Abstract
Recent advances in multiple sclerosis (MS) management have shifted perspectives on treatment strategies, advocating for the early initiation of high-efficacy disease-modifying therapies (heDMTs). This perspective review discusses the rationale, benefits, and challenges associated with early heDMT initiation, reflecting on the obsolescence of the traditional “first-line” and “second-line” treatment classifications. The article emerges from the last update of the consensus document of the Spanish Society of Neurology on the treatment of MS. During its development, there was a recognized need to further discuss the concept of treatment lines and the early use of heDMTs. Evidence from randomized controlled trials and real-world studies suggests that early heDMT initiation leads to improved clinical outcomes, including reduced relapse rates, slowed disease progression, and decreased radiological activity, especially in younger patients or those in early disease stages. Despite the historical belief that heDMTs involve more risks and adverse events compared to moderate-efficacy DMTs (meDMTs), some studies have reported comparable safety profiles between early heDMTs and meDMTs, though long-term safety data are still lacking. The review also addresses the need for a personalized approach based on patient characteristics, prognostic factors, and preferences, explores the importance of therapeutic inertia, and highlights the evolving landscape of international and national guidelines that increasingly advocate for early intensive treatment approaches. The article also addresses the challenges of ensuring access to these therapies and the importance of further research to establish long-term safety and effectiveness of DMTs in MS.
Plain language summary
Recent progress in treating multiple sclerosis (MS) has changed how doctors think about starting treatments, with more support now for using high-efficacy disease-modifying treatments (heDMTs) early on. This article talks about why starting heDMTs early can be good, what benefits it might bring, and what challenges there might be. It also mentions how the old way of categorizing treatments into “first-line” and “second-line” is becoming outdated. This discussion is based on the latest recommendations from the Spanish Society of Neurology. The article explains that starting heDMTs early can lead to better results for patients, like fewer relapses, slower progression of the disease, and less damage seen on magnetic resonance imaging (MRI). This is particularly true for younger patients or those who are in the early stages of MS. Even though there was a concern that these heDMTs might have more side effects compared to other treatments, recent studies show that they could be just as safe, though more research is needed to be sure about their safety in the long run. The review suggests that treatment should be tailored to each patient, considering their specific situation, what they prefer, and the urgency to start treatment. It also discusses the need to overcome delays in starting these treatments and how treatment guidelines are changing to support starting strong treatments earlier. Finally, the article points out that it is still important to make these treatments accessible to everyone who needs them and to keep researching to understand their long-term safety and effectiveness.
Introduction
Multiple sclerosis (MS) is a complex and unpredictable neurological disorder affecting millions of people worldwide. 1 The treatment landscape for MS has evolved considerably over the past two decades, with the introduction of many disease-modifying therapies (DMTs) that significantly reduce the frequency and severity of relapses, slow disease progression, and improve quality of life and other patient-reported outcomes (PROs). The importance of early treatment has been recognized,2–4 but the type of treatment to provide at onset is still under debate.
