisciplinary approaches to complex pathology, including genital lymphedema, abdominal wall and perineal hernias, and pelvic flap coverage may warrant urological expertise, to which robotic-assisted modalities are a welcome addition. An array of minimally invasive iterations of previously highly morbid or technically challenging open operations has emerged specifically to address these clinical entities. For example, the mobilization of omentum for vascularized lymph node transfer historically was performed via laparotomy. Frey et al. 44 reported a technique for omental free flap harvest in which the desired tissue is mobilized off the transverse colon greater curvature of the stomach while preserving its blood supply. Intraoperative ICG confirms the direction of dissection and the viability of the resultant free flap, which is then delivered to the recipient vascular bed extracorporeally. While the aid of a microvascular surgeon is needed for the microvascular anastomosis, the robotic flap harvest can be accomplished with either the multiport or single port platform.
A pedicled rectus abdominis flap, which similarly occasions significant morbidity if performed open due to the need for a large abdominal scar and potential for hernia, can be harvested robotically as described by Hammond et al. 45 The application here is in the setting of abdominoperineal resection, to provide vascularized coverage of dead space: via a peritoneal incision the posterior rectus sheath alone can be opened, the rectus body separated from the anterior sheath and mobilized to the level of the inferior epigastric artery, yielding a long muscle flap which easily reaches the deep pelvis. One potential application of rectus flaps is the management of perineal hernias. Traditional open mesh-based repairs of this entity are technically demanding and suffer from a high rate of recurrence. A putative advantage of a robotic-assisted approach, first described in a case report by Rajabaleyan et al., 46 is the relative ease of precise suturing with magnification in the pelvis. Similarly, robotic-assisted management of ileal conduit parastomal hernias has been reported with excellent short-term outcomes and no erosion of mesh into the bowel segment, or need for stoma re-siting. 47 For these and other pathologies, the intersection of urology, colorectal surgery and plastic surgery will continue to yield novel and efficacious surgical approaches which can be adapted and improved via robotic techniques. Table 2 demonstrates benchmark procedures described in this review.
Benchmark reconstructive procedures discussed in this paper.

BNC, bladder neck contracture; MP, multiport; SP, single port; VUAS, vesicourethral anastomotic stenosis.
Training and education
While learning curves for certain benchmark procedures such as robotic radical prostatectomy or cystectomy are established, as discussed previously, structured curricula and training models as they apply specifically to reconstruction have not yet been published. Dual-console based systems may lend themselves to real-time feedback and technical guidance. 48 However, the SP platform does not presently feature this. Studies to date suggest that the learning curve for most procedures (albeit in expert hands) may be similar to that of multiport robotic surgery.2,49 The cognitive burden of operating the SP platform has also been evaluated, with findings again comparable to more established approaches. With further dissemination of various techniques and approaches for robotic-assisted reconstruction, standardizing the training and grading of surgeons will become paramount – as well as the environment in which learning occurs. 50
Limitations of robotic platforms
In addition to the need for standardized training, currently available robotic platforms have both device-inherent and systemic limitations. In particular, lack of haptic feedback and prolonged operative times, especially early in the learning curve of any robotic-assisted procedure, may pose impediments to efficient operations. While no studies to date have specifically evaluated whether tactile feedback is in fact necessary to attain robotic surgical proficiency, future iterations of these platforms may include such functionality and may improve safe tissue handling. 51 A further gap in the evidence base is the lack of comparative analyses directly evaluating robotic versus open procedures, obscuring exact comparison of complications and outcomes, not to mention downstream considerations such as reimbursement and overall financial impact to healthcare systems. Nevertheless, trials such as RAZOR are an excellent model to follow for future investigation. In particular, the reconstructive surgical space lends itself to a toolbox or armamentarium mentality: surgeons avail themselves of techniques and instrumentation with which they are most proficient, and which can achieve the desired operative outcome as efficiently as possible. As more surgeons adopt robotic-assisted procedures in their practice, critical questions may include the possibility of degraded open surgical expertise, reduced cost-effectiveness and uncertain comparison of outcomes. 52 While the initial investigations detailed in this review are promising, only head-to-head comparative analysis will elucidate the overall value of robotic platforms in reconstructive urological procedures.
Conclusion
As robotic proficiency becomes more widespread, analogues and extensions of open procedures and operative maneuvers, previously perceived as technically challenging, will become more commonplace. Robotic-assisted procedures carry putative advantages in terms of anastomotic patency, identification and preservation of blood supply, and reduced perioperative morbidity. The role of the reconstructive expert who can leverage both robotic and traditional techniques in the management of complex upper and lower urinary tract pathology will be at the forefront of multidisciplinary management.
Footnotes
Author contributions
All persons who meet authorship criteria are listed as authors, and all authors certify that they have participated sufficiently in the work to take public responsibility for the content, including participation in the concept, design, analysis, writing, or revision of the manuscript. Furthermore, each author certifies that this material or similar material has not been and will not be submitted to or published in any other publication.
Conception and design of study: Shakir NA, Zhao LC
Data analysis and/or interpretation: Shakir NA, Zhao LC
Drafting of manuscript and/or critical revision: Shakir NA, Zhao LC
Approval of final version of manuscript: Shakir NA, Zhao LC
Conflict of interest statement
The authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
