Abstract
Background:
Fingolimod slows heart rate (HR) due to vagomimetic effects and might cause additional cardiovascular autonomic changes. While the time course of HR changes is well described, the extent and course of cardiovascular autonomic changes upon fingolimod initiation has not yet been evaluated. This study, therefore, intended to assess cardiovascular autonomic changes during the first 6 h after fingolimod initiation.
Methods:
In 21 patients with relapsing-remitting multiple sclerosis (RRMS), we recorded respiration (RESP), electrocardiographic RR interval (RRI), systolic and diastolic blood pressure (BPsys, BPdia) at rest, before and 0.5, 1, 2, 3, 4, 5, and 6 h after fingolimod initiation. We calculated parameters of total autonomic modulation [RRI standard deviation (RRI-SD), RRI coefficient of variation (RRI-CV), RRI-total powers], mainly sympathetic cardiac modulation [RRI low frequency (LF) powers], sympathetic BP modulation (BPsys-LF powers), parasympathetic modulation [square root of the mean squared difference of successive RRIs (RMSSD), RRI high frequency (HF) powers], sympatho-vagal cardiac balance (RRI-LF/HF ratios), and baroreflex sensitivity (BRS). We compared parameters between the eight measurements [analysis of variance (ANOVA) or Friedman test with post-hoc analysis; significance: p < 0.05].
Results:
After fingolimod initiation, RESP, BPsys, and BPsys-LF powers remained unchanged while RRIs, RRI-CV, RRI-SD, RRI-total powers, RRI-LF powers, RMSSD, RRI-HF powers, and BRS increased after 1 h and rose to peak values occurring after 5, 1, 2, 2, 1, 4, 4, and 4 h, respectively. After 3 h, BPdia had decreased significantly and was lowest after 5 h. RRI-LF/HF ratios decreased to a nadir after 4 h.
Conclusions:
The increases in parasympathetic and overall cardiac autonomic modulation and in BRS seen with fingolimod initiation are theoretically beneficial for the MS patient’s cardiovascular system. However, long-term studies must show whether these effects persist or are attenuated (e.g. due to S1P1 receptor down-regulation upon continued fingolimod therapy).
Introduction
Fingolimod, a sphingosine-1-phosphate (S1P) receptor modulator [Camm et al. 2014; Gold et al. 2014] approved as the first new oral disease-modifying therapy for relapsing-remitting multiple sclerosis (RRMS) [Pelletier and Hafler, 2012], causes transient heart rate (HR) slowing due to its vagomimetic cardiac activity [Dimarco et al. 2014; Gold et al. 2014]. HR-slowing upon fingolimod initiation is due to the activation of G-protein-gated potassium channel IKACh on atrial myocytes, based on a structural similarity of fingolimod to S1P opening these potassium channels [Kovarik et al. 2008; Camm et al. 2014]. Potassium efflux from myocytes and cell membrane hyperpolarization result in slowed depolarization and HR-slowing [Kovarik et al. 2008; Camm et al. 2014].
Recently, Simula and colleagues reported an increase in parasympathetic HR modulation averaged during the first 24 h after fingolimod initiation [Simula et al. 2016]. However, the specific time course of cardiovascular autonomic changes after fingolimod initiation has not yet been evaluated. Fingolimod may cause bradycardia in 0.5–2.4% of patients, and even cardiovascular serious adverse events (0.9% of patients), including atrioventricular blocks (0.4% of patients) [Pelletier and Hafler, 2012; Gold et al. 2014; Hilz et al. 2015]. Therefore, it is of relevance to better identify fingolimod-related cardiovascular autonomic changes and their time course.
Since atropine blocks fingolimod-related HR-slowing [Kovarik et al. 2008], fingolimod seems to primarily affect parasympathetic control. So far, it seems unclear whether fingolimod also has clinically relevant direct effects on sympathetic ganglia. Theoretically, S1P1, S1P2, and S1P3 receptors are ubiquitously distributed [Camm et al. 2014]. S1P4 receptors are mainly found in lymphocytes, and S1P5 receptors are mainly found in white-matter oligodendrocytes [Camm et al. 2014]. To our knowledge, there is only the autopsy study by Nishimura and colleagues that showed that S1P1 receptors are not expressed in human sympathetic ganglia [Nishimura et al. 2010]. Therefore, the effects of fingolimod on the sympathetic nervous system still seem to require further study. However, HR changes may also alter sympathetic modulation (e.g. via the baroreflex loop) [Eckberg and Sleight, 1992; Hilz et al. 2010, 2011] and may even change baroreflex sensitivity (BRS) which is considered a measure of cardiovascular prognosis [La Rovere et al. 1998; Hilz et al. 2010, 2011].
