Abstract
Background and Methods:
Limited data are available on the safety of fingolimod in pregnant women. We estimated the risk of adverse pregnancy outcomes in women with multiple sclerosis (MS) exposed to fingolimod either shortly before or during pregnancy in prospectively collected cases from clinical trials, observational studies, surveillance programs, and spontaneous reports.
Results:
The prevalence of major malformations among live births does not appear to be significantly higher than those in the general population and the unexposed MS population. Similarly, the prevalence of cardiac malformations observed in this analysis was not significantly different from that of the general population. Proportions of miscarriage were in line with those of the general and unexposed MS population and no specific pattern of birth defects was identified.
Conclusions:
These data can help inform healthcare professionals and women with MS exposed to fingolimod during conception.
Introduction
Multiple sclerosis (MS) affects more women than men, 1 and at the time of diagnosis many women are of child-bearing age. Patients, partners and healthcare professionals need to consider the benefits and risks of continuing MS treatment when planning a pregnancy and in the case of unplanned pregnancy. Documentation of fetal and maternal risks of fingolimod exposure periconceptionally and in early pregnancy will help inform pregnancy management choices.
Fingolimod (FTY720; Gilenya®) is a sphingosine 1-phosphate (S1P) receptor modulator approved for the treatment of relapsing MS. During embryonic development, S1P1, S1P2 and S1P3 receptors, which are expressed within the endothelial cells and vascular smooth muscle cells, regulate vascular development and its function. S1P 1 signaling plays an important role during embryonic neurogenesis and subsequent development of nervous system.2,3 Preclinical studies in rabbits and rats have demonstrated reproductive toxicity of fingolimod, including fetal loss and congenital defects (notably persistent truncus arteriosus and ventricular septal defect). 4 At the time of the launch of fingolimod, there were sparse data available on the safety of fingolimod in pregnant women. After discontinuation of fingolimod, it takes approximately 2 months to eliminate the drug from the body; 5 hence, the label recommends that all women of child-bearing potential should use effective contraception during and for 2 months following discontinuation of fingolimod treatment. 5 Additional prospective data on pregnancy outcome are necessary to provide more reliable information for patients, their families, and prescribers.
The objective of this report is to describe the estimated risk of adverse pregnancy outcome following fingolimod exposure, based on all data captured in the Novartis safety database (NSDB) to date from a variety of sources.
Methods
Data sources and definition of cases
The NSDB includes pregnancy outcomes from the following data sources with a database that was locked at the end of February 2017: (a) the Gilenya® Pregnancy Exposure Registry (referred to as the Registry), launched in May 2011 to prospectively collect safety data on maternal, fetal and infant outcomes associated with fingolimod exposure during pregnancy and up to 8 weeks before the last menstrual period (LMP); (b) PRegnancy outcomes Intensive Monitoring (PRIM), which was instituted on the 1 March 2014 to collect additional data from pregnant patients prospectively reported to Novartis who were not enrolled in the Registry; (c) other pregnancy cases reported to Novartis outside of these programs from a variety of sources including clinical trials, observational studies, surveillance programs, and spontaneous reports.
For the Registry, cases were considered prospective if at the time of enrollment, no prenatal testing had been done and pregnancy outcome was not known. Cases were considered retrospective if prenatal tests had been performed at enrollment, while the patient is still pregnant, regardless of their results. For PRIM and NSDB, prospective cases included those for which prenatal testing was performed and in which the results were either normal or not known. When analyzed as part of the NSDB, the NSDB definition of ‘prospective cases’ was applied to Registry cases. Prenatal testing was conducted according to the routine medical practice in the corresponding countries. Tests included but were not limited to targeted ultrasound, amniocentesis, nuchal translucency scan or chorionic villus sampling.
Data reported here are obtained from prospective cases unless otherwise specified.
The protocol for the Registry was approved by the local Institutional Review Boards in all the countries where the Registry was launched and written informed consent was obtained from all the patients.
Pregnancy outcomes from the above sources are put into context with MS patients who were not previously exposed to any disease-modifying therapy (unexposed MS patients) and with the general population.
Pregnancy outcomes
The primary outcome measure was the prevalence of major congenital malformations, defined as any structural defects with recognized surgical, medical, or cosmetic importance as per the European Surveillance of Congenital Anomalies (EUROCAT) guidelines, while anomalies were qualified as minor if they were of no serious medical or cosmetic consequence to the child. Other outcome measures included miscarriages, elective abortions, stillbirths, neonatal deaths (counted in live births), and term and preterm live births with or without congenital malformations.
