Abstract
Background:
Pediatric-onset multiple sclerosis (POMS) is the onset of MS before the age of 18 and accounts for 3%–5% of all multiple sclerosis (MS) cases. Natalizumab (NTZ) is among the higher-efficacy disease-modifying treatments (HETs) in MS and is increasingly used for POMS.
Objectives:
In this systematic review and meta-analysis, we aimed to discuss the debate on the efficacy and safety of natalizumab use in POMS, providing quantitative results on relapse rate, disability progression, adverse events (AEs), and JC virus seropositivity.
Design:
The primary endpoint for meta-analysis was the mean difference (MD) in annualized relapse rate (ARR) after natalizumab compared to before treatment. Secondary outcomes were the MD of Expanded Disability Status Scale (EDSS) and the proportion of POMS patients experiencing AEs and JC virus seropositivity after natalizumab treatment.
Data sources and methods:
We performed a comprehensive search of PubMed, Embase, Web of Science, and Scopus between January 1, 1991 and May 1, 2025.
Results:
In this systematic review, 18 non-randomized interventional studies including 922 patients with POMS were included. Natalizumab therapy was associated with a mean reduction in ARR of −1.962 relapses per patient-year from baseline (95% confidence interval (CI): −2.449 to −1.475;
Conclusion:
Natalizumab may represent a viable therapeutic option for POMS patients exhibiting highly active disease or serves as an effective alternative in those with inadequate response to initial treatment. The safety profile remains acceptable, with most AEs being manageable.
Registration:
PROSPERO (CRD42024583911). This study follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses reporting guidelines.
Keywords
Introduction
Pediatric-onset multiple sclerosis (POMS) refers to multiple sclerosis that begins before 18 years of age and accounts for roughly 3%–10% of all multiple sclerosis (MS) patients.1,2 The global burden of POMS appears to be increasing. Reported annual incidence lies between 0.05 and 2.85 per 100,000 individuals, and current prevalence estimates range from 0.69 to 26.92 per 100,000 children.1,2
MS profoundly interferes with daily life. In children, MS requires particularly careful clinical attention because cognitive decline tends to emerge earlier than in adults, and relapse rates are higher.2,3 Compared with adult-onset MS (AOMS), POMS shows a prominent inflammatory activity in the central nervous system (CNS) early in the disease course. 3 Early initiation of effective disease-modifying therapy (DMT) is, therefore, essential to safeguard cognitive development in school-aged children and to preserve their overall quality of life.
Selecting an optimal DMT regimen for POMS remains a major clinical challenge and demands careful consideration. On one hand, POMS typically follows a more aggressive clinical trajectory, so initiating highly effective agents early in the disease course may delay CNS damage and attenuate long-term cognitive sequelae. 3 On the other hand, these more potent treatments raise greater safety concerns, and in children, treatment decisions must place particular weight on short and long term tolerability.3,4
Evidence-based decision making is further constrained by the rarity of POMS and ethical as well as practical barriers to conducting adequately powered randomized clinical trials (RCTs), which together limit the volume and robustness of available comparative data. 5
In AOMS, clinicians often turn to higher-efficacy DMTs (higher-efficacy disease-modifying treatments, HETs) such as natalizumab (NTZ) and anti-CD20 monoclonal antibodies for individuals with a more aggressive disease course who prioritize effectiveness despite increased safety risks.6,7 To date, fingolimod is still the sole Food and Drug Administration-approved DMT for POMS. 5 In AOMS trials, fingolimod is usually considered a moderate efficacy DMT, and its approval for POMS likely reflects its comparatively favorable safety profile together with low long term discontinuation rates.8,9 However, current evidence indicates that fingolimod does not substantially reduce scores on the expanded disability status scale (EDSS), which reflects disability progression over time. 10 In POMS, there may therefore be a need for HETs that both decrease clinical relapses and improve EDSS, particularly during critical years of brain growth. 11
NTZ, a monoclonal antibody to integrin α4β1, restricts T- and B-lymphocyte migration across the blood-brain barrier. 12 In AOMS, it is approved as a potent DMT for adults with strong clinical efficacy. 12 Its main safety concern is progressive multifocal leukoencephalopathy (PML), a rare but severe JC virus brain infection with a risk of 0.33 cases per 100 treated adults, which rises in patients with baseline JCV seropositivity and with longer exposure to NTZ.13,14 To the best of our knowledge, no PML has been reported in NTZ-treated MS patients under 18, possibly because the limited use of NTZ in POMS and latent JCV infection is less frequent in children than in adults. 15
This systematic review and meta-analysis examines the efficacy of NTZ in POMS, while also addressing its safety profile and the prevalence of JCV seropositivity, to offer clinicians more solid guidance for treatment decisions in this particularly vulnerable population.
