Abstract
Background:
Gaps in current product labels and a lack of detailed clinical guidelines leaves clinicians’ questions on the practical management of patients receiving cladribine tablets for the treatment of relapsing multiple sclerosis (MS) unanswered. We describe a consensus-based programme led by international MS experts with the aim of providing recommendations to support the use of cladribine tablets in clinical practice.
Methods:
A steering committee (SC) of nine international MS experts led the programme and developed 11 clinical questions concerning the practical use of cladribine tablets. Statements to address each question were drafted using available evidence, expert experiences and perspectives from the SC and an extended faculty of 33 MS experts, representing 19 countries. Consensus on recommendations was achieved when ⩾75% of respondents expressed an agreement score of 7–9, on a 9-point scale.
Results:
Consensus was achieved on 46 out of 47 recommendations. Expert-agreed practical recommendations are provided on topics including: the definition of highly active disease; patterns of treatment response and suboptimal response with cladribine tablets; management of pregnancy planning and malignancy risk, infection risk and immune function, and switching to and from cladribine tablets.
Conclusion:
These expert recommendations provide up-to-date relevant guidance on the use of cladribine tablets in clinical practice.
Keywords
Introduction
A considerable number of disease modifying drugs (DMDs) with different properties have been approved for the treatment of relapsing multiple sclerosis (RMS) over the two last decades (recently reviewed by Cree et al.). 1 This evolving treatment landscape requires physicians to have a comprehensive understanding of the various treatment options, including their relevant mechanisms of action, clinical efficacy and safety, mode of administration and monitoring requirements, in order to be able to offer optimal care. 2 The increasing number of treatment options also pose new challenges for neurologists, including when to start, switch and stop treatments, and other important issues around pregnancy. 3
Clinical guidelines and treatment labels provide a framework, but rarely provide specific, detailed information on real-life usage, and are of limited help for individual decision making. 3 This means that there are many unanswered questions about the practical use of these treatments.
Expert consensus recommendations can be developed to aid treatment decision-making when clinical recommendations are limited and there are gaps in guidance due to limited experience. 4 Such consensus recommendations typically summarise opinions from an expert panel on particular, focussed topics and suggest treatment strategies based on collective knowledge and clinical experience. 4
Cladribine tablets (MAVENCLAD®) are a short-course oral DMD for use in MS.5,6 At a cumulative dose of 3.5 mg/kg over 2 years, cladribine tablets were associated with significant improvement in clinical and imaging parameters in patients with highly active RMS in clinical trials (CLARITY and CLARITY Extension). Cladribine tablets were recently approved by the European Medicines Agency (EMA) and the United States (US) Food and Drug Administration (FDA), and various other regulatory authorities throughout the world.7,8 Cladribine is a deoxyadenosine analogue that selectively reduces B and T lymphocytes and is thought to interrupt the cascade of immune events central to the pathogenesis of MS. 6
In order to address some of the unanswered questions relating to the use of cladribine tablets, we describe a consensus-based programme led by international MS experts with the aim of providing practical recommendations to support its use in real-life clinical practice. The questions focussed on six topics:
The definition of highly active disease;
The patterns of treatment response in patients treated with cladribine tablets;
Management of patients with evidence of disease activity while being treated with cladribine tablets;
Infection risk and immune function in patients being treated with cladribine tablets;
Management of pregnancy planning and malignancy risk in patients being treated with cladribine tablets;
Treatment switching to and from cladribine tablets and monitoring considerations.
The objective of the programme was to provide consensus-based practical recommendations on the use of Cladribine tablets in real-life practice addressing gaps not covered in current guidelines and labels.
Materials and methods
The consensus programme was based on a multi-step modified Delphi methodology, which took place between April 2018 and June 2019. The process is outlined in Figure 1. A steering committee (SC) of nine international MS experts with well-documented experience with cladribine tablets led the programme, co-chaired by P.S.S. and P.R. The SC in turn was supported by the identification of an extended faculty (EF) through the nomination of additional internationally recognised practicing neurologists with experience of caring for people with MS. A total of 76 international experts were invited to participate in the EF, with 33 taking part. A total of 19 countries were represented in the programme. The role of the EF was to review the available evidence, complete a questionnaire and finally vote on draft recommendations.

Overview of the modified Delphi process for achieving consensus.