Historically, DMTs were classified into first-line and second-line therapies, with the former being moderately effective (moderate-efficacy; meDMT) and associated with a relatively safe risk profile and a lower cost, and the latter being highly effective (high-efficacy; heDMT) but presenting a higher risk of adverse events (AEs) and a higher cost. Classification of DMTs into “lines” of treatment has shaped the escalation-based treatment approach, which has been the most popular strategy in the management of MS patients. In the escalation approach, initial treatment involves meDMT (“first-line”) with a known and relatively safe risk profile; if disease activity persists or recurs despite sufficiently long and regular treatment, therapy is escalated to an heDMT (“second-line”). Escalation can be based on conventional step-care and watchful waiting or rapid, depending on the tolerated disease activity. 5 Alternatively, treatment with an heDMT can be initiated early, known as induction or early intensive approach, at the time of diagnosis or as soon as possible. Both strategies are based on assessing the patient’s characteristics and prognostic factors,6–9 and considering the risks and efficacy of available DMTs. One proposed classification of DMTs according to their efficacy has been monoclonal antibodies (alemtuzumab, natalizumab, ocrelizumab, ofatumumab), sphingosine-1-phosphate (S1P) receptor modulators (fingolimod, siponimod, ozanimod, and ponesimod), cladribine, and mitoxantrone as heDMT, and dimethyl fumarate, glatiramer acetate, interferons, teriflunomide, and diroximel fumarate as meDMT. 10 However, this classification varies between publications.5,11–14
We consider that this dichotomous classification system in lines of treatment is now obsolete and does not reflect the latest scientific evidence. In this article, we aimed to discuss the rationale, benefits, and challenges of early initiation of heDMTs in patients with MS, based on available data and guidelines, and to provide a position statement on revising the concept of first- and second-line therapies. This perspective review article emerges from a comprehensive discussion and exchange of ideas during the preparation of the last update of the consensus document of the Spanish Society of Neurology on the treatment of MS. 15
Early initiation of high-efficacy disease-modifying treatments
Scientific support for heDMT at early disease stages
A growing body of evidence demonstrates that early initiation of heDMTs may be a more effective approach to managing MS (Table 1). Current randomized controlled trials (RCT) are evaluating the efficacy and safety of early intensive and escalation approaches for relapsing MS (RMS) (ClinicalTrials.gov identifier: NCT03500328), 14 but the results are not yet available. To date, only the findings from pairwise comparisons in RCT (one meDMT vs one heDMT)16–18 or observational studies assessing the results with early heDMT compared to meDMT have been published thus far.11,13,19–27 These studies have consistently reported improved clinical outcomes in MS patients treated with early heDMTs compared to those following escalation treatment strategies, including reduced relapse rates, slower disease progression, and decreased radiological activity.
Summary of real-world studies on the use of early heDMT in MS patients.

This study analyzed the effect of treatment on nonresponders and treatment-naïve patients, but only data from treatment-naïve patients is reported here.
ALZ, alemtuzumab; ARR, annualized relapse rate; AZA, azathioprine; CDP, confirmed disability progression; CDW, confirmed disability worsening; CI, confidence interval; CLB, cladribine; CYC, cyclophosphamide; DAC, daclizumab; DMF, dimethyl fumarate; EDSS, Expanded Disability Status Scale; EHE, early high-efficacy; EIT, early intensive treatment; ESC, escalation; FNG, fingolimod; GA, glatiramer acetate; heDMT, high-efficacy disease-modifying therapies; HR, hazard ratio; INF-B, interferon beta; MRI, magnetic resonance imaging; MTX, mitoxantrone; NTZ, natalizumab; OCR, ocrelizumab; PS, propensity score; RRMS, relapsing-remitting multiple sclerosis; RTX, rituximab; SPMS, secondary progressive multiple sclerosis; TFN, teriflunomide.