Congenital malformations were judged by external adjudicators (qualified pediatrician, clinical geneticist, teratologist, pediatric neurologist, nephrologist, toxicologist, or clinical pharmacologist), according to the EUROCAT or other guidelines. 6 In cases of discrepancy, cases were classified as the more severe of the two adjudications.
Statistical analysis
Descriptive statistics, including mean ± standard deviation, median (range) and proportions [95% confidence interval (CI)] were used to describe the data. CIs for the proportions of pregnancies were calculated using the Clopper–Pearson method. The prevalence of major congenital malformations in live births and in live births, stillbirths and terminations of pregnancy due to fetal anomaly (TOPFA) cases was reported for the Registry, PRIM and NSDB, and the prevalence of major cardiac malformations was reported for the NSDB.
Results
By the end of February 2017, a total of 1246 prospectively collected pregnancies with 1255 infants were recorded in the NSDB (Figure 1).
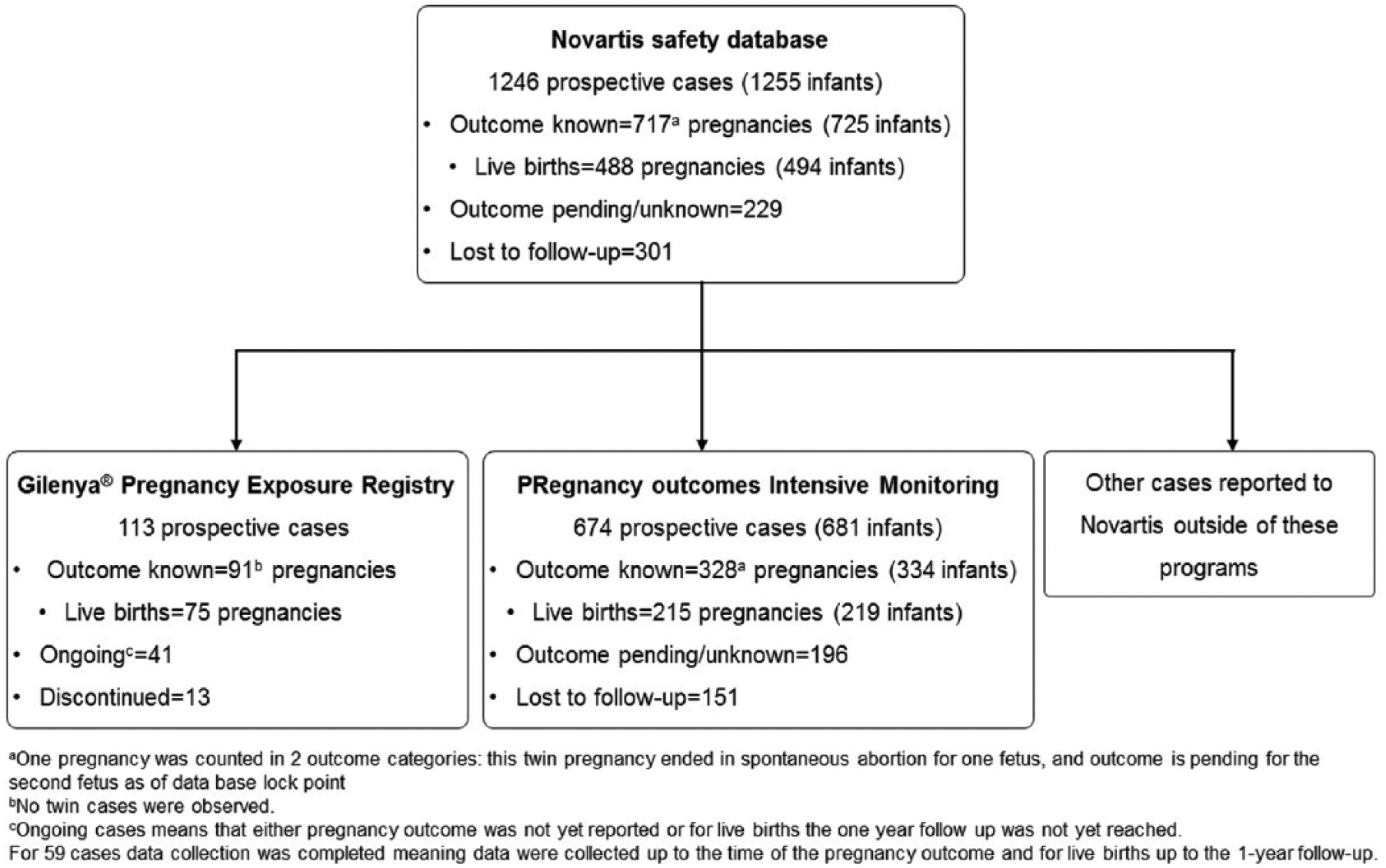
Disposition of cases collected in the Novartis safety database from various data sources.