The SC outlined the areas of clinical focus and drafted 21 clinical questions to be addressed. These were ranked using a web-based platform, resulting in 11 prioritised questions, categorised into six topics. The questions included both contextual questions, relating to the definition of highly active RMS, and practical questions, relating to the use of cladribine tablets in different clinical scenarios. A comprehensive literature review was performed using the PICO (population, intervention, comparison, outcome) framework for each of the 11 questions. The level of evidence was assessed and agreed by the SC using the GRADE (grading of recommendations assessment, development, and evaluation) level of evidence ratings scale. 9
A questionnaire was developed by the SC with draft answers based on the available evidence from the literature review, combined with their expert opinion, where evidence was lacking. The questionnaire was completed remotely via the on-line platform by the EF, following a review of the available evidence. The results from the questionnaire were used to develop draft clinical recommendations, which were then voted on by the SC and EF members. Consensus was achieved when ⩾75% of respondents agreed in the range 7–9 (on a 9-point scale). Each statement/recommendation was assigned a strength score (i.e. the median score) and a level of consensus, defined as the percentage of votes with a score of 7–9.10–14
Results
In total, 47 recommendations were drafted by the SC and voted on. Consensus was achieved on 46 of these recommendations. Not all EF members voted on all questions. The exact reasons for the few abstentions were unclear but could have been due to time constraints and the length of the on-line questionnaire and voting procedure. However, this did not affect the overall strength of the recommendations. A summary of the available evidence for each topic is provided in the following, and the recommendations for each question provided in Tables 1–6.
Defining highly active disease.

Median score on a 1–9 scale (mean score in brackets).
Percentage of votes with 7–9 on a 9-point scale.
DMD, disease modifying drug; Gd+, presence of gadolinium; MRI, magnetic resonance imaging.
Patterns of treatment response in patients treated with cladribine tablets.

Median score on a 1–9 scale (mean score in brackets).
Percentage of votes with 7–9 on a 9-point scale.
DMD, disease modifying drug.
Managing patients with evidence of disease activity while being treated with cladribine tablets.

Median score on a 1–9 scale (mean score in brackets).
Percentage of votes with 7–9 on a 9-point scale.
DMD, disease modifying drug.
Infection risk and immune function in patients being treated with cladribine tablets.
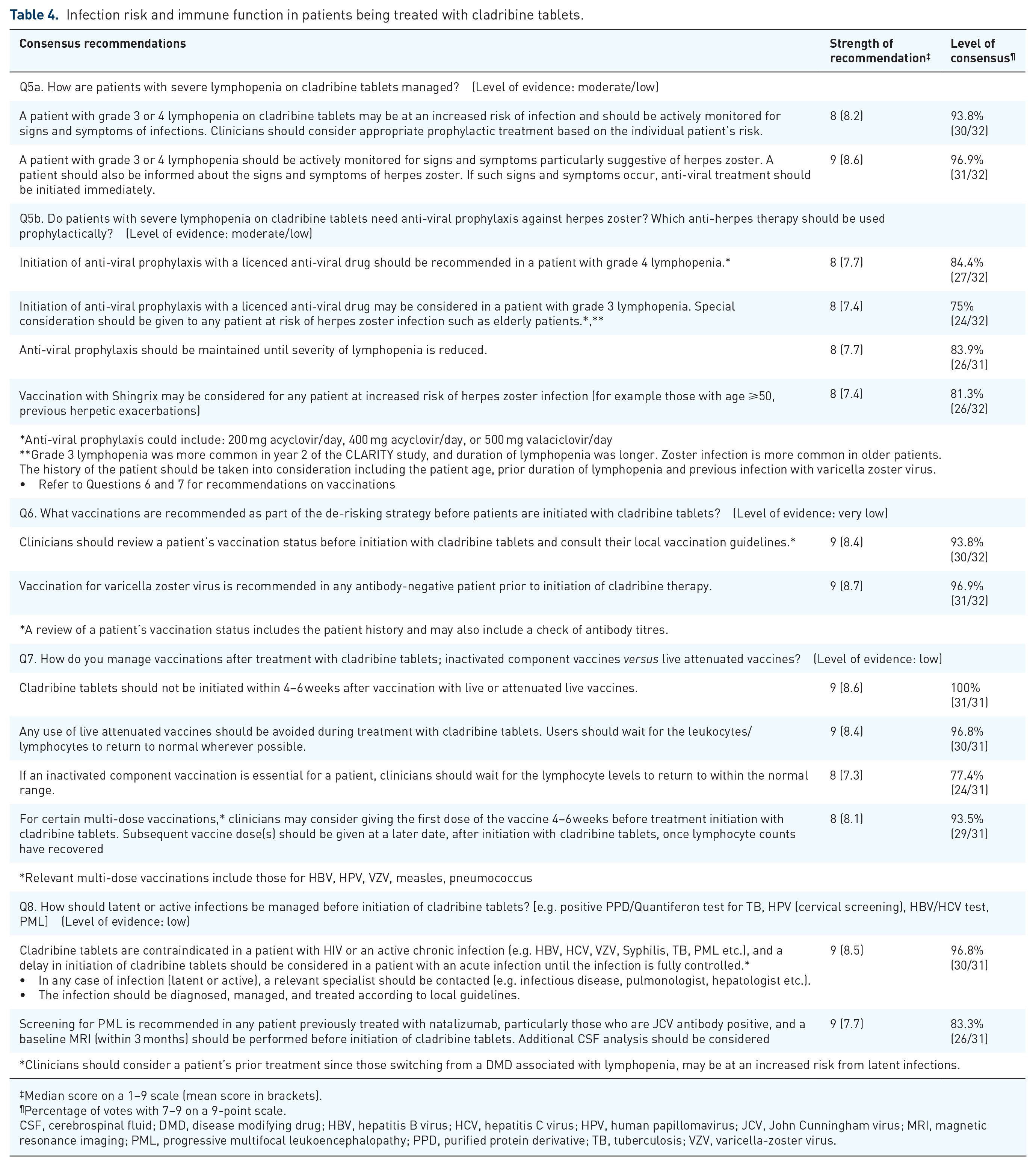
Median score on a 1–9 scale (mean score in brackets).
