Abstract
Background:
Cladribine (CLAD) stands as an oral disease modifying treatment (DMT) for multiple sclerosis (MS) patients, distinguished by its unique dosing regimen and mechanism of action. However, real-world data on its effectiveness remain limited, particularly regarding the clinical and therapeutical management beyond the 2-year treatment schedule.
Objectives:
The aim of our study was to explore the effectiveness profile of CLAD in individuals with MS (pwMS). We assessed the proportion of patients achieving no evidence of disease activity (NEDA-3) status and identified variables associated with better outcomes.
Design:
In this retrospective study, we collected clinical and magnetic resonance imaging (MRI) data of MS patients across 10 MS Clinics in Central Italy who started CLAD between 2018 and 2023.
Methods:
We evaluated the annualized relapse rate (ARR) during treatment, and the proportion of patients who experienced relapses, radiological activity, and confirmed disability progression. Additionally, we estimated the proportion of patients achieving NEDA-3 among those with a minimum follow-up of 3 months and explored baseline variables associated with NEDA status.
Results:
We collected data from 1094 patients with a mean follow-up of 25.1 months, of whom 79% completed the second CLAD cycle. The mean age was 37.7 years (SD 9.7), and the mean disease duration was 6.5 years, with 40.5% being treatment naïve. Despite a significant reduction of the ARR from 0.91 to 0.04 (p < 0.01) following CLAD treatment, 8.9% of patients presented at least one relapse, while 22.0% and 7.9% of patients experienced radiological activity or disability progression, respectively. Across the entire study cohort, 70.2% of patients maintained the NEDA-3 status. Younger age (HR = 0.98, p < 0.001) and higher expanded disability status scale score (HR = 1.11, p = 0.049) were associated with a higher risk of not achieving the NEDA-3 status. Additionally, we included 131 patients who were older than 50 years at the time of CLAD initiation. Among the cohort, 116 patients switched to another DMT after CLAD, primarily anti-CD20 monoclonal antibodies following disease reactivation.
Conclusion:
This postmarketing experience confirms the effectiveness of CLAD in the treatment of pwMS, with a significant reduction in ARR and a high proportion of patients remaining free from disease activity. By contrast, some patients required an escalation strategy mainly with anti-CD20 monoclonal antibodies because of persisting disease activity.
Introduction
Multiple sclerosis (MS) is the most frequent demyelinating Central Nervous System (CNS) disorder and the second leading cause of permanent neurological disability in young adults, following traumatic injuries.1,2 Neuroinflammation and neurodegeneration together contribute to MS-specific pathological processes, respectively, leading to subacute potentially reversible worsening and chronic permanent disability.3–5 Although the classification of MS phenotype continues to evolve, the traditional distinction remains between two main categories: relapsing multiple sclerosis (RMS) with more frequent relapses and accumulation of new/enlarging MRI lesions and progressive multiple sclerosis marked by chronic disability accumulation largely independent from relapses.6,7 Several disease-modifying treatments (DMTs) are approved for RMS, having demonstrated effectiveness in reducing the annualized relapse rate (ARR) and the formation of new focal CNS lesions on MRI. 8 However, their impact on disability outcomes remains more limited. 9 Each DMT has unique targets, specific targets within the immune system, implying different mechanisms of action and treatment regimens. 10
Cladribine (CLAD) is an oral DMT for MS patients with unique dosing schedule and mechanism of action. CLAD is a synthetic chlorinated deoxyadenosine analog that is biologically active in selected cell types and provides targeted reduction of circulating T and B lymphocytes. The treatment schedule encompasses two treatment courses separated by 12 months for a total dosage of 3.5 mg/kg. Each treatment course is given over four to five consecutive days in the first month, followed by an equivalent dose over four to five consecutive days in the second month. This treatment schedule leads to a short-term selective immune reconstitution therapy. 11
CLARITY and CLARITY EXTENSION trials compared CLAD treatment with placebo demonstrating the efficacy of this therapeutic approach in reducing the ARR and the accumulation of new brain MRI lesions.12,13 Despite the growing real-world data on CLAD’s effectiveness, the identification of optimal treatment candidates and the management of patients beyond the 2-years treatment schedule remain controversial and insufficiently documented.14–17
In this retrospective study, we report clinical and radiological data from a very large multicenter real-world cohort from Central Italy including over one thousand people with MS (pwMS) treated with CLAD.
Materials and methods
Study design
This is an independent, multicenter, retrospective, postmarketing study including people with RMS starting CLAD and regularly attending ten MS outpatient clinics in Central Italy (S. Andrea Hospital, S. Camillo-Forlanini Hospital, Policlinico Umberto I, Fondazione Policlinico A. Gemelli IRCCS, Policlinico Tor Vergata, S. Filippo Neri Hospital, San Pietro Fatebenefratelli Hospital, Rome; IRCCS Neuromed, Pozzilli, Fabrizio Spaziani Hospital, Frosinone; Santa Maria Goretti Hospital, Latina). 18 Clinical and MRI data were collected by each MS center following the local medication monitoring plan and hospital guidelines. Given the specific nature of the protocol, the study did not interfere in the care received by patients.