In terms of safety, RCT and extended studies have reported several risks of heDMTs including hematologic abnormalities, infections, malignancies, secondary autoimmunity, cardiovascular disease, neurovascular events, and teratogenic effects.28–30 For instance, ocrelizumab has led to infusion reactions and reductions in immunoglobulin (Ig) levels (particularly IgM), but for the majority of patients Ig levels remained above lower limit of normal (LLN) during 6 years of treatment. 31 Alemtuzumab have resulted in thyroid alterations and in rare cases of immune thrombocytopenic purpura, and lymphopenia, raising the risk of serious infections.32–34 In patients treated with ofatumumab for up to 5 years, mean IgG levels remained stable, while mean IgM levels decreased but remained above the LLN. 35 The threat of progressive multifocal leukoencephalopathy (PML) is elevated particularly with natalizumab. 36 PML risk can be mitigated by identifying factors such as positive John Cunningham virus antibody status that increase PML risk. 37 Cladribine tablets has resulted in transient decreases in absolute lymphocyte counts. 38 Lymphopenia is a well-known AE of fingolimod, 39 and other S1PR modulators. 40 Also, increased transaminases, heart blocks, infections, and potential teratogenicity have been associated with S1PR modulators,30,41,42 which should be paused before pregnancy.41,43 The OPERA, ASCLEPIOS, and ULTIMATE trials demonstrated that, except for infusion- or injection-related reactions, the safety profiles of ocrelizumab, ofatumumab, and ublituximab were comparable to those of IFN beta and teriflunomide.17,44,45
Several real-world studies have also reported similar rates of AEs or serious AEs (SAEs) between patients receiving early heDMT or meDMT.11,25 For instance, Harding et al. 11 reported that patients receiving alemtuzumab and natalizumab presented no SAEs, but 1.4% of patients with meDMT reported SAEs. In another study, patients on meDMT were more likely to discontinue the therapy due to AEs than those receiving heDMTs. 26 However, other studies revealed a worse safety profile with early use of heDMT. For example, Prosperini et al. 22 found that SAEs occurred significantly more frequently after induction (10.7%) than escalation (2.4%). Note that some of the observational studies aimed at comparing the effectiveness of early use of heDMT and meDMT (included in Table 1) did not report data on safety20,21,23,24 and, therefore, information on safety when these strategies are compared is limited.
Most of the available safety data for heDMTs are derived from RCT, which had a relatively short follow-up. Longer-term observational studies are needed to fully characterize the safety profiles of these treatments, particularly concerning rare SAEs such as malignancies, which may have a delayed onset. A systematic review with network meta-analysis concluded that commonly reported AEs were overall similar among heDMTs. 46 However, considering the limitations of indirect comparisons, head-to-head comparisons of the safety profiles of different heDMTs should be conducted. Since the main argument for choosing an escalation approach is stronger evidence on meDMT safety profile, evidence pointing to a similar profile would strongly challenge this classical argument. It is important to mention as well that potential undertreatment of MS with meDMTs is not exempt from risks. Most patients who undergo escalation already had sustained accumulation of disability while receiving meDMTs. 11
Despite differences in study design, patient populations, and specific heDMTs evaluated, real-world studies collectively support the early use of heDMTs in MS patients, who qualify for heDMT, to maximize treatment outcomes. 47 By intervening early in the disease course, heDMTs may mitigate irreversible neurological damage, reduce conversion from relapsing to progressive MS, and improve long-term disability outcomes for MS patients. 20 Data from recent meta-analyses have shown that early initiation of heDMTs, compared to escalation, had higher efficacy in preventing disability progression and a similar safety profile in the short-term. 48 Other meta-analyses concluded that, among all DMT, an meDMT (interferon beta-1b subcutaneous) had the highest probability of the best safety for SAEs, whereas an heDMT (alemtuzumab) showed the highest probability of the best safety for discontinuation due to AEs. 49 Further studies examining real-world effectiveness and safety outcomes with early use of heDMT in the long-term are warranted.
There are data showing that the most active patients, 12 the youngest, 23 and those with shorter disease duration 23 may benefit most from early initiation with heDMT. The impact of age on DMT efficacy has also been studied by a meta-analysis of randomized trials, which showed that the efficacy of immunomodulatory DMTs on MS disability strongly decreased with advancing age. 50 Specifically, heDMTs outperformed meDMTs in reducing disability only for patients younger than 40.5 years. 50 However, the results of this study are controversial due to methodological limitations. The model evaluated efficacy on disease progression based on the limited duration of RCT, and it only considered age at initiation of DMTs. Furthermore, other variables such as baseline clinical–radiological activity or disease duration were not considered. Moreover, the clinical course after discontinuation of treatment was not evaluated.