Percentage of votes with 7–9 on a 9-point scale.
CSF, cerebrospinal fluid; DMD, disease modifying drug; HBV, hepatitis B virus; HCV, hepatitis C virus; HPV, human papillomavirus; JCV, John Cunningham virus; MRI, magnetic resonance imaging; PML, progressive multifocal leukoencephalopathy; PPD, purified protein derivative; TB, tuberculosis; VZV, varicella-zoster virus.
Pregnancy planning management and malignancy risk in patients being treated with cladribine tablets.

median score on a 1–9 scale (mean score in brackets).
percentage of votes with 7–9 on a 9-point scale.
CI, confidence interval; DMD, disease modifying drug; MS, multiple sclerosis; SIR, standardised incidence ratio.
Treatment switching to and from cladribine tablets and monitoring considerations.

Median score on a 1–9 scale (mean score in brackets).
percentage of votes with 7–9 on a 9-point scale.
CSF, cerebrospinal fluid; DMD, disease modifying drug; FLAIR, fluid-attenuated inversion recovery; JCV, John Cunningham virus; LLN, lower limit of normal; MRI, magnetic resonance imagining; MS, multiple sclerosis; PCR, polymerase chain reaction; PML, progressive multifocal leukoencephalopathy; TSH, thyroid stimulating hormone.
Definition of highly active disease
Currently, there is no universally accepted definition of highly active MS, yet many MS treatments include this in their labelled indication. Clinical trials lack consistent definitions for ‘highly active’ MS in subgroup analyses (reviewed by Fernandez). 15 In general, patients with highly active MS will have frequent relapses and/or an increasing burden of brain magnetic resonance imaging (MRI) lesions. 16 Other features that may be taken into consideration include severity and localization of relapses, clinical or MRI disease activity on treatment, burden of T2 lesions and presence of gadolinium (Gd+) enhancing lesions.
The consensus recommendation for the definition of highly active MS is provided in Table 1 for both treatment naïve patients (Question 1a), and those who have received an appropriate course of a DMD (Question 1b). Additional factors associated with poor prognosis may also be taken into consideration, alongside activity metrics, when deciding whether to initiate a high-efficacy therapy in treatment naïve patients with MS including those with ⩾1 spinal cord lesions, incomplete recovery from relapses, accrual of physical or cognitive impairment and a short inter-attack interval.16,17
Patterns of treatment response in patients treated with cladribine tablets
Clinical judgement about response to treatment and acceptance of any on-treatment disease activity is often subjective and can vary between individual physicians. There is no consensus on the definition of treatment failure, or suboptimal treatment response in MS, nor indeed specifically for patients receiving cladribine tablets.