Participants
We retrospectively included all RMS patients starting CLAD as first treatment (treatment-naïve) or after switching from other DMTs. As per CLAD technical data sheet, each patient took one or two cladribine 10-mg tablets daily over 4–5 days in either weeks 1 and 5 of years 1 and 2 (to give a cumulative dose of 3.5 mg/kg). Baseline characteristics collected included sex, age, time since first symptom, EDSS score, relapses in the previous year, absence/presence of Gd+ lesions, and previous treatment history (naïve or switchers). Switching patients were further divided into platform and highly effective DMT groups. Platform DMT group included patients switching from interferons, glatiramer acetate, teriflunomide, and dimethyl fumarate. Highly effective DMT group comprised patients switching from fingolimod, siponimod, natalizumab, ocrelizumab, alemtuzumab. Patients who received the first treatment prescription by one MS center but continued follow-up elsewhere, were excluded.
Follow-up assessments
Despite the multicenter study design, monitoring and management of patients with RMS on disease-modifying treatment are highly standardized in Central Italy. 19 For each patient, clinical visits were scheduled every 6 months after treatment start and included disability scoring using the EDSS performed by certified neurologists (www.neurostatus.net). 20 Each patient underwent brain and spinal cord MRI scans at baseline, every 6 months in the first year of treatment and on a yearly basis thereafter according to standardized procedures. 21 MRI scans were performed using 1.5T or 3.0 magnets before and after gadolinium with <5-mm slice thickness and included axial T2 (proton density)-weighted fast spin echo, fluid-attenuated inversion recovery sequences, and T1-weighted spin echo post–Gd administration sequences (for brain imaging), and sagittal and axial T2-weighted fast spin echo, T2-short tau inversion recovery, and T1-weighted spin echo post–Gd administration sequences (for spinal cord imaging). 21
MRI images were locally reviewed by each involved center. In the case of atypical or uncertain images, the findings were discussed with the most experienced investigators of each center jointly. Additionally, unscheduled visits, laboratory exams, and/or MRI scans were also performed in case of relapse or any other clinically relevant condition, including adverse events.
Efficacy measure definitions and safety data
For efficacy outcomes, a minimum of 3 months of follow-up was required to reduce a potential bias including patients with short follow-up and consequently, a lower probability of disease activity occurrence. The primary clinical effectiveness outcomes included the annualized relapse rate (ARR), the number of patients experiencing a relapse, the mean change of EDSS, and the number of patients with disability worsening. A relapse was defined as any new neurological symptom, not associated with fever or infection, lasting for at least 24 h and accompanied by new neurological signs. 18 ARR was calculated by dividing the number of relapses by the number of years on drug for each patient. Confirmed disability worsening (CDW) was defined as 1.5-point increase (if baseline EDSS score was 0), 1.0-point increase (if baseline EDSS score was <5.5), or 0.5-point increase (if baseline EDSS score was ⩾5.5) confirmed 6 months apart. 22
CDW was further divided into relapse-associated worsening (RAW) or independent from a clinical relapse (PIRA) if CDW occurred because of or not related to a relapse, respectively. 23
Radiological outcomes were represented by the presence of Gd+ lesions on T1-weighted images and/or the occurrence of new hyperintense lesions on T2-weighted images (compared to baseline scan) after checking the Fluid Attenuated Inversion Recovery (FLAIR) and Short-time Inversion Recovery (STIR) sequences for brain and spinal cord, respectively.
As a composite effectiveness outcome, we evaluated the proportion of patients who maintained NEDA-3 status, a combined measure defined as absence of clinical relapses, disability worsening, and radiological activity. 24
Considering the specific CLAD treatment schedule, we collected data on the eventual use of CLAD or other DMTs after the 2-year treatment course.
Regarding safety aspects, we recorded complete blood count and blood chemistry test data to evaluate the prevalence of lymphopenia or elevation of liver function enzymes. As per the drug’s technical data sheet, CBC and liver function enzymes are required at least 2, 6, and 12 months after the start of each treatment cycle. 25 Any alterations were classified according to the Common Terminology Criteria for Adverse Events. 26
Statistical analysis
Continuous variables were described as mean ± standard deviation unless otherwise specified. Dichotomic or categorical variables were expressed as frequencies.
ARR on CLAD treatment was calculated dividing the total number of relapses by the total duration of follow-up. Eventual differences between ARR in the year before enrollment and on CLAD treatment was evaluated through a Poisson regression model.
Through a Cox proportional hazards model (stratified by center and including the single patient follow-up duration), we investigated which baseline (i.e., at treatment start) variables were associated with the occurrence of clinical activity, expressed as at least one relapse or confirmed disability progression, and of radiological activity, defined as the occurrence of Gd+ lesions on T1 weighted images or new hyperintense lesions on T2 weighted images compared to baseline scan. Lastly, we performed the same Cox proportional hazards model to explore baseline features associated with the maintenance of the NEDA-3 status. We then performed a logistic binary regression to explore the eventual presence of variables associated with the risk of developing grades 3–4 lymphopenia or infections during follow-up.