Methods
Protocol and design
After a preliminary search, we registered the protocol in PROSPERO (CRD42024583911) to ensure our prospective design. This study is reported based on the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) 2020 guideline. 16
Data sources, searches, and study selection
A comprehensive search of the literature was performed in PubMed, Embase, Scopus, and Web of Science from January 1, 1991 to May 1, 2025. In addition, the International Clinical Trials Registry Platform, the Cochrane Central Register of Controlled Trials, and ClinicalTrials.gov were examined manually to capture further eligible studies. For each database, the search strategy combined controlled vocabulary terms with free-text keywords reflecting the study population and intervention. The main terms were “pediatric,” “multiple sclerosis,” and “natalizumab.” The full search strategy is reported in the Supplemental Material.
Quantitative synthesis focused on the primary endpoint of the mean difference (MD) in annualized relapse rate (ARR) before and after the start of NTZ therapy. Secondary endpoints included the mean change in EDSS and the proportion of participants who experienced adverse events (AEs) or had JCV positivity after treatment.
Studies were considered eligible if they fulfilled all of the following criteria. First, the study design had to be a peer-reviewed interventional study (randomized controlled trials or non-randomized study of intervention (NRSI)). Second, the target population consisted of patients younger than 18 years with a definite diagnosis of MS based on the McDonald criteria and/or the International Pediatric Multiple Sclerosis Study Group. Third, NTZ had to be initiated before 18 years of age, and treatment outcomes were required to be evaluated only up to 18 years of age, regardless of the specific dosing regimen used. Fourth, each study needed to report at least one of the following outcomes: ARR, EDSS, MRI activity, No Evidence of Disease Activity (NEDA), JCV positivity, or AEs, including Serious Adverse Events (SAEs).
All retrieved records underwent deduplication before being imported into Rayyan. Two authors (A.R., Y.E.-A.-H.) evaluated titles and abstracts against predefined eligibility criteria. Subsequently, they performed independent full-text assessments of potentially relevant articles. Selection proceeded without imposing any language constraints. A third author (H.K.) resolved conflicts when consensus could not be reached between the initial pair of reviewers. Whenever conference abstracts appeared relevant, we attempted to locate their corresponding full-text publications for inclusion. If multiple studies presented overlapping participant populations, we selected the one reporting the most comprehensive dataset.
Risk-of-bias assessment and data extraction
Each included study underwent risk-of-bias assessment by two authors working independently (Y.E.-A.-H., A.R.) using the NIH Quality Assessment Tool for Before-After (Pre-Post) Studies with No Control Group. 17 The NIH tool has 12 questions assessing study design, participant selection, data collection, and statistical analysis. Based on the 12 responses, each study is then characterized as high risk of bias, moderate, or low. A third author (H.K.) resolved any discrepancies.
Data extraction from eligible studies was performed independently by two authors (Y.E.-A.-H., A.R.) with subsequent verification by a third author (H.K.). When numerical outcome data were not fully reported in the text or tables, we screened Supplemental Materials and extracted the required values from published figures using the Automeris WebPlotDigitizer (Automeris LLC) tool. The extraction protocol encompassed the following variables: lead author name, year of publication, patient count, sex distribution, baseline age, age when disease began, age at treatment initiation with NTZ, dosing protocol for NTZ, disease duration, length of therapy, follow-up period, therapeutic agents administered prior to NTZ, radiological activity on MRI, ARR, EDSS, additional clinical endpoints such as NEDA and treatment discontinuation rates, JCV seropositivity status, and safety endpoints encompassing AEs and SAEs.