There are published decision models that may support the identification of patients failing on current treatment including the Canadian MS Working Group model 20 and the multifactorial MS decision model. 21 Both of these use a traffic light-based system to flag whether a change of therapy should be considered for a given patient. However, there has been no wide implementation of these models across the MS community.
The European Academy of Neurology/European Committee of Treatment and Research in Multiple Sclerosis (EAN/ECTRIMS) recommendation suggests ‘combining MRI with clinical measures to evaluate disease evolution’. 18 The American Academy of Neurology (AAN) recommends including ⩾1 relapses, ⩾2 MRI lesions or increased disability when assessing suboptimal treatment response. 19 There is a strong debate in the MS community whether physicians should treat to ‘no evidence of disease activity’ (NEDA), defined as an absence of relapses, disability worsening and MRI activity.17,22,23 However, NEDA can be difficult to sustain in the long term, and can depend on the mechanism of action of the DMD and timing of the re-baseline scan.22,23
Consensus recommendations for the definitions of optimal (Question 2a) and suboptimal (Question 2b) treatment responses with cladribine tablets are provided in Table 2.
Management of patients with evidence of disease activity while being treated with cladribine tablets
Disease activity in the first 2 years
The full recommended dose of cladribine tablets is 3.5 mg/kg given as two courses 12 months apart. 24 Following completion of the two treatment courses in years 1 and 2, The patient is observed in years 3 and 4 without any additional planned treatment with cladribine tablets. Any patient with residual activity in the first 12 months should receive the full dose of cladribine tablets, since a lower cumulative dose appears to result in significantly lowered efficacy. 5 In CLARITY, 56.1% of patients receiving cladribine tablets were disease activity-free during year 1 (defined as having no relapses, no 6-month sustained change in EDSS score, no new T1 gadolinium-enhancing lesions and no active T2 lesions). 25
When considering disease activity in the first year, the timing of the maximal effect of cladribine tablets on B and T cells may need to be considered. 24 This is recognised in the AAN guidelines, which state that relapses or new MRI lesions may develop after initiation of a disease-modifying therapy but before it becomes effective. 19 Across clinical studies, the largest proportion of patients with grade 3 or 4 lymphopenia was seen 2 months after the first cladribine dose in each year. The greatest reduction in lymphocyte count is reached at 4 months after the first dose.7,24 It is a possibility that residual disease activity at less than 6 months may indicate that the maximum effect on lymphocytes has not been reached.
The CLARITY study protocol allowed relapses in years 1–2 to be treated as per clinical practice with short-term systemic corticosteroid therapy. This occurred in 23% of patients receiving cladribine tablets versus 46% of patients receiving placebo [odds ratio (OR) 95% confidence interval (CI): 0.34 (0.26, 0.46) p < 0.001].7,26,27
Disease activity in years 3, 4 or beyond
Cladribine tablets are efficacious in patients with MS for up to 4 years after the initial dose in clinical trials; re-initiation of therapy after year 4 has not been investigated. 24 The CLARITY extension study (CLARITY EXT) was a safety study in which 867 patients from the original CLARITY trial were enrolled. Of these, 98 patients who received cladribine tablets during CLARITY, received placebo in CLARITY EXT (CP group), and no further doses of cladribine tablets, and 186 patients received an additional two treatment courses of cladribine tablets, 12 months apart in years 3 and 4 (CC group; 7 mg/kg cumulative dose). All efficacy analyses were exploratory. The incidence of lymphopenia events during CLARITY EXT increased in the CC group compared with the CP group (36.6% versus 9.2%, respectively). 8 Furthermore, there was an increase in lymphopenia leading to discontinuation (11.8% versus 0%, respectively). However, these findings occurred in the absence of the requirement to achieve a lymphocyte count of at least 800 cells/mm3 before initiation of cladribine tablets in year 2, as per current practice. 24 In patients with ⩾800 cells/mm3 prior to administration of subsequent courses in years 2, 3 and 4, the incidence of lymphopenia dropped to 11% and 12% in years 3 and 4, respectively. 28 During the extension period, 75.6% of patients in the CP group stayed relapse free, compared with 81.2% of patients in the CC group. 8 Additional exploratory MRI analyses demonstrated that cladribine tablets had a durable effect on MRI outcomes in the majority of patients, an effect that was sustained up to the end of the extension period.