All two-tailed p-values < 0.05 were considered as significant, without correction for multiple comparisons considering the exploratory study design. Data were analyzed by using the Statistical Package for Social Sciences, version 22.0 (IBM SPSS, Inc., Chicago, IL, USA).
Results
Participants
From April 2018 to December 2023 (with the database closed in March 2024), 1094 patients started CLAD. After excluding 62 patients with an observation period less than 3-months, 1032 patients were eligible for effectiveness analysis. Table 1 shows the baseline demographic and clinical characteristics of the cohort. As expected, there was a female predominance with 834 women included (76.2%). Mean age and mean disease duration were 37.7 (interquartile range (IQR) 30–45) and 6.53 (IQR 1–10) years, respectively. Median EDSS score was 2.0 (IQR 1–3). Relapse rate during the year before starting CLAD was 0.91 (95% CI 0.86–0.98). Only 285 (26.1%) patients were relapse-free in the previous year, with nearly half of the patients experiencing a single relapse in the year before starting CLAD (n = 619, 56.6%), and only a minority with two or more relapses in the same timeframe (n = 189, 17.2%). Baseline brain and spinal MRI documented a mean of 0.83 (range 0–11) Gd+ lesions. Mean observation period after CLAD initiation was 25.2 months (IQR 12–36).
Baseline features.
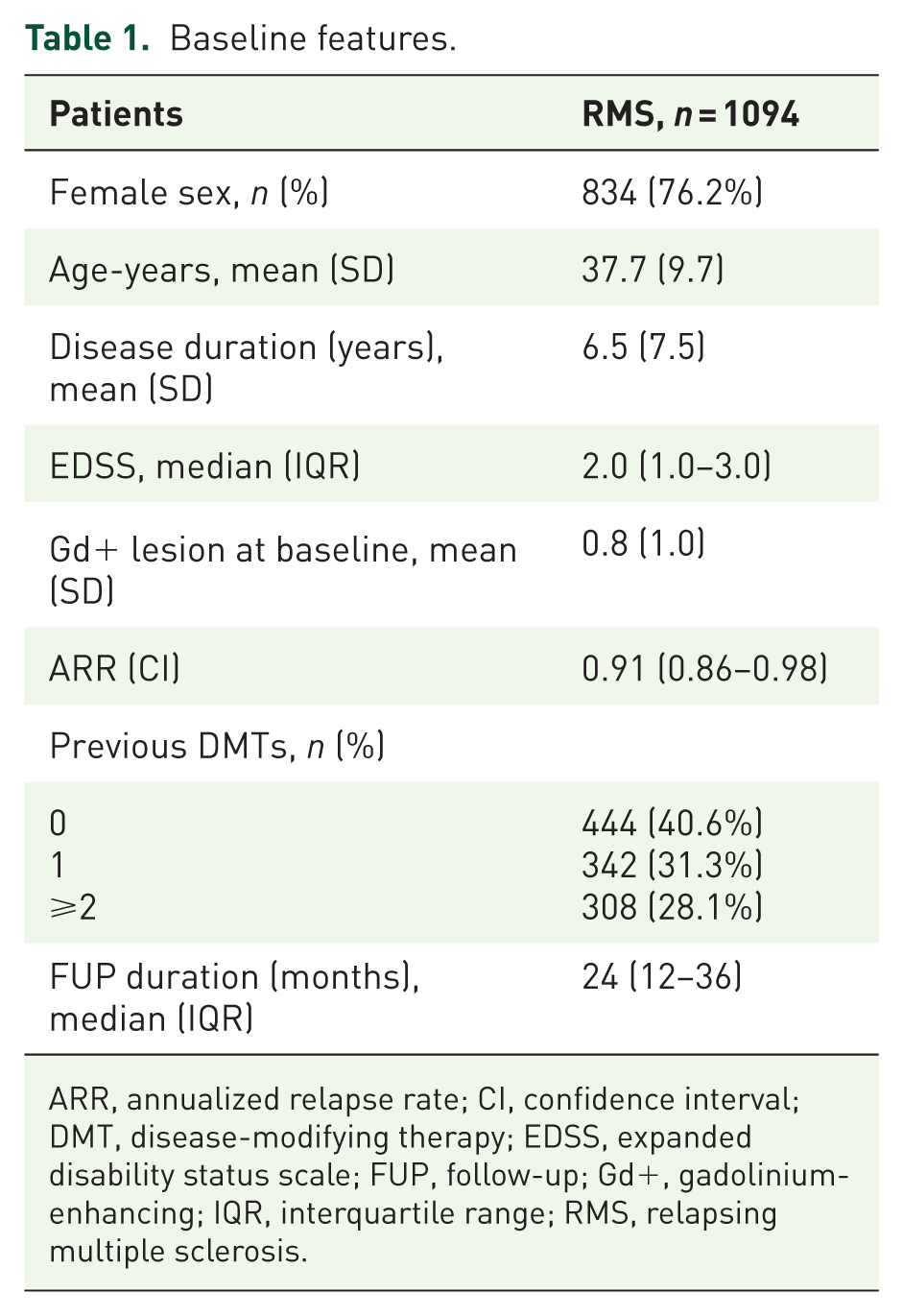
ARR, annualized relapse rate; CI, confidence interval; DMT, disease-modifying therapy; EDSS, expanded disability status scale; FUP, follow-up; Gd+, gadolinium-enhancing; IQR, interquartile range; RMS, relapsing multiple sclerosis.