Statistical analysis
All quantitative syntheses were carried out using Stata software (version 16; Statacorp, College Station, TX, USA). In view of the considerable methodological, clinical, and setting-related heterogeneity across the included studies, pooled estimates were generated with random-effects models. Between-study variance (τ
2
) was estimated using the restricted maximum likelihood (REML) estimator, and confidence intervals (CIs) were calculated with the Hartung–Knapp method. Heterogeneity was summarized using the
The random-effects model assumes that the true effects across studies follow a normal distribution. Continuous outcomes (ARR and EDSS) were assumed to be approximately normally distributed after conversion of reported summary statistics to means and standard deviations.
For continuous outcomes, including ARR and EDSS, all available summary data were converted to means and standard deviations before pooling, using established transformations from CIs, standard errors, interquartile ranges, or ranges. Because the same measurement scales were applied across studies, treatment effects were synthesized as MDs with inverse-variance weighting. To avoid multiplicity, when more than one study was available from a center, we selected the study with the greatest sample size for our qualitative synthesis.
For AEs and JCV seropositivity, proportional meta-analyses were undertaken to derive pooled prevalence estimates across studies. To address skewness and stabilize variances, especially for proportions close to zero or one, the Freeman–Tukey arcsine transformation was applied. Study-specific prevalence estimates and their 95% CIs were then combined in random-effects models using the DerSimonian–Laird approach to account for between-study heterogeneity.
For outcomes with more than 10 studies included in the quantitative synthesis (ARR, AEs, and JCV seropositivity), publication bias was explored visually using funnel plots. For EDSS, where fewer than 10 studies contributed to any given analysis, funnel plots were not presented; instead, small-study effects were evaluated with Egger’s regression test and Begg’s rank-correlation test, and these results were interpreted cautiously in light of their limited power and the potential for inflated error rates when the number of studies is small.
The prespecified sensitivity analysis used a leave-one-out framework, in which the pooled estimate was recalculated after omitting each study in turn. To investigate residual heterogeneity, subgroup analyses and meta-regression with Knapp–Hartung inference were performed. Candidate moderators were defined a priori and consisted of study-level characteristics available across the dataset (e.g., follow-up duration, age at onset, and risk-of-bias assessments). Unless otherwise specified, statistical tests were two-sided with a significance level of
Results
Study selection and baseline characteristics
The initial search identified 3092 records. Subsequently, 767 duplicates were removed, and 2325 articles went through screening. During the study selection, 74 full texts were reviewed, and 18 studies met the inclusion criteria for qualitative synthesis (Figure 1).

PRISMA flow diagram.
Across 18 studies, a total of 922 POMS patients were included. The mean age at disease onset ranged from 12.9 to 15.8 years, and follow-up durations spanned 12–66 months. All studies administered NTZ 300 mg IV every 4 weeks. Baseline study and patient characteristics are summarized in Table 1.
Demographic and outcomes of included studies.