Mostly, fingolimod-induced HR-slowing returns to baseline values within 4–6 h in healthy persons [Kovarik et al. 2008; Schmouder et al. 2012] and MS patients [Dimarco et al. 2014; Gold et al. 2014]. Thus, the European Medicines Agency recommends HR monitoring upon fingolimod initiation for at least 6 h or longer if HR is at the lowest value 6 h after the first dose [http://www.ema.europa.eu/ema].
To better assess cardiovascular autonomic changes in MS patients during the initial 6 h after fingolimod initiation, we monitored HR, blood pressure (BP), and respiration in RRMS patients who had no clinical signs or symptoms of autonomic dysfunction and determined autonomic modulation before and during the first 6 h of fingolimod treatment.
Patients and methods
Among outpatients seen at the Multiple Sclerosis Clinic of the Department of Neurology, University of Erlangen-Nuremberg, Germany, we screened patients who had been diagnosed with RRMS according to the 2010 revised McDonald criteria [Polman et al. 2011] and had been recommended for new treatment with fingolimod.
In an effort to rule out that other influences on autonomic modulation might confound possible fingolimod effects on cardiovascular autonomic modulation, we only included patients in whom we had ruled out effects on the autonomic nervous system due to other diseases, medications or MS itself. Consequently, we were only able to include a rather small number of 21 RRMS patients in whom we had assured that there were no other diseases or risk factors possibly afflicting the autonomic nervous system, such as diabetes, hypertension, depression, epilepsy, hyper- or hypothyroidism, and no clinically overt signs or symptoms of cardiovascular autonomic dysfunction. We also excluded patients who were on any medication that might alter autonomic function, such as antihypertensive medications, antidepressants, oral contraceptives, antihistamines, asthma medications, medications for hyper- and hypothyroidism, and cholinesterase inhibitors. Since autonomic dysregulation is a frequent finding in MS [Hengstman and Kusters, 2011; Razazian et al. 2014; Racosta et al. 2015; Hilz, 2016] and might intertwine with the effects of fingolimod, we excluded patients in whom the examination prior to fingolimod initiation unveiled abnormal results of autonomic testing, including uroflowmetry, assessment of residual urine volume, and abnormal results in the Composite Autonomic Symptom Score 31, an autonomic questionnaire [Sletten et al. 2012].
In the 21 RRMS patients (14 women and 7 men, mean age 33.5 ± 1.8 years, time since diagnosis 6.0 ± 1.1 years), we monitored HR as electrocardiographic RR intervals (RRIs), systolic and diastolic beat-to-beat blood pressure (BPsys, BPdia), and respiration under supine resting conditions before and 0.5, 1, 2, 3, 4, 5, and 6 h after fingolimod initiation.
The study was approved by the Ethics Committee of the University of Erlangen-Nuremberg, Germany, and registered at the German Clinical Trial Register (DRKS00004548). Prior to the study, all patients had given their written informed consent according to the declaration of Helsinki.
Patients who had received previous disease-modifying treatments were taken off their previous medication for at least the period consistent with current recommendations [Pelletier and Hafler, 2012].
We tested the patients between 9 a.m. and 4 p.m., after a resting period of at least 40 min that ensured a stable cardiovascular situation. Cardiovascular autonomic testing was performed in a quiet room with an ambient temperature of 24°C and stable humidity. Study conditions were standardized throughout the entire observational period. None of our patients had any electrocardiographic abnormalities prior to fingolimod initiation.
We estimated MS severity using the expanded disability status scale (EDSS) [Kurtzke, 1983] and the MS functional composite (MSFC) score [Cutter et al. 1999]. MSFC consists of three parts: a timed 25-foot walk, the 9-hole peg test, and the paced auditory serial addition test. The raw scores for each of the three components are converted to z-scores using the National Multiple Sclerosis Society Task Force MS population as the reference population [Fischer et al. 1999; Fox et al. 2007b], then the three z-scores are averaged to calculate the overall z-score of MSFC [Fischer et al. 1999; Fox et al. 2007b].