A total of 444 patients (40.6%) started CLAD as their first DMT, while the remaining patients (n = 650, 59.4%) switched from other DMTs, primarily from other oral agents, including 275 (25.1%) patients who switched after suspension of dimethyl fumarate (detailed information in Table 2). In nearly half of the switchers (n = 342, 52.6%), CLAD represented the first switching DMT. The main reasons for discontinuation of previous treatments were lack of efficacy (n = 457, 70.3%), poor tolerability (n = 85, 13.1%), and safety concerns (n = 80, 12.3%).
Last DMT.

DMT, disease-modifying therapy.
Effectiveness outcomes
The ARR decreased from 0.91 (95% CI 0.86–0.98) to 0.04 (95% CI 0.03–0.05) with a relative reduction of 95.3% (95% CI 94.2–96.2, p < 0.01) (Figure 1(a)). During follow-up, 91 patients (8.9%) presented at least one relapse, with the mean time to first relapse being 16.8 (SD ± 17.7) months (Figure 2(a)). The occurrence of at least one relapse during CLAD treatment was associated with younger age (HR = 0.96, 95% CI 0.94–0.99, p < 0.01) and female sex (HR 2.30, 95% CI 1.26–4.20, p < 0.01) (Supplemental Table 1).

Histograms showing the ARR at the start cladribine and during follow-up. In the overall cohort (a), the ARR significantly decreased from 0.91 to 0.04 (p < 0.01). In the treatment-naïve group (b), the ARR decreased from 1.29 to 0.04 (p < 0.01). In the platform and highly effective DMT switch groups, we found a significant ARR reduction from 0.72 to 0.04 (p < 0.01) and from 0.44 to 0.06 (p < 0.01), respectively. Platform DMT group included patients switching from interferons, glatiramer acetate, teriflunomide, and dimethyl fumarate. Highly effective DMT group comprised patients switching from fingolimod, siponimod, natalizumab, ocrelizumab, and alemtuzumab.

Survival curve showing the occurrence of clinical and radiological activity during follow-up. Clinical outcomes included time to first relapse (a) and time to confirmed disability worsening (b), while the radiological outcome was the first occurrence of any new/enlarging T2 lesion or gadolinium enhancing lesion (c) on follow-up MRI. As a composite outcome, we reported the occurrence of any clinical or radiological disease activity (d).
Eighty-two (7.9%) patients experienced a 6-month CDW with a mean time to CDW of 16.2 (SD ± 10.6) months (Figure 2(b)). Older age at CLAD initiation (HR 1.03, 95% CI 1.00–1.06, p = 0.025), and higher baseline EDSS score (HR 1.23, 95% CI 1.03–1.47, p = 0.02) were associated with CDW (Supplemental Table 2). In our cohort, PIRA was more frequent than RAW with 60 (73.2%) of CDW events categorized as PIRA.
At follow-up, 227 (22.0%) patients presented radiological activity, defined as the occurrence of Gd+ or new T2/FLAIR lesions compared to baseline scan (Figure 2(c)). The mean time to first radiological activity was 12.7 (SD ± 12.1) months. Radiological activity during CLAD treatment was associated with younger age (HR = 0.96, 95% CI 0.95–0.98, p < 0.001) (Supplemental Table 3).
During follow-up, 308 (29.8%) patients showed evidence of any disease activity, resulting in the loss of NEDA3 status (Figure 2(d)). The mean time to loss of NEDA3 status was 13.1 (SD ± 11.4) months. Lastly, higher risk of the occurrence of any clinical and/or radiological activity during follow-up was associated with younger age (HR = 0.98, 95% CI 0.96–0.99, p < 0.001) and higher EDSS score (HR = 1.11, 95% CI 1.00–1.23, p = 0.049) (Table 3).
Cox regression models (stratified by MS center) to achieve NEDA-3 status.

95% CIs, 95% confidence intervals; EDSS, Expanded Disability Status Score; Gd+, gadolinium enhancing; HR, hazard ratio; MS, multiple sclerosis; NEDA-3, no evident disease activity. In bold p value <0.05.
Naïves versus switchers
Table 4 shows the baseline demographic and clinical characteristics of the cohort divided into three groups based on the previous exposure to DMT.
Baseline features based on previous DMT exposure.