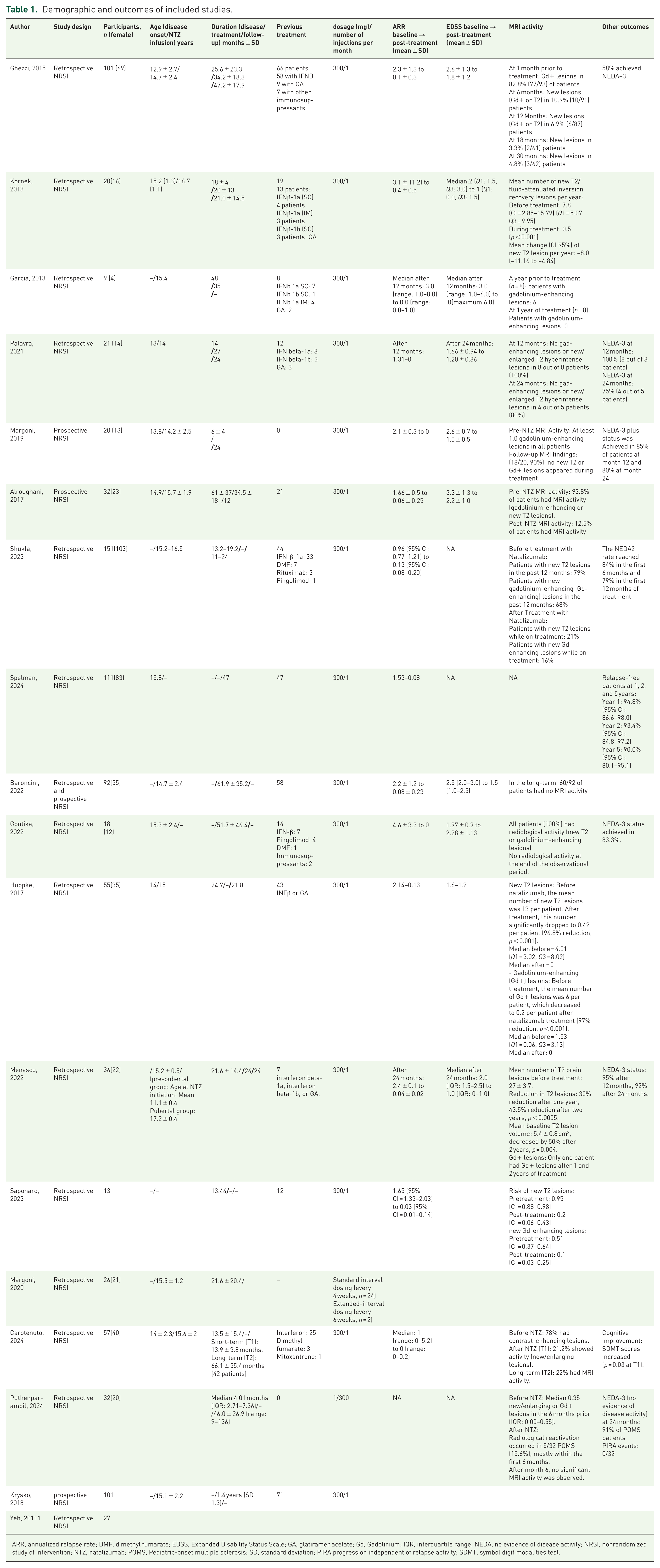
ARR, annualized relapse rate; DMF, dimethyl fumarate; EDSS, Expanded Disability Status Scale; GA, glatiramer acetate; Gd, Gadolinium; IQR, interquartile range; NEDA, no evidence of disease activity; NRSI, nonrandomized study of intervention; NTZ, natalizumab; POMS, Pediatric-onset multiple sclerosis; SD, standard deviation; PIRA,progression independent of relapse activity; SDMT, symbol digit modalities test.
The results of NIH Quality Assessment Tool for Before-After (Pre-Post) Studies with No Control Group showed that 13 of 18 included articles had moderate risk of bias, 2 had low, and 3 had high risk of bias. Our systematic search did not identify any RCTs and all 18 studies were NRSIs, 14 with retrospective data collection, 3 with prospective, and 1 with a mix of retrospective and prospective approach. The NIH risk-of-bias summary is shown in Figure 2.

Traffic light plot of ROB with NIH tool.
Annualized relapse rate
Ten studies (

Forest plot for MD (post-treatment compared to pretreatment) of ARR.
Expanded disability status scale
Five studies (

Forest plot for MD (post-treatment compared to pretreatment) of EDSS.
Meta-regression, subgroup analyses, and sensitivity analysis
Meta-regression and subgroup analyses were conducted to explore moderators of between-study heterogeneity. Prespecified covariates were follow-up duration, age at NTZ initiation, and risk of bias.