Additionally, AEs with heDMT, including infections, are typically more common in older patients.19,51 The initial phase following MS onset appears to be critical, as during this period heDMTs can notably impact the disease course and decelerate MS progression. The benefits of initiating heDMTs early during the disease course likely stem from reducing neuroinflammation 52 and brain volume loss 53 during the earliest stages, thereby helping to control disease progression. By impeding early inflammatory damage and controlling relapses at the beginning of the disease course, early heDMT use may seize the “window of opportunity” for preventing accumulation of irreversible neuronal damage and achieving more favorable long-term outcomes. On the other hand, the possible reduced efficacy and higher risk for AEs of heDMTs in older patients could be linked to two interconnected physiological processes associated with age: immunosenescence and inflamm-aging. Immunosenescence refers to the gradual deterioration of the immune system with advanced age, and inflamm-aging is the result of this low-grade proinflammatory state. 54 The age-related immune decline could potentially diminish the target of the heDMTs, resulting in poorer disease control and a higher likelihood of complications. Furthermore, age-associated comorbidities and concurrent medications could contribute to a heightened risk of AEs. Figure 1 presents classical arguments for the escalation approach and current arguments for early use of heDMTs.

Arguments for the escalation approach and for early use of heDMTs.
Timing and patient profile
Considering all the abovementioned evidence, reserving heDMT only for those patients perceived to have the most active disease does not seem to be justified. However, the exact timing for starting the heDMT is a matter of debate. International and national guidelines play a crucial role in defining this “window of opportunity,” in particular, and in tailoring treatment approaches, in general. By synthesizing a vast body of research and consensus among experts, these guidelines aim to ensure consistency in patient care, reducing variability in outcomes and promoting best practices based on the latest scientific research.
The current version of the European guidelines (European Committee for Treatment and Rehabilitation in Multiple Sclerosis, together with the European Academy of Neurology) and American guidelines (American Academy of Neurology) for treating patients with MS, do not explicitly address whether to use an escalation strategy or to opt for early use of heDMTs.55,56 The European guidelines state that the choice between available treatments should be based on the patient characteristics, comorbidities, disease severity and activity, as well as the drug safety profile and accessibility. 55 The American guidelines also highlight that patient preferences should be respected and taken into consideration when choosing the DMT. 56 Both guidelines recommend monitoring programs for increased safety with DMTs. The updated version of these guidelines will address early treatment decisions, supporting the early start of heDMTs, depending on patient characteristics and disease activity.57,58
At a national level, the guidelines for the management of MS have recently been updated by the Spanish Society of Neurology. The previous version of these guidelines stated that patients with relapsing-remitting multiple sclerosis (RRMS) were candidates for any of the “first-line” DMTs and that patients with clinically aggressive MS may benefit from “second-line” DMTs approved at the time the guidelines were established (fingolimod, natalizumab, or alemtuzumab). 59 The recently updated guidelines 15 advocate for treating with heDMTs as a first therapeutic option, after considering the characteristics and disease of the patient and evaluating the risks and benefits of the treatment. Specifically, the early use of heDMTs may be considered in patients with a first MS relapse or in patients with factors associated with a poor prognosis,6,7 based on the demographic, clinical, and radiological characteristics of the patients and on available biomarkers (Table 2). Here, it is worth noting that not all prognostic factors, however, predict disease worsening equally. A study with 1058 patients with clinically isolated syndrome found that demographic and topographic characteristics were low-impact prognostic factors, the presence of oligoclonal bands was a medium-impact prognostic factor, and the number of lesions on brain MRI was a high-impact prognostic factor.15,60 The use of tools designed to identify the presence or absence of specific variables may help in the evaluation of these prognostic factors at the individual level. 6 A detailed and individualized analysis of prognostic factors could be very useful in making treatment decisions.
Factors and biomarkers of poor prognosis.

CSF, cerebrospinal fluid; EBV, Epstein–Barr virus; EDSS, Expanded Disability Status Scale; GCL, ganglion cell layer; Gd+ gadolinium-enhancing; GFAP, glial fibrillary acidic protein; IgM, immunoglobulin M; MS, multiple sclerosis; NfL, neurofilament light; OCB, oligoclonal bands; OCT, optical coherence tomography; PRLs, paramagnetic rim lesions, RNFL, retinal nerve fiber layer; SELs, slowly expanding lesions.