Consensus recommendations for the management of patients with evidence of disease activity during: Year 1 (Question 3a); the first 2 years (Question 3b); in years 3 or 4 (Question 4a); or beyond year 4 (Question 4b) while being treated with cladribine tablets are provided in Table 3. Consensus was not achieved for one recommendation in answer to Question 4a. The reasons provided by the EF for this was that they would consider re-treatment with cladribine tablets in this instance. Re-treatment in year 3 or 4 has not been formally investigated in a clinical trial setting, in addition to an increased incidence of lymphopenia and other adverse events following additional cladribine tablets treatment in year 3 or 4 in the CLARITY EXT study was observed.
Infection risk and immune function in patients being treated with cladribine tablets
Lymphopenia and infection risk (including herpes zoster)
A pooled safety analysis showed the incidence of infection was similar between cladribine tablet-exposed cohort and placebo groups, except for herpes zoster. 30 Infections or infestations, including herpes zoster were more common in patients receiving cladribine tablets during periods with Grade 3 or 4 lymphopenia. 30
Guidance on the management of lymphopenia and herpes zoster is provided in the Mavenclad Summery of Product Characteristics (SmPC) and in the German Competence Network guidance.24,31 Consensus recommendations on the management of lymphopenia and infections in patients on cladribine tables are provided in Table 4 (Question 5). Whereas consensus was reached on recommendations regarding use of antiviral prophylaxis in patients with lymphopenia (Question 5b), five EF members (16%) did not agree that antiviral treatment should be considered in patients with grade 4 lymphopenia. Reasons provided for this included ‘a lack of indication for prophylaxis with anti-viral treatments in patients with lymphopenia’, ‘no scientific evidence’, ‘more evidenced on risk/benefit ratio is needed for “continuous” antiviral therapy’, ‘lack of evidence based documentation for efficacy and duration of anti-viral prophylaxis’ and ‘lack of sufficient evidence that prophylactic antivirals significantly reduces the risk of shingles to justify routine use’.
Vaccinations
There are no comprehensive studies available on vaccines and cladribine tablets, and the effects of cladribine tablets on the immunological memory acquired by previous vaccinations have not been studied. Certain vaccines may be recommended by physicians to their patients before initiation of cladribine tablets as part of a de-risking strategy; however, this may vary by physician and by country.
Guidance on vaccinations is provided in the Mavenclad SmPC and in the German Competence Network guidance.24,31 Consensus recommendations on vaccinations for patients receiving cladribine tables are provided in Table 4 (Questions 6 and 7).
Management of latent or active infections
Active infection with HIV, tuberculosis or hepatitis must be excluded before initiation of cladribine tablets. 24 Latent infections may be activated upon treatment with cladribine tablets, therefore screening must be performed. 24
Guidance on the management of latent and active infections is provided in the Mavenclad SmPC and in the German Competence Network guidelines.24,31 Guidance on John Cunningham virus activation and screening for progressive multifocal leukoencephalopathy is also provided in EAN/ECTRIMS recommendations. 18 Consensus recommendations for the management of latent or active infections are provided in Table 4 (Question 8).
Management of pregnancy planning and malignancy risk in patients being treated with cladribine tablets
Management of pregnancy planning
There are no clinical studies that have investigated the effect of cladribine tablets on pregnancy outcomes. There are limited data from the cladribine clinical programme on outcomes from women exposed to cladribine (n = 44) and from women whose partners had been exposed to cladribine (n = 10); however, the numbers are too small to draw conclusions. 32
Guidance on the management of pregnancy planning is provided in the Mavenclad SmPC and in the German Competence Network guidelines.24,31 Consensus recommendations on the management of pregnancy planning are provided in Table 5 (Question 9a). Two EF members did not strongly agree with the recommendation on breast feeding, both giving a score of 6. The reason provided for not scoring in the range 7–9 was the duration of abstaining from breast feeding; 7 days was seen to be sufficient.