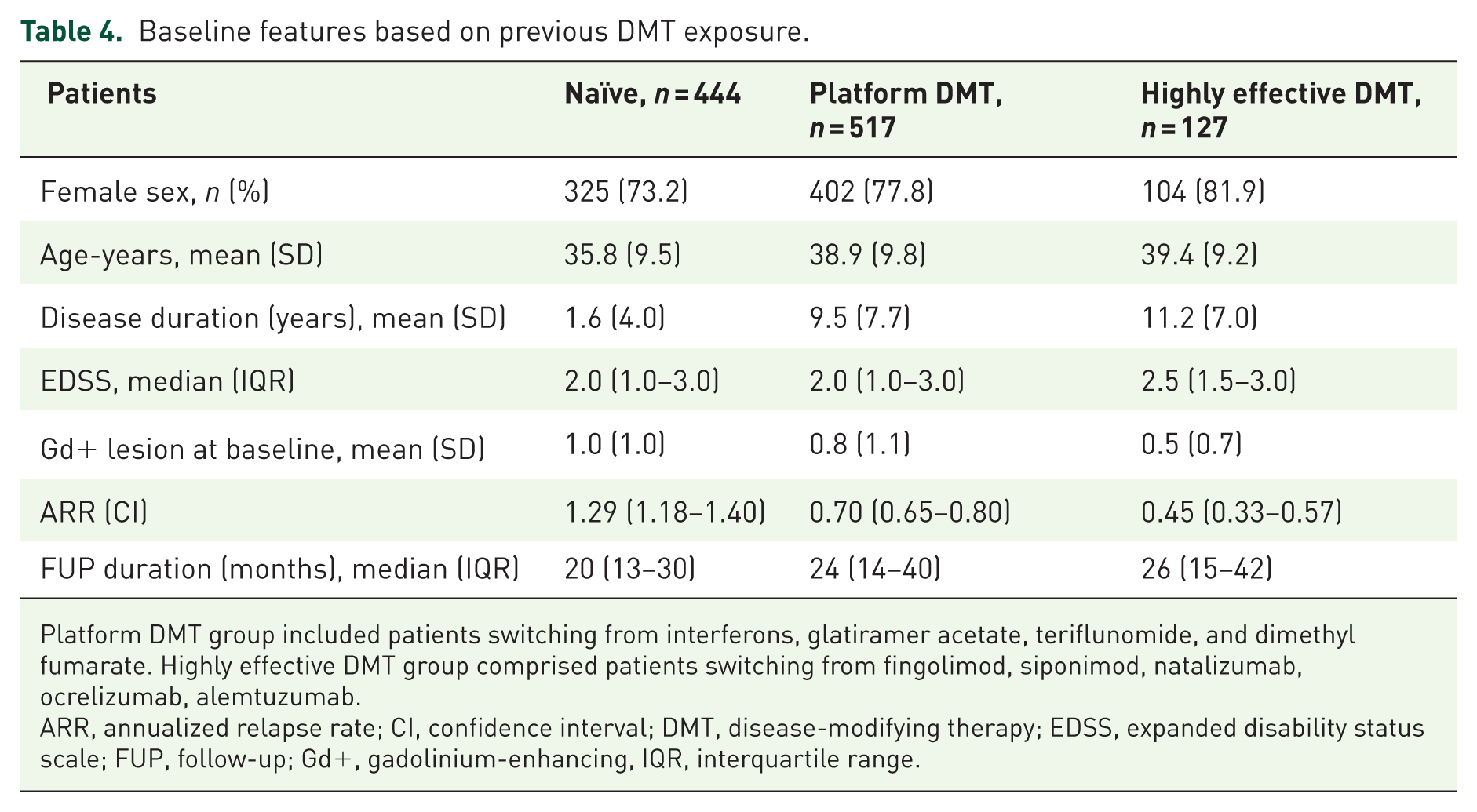
Platform DMT group included patients switching from interferons, glatiramer acetate, teriflunomide, and dimethyl fumarate. Highly effective DMT group comprised patients switching from fingolimod, siponimod, natalizumab, ocrelizumab, alemtuzumab.
ARR, annualized relapse rate; CI, confidence interval; DMT, disease-modifying therapy; EDSS, expanded disability status scale; FUP, follow-up; Gd+, gadolinium-enhancing, IQR, interquartile range.
In the treatment-naïve group, the ARR decreased from 1.29 (95% CI 1.18–1.40) to 0.04 (95% CI 0.03–0.06) with a relative reduction of 97.0% (95% CI 95.7–97.9, p < 0.01) (Figure 1(b)). Thirty patients (7.2%) presented a new relapse while 25 (6.0%) experienced a CDW event. Radiological activity was the most frequent disease activity occurring in 78 (18.7%) patients. Aggregating these effectiveness outcome measures, 23.7% naïve patients presented any disease activity during follow-up (Supplemental Figure 1).
The switch cohort was divided into two groups: platform DMT switchers and highly effective DMT switchers. In the platform DMT switch group, we found a significant reduction in the ARR from 0.72 (95% CI 0.65–0.80) to 0.04 (95% CI 0.03–0.05) with a relative reduction of 94.4% (95% CI 92.4–95.9%, p < 0.01) (Figure 1(b)). Any relapse or CDW event occurred in 44 (9.1%) and 45 (9.3%) patients, respectively. Radiological activity was documented in 117 (24.1%) patients resulting in 162 (33.4%) patients with evidence of disease activity during the observation period (Supplemental Figure 1).