In patients recently diagnosed and treatment-naïve, heDMTs could be the first treatment option if the patient presented two or more relapses in the previous year or had one relapse in the previous year and poor prognosis (due to incomplete remission and/or relapse followed by disability or other factors) or two or more gadolinium-enhancing (Gd+) lesions on T1, or spinal cord or infratentorial lesions, or 9 or more T2 lesions 15 (Figure 2). These recommendations on early heDMT in newly diagnosed treatment-naïve patients are in line with the opinion-based orientations previously reported by another group aimed at establishing criteria to identify patients with RMS eligible for heDMT. 84 In the updated Spanish guidelines, 15 a decision was made not to classify specific DMTs as either meDMT or heDMT. This approach aimed to maintain flexibility and allows neurologists to categorize the treatments based on their individual judgment. Recommendations for specific classes of treatments were not included in the guidelines. However, we would like to comment that for some classes of treatments such as S1P modulators, only one relapse and no poor prognosis factors6,7 would be required to start treatment.41,43,85
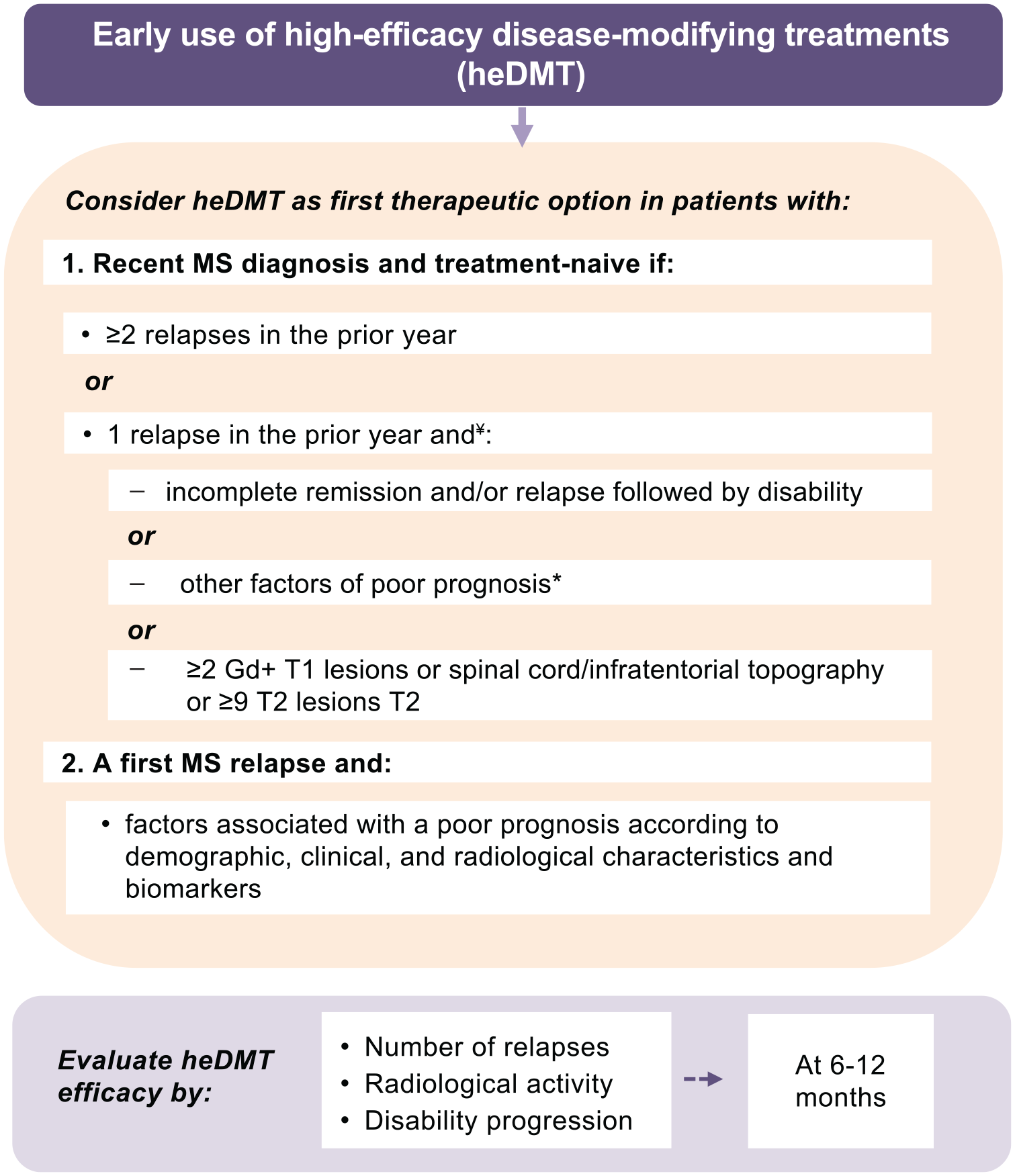
Considerations for an early use of heDMTs in patients with MS as recommended by the Spanish MS guidelines.
In addition to the recommendations in national and European guidelines regarding early use of heDMT,57,86 several worldwide experts have advocated for adopting this approach in the last few years.10,84,87–90 All experts agree that the use of heDMTs at the disease’s onset must be tailored to each case, considering the unique preferences and needs of the individual patient. In this regard, when it comes to patient preferences, patients with MS have reported the drug administration mode as one of the most important attributes of a DMT. 91 Patients with MS have different preferences for the route of administration of DMTs, influenced by factors such as treatment frequency, outcomes, safety profiles, and individual considerations. Some studies reported a general preference for oral DMTs over injectable options when other attributes were held constant.92,93 However, the preferred route of administration may change with variations in treatment frequency, as the route of administration is closely related to the frequency of treatment. For instance, when the treatment frequency and AEs remained constant, most patients preferred oral DMTs over injections. 93 However, this preference shifted to injections when pills had to be taken three times a day and injections were required only once a week. 93 It is important to consider, however, that these studies were conducted more than 10 years ago, when several currently available heDMT had not yet been approved.