Risk of malignancy with cladribine tablets
In a pooled analysis of clinical studies and long-term follow up of patients treated with cladribine tablets 3.5 mg/kg, events of malignancies were observed more frequently in cladribine tablets-treated patients compared with patients who received placebo (0.29 versus 0.15 events per 100PY, respectively) (included all studies that used cladribine tablets monotherapy, matching the recommended dose: CLARITY, CLARITY EXT and ORACLE-MS, plus follow-up in PREMIERE). 30 However, in an independent analysis of key MS clinical trials, there was no observed increased incidence of malignancy associated with cladribine tablets, and the incidence rate was similar to that reported for other DMDs.30,33 Furthermore, the incidence of malignancies observed with cladribine tablets 3.5 mg/kg was almost identical to the expected rate of malignancies from the GLOBOCAN matched reference population (0.97, 95% CI 0.44–1.85). 30 There was no increase in malignancies over time in patients treated with cladribine tablets, and no increase in the types of malignancies known to be associated with severe immunosuppression (e.g. non melanoma skin cancer, virally associated tumours and haematological malignancies). 30
Consensus recommendations regarding the risk of malignancy with cladribine tablets are provided in Table 5 (Question 9b). Consensus was achieved on all three recommendations around the risk of malignancy. Three EF members (10%) did not vote in the range 7–9 regarding contraindicated use of cladribine tablets in active malignancy. Two of these EF members provided a score of 5 (neither agree nor disagree) and one gave a score of 3. Reasons for this included ‘there is no evidence that cladribine tablets are contraindicated in active malignancy – it should be at clinician’s discretion depending upon the malignancy’ and ‘I would think it would depend on the type of malignancy’. One voting expert gave no reason for their allocated score.
Treatment switching to and from cladribine tablets and monitoring considerations
Switching to cladribine tablets
There are no randomised clinical studies investigating a switch in DMD to cladribine tablets. However, there are currently a number of treatments for RMS with different mechanisms of action that are candidates for treatment sequencing with cladribine tablets; therefore, advice on switching is necessary and important.
Specific guidance on switching to cladribine tablets is limited. The German Competence Network provide the most comprehensive guidance to date, detailing individual treatments and suggested washout/safety interval times. 31 This has been used as a basis for the development of the consensus recommendations on switching to cladribine tablets as provided in Table 6 (Question 10), which may act as a guide to support switching decisions; however, individual patients may vary and decisions should be made on the specific case.
Switching from cladribine tablets
There are no Class 1 clinical trials specifically investigating a treatment switch from cladribine tablets to other DMDs. In patients treated with cladribine 3.5 mg/kg in CLARITY, approximately 75% remained relapse-free when given placebo during CLARITY Extension. 8 Indeed, in one study, 4 years after the last dose of cladribine tablets 3.5 mg/kg, approximately 25% of patients had switched to another DMD. 34 Across the clinical development programme, 21.5% (124/576) of patients had a record of using another DMD post-treatment with cladribine tablets; the majority of these (56%) received treatment with interferon β-1a. 34
There are very few data on the outcomes of patients who have switched from cladribine tablets to other DMDs, coming only from real-world observations, therefore guidance on the management of this switch is lacking. The PREMIERE long-term follow-up registry includes some patients that have been treated with other DMDs following treatment with cladribine tablets (n = 941). 5 In a snapshot analysis before the study end, the highest proportion of patients switched to IFN β: 23.0%, followed by glatiramer acetate: 9.7%. 5 No specific pattern in the reported serious adverse events and no unexpected safety findings were observed.
Consensus recommendations for switching from cladribine tablets to another DMD are provided in Table 6 (Question 11).
Conclusion
The recommendations described here are the collective opinions of an international group of MS experts. The SC identified and prioritised a number of essential questions concerning the practical use of cladribine tablets in real-life clinical situations. Recommendations were drafted based on a review of the literature and expert opinion and were voted on through a rigorous and transparent process.
The strengths of the recommendations provided here are the result of the large number of experts involved (both SC and EF) and the breadth of geographical representation. Limitations include the lack of available class 1 evidence to support the development of many recommendations.
The recommendations reflect status of knowledge in 2019 and will be updated in a timely fashion when new evidence and/or novel data emerges. The consensus recommendations should provide practical, specific advice to all health-care providers (involved in the treatment and management of patients with MS, address gaps in existing guidance and ultimately improve care.