During each time point of interest (i.e. before and 0.5, 1, 2, 3, 4, 5, and 6 h after fingolimod initiation), we recorded RRIs (ms) by 3-lead electrocardiography, BPsys and BPdia (mmHg) by finger-pulse photoplethysmography (Portapress; TPD-Biomedical Instrumentation, Amsterdam, the Netherlands), and respiratory frequency [RESP, (min−1)] by chest impedance measurements [Hilz et al. 2015, 2016] for 3 min. From each of the 3-min recordings, we extracted the most stationary and artefact-free 120 s epochs to average values of RRIs, BPsys, BPdia, and RESP and to calculate the autonomic parameters described below. Biosignal data were digitized and displayed on a personal computer and a custom designed data acquisition and analysis system (SUEmpathy™, SUESS-Medizintechnik, Germany) and stored for offline analysis [Hilz et al. 2015, 2016].
We calculated autonomic parameters in the time and frequency domains [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2015, 2016].
As time domain parameters, we determined RRI standard deviation (RRI-SD) and the coefficient of variation of the RRI (RRI-CV), both reflecting cardiac sympathetic and parasympathetic modulation [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2015, 2016]. Moreover, we calculated the square root of the mean squared differences of successive RRIs (RMSSD), reflecting cardiac parasympathetic modulation [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2015, 2016].
To assess cardiovascular sympathetic and parasympathetic modulation in the frequency domain, we performed trigonometric regressive spectral analyses (TRSs) of RRI and BPsys values sampled during the 120 s epochs at the eight time points of interest.
We determined sympathetic and parasympathetic modulation of RRI and BP in the low frequency (LF; 0.04–0.14 Hz) and high frequency (HF; 0.15–0.50 Hz) ranges [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2011, 2015, 2016].
LF oscillations of RRI at rest reflect sympathetic outflow and, to an undetermined degree, also parasympathetic modulation; LF oscillations of BP are related to sympathetic outflow only [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2015]. HF oscillations of RRIs reflect parasympathetic modulation [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2015], whereas BP fluctuations in the HF range are primarily a mechanical consequence of respiration-induced fluctuations in venous return and cardiac output [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2015, 2016]. The magnitude of LF and HF oscillations was determined as the integral under the power spectral density curves of RRI (ms2/Hz) and BP (mmHg2/Hz) for the LF and HF frequency bands, and was expressed as LF and HF powers of RRI (ms2) and BP (mmHg2) [Hilz et al. 2015, 2016].
As an approximation of the total power (TP) of RRI oscillations and an index of overall cardiac autonomic modulation, we calculated the sum of LF and HF powers in the range from 0.04–0.5 Hz [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2010, 2011, 2016]. Moreover, we also calculated the ratios between RRI oscillations in the LF and HF ranges, and used the LF/HF ratios of RRI as index of the balance between sympathetic and parasympathetic influences on HR modulation [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2016]. To adjust for inter-individual differences in sympathetic or parasympathetic modulation and to assess relative changes, we normalized LF and HF powers of RRI by calculating percentage values of LF and HF powers in relation to TP of RRI fluctuation, with RRI-LFnu = [LF/(LF + HF)] × 100%, and RRI-HFnu = [HF/(LF + HF)] × 100% [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996].
Finally, we determined BRS using the TRS software which selected pairs of LF and HF oscillations of BPsys and RRI with high coherence [Hilz et al. 2011, 2016]. Coherence spans from 0 (i.e. no association), to 1 (i.e. maximum association). High coherence at a specific frequency (e.g. >0.7), indicates a stable-phase relation, and thus synchronization, between two signals oscillating at this frequency [Hilz et al. 2011, 2016]. Then, the sensitivity of the baroreflex loop (ms mmHg−1) was derived as gain-values from changes in RRIs (ms) in relation to changes in BPsys (mmHg) [Hilz et al. 2011, 2016].