If patients had to choose between parental routes, some studies have shown that more than twice as many patients preferred infusions to self-injections, 94 whereas other studies reported equal preference for intravenous or subcutaneous injections 95 or a preference for subcutaneous administration. 96 Among the arguments provided by patients who preferred infusions were that they did not need to administer it themselves and that they felt looked after when integrated in an infusion scheme. 97 In contrast, patients who preferred subcutaneous injections mentioned the convenience and comfort of home treatment and appreciated the time-saving and increased independence that this administration offered.96,98,99 Lifestyle and personal preferences, such as the desire for minimal disruption to daily activities and reduced hospital visits, are therefore important factors. The higher monitoring requirements and administration time associated with infused heDMTs can be burdensome not only for some patients but also for healthcare providers, 100 although they improve treatment adherence.
In terms of treatment outcomes, relapse-free rate and symptom progression have been considered determinants of preference by patients.91,92 In this sense, it is worth noting that most heDMTs align with the first preference, by having stronger efficacy to reduce relapses and symptoms progression. However, not all studies on patients DMTs preferences have yielded the same results. Other studies have shown that patient’s preferences were mainly driven by risk minimization.101,102 A study that assessed patient preferences for features of injectables showed that treatment efficacy was rated as important as a reduction in injection frequency and in some AEs. 103 Since studies were conducted in different countries, patient profiles, and at different time points, discrepancies in treatment preferences may be due to cultural differences between countries, the methodology used to assess these preferences in the studies, or a limited availability of heDMTs in less recent studies.91,92,94,97,101,102 Qualitative research can complement quantitative data to assess treatment preferences. 104 Both types of studies should be conducted to better understand patient preferences of the administration route in the present.