Footnotes
Acknowledgements
Emma East of Vivid Medical Communications provided editorial assistance, funded by Merck KGaA, Darmstadt, Germany. The SC would like to thank all the experts who contributed their knowledge to this programme by completing the questionnaire and voting on the draft recommendations. Listed below are 29 EF members who are happy to be cited in this document:
Mohammed Al Jumah1, Thomas Berger2, Alexey Boyko3, Helmut Butzkuven4, Marinella Clerico5, Joao Correia de Sa6, Sara Eichau7, Juha-Pekka Erälinna8, Oscar Fernández9, Mark Freedman10, Claudio Gasperini11, Joachim Havla12, Eva Havrdová13, Dana Horáková14, Gunnar Juliusson15, Juerg Kesselring16, Natalia Khachanova17, Joanna Kitley18, Myhr Kjell-Morten19, Marcello Moccia20, Ester Moral21, Thomas Mueller22, Jiwon Oh23, David Paling24, Tobias Ruck25, Johann Sellner26, Aksel Siva27, Natalia Totolyan28, Tjalf Ziemssen29
1National Neuroscience Institute, King Fahad Medical City (KFMC), Riyadh, Kingdom of Saudi Arabia
2Department of Neurology, Medical University of Vienna, Waehringer Guertel 18-20, 1090 Vienna, Austria
3Department of Neurology, Neurosurgery & Medical Genetics of the Pirogov Russian National Research Medical University (RNRMU), Department of Neuroimmunology of the Federal Center of Cerebrovascular Pathology and Stroke, Moscow, Russia
4MS and Neuroimmunology Unit, Department of Neuroscience, Alfred Centre, Monash University, Melbourne, Australia
5Department of Clinical and Biological Sciences, University of Torino, Torino, Italy
6CHLN Hospital de Santa Maria, Faculdade Medicina, Lisboa, Portugal
7Multiple Sclerosis Unit (UEMAC), Virgen Macarena Hospital, Seville, Spain
8University of Helsinki, Finland and University of Turku, Finland
9Instituto de Investigación Biomédica de Málaga (IBIMA), Hospital Regional Universitario de Málaga, Málaga 29010, Spain
10Multiple Sclerosis Research Unit, Professor of Neurology, Division of Neurology, Department of Medicine, The Ottawa Hospital, Canada
11Department of Neuroscience, San Camillo Forlanini Hospital, 00152 Rome, Italy
12Institute of Clinical Neuroimmunology, Ludwig-Maximilians University, Marchioninistr. 15, 81377, Munich, Germany
13Department of Neurology and Center of Clinical Neuroscience, First Faculty of Medicine Charles University and General University Hospital, Katerinska 30, Prague 2, 12000, Czech Republic
14Department of Neurology and Center of Clinical Neuroscience, First Faculty of Medicine Charles University and General University Hospital, Katerinska 30, Prague 2, 12000, Czech Republic
15Department of Hematolog, Skåne University Hospital, Lund University, Lund, Sweden
16Department of Neurology & Neurorehabilitation, Kliniken Valens, Taminaplatz 1, CH-7317 Valens, Switzerland
17Russian National Research Medical University, Department of Neurology, Neurosurgery and Medical Genetics, Moscow, Russia
18Wessex Neurological Centre, Southampton General Hospital, Southampton, SO16 6YD, UK
19Department of Neurology, Haukeland University Hospital, and the Department of Clinical Medicine, University of Bergen, Bergen, Norway
20Department of Neurosciences, Federico II University of Naples, Italy
21Department of Neurology, Hospital de Sant Joan Despí Moises Broggi, Sant Joan Despí, Spain
22Department of Neurology, St. Joseph Hospital Berlin-Weißensee, Gartenstr. 1, 13088 Berlin, Germany
23St. Michael’s Hospital, Keenan Research Centre of the Li Ka Shing Knowledge Institute, Division of Neurology, University of Toronto, Canada
24Royal Hallamshire Hospital, Sheffield and NIHR Sheffield Biomedical Research Centre (BRC), University of Sheffield, Sheffield, UK
25Department of Neurology with Institute of Translational Neurology, University Hospital Muenster, Albert-Schweitzer-Campus 1, 48149 Muenster, Germany
26Neurology and Multiple Sclerosis Center, Landeskrankenhaus Mistelbach-Gänserndorf, Liechtensteinstr. 67, 3120 Mistelbach, Austria
27Istanbul University Cerrahpaşa School of Medicine, Department of Neurology, Clinical Neuroimmunology Unit and MS Clinic, Istanbul, Turkey
281st Saint Petersburg Pavlov State Medical University, Neurology Department, 6-8 Lva Tolstogo Street, St Petersburg, 197022, Russia
29Center of Clinical Neuroscience, University Hospital Carl Gustav Carus, Dresden University of Technology, Germany
Conflict of interest statement
PSS has served on advisory boards for Biogen, Merck Healthcare KGaA, Novartis, Teva, MedDay Pharmaceuticals and GSK; on steering committees or independent data monitoring boards in trials sponsored by Merck Healthcare KGaA, Teva, GSK and Novartis; has received speaker honoraria from Biogen Idec, Merck Healthcare KGaA, Teva, Sanofi-Aventis, Genzyme, Celgene and Novartis. His department has received research support from Biogen, Merck Healthcare KGaA, Teva, Novartis, Roche and Genzyme.