To evaluate influences of age, disease duration, and body mass index (BMI) on cardiovascular autonomic modulation prior to fingolimod treatment and on any possible autonomic effects of fingolimod, we correlated the patients’ age, disease duration, and BMI with baseline cardiovascular autonomic parameters assessed prior to fingolimod initiation, and with the maximum changes of those biosignals and autonomic parameters that increased or decreased significantly from respective baseline values within the first 6 h after fingolimod initiation.
Statistical analysis
Data were tested for normal distribution using the Shapiro–Wilk test.
We assessed differences between biosignals and autonomic parameters determined at the eight time points. If data were normally distributed, we performed analysis of variance (ANOVA) for repeated measurements (general linear model), with ‘assessments’ (before, 0.5, 1, 2, 3, 4, 5, and 6 h after fingolimod initiation) as the within-subject factor. Suitability of the ANOVA was assessed by Mauchly’s test of sphericity. In case of violation of the sphericity assumption, the Greenhouse–Geisser correction was employed. We performed post-hoc paired Student’s t-tests to evaluate differences between values sampled prior to fingolimod initiation and values sampled at each of the seven time points after fingolimod intake.
If data were not normally distributed, we performed Friedman tests to assess differences between values sampled at the eight time points, and Wilcoxon tests to evaluate differences between values sampled prior to fingolimod and values sampled at each of the seven time points after fingolimod intake.
To correlate patient age, disease duration, and BMI with biosignals and autonomic parameters assessed prior to fingolimod treatment and with their maximum changes upon fingolimod initiation, we used the Spearman rank correlation test for non-normally distributed data and the Pearson test for normally distributed data.
Data were expressed as mean ± standard error of the mean (SEM). EDSS values were expressed as median (interquartile range). Significance was set at p < 0.05. Due to the exploratory nature of the study, no adjustment for multiple comparisons was made. For data analysis, we used a commercially available statistical program (IBM SPSS Statistics for Windows, version 20.0. Armonk, NY, USA).
Results
All 21 RRMS patients completed the biosignal recordings at the eight time points of interest. Their EDSS scores were 2.0 (1.5–3), MSFC z-scores were 0.16 ± 0.09.
Biosignals at the eight time points of interest
Respiratory frequency before and at the seven time points after fingolimod intake remained unchanged (p > 0.05).
Compared with RRI values before fingolimod treatment, RRIs were significantly higher; HR was significantly lower 1, 2, 3, 4, 5, and 6 h after fingolimod initiation and the highest RRI was reached after 5 h (Table 1). None of the participants developed bradycardia with HR values below 60 bpm or any electrocardiographic abnormalities.
Time course of biosignals and autonomic time domain parameters in 21 patients with RRMS before and within 6 h after fingolimod initiation.

Data are expressed as mean ± SEM. Significant differences between before and after fingolimod initiation are highlighted as
ANOVA, analysis of variance; BPdia, diastolic blood pressure; BPsys, systolic blood pressure; HR, heart rate; RESP, respiration; RMSSD, square root of the mean squared differences of successive RRIs; RRI, electrocardiographic RR interval; RRI-CV, coefficient of variation of RRIs; RRI-SD, standard deviation of RRIs; RRMS, relapsing-remitting multiple sclerosis; SEM, standard error of the mean; TP, total power.
Upon fingolimod initiation, BPsys and BPdia had increased slightly though not significantly after 30 min. After 1 h, BP values reached a peak with a nonsignificant average increase in BPsys by 6.7 mmHg (BPsys at baseline versus BPsys after 1 h: 116.6 ± 2.4 versus 123.3 ± 3.5 mmHg; p = 0.053). Then, BPsys and BPdia decreased until the fifth hour and re-increased in the sixth hour. However, only BPdia values had decreased significantly 3, 4, 5, and 6 h after fingolimod intake (Table 1 and Figure 1).