Challenges, controversies, and future directions in MS management
Despite the compelling evidence supporting the early initiation of heDMTs, several challenges and controversies remain in the management of MS and may prevent the early use of these therapies. One of the main challenges is therapeutic inertia. In the context of MS, therapeutic inertia is defined as the failure to initiate or intensify treatments despite evidence of disease activity. 105 The presence of therapeutic inertia has been observed in almost one-fifth of patients with MS in Portugal. 105 Among factors leading to therapeutic inertia are errors in risk assessment, low tolerance to uncertainty, status quo, underestimation of patient’s needs, herding, clinician’s limited education in decision-making, and formal training in risk management. 106 Educational intervention to facilitate decisions to recognize scenarios where MS patients would require heDMT have been proven feasible and useful to reduce therapeutic inertia among neurologists. 107 Acknowledging patient-related factors to therapeutic inertia (e.g., misinterpretation of clinical activity, AEs of new DMTs, aversion to change, poor communication) is also key. 106 Ensuring that patients have access to available data of current DMTs, and that they understand these data, will provide them with tools and more confidence to participate in shared decision-making. In line with these, patients with MS consider that healthcare professionals spend too much time focusing on the risks of DMTs, and have expressed their interest to discuss both risks and benefits. 108
On another note, limited access to heDMT imposed by reimbursement authorities affects the use of these treatments as well. For instance, despite the European Medicines Agency indication statement for ocrelizumab, cladribine, or ofatumumab for use in all patients with RMS with active disease,109–111 these treatments have been restricted to a second or later line of treatment for patients with RMS, or as a first-line exceptionally in patients with an aggressive and fast course, by the health authorities of some European countries like Spain.112–114 Future evidence confirming the benefits of early use of heDMT, together with recommendations from clinical guidelines, might help to modify the reimbursement conditions in the future. Also, recommendations from international organizations may influence local authorities. The lists of essential medicines by the World Health Organization (WHO) is aimed at providing guidance to governments and enhance global access to treatments within health systems. 115 In the last update in 2023, the WHO has added cladribine to the list of essential medicines, which represents a milestone in MS treatment, especially in developing countries.
Moreover, cost savings is crucial not only for health authorities but also for the families of patients with MS and for the society as a whole. Cost-effectiveness analyses have concluded that initial investment in the early use of heDMTs may lead to long-term savings by slowing the progression of the disease. 116 Cost-effectiveness can be detected in a 5-year time horizon 48 or even sooner. 117 Early initiation of ofatumumab, for instance, has been estimated to save (excluding the costs of acquiring the DMTs) €35,328 per patient annually when compared to teriflunomide and €24,373 per patient annually when compared to a 3-year delayed start of ofatumumab. 117 Ofatumumab was associated with reduced informal care time and fewer disability-adjusted life years than teriflunomide. 117 Indirect costs, such as costs related to lost productivity or absenteeism due to MS, have also been observed to improve with heDMTs. In Australia, patients treated with heDMTs were two to three times more likely to have improvements in amount of work, work attendance, and work productivity compared with those treated with classical injectables (glatiramer acetate and interferons). 118 More economic evaluations and cost-effectiveness analyses are needed to support the value of early heDMT initiation in the context of healthcare resource allocation. In line with this, patients with MS have highlighted the need to educate a broader range of stakeholders about the personal impact of MS, enhancing their awareness of how reimbursement limitations affect individuals. 108
Another challenge is the heterogeneity of MS, which makes it difficult to establish a one-size-fits-all approach to treatment. While early heDMT initiation may be beneficial for most patients, some might not benefit as much as expected from this approach. Identifying which patients would benefit from early heDMT initiation the most remains an ongoing challenge. Further research is needed to validate and implement reliable prognostic biomarkers of treatment response and disease course. These biomarkers will help to refine personalized treatment approaches in MS.