Baseline characteristics
Maternal baseline demographics and MS disease characteristics are presented for the Registry and PRIM cases (Table 1). Availability of this information for other cases in the NSDB was limited. The mean age at LMP was comparable between the Registry and PRIM (31.7 versus 31.6 years). In both data sources, more than 75% of the patients were exposed to fingolimod either within 8 weeks before LMP or during the first trimester.
Baseline characteristics and pregnancy information at enrollment/reporting.

This is based on 88% of PRIM cases.
Extreme range in the Registry is attributed to a case with an informed consent date on 20 January 2015 and an estimated delivery date on 14 December 2015, resulting in a negative gestational age at enrollment and another case which did not report any prenatal test and therefore is considered prospective; in PRIM outliers may be due to technical issues with how manufacturer’s received date is determined.
This is based on 75.9% of PRIM cases with known outcomes that provided data.
In PRIM, patients are categorised as per fingolimod discontinuation, therefore the categories are mutually exclusive; in the Registry, patients are included in all categories in which fingolimod was taken.
LMP, last menstrual period; MS, multiple sclerosis; N, number of cases enrolled; NA, not available; PRIM, PRegnancy outcomes Intensive Monitoring; SD, standard deviation.
Pregnancy outcome
The prevalence of major congenital malformations among live births was 4.0% in the Registry, 2.7% in PRIM, and 3.9% overall in NSDB. The prevalence of major congenital malformations (excluding chromosomal anomalies) among live births, stillbirths and cases with TOPFA was 5.3% in the Registry, 3.2% in PRIM and 4.5% overall in the NSDB (Table 2). In the NSDB, the prevalence of major cardiac malformations was 1.5% in live births and 1.6% in live births, stillbirths and TOPFA cases.
Prevalence of major congenital malformations among the prospective cases with known outcomes.

Number of term and preterm live births (including neonatal deaths) with major congenital malformations as the numerator; all term and preterm live births (including neonatal deaths) as the denominator. There were no major chromosomal anomalies reported in live births.
Number of cases with major congenital malformations among all term and preterm live births (including neonatal deaths) plus stillbirths plus TOPFA as the numerator; number of all term and preterm live births (including neonatal deaths) plus stillbirths plus TOPFA as the denominator.
482 infants (476 pregnancies) are considered for the prevalence calculation following the exclusion of 12 pregnancies for which the specific timing of exposure was not confirmed.
In the Registry, no chromosomal abnormality was reported.
CI, confidence interval; NSDB, Novartis safety database; PRIM, PRegnancy outcomes Intensive Monitoring; TOPFA, terminations of pregnancy due to fetal anomaly.
All pregnancy outcomes observed separately in the Registry and PRIM as well as for all NSDB cases are presented in Table 3. Miscarriages were reported in 6.6%, 14.7% and 13.5% of fetuses in the Registry, PRIM, and NSDB, respectively. Elective terminations were reported in 9.9%, 19.8% and 17.4% of fetuses, respectively. No specific pattern of malformations was identified among the observed cases. A description of major, minor malformations and anomalies that were not otherwise specified (NOS), observed in prospective cases, is provided in Table 4. Cardiac anomalies were reported in 10 infants, musculoskeletal anomalies in 9 infants and renal anomalies in 7 infants (Table 4).
All pregnancy outcomes among prospective cases with known outcomes.

Data are presented as n (%), unless specified otherwise.
Number of infants is used as the denominator to calculate the percentages. In the Registry, this is equal to the number of pregnancies.
For the NSDB, infants may appear in two categories (such as live birth with subsequent neonatal death); therefore, the sum will be more than the actual number of infants.
Includes all malformations (major, minor, chromosomal, and not otherwise specified).
NSDB, Novartis safety database; PRIM, PRegnancy outcomes Intensive Monitoring.
Description of anomalies observed among prospective cases.

ASD, atrial septal defect; NOS, not otherwise specified; TOPFA, terminations of pregnancy due to fetal anomaly; VSD, ventricular septal defect.
Of the 58 pregnancies enrolled retrospectively in the Registry, the outcome was known for 53 cases: 45 full-term live births including one infant with congenital malformation and eight preterm live births including one with congenital malformation.