DC is an Advisory Board member of Almirall, Bayer Schering, Biogen, GW Pharmaceuticals, Merck Serono, Novartis, Roche, Sanofi-Genzyme and Teva and received honoraria for speaking or consultation fees from Almirall, Bayer Schering, Biogen, GW Pharmaceuticals, Merck Serono, Novartis, Roche, Sanofi-Genzyme and Teva. He is also the principal investigator in clinical trials for Bayer Schering, Biogen, Merck Serono, Mitsubishi, Novartis, Roche, Sanofi-Genzyme and Teva. His preclinical and clinical research was supported by grants from Bayer Schering, Biogen Idec, Celgene, Merck Serono, Novartis, Roche, Sanofi-Genzyme and Teva.
GG has received speaker honoraria and consulting fees from Abbvie, Actelion, Atara Bio, Almirall, Bayer Schering Pharma, Biogen Idec, FivePrime, GlaxoSmithKline, GW Pharma, Merck & Co., Merck Healthcare KGaA, Pfizer Inc, Protein Discovery Laboratories, Teva Pharmaceutical Industries Ltd, Sanofi-Genzyme, UCB, Vertex Pharmaceuticals, Ironwood and Novartis; and has received research support unrelated to this study from Biogen Idec, Merck & Co., Novartis and Ironwood.
XM has received speaking honoraria and travel expenses for participation in scientific meetings, has been a steering committee member of clinical trials or participated in advisory boards of clinical trials in the past years with Actelion, Bayer, Biogen, Celgene, Genzyme, Merck, Novartis, Roche, Sanofi-Genzyme, Teva Pharmaceutical, Excemed, MSIF and NMSS
DS has received grants and/or personal fees from Teva, Merck Serono, Novartis, Roche, Genzyme, Sanofi-Genzyme, Biogen Inc. and Bayer HealthCare.
PV has received honoraria or consulting fees from Biogen, Sanofi-Genzyme, Servier, Novartis, Merck KGaA, Celgene, Roche, MedDay and Almirall; and research support from Biogen, Sanofi-Genzyme, Bayer and Merck KGaA.
HW is a member of Scientific Advisory Boards/Steering Committees for Bayer Healthcare, Biogen Idec, Sanofi Genzyme, Merck Serono, Novartis, Roche and Teva. He received speaker honoraria and travel support from Bayer Vital GmbH, Bayer Schering AG, Biogen, CSL Behring, EMD Serono, Fresenius Medical Care, Genzyme, Merck Serono, Omniamed, Novartis, and Sanofi Aventis and Teva. He received compensation as a consultant from Biogen Idec, Merck Serono, Novartis, Omniamed, Roche and Sanofi Genzyme. He has received research supports from Bayer Healthcare, Bayer Vital, Biogen Idec, Merck Serono, Novartis, Sanofi Genzyme, Sanofi US, and Teva Pharma as well as German Ministry for Education and Research (BMBF), German Research Foundation (DFG), Else Kröner Fresenius Foundation, Fresenius Foundation, Hertie Foundation, Merck Serono, Novartis, NRW Ministry of Education and Research, Interdisciplinary Center for Clinical Studies (ISKF) Muenster and RE Children’s Foundation.
BY has received honoraria for lectures and advisory boards from Bayer, Biogen, Genpharm, Genzyme, Merck-Serono and Novartis; and has received research grants from Bayer, Biogen, Merck-Serono, Novartis and Pfizer.
HS is an employee of Ares Trading S.A., and affiliate of Merck Serono S.A., Eysins, Switzerland.
PR has received honoraria for lectures/steering committee meetings from Merck, Biogen Idec, Bayer Schering Pharma, Boehringer-Ingelheim, Sanofi-Aventis, Genzyme, Novartis, Teva Pharmaceutical Industries, and Serono Symposia International Foundation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Merck KGaA, Darmstadt, Germany who provided funding for the project. The SC members received financial compensation for attendance at two advisory board meetings as part of this programme. No payments were made to the authors for the writing of this manuscript. The Extended Faculty received no financial compensation for the completion of the on-line questionnaire, or the consensus voting.