Importantly, recent advances in digital technologies and artificial intelligence (AI) are opening up new possibilities for MS research and patient management. Studies using AI-based approaches, digital twins—virtual representations of patients that incorporate detailed medical history, clinical data, and other relevant information—or the combination of both represent promising avenues for understanding which patients are most likely to benefit from early heDMTs.119,120 The concept of digital twins allows for the simulation of treatment scenarios and the prediction of individual patient outcomes, favoring more personalized data-driven medicine. 121 Although the application of digital twins and AI-based methodologies is still in its infancy in the context of MS management, these approaches hold great potential for refining treatment selection and optimizing patient outcomes. The use of machine learning algorithms can help identify patterns in clinical data and biomarkers to predict treatment response and monitoring, among other applications.122,123 These predictive models can then be used to inform clinical decision-making, facilitating the selection of optimal treatment strategies for individual patients. As our understanding of MS and the capabilities of AI continue to advance, it is expected that these novel methodologies will play an increasingly important role in the management of MS and the evaluation of early heDMTs.
Ongoing research and RCT will help address some of the challenges and controversies surrounding the early initiation of heDMTs in MS management. For example, the DELIVER-MS trial (ClinicalTrials.gov identifier: NCT03535298) is an ongoing RCT comparing early initiation of heDMTs to an escalation treatment approach in patients with RRMS. The study will follow-up patients for 36 months and assess the efficacy of these approaches in terms of brain volume loss (primary endpoint), progression, and PROs. 14 Another pragmatic RCT evaluating the efficacy of the escalation approach or early heDMT on disability accumulation is the TREAT-MS trial (ClinicalTrials.gov identifier: NCT03500328). The primary outcome measures are time to sustained disability progression (Expanded Disability Status Scale) up to 75 months (ClinicalTrials.gov identifier: NCT03500328). Other outcome measures include disability measured with other tools (Timed 25 Foot Walk Test, Nine-hole Peg Test), cognition, PROs, brain volume loss, lesions on MRI, and retinal layer thickness by optical coherence tomography, among others. These RCT comparing early initiation of heDMTs to the escalation approach will provide high-quality data that are often required to support changes in clinical practice guidelines and reimbursement policies by regulatory agencies and payers. Conducting RCTs could also help overcome the therapeutic inertia that may exist among some clinicians who may prefer meDMTs due to familiarity and perceived safety concerns with heDMTs,105,106,124 and which cannot be disregarded. Also, the close monitoring of AEs in the controlled environment of RCT will help confirm whether the safety profile of both approaches are comparable, as suggested by prior observational studies. 23
Conclusion
The traditional classification of “first-line” and “second-line” treatments in MS is no longer relevant in the era of early heDMT. The reviewed evidence and the last update of the consensus by the Spanish Society of Neurology on the treatment of MS support the notion that considering early initiation of heDMTs can optimize clinical outcomes, delay disease progression, and improve the quality of life and long-term prognosis for MS patients. We join other voices advocating that initiating heDMT early is the optimal strategy for achieving the most favorable long-term outcomes for individuals with MS. However, the decision to initiate heDMT early is complex and depends on several factors, such as the expertise of the neurologist with these therapies, long-term safety data, prognostic factors, the information that patients have and their preferences, availability of therapies and disease monitoring, local regulatory requirements, and national and international guidelines. Future studies should further compare the short- and long-term safety of the two approaches. Results from the ongoing RCT that directly compared early versus delayed start of heDMTs will shed light on the effect of using each of these approaches in the disease course of MS.
Footnotes
Acknowledgements
We would like to express our gratitude to Laura Prieto del Val from Evidenze Health Spain S.L.U. for her support as a scientific advisor and medical writer during the elaboration of this article (writing of first the draft of the manuscript, inclusion of inputs from authors, preparation of the final version for submission, and submission to the journal), funded by Novartis S.A. in accordance with Good Publication Practice (GPP3) guidelines.