A significant reduction of ARR was also observed in the highly effective DMT group. ARR decreased from 0.44 (95% CI 0.33–0.57) to 0.06 (95% CI 0.04–0.10) with a relative reduction of 86.2% (95% CI 76.5–91.9%, p < 0.01) (Figure 1(b)). Forty-four patients (35.8%) experienced any disease activity with radiological activity (n = 30, 24.4%) being the most common. Additionally, 16 (13.0%) patients experienced a new relapse, and 10 patients had a (8.1%) CDW event (Supplemental Figure 1).
Patients with an observation period ⩾24 months
We performed a subanalysis including patients with a minimum follow-up of 2 years evaluating the percentage of patients with clinical and radiological activity within the first 24 months after starting CLAD.
We included 508 patients (46.4%) whose baseline characteristics were comparable to the overall cohort: the mean age was 37.2 years, the mean disease duration was of 6.8 years, the median EDSS score was 2.0, and the mean baseline ARR was 0.9. Females accounted for 66.3% of the cohort, with 33.7% being treatment-naïve.
As for the whole cohort, ARR significantly decreased from 0.90 (95% CI 0.82–0.98) to 0.04 (95% CI 0.03–0.05). The most frequently detected disease activity was the presence of Gd+/new T2 lesion on follow-up MRIs in 126 patients (24.8%). Forty-three (8.5%) patients presented at least one relapse, while 38 patients (7.5%) experienced a CDW. Within the first 24 months of follow-up, 163 patients showed any disease activity thus resulting in a 2-year NEDA3 status of 67.9% (Figure 3).

Proportion of patients who remained free from clinical relapse, CDW, and MRI activity, respectively, with a minimum follow-up of 24 months. As a composite outcome, we reported the NEDA3 status.
Patients older than 50 years
A total of 131 patients older than 50 years at the time of CLAD exposure were enrolled. Main clinical baseline features are summarized in Table 5. The mean age was 54.6 years and mean disease duration was 13.0 years. Thirty-six (27.5%) were treatment-naïve, while 95 patients had previously been exposed to at least one DMT, with most switching from a platform therapy (n = 76, 58.9%). The median baseline EDSS was 2.5, and the ARR in the previous year was 0.79. After a mean follow-up of 23.4 months, only two patients presented a new relapse with the ARR on CLAD reduced to 0.01. Respectively, MRI activity was documented in 14 (10.7%) cases and 11 (8.4%) patients experienced a CDW.
Baseline features of ⩾50 years-old patients.

ARR, annualized relapse rate; CI, confidence interval; DMT, disease-modifying therapy; EDSS, expanded disability status scale; FUP, follow-up; Gd+, gadolinium-enhancing; IQR, interquartile range; RMS, relapsing multiple sclerosis.
DMTs after CLAD 2-years treatment course
During the observation period, 116 patients (10.6%) switched to a new DMT after the completion of the CLAD 2-years treatment course with a mean time to switch of 33.8 (SD ± 12.6) months (Supplemental Table 4). Anti-CD20 monoclonal antibodies represented the most common choice after CLAD (63 patients), followed by the sphingosine-1-phosphate modulators (18 patients). Interestingly, 14 patients underwent another CLAD treatment course.
Moreover, 39 patients, following a clinical (relapse) and radiological reactivation, started a new highly effective DMT before the end of the fourth year. Most (n = 33) of these patients started a monoclonal antibody (n = 21 ocrelizumab, n = 7 natalizumab, and n = 5 ofatumumab), while the remaining six switched to a sphingosine-1-phosphate modulator.
Safety aspects
Complete CBC data were available for 616 patients. A total of 30 patients started CLAD with a lymphocyte count between 800 and 1000 cells/mm3, 16 of whom switched from fingolimod or dimethyl fumarate. Grade 2 to grade 4 lymphopenia was recorded in 44.5% of patients. Only a minority of patients developed grade 4 lymphopenia (n = 3, 0.5%), while the majority had grade 2 lymphopenia (n = 200, 32.5%). In patients who developed grade 4 lymphopenia, CBC was strictly monitored weekly and prophylactic antiviral therapy was given until reaching a lymphocyte count greater than 500/mm3. In patients with grades 3–4 lymphopenia, lymphocyte count nadir occurred after the second year in 61.3% of patients, and after the first treatment cycle in the remaining 38.7%. In these patients, the mean time between the most recent CLAD cycle and lymphocyte count nadir was 4.5 (SD ± 2.4) months. The second year of therapy was delayed in 26 patients because of persistent grade ⩾2 lymphopenia with a mean delay of 96 (SD ± 45) days. Female sex resulted as the only baseline factor associated with developing grades 3–4 lymphopenia (OR 2.87, 95% CI 1.34–6.16).
We observed an elevation of serum aminotransferases greater than 5-fold the upper limit of normal (grade 3) in 4 patients (0.37%). Of these, only one patient discontinued CLAD following a grade 4 (>20 ULN) elevation of serum aminotransferases.