For ARR, none of the moderators were statistically significant in meta-regression; however, age at initiation explained 52.76% of the between-study variance (
Leave-one-out sensitivity analyses confirmed robustness, as no individual study materially changed the pooled ARR or EDSS estimates. Leave-one-out analyses for ARR ranged from −1.927 to −2.039, and for EDSS from −0.727 to −0.901.
Publication bias
Publication bias analysis suggested possible small-study effects for ARR. Egger’s test (intercept β = −2.86, SE = 0.96;
MRI outcomes
Long-term MRI data were primarily derived from an Italian registry, which collectively followed 188 pediatric patients across 4 sequential Ghezzi studies7,22,30,31 and a continuation cohort by Baroncini in 2022.
In the 2010 study (
In 2013 (
The 2015 multicenter study (
Baroncini et al. 20 followed 92 of these patients for a mean of 56.8 ± 34.7 months. At baseline, median Gd+ lesions were 2 (IQR 1–5). Residual activity was detected in 12 patients early on but subsequently stabilized, with 65% remaining MRI-inactive through therapy. However, after NTZ discontinuation, 59% experienced clinical or radiologic reactivation, underscoring rebound risk. 32
Independent cohorts confirmed these findings. Kornek et al.
24
observed a reduction in new T2 lesions from 7.8/year (95% CI: 2.85–15.79) to 0.5/year (
In Alroughani et al.,
18
pre-treatment MRI activity (93.8%) fell to 12.5% after 12 months, confirming marked suppression of subclinical inflammation. The large Malani Shukla et al.
25
cohort (
Long-term data from Spelman et al.
27
(
Huppke et al.
33
documented a 96.8% reduction in new T2 lesions (13.0 → 0.42 per patient) and a 97% reduction in Gd+ lesions (6.0 → 0.2;
Saponaro et al.
34
observed a drop in new T2-lesion from 0.95 (CI: 0.88–0.98) to 0.20 (CI: 0.06–0.43) and in Gd+ lesions from 0.51 (CI: 0.37–0.64) to 0.10 (CI: 0.03–0.25). Similarly, Carotenuto et al.
21
reported a fall in contrast-enhancing lesions from 78% to 21%–22%, paralleled by improved cognitive performance (SDMT;
No evidence of disease activity
NEDA represents a composite measure of disease suppression, with NEDA-2 encompassing the absence of clinical relapses and MRI activity, while NEDA-3 extends this definition to include the additional criterion of no confirmed disability progression on the EDSS. 35 NTZ consistently induced high NEDA rates across cohorts. In the Ghezzi multicenter study, 58% achieved NEDA-3 over ~70 months. 22 Palavra et al. 29 found 100% NEDA-3 at 12 months and 75% at 24 months; Margoni et al. 28 reported 85% and 80% at 12 and 24 months. Malani Shukla et al. 25 observed NEDA-2 rates of 84% and 79% at 6 and 12 months, while Spelman et al. 27 documented relapse-free survival of 90% at 5 years. Menascu et al. 26 found NEDA-3 in 95% and 92% of patients at 12 and 24 months after NTZ treatment, respectively, and Puthenparampil et al. 35 reported 91% NEDA-3 at 24 months with no PIRA. Gontika et al. 23 noted NEDA-3 in 83.3% of adolescents versus 58.3% of adults, suggesting younger patients may sustain stronger disease control.
Adverse events
Fifteen studies (

Forest plot for the prevalence of adverse events.
JC virus seropositivity and PML risk
Ten studies (

Forest plot for the prevalence of JC virus seropositivity after natalizumab treatment.