Discussion/conclusion
The prevalence of major congenital malformations (excluding chromosomal anomalies) of all three prospective data sources observed in this analysis was 2.7% (95% CI: 1.0; 5.9) to 4.0% (0.8; 11.2) in live births) and 3.2% (1.3; 6.4) to 5.3% (1.5; 12.9) in live births, stillbirths and TOPFA. In the general population, the prevalence of major malformations varied between 2.0% (2.0; 2.1) 7 and 4.5% (4.5; 4.5) 8 for live births, and 2.6% (2.6; 2.6) 7 and 6.9% (6.6; 7.2) 9 for live births, stillbirths and TOPFA. In the unexposed MS population, the prevalence of major congenital malformations in live births ranged between 1.4% (0.0; 7.8) 10 and 6.8% (2.5; 14.3; Note: Wherever the CIs were not available in the published sources, they were calculated by Clopper–Pearson method). 11 The prevalence of major congenital malformations do not appear to be significantly more prevalent among pregnant women exposed to fingolimod 0.5 mg than what would be expected in the general7–9 and unexposed MS population.10,11 There was a trend towards lower prevalence of major congenital malformations in prospectively collected data from PRIM compared with the Registry, potentially due to the different nature of data collection and variation in the definition of prospective cases. We did not identify any specific patterns of birth defects supporting fingolimod-induced effects. Our sample size, the largest for fingolimod exposure during pregnancy to date, is adequate to detect an 80% increase in the prevalence of major malformations (assuming a background prevalence of 3%; 80% power; two-sided test α = 0.05).
A total of 11 cardiac defects (including major and minor) were identified in 10 infants. Of those, 8 were considered as major (7 defects in 6 live births and 1 defect in TOPFA) and were included in prevalence calculations (Table 4). The prevalence of major cardiac malformations in the NSDB was 1.5% (0.6; 3.0) in live births and 1.6% (0.7; 3.2) in live births, stillbirths and TOPFA cases. In the general population, the prevalence varied from 0.2% 12 to 1.1% 13 in live births and from 0.8% 14 to 1.1% (1.0; 1.2) 15 in live births, stillbirths and TOPFA. Overall, major cardiovascular anomalies reported with fingolimod showed a point estimate of reporting rate that is higher than prevalence estimates from the general community; however, the community prevalence estimates are contained within the 95% CIs, indicating these are not statistically significant. Given the small number of cases with confirmed diagnosis, and confounding factors in the presentation of cases with congenital cardiac defects, this is not considered a safety signal for fingolimod at this point in time but will be re-evaluated as new cases are reported.
We also found that the proportions of miscarriages from the three data sources (6.6−14.7%) were within the range reported for the general population [14.2 (13.2; 15.3) 16 to 20.9 (18.9; 23.0) 17 ] and the unexposed MS population [4.3% (0.1; 22.0) 18 to 21.1% (13.4; 30.6) 10 ]. A higher proportion of miscarriages was observed in PRIM compared with the Registry. This may be because patients in the Registry were enrolled later than they were reported in PRIM due to the informed consent process, which may have led to a few cases of abortions in the early first trimester being missed. The same has been evidenced by the gestational age at the time of enrollment/reporting.
Data from populations of women with MS exposed to other MS-specific medications are limited. The prevalence of major congenital malformations observed in cases exposed to natalizumab ranged from 3.9% (0.8; 11.0) 10 to 7.6% (4.9; 11.1) in live births and 7.9% (5.2; 11.4) among live births, stillbirths and elective terminations. 19 In cases exposed to glatiramer acetate, prevalence was 8% (0.9; 26.0) in live births 20 and 1.5% (0.2; 5.2) in live births, stillbirths and TOPFA. 11 The prevalence in cases exposed to interferon β was 8.3% (0.2; 38.5) in live births 21 and ranged from 3.1% (1.2; 6.3) 22 to 14.3% (1.8; 42.8) in live births, stillbirths and TOPFA. 21 No birth defects were observed in cases exposed to teriflunomide in a limited number of pregnancies.
The main limitation of this analysis is the high number of lost to follow-up cases in the NSDB, including a number of cases in PRIM, which is typical of voluntary postmarketing reporting systems. Furthermore, a slow enrollment rate restricted the amount of information available from the Registry. Enrollment in registries, and prompt reporting and follow up in surveillance programs are critical to the evaluation of pregnancy outcomes, and healthcare professionals who treat women of child-bearing age are urged to participate in this ongoing important effort. The current data do not warrant a change in the recommendation provided in the label due to relatively limited numbers. However, this report will provide useful information for women who become pregnant while taking fingolimod, their families and healthcare professionals.
Footnotes
Acknowledgements
The authors would like to thank all the patients and physicians who contributed to the Registry; Sreelatha Komatireddy and Marie-Catherine Mousseau (Novartis) for medical writing assistance. All authors edited the manuscript for intellectual content and approved the final version. The final responsibility for the content lies with the authors.
Funding
The Registry was funded by Novartis Pharma AG, Basel, Switzerland.