The occurrence of any infection during follow-up was reported in 64 (5.9%) patients. The most common infections were herpetic with 21 cases (including six shingles cases, all without CNS complications), followed by urinary and upper respiratory tract infections, with 18 and 16 cases, respectively. All reported infections resolved following specific antiviral or antibiotic therapy. Grades 3–4 lymphopenia (OR 3.63, 95% CI 1.85–7.11) and higher EDSS score (OR 1.51, 95% CI 1.19–1.94) were associated with a higher risk of infection while patients switching from platform DMT had the lowest rate of infection (OR 0.38, 95% CI 0.19–0.78) (Supplemental Table 5).
Discussion
In the complex and progressively broader scenario of DMTs, CLAD stands out due to its unique dosing schedule and mechanism of action. 11 Following CLAD administration, there is a slow but transient lymphopenia with blood count lymphocyte values returning to normal values by the end of each cycle in most individuals. 27 This more gradual development of lymphopenia sets cladribine apart from alemtuzumab, another selective immune reconstitution therapy, which has a more immediate and intense effect on lymphocyte depletion.28,29
This study provides valuable insights into the clinical and radiological outcomes in a large cohort of RMS patients treated with CLAD. Notably, our cohort included a significant proportion of treatment-naïve patients as well as patients transitioning from platform DMTs. After starting CLAD, we found a significant reduction in the ARR. Despite the difficulty of comparing different real-world studies, our ARR data is in line with what has been reported in several similar studies.16,30 Randomized clinical trials (RCTs) and other real-world studies reported a slightly higher ARR with similar relative reduction data with these mild differences explained by the different degree of disease activity in the included population with higher baseline ARR.12,31
The most common disease activity during follow-up was radiological (22.0% in the total cohort), followed by relapses (8.9%) and by CDW (7.9%) events. Among the CDW events, the majority were classified as PIRA events (73.2% of the CDW events). 32 Younger age resulted in greater risk of relapse and radiological activity, while older age was linked to higher risk of CDW.
To assess CLAD’s effectiveness across different patient groups, we stratified our cohort based on prior DMT exposure. The treatment-naïve group had the highest baseline ARR, followed by the platform-switching group, and the lowest in the highly effective DMT-switching group. This pattern reflects the switching reasons: efficacy or tolerability issues in the platform group, and safety concerns in the highly effective DMT group. While CLAD showed effectiveness in all three groups, the treatment-naïve group showed the most favorable outcomes, with the greatest reduction of ARR and the highest percentage of patients maintaining the NEDA3 status. 33
The analysis focusing solely on patients with a minimum of 2 years of observation, which included approximately half of the study population, showed that 67.9% of patients maintained NEDA-3 status. Interestingly, most patients lost NEDA3 status due to isolated radiological activity.34,35 Given the paucity of real-world data on CLAD’s effectiveness beyond the first 2 years, several therapeutic algorithms have been proposed, with exclusive radiological activity in the first 2 years of treatment not considered a treatment failure. 14 Extending the clinical and radiological follow-up in this cohort could provide further insights into the significance of isolated radiological activity during the first 2 years of treatment.
Over the last years, there has been a progressive increase in the proportion of late-onset MS patients. 36 Several aspects highlight the need for more attention to this group, as data safety and efficacy of DMTs are scarce, partly due to their underrepresentation in RCTs.37,38 Furthermore, advanced age is an important risk factor for the development of tumors and severe infections both in the general population and in MS patients exposed to DMT. 39
In our study, we included 131 patients over 50 years of age with a relatively low median EDSS score. About a quarter of these were treatment-naïve and over half have switched from platform DMTs. In this population, we found a marked reduction in the risk of relapse and a reduced radiological activity compared to the overall cohort with a limited risk of CDW. The peculiar mechanism of action of the drug could provide to this population the benefit of long-lasting clinical-radiological effectiveness, eliminating the need for continuous immunosuppression. 40
Among the unresolved questions related to CLAD use, it stands out the subsequent choice of a new DMT at the end of the 4-year observation period, or after the 2-year treatment cycle in case of persistent disease activity.14,41,42 In our cohort, 10.6% of patients started a new DMT after an average of 3 years from starting CLAD. More than half of these patients switched to an anti-CD20 monoclonal antibody following an insufficient clinical-radiological response to CLAD. 16
This work adds valuable real-world evidence on the use of CLAD in MS patients, representing, to the best of our knowledge, the largest cohort studied to date, including both clinical and radiological data.