Discussion
This systematic review and meta-analysis evaluated the safety and efficacy of NTZ in POMS. Across included studies, NTZ demonstrated favorable therapeutic benefits. The pooled analysis revealed a statistically significant decrease in ARR from baseline (MD = −1.962, 95% CI: −2.449 to −1.475,
MRI findings across longitudinal and multicenter cohorts confirmed consistent and durable suppression of inflammatory activity. New or enlarging T2 and Gd+ lesions markedly declined within 6 months of therapy, with many studies reporting near-complete radiologic remission after 1 year. NEDA outcomes were achieved in 58%–100% of patients at 12 months and 75%–92% at 24 months, underscoring sustained control of both clinical and subclinical disease.
AEs occurred in approximately 18% of treated patients (95% CI: 11%–25%;
Approach to DMTs and NTZ’s therapeutic position
Management of POMS increasingly prioritizes early initiation of high- or intermediate-efficacy DMTs over traditional escalation approaches. The treatment choice is influenced by disease activity, patient and family preferences, and tolerance for risk. Current expert recommendations advocate starting potent agents such as Rituximab (RTX), Fingolimod (FTY), dimethyl fumarate (DMF), or NTZ rather than conventional injectables (IFNs or glatiramer acetate (GA)), which offer modest efficacy but better tolerability. 37 For patients with low disease activity or safety concerns, IFNs or GA remain among reasonable initial choices due to well-characterized safety and ease of dosing (e.g., peginterferon every 14 days). Escalation to higher-efficacy therapy is warranted for breakthrough disease. NTZ, in particular, is favored for patients with highly active or refractory MS unresponsive to prior agents, though its use is guided by PML risk assessment in JCV-positive patients. 37
Comparative efficacy and safety across DMTs
Although comparative data interpretation between NTZ and other DMTs is still limited, accumulating evidence positions NTZ among the most potent therapies for relapse and MRI suppression. The PARADIGMS RCT 38 established FTY as superior to IFN-β, reducing relapse rates by 82% and halving the number of new T2 lesions. Fingolimod requires first-dose cardiac monitoring and carries risks of bradyarrhythmia, macular edema, and lymphopenia. NTZ has been observed to show high levels of relapse suppression and fast clinical stabilization, comparable to or potentially exceeding FTY in POMS observational cohorts, but demands intravenous infusions and ongoing safety surveillance. 27 Large registry studies support this finding. Spelman et al. 27 observed a lower relapse hazard under NTZ than FTY, and Carotenuto et al. 21 reported greater EDSS improvement with NTZ, despite both agents markedly reducing relapse rates. While these comparisons are indirect, the consistency across multicenter data highlights NTZ’s superior disease control.
NTZ’s MRI effects remain highly robust in pediatric populations, with over 90% reductions in new T2 and Gd+ lesions, an outcome rarely achieved with oral or moderate-efficacy therapies. DMF and TERI yield more modest benefits, with ARR reductions of 0.8–1.2 and limited MRI control.8,39 IFN-β and GA typically reduce relapse rates by only 25%–35%, with minimal impact on MRI lesion burden. In the Italian multicenter series, IFN-treated children demonstrated ARR declines from 2.4–3.2 to 0.4–0.8, whereas NTZ reduced ARR from 2.3 to 0.1, 22 highlighting its potential superior efficacy.
RTX and other anti-CD20 agents are gaining traction off-label in POMS. Data from nonrandomized pediatric cohorts and adult RCTs with OCR suggest comparable relapse suppression, though NTZ often achieves faster disease control.40,41 The primary limitations of RTX in children include infection risk and hypogammaglobulinemia, occasionally necessitating IVIG supplementation.37,40–42 NTZ, conversely, provides rapid clinical benefit without systemic immunosuppression but requires continuous monitoring for JCV seroconversion.