Our data confirm CLAD effectiveness on both clinical and radiological outcomes, providing insights into identification of the best candidates for this treatment. Specifically, treatment-naïve patients exhibit better effectiveness outcomes compared to those transitioning from highly effective therapies.43,44 The positive results are also observed in patients over 50 years of age, suggesting that CLAD could be a valid therapeutic option for this population. 45
Over the past few years, there have been a growing number of real-world data reports on CLAD effectiveness in MS trying to confirm RCTs data and to characterize the best candidates for this drug.30,44,46,47 However, some aspects still need to be better clarified such as the therapeutical management after the completion of the treatment cycle (opportunity and optimal timing of a CLAD re-treatment or of a new DMT start), the definition of a patient as CLAD partial or full responder, and CLAD effectiveness in special populations. We tried to enlighten some of those aspects; hence, confirming the better clinical and radiological response for naïve and platform-switching patients, and reporting the use of anti-CD20 monoclonal antibodies in case of treatment failure.16,33,48 Moreover, we included a wide cohort of patients older than 50 years, thus, reporting CLAD effectiveness in this peculiar population. Despite the lack of specific reports for this aged population, a recent paper described positive data on clinical outcomes and patient reported outcomes (PROs) in 117 pwMS exposed to CLAD with a mean age of 46.7. 49 Another very recent article described the clinical-radiological outcomes of 129 pwMS aged >45 years at the time of initiating CLAD with only a minority experiencing a clinical relapse (three patients) or MRI activity. 50 Moreover, a recent study from MSBase registry compared ocrelizumab with injectables platform DMTs in patients >60 years with significantly lower risk of relapse in the ocrelizumab group. Ocrelizumab group had a very low ARR comparable to our findings with CLAD in older patients. 51 Given those findings, CLAD seems a rational option for reducing the burden of continuous treatment and monitoring for older people with RMS while maintaining a high degree of disease control. 52
Finally, our data confirmed the overall safety of CLAD use in pwMS. We also highlighted the need for strict monitoring CBC to implement preventing strategies in patients who develop grades 3–4 lymphopenia having a higher risk of infection. 53
Despite the strengths of our study, including the large multicenter real-world cohort and comprehensive follow-up data, limitations must be acknowledged. First, the retrospective nature of the study introduces potential selection and reporting biases, as data collection was dependent on clinical records rather than a predefined protocol. Second, while our cohort includes over 1000 patients, the follow-up duration varies, with some participants having a relatively short observation period, potentially limiting the assessment of long-term CLAD effectiveness and safety. Third, the definition of NEDA-3, while widely used in MS research, does not capture other important aspects of disease progression, such as brain volume loss or cognitive decline, which were not systematically assessed in our study. Lastly, the study lacks a direct comparator group, making it challenging to contextualize CLAD’s effectiveness against other DMTs in a controlled manner. Future prospective studies with longer follow-up and additional outcome measures will be crucial to further elucidate CLAD’s long-term impact and optimal positioning within the MS treatment landscape.
Further studies are needed to clarify the effectiveness of CLAD after the end of the 2-year treatment cycle, since identification of patients at greater risk for inadequate clinical response will be crucial to determine those who require an early change to other highly effective DMTs.
Conclusion
This postmarketing experience confirms the effectiveness of CLAD in the treatment of pwMS, with a significant reduction in ARR and a high proportion of patients remaining free from disease activity. By contrast, some patients required an escalation strategy mainly with anti-CD20 monoclonal antibodies because of persisting disease activity.
Supplemental Material
sj-doc-1-tan-10.1177_17562864251360047 – Supplemental material for Real-world experience with cladribine tablets in people with multiple sclerosis: effectiveness data from a multicenter Italian study
Supplemental material, sj-doc-1-tan-10.1177_17562864251360047 for Real-world experience with cladribine tablets in people with multiple sclerosis: effectiveness data from a multicenter Italian study by Matteo Lucchini, Giovanna Borriello, Shalom Haggiag, Carolina Gabri Nicoletti, Roberta Fantozzi, Maria Chiara Buscarinu, Gina Ferrazzano, Antonio Cortese, Fabiana Marinelli, Fabrizia Monteleone, Diego Centonze, Antonella Conte, Elisabetta Ferraro, Claudio Gasperini, Girolama Alessandra Marfia, Carlo Pozzilli, Marco Salvetti, Elena Barbuti, Gianmarco Bellucci, Assunta Bianco, Vincenzo Carlomagno, Alessandro Cruciani, Laura De Giglio, Claudia Dionisi, Antonio Ianniello, Leonardo Malimpensa, Martina Nasello, Viviana Nociti, Luca Prosperini, Carla Tortorella and Massimiliano Mirabella in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-2-tan-10.1177_17562864251360047 – Supplemental material for Real-world experience with cladribine tablets in people with multiple sclerosis: effectiveness data from a multicenter Italian study
Supplemental material, sj-tif-2-tan-10.1177_17562864251360047 for Real-world experience with cladribine tablets in people with multiple sclerosis: effectiveness data from a multicenter Italian study by Matteo Lucchini, Giovanna Borriello, Shalom Haggiag, Carolina Gabri Nicoletti, Roberta Fantozzi, Maria Chiara Buscarinu, Gina Ferrazzano, Antonio Cortese, Fabiana Marinelli, Fabrizia Monteleone, Diego Centonze, Antonella Conte, Elisabetta Ferraro, Claudio Gasperini, Girolama Alessandra Marfia, Carlo Pozzilli, Marco Salvetti, Elena Barbuti, Gianmarco Bellucci, Assunta Bianco, Vincenzo Carlomagno, Alessandro Cruciani, Laura De Giglio, Claudia Dionisi, Antonio Ianniello, Leonardo Malimpensa, Martina Nasello, Viviana Nociti, Luca Prosperini, Carla Tortorella and Massimiliano Mirabella in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