DMF offers moderate efficacy (≈50% relapse reduction) but may cause gastrointestinal symptoms and lymphopenia.43,44 Teriflunomide demonstrated modest benefit in patients (34% relapse risk reduction). 38 Injectable IFNs and GA remain options for mild disease or contraindications to stronger agents, though they provide only one-third relapse reduction and limited protection from long-term disability. 22
Overall, NTZ’s safety profile (~18% AEs) compares favorably with other monoclonal and oral DMTs. Most events were mild, and unlike sphingosine-1-phosphate modulators, NTZ lacks cardiac, ophthalmologic, or pulmonary toxicities. Its principal safety concern remains PML, determined by JCV status, prior immunosuppression, and therapy duration. 47
Comparison with adult NTZ data
Adult data firmly support NTZ’s classification as a high-efficacy therapy. The AFFIRM trial demonstrated a 68% ARR reduction versus placebo, and the SENTINEL trial reported a 54% reduction compared with interferon-β.39,46 Long-term extension studies confirmed sustained suppression of disease activity for over 6 years. 47
Adult experience has also refined risk stratification for PML, linking risk to JCV index values, prior immunosuppressant exposure, and treatment duration. Registry data estimate a PML incidence of 0.4–1.2 per 1000 treated adults after 8–10 years. 45 To date, no pediatric PML cases have been reported, likely due to shorter treatment duration and lower cumulative JCV exposure. Nonetheless, vigilant surveillance remains essential, as JCV seroconversion increases with age. Strategies adopted from adult practice, such as regular JCV testing, serial MRI monitoring, and extended-interval dosing, have reduced adult PML risk by up to 88% without loss of efficacy and are increasingly utilized in pediatric programs. 47
Clinical implications
These findings underscore NTZ’s value as an early intervention for children and adolescents with aggressive POMS characterized by frequent relapses, extensive MRI lesion load, or early disability progression. Its relapse suppression, MRI stability, and NEDA rates mirror those observed in adult high-efficacy therapy, suggesting that early initiation could help delay disease progression and accumulation of disability. The decision to initiate NTZ should balance its potent efficacy against the logistical and safety requirements of intravenous infusions, regular JCV antibody monitoring, and MRI follow-up. Importantly, the absence of pediatric PML cases supports its safe use under specialized supervision. Early use in appropriately selected patients could meaningfully alter long-term disease trajectories. 48
Strengths, limitations, and future directions
This systematic review and meta-analysis offer a comprehensive synthesis of NTZ outcomes in POMS. We gathered evidence from 18 NRSIs involving 922 patients, drawn from a multi-database search conducted under a prespecified protocol registered in PROSPERO, with reporting aligned to PRISMA 2020. We reinforced through duplicate screening and data extraction with adjudication, a structured ROB evaluation using the NIH before-and-after tool, plus prespecified sensitivity, subgroup, meta-regression, and leave-one-out analyses.
This analysis has several limitations. Most included studies were observational and thus susceptible to confounding and selection bias. Although random-effects modeling and sensitivity analyses were applied, high heterogeneity (
Future RCTs should prioritize direct comparison of NTZ and other high-efficacy options (e.g., anti-CD20 therapies and fingolimod) using standard follow-up and large samples. Long-term registries for POMS treatment are needed to help us better understand the safety concerns over the long-term use of each DMT.
Conclusion
Natalizumab showed high efficacy in POMS with notable reductions in relapse rates, MRI activity, and disability progression while achieving high NEDA rates and maintaining an acceptable safety profile. Although long-term safety data remain limited, current evidence supports NTZ as a valuable high-efficacy therapy for active POMS when administered within structured monitoring frameworks. Future research should focus on head-to-head randomized trials against other potent agents, longitudinal registries evaluating immune evolution and PML risk, and optimization of early biologic initiation strategies in this vulnerable patient population.
Supplemental Material
sj-docx-1-tan-10.1177_17562864261440657 – Supplemental material for Natalizumab for pediatric multiple sclerosis: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tan-10.1177_17562864261440657 for Natalizumab for pediatric multiple sclerosis: a systematic review and meta-analysis by Yasin Ebne-Ali-Heydari, Aryana Ramezani, Amirmohammad Jozayi, Mahdi Norouzi, Parto Zohrabi and Hasan Kaveyee in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
Language editing was performed in part with AI tools (e.g., ChatGPT 5). The authors then manually double checked the text, made any necessary revisions, and approved the AI edits.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