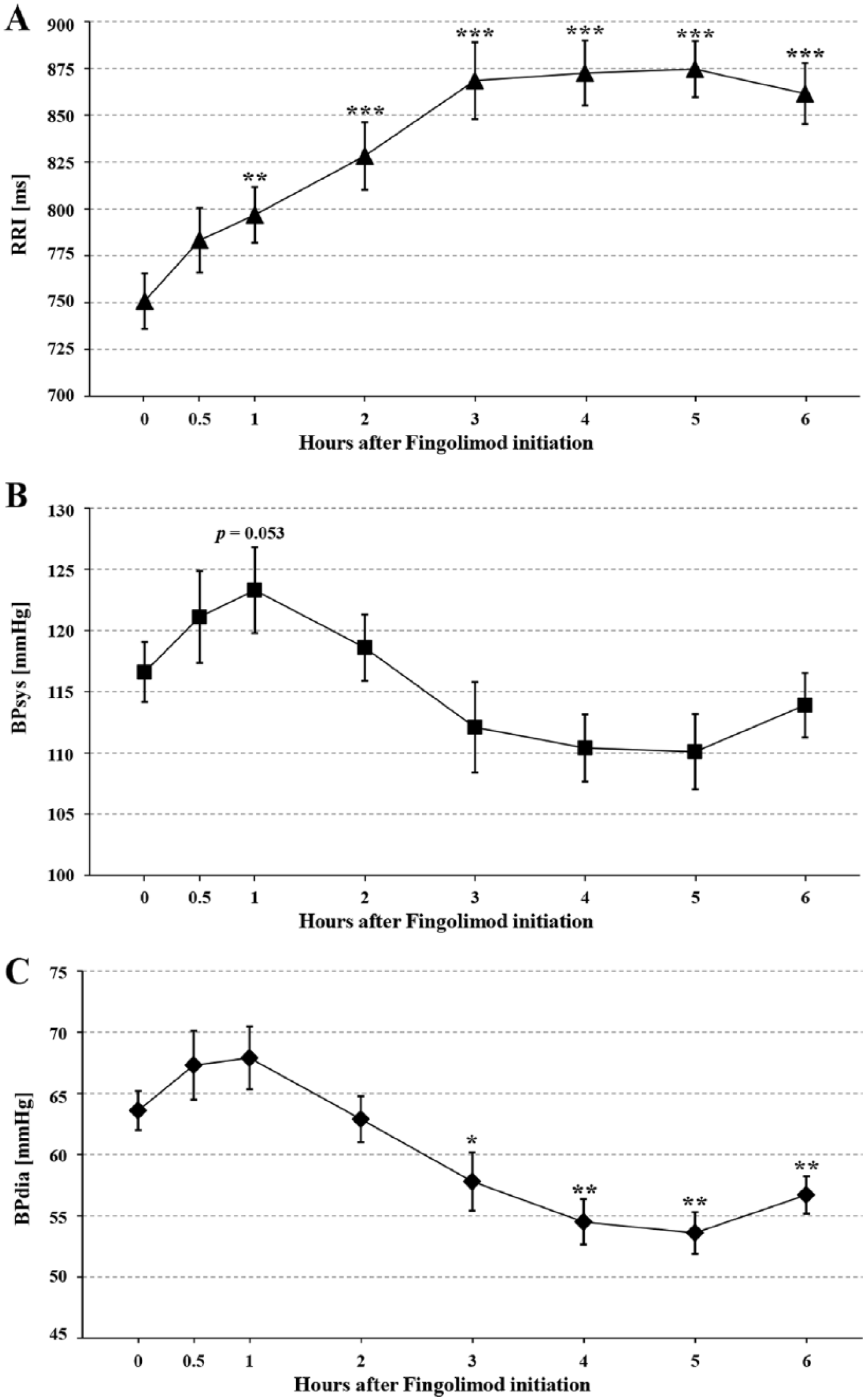
RRI (panel A), BPsys (panel B) and BPdia (panel C) in 21 patients with RRMS before and 0.5, 1, 2, 3, 4, 5, and 6 h after fingolimod initiation.
Time domain parameters at the eight time points
The three time domain parameters of autonomic RRI modulation slightly increased after fingolimod initiation (Table 1, Figure 2). However, the increase in parameters of total autonomic modulation was significant only after the first hour for RRI-CV, and after the first, second, third and fourth hour for RRI-SD. Values were highest after 1 h for RRI-CV and after 2 h for RRI-SD. In contrast, parasympathetic RMSSD values steadily increased until the fourth hour and then started to slightly decrease. RMSSD values were significantly higher than at baseline even after the first hour, and remained higher until the sixth hour (Table 1, Figure 2).
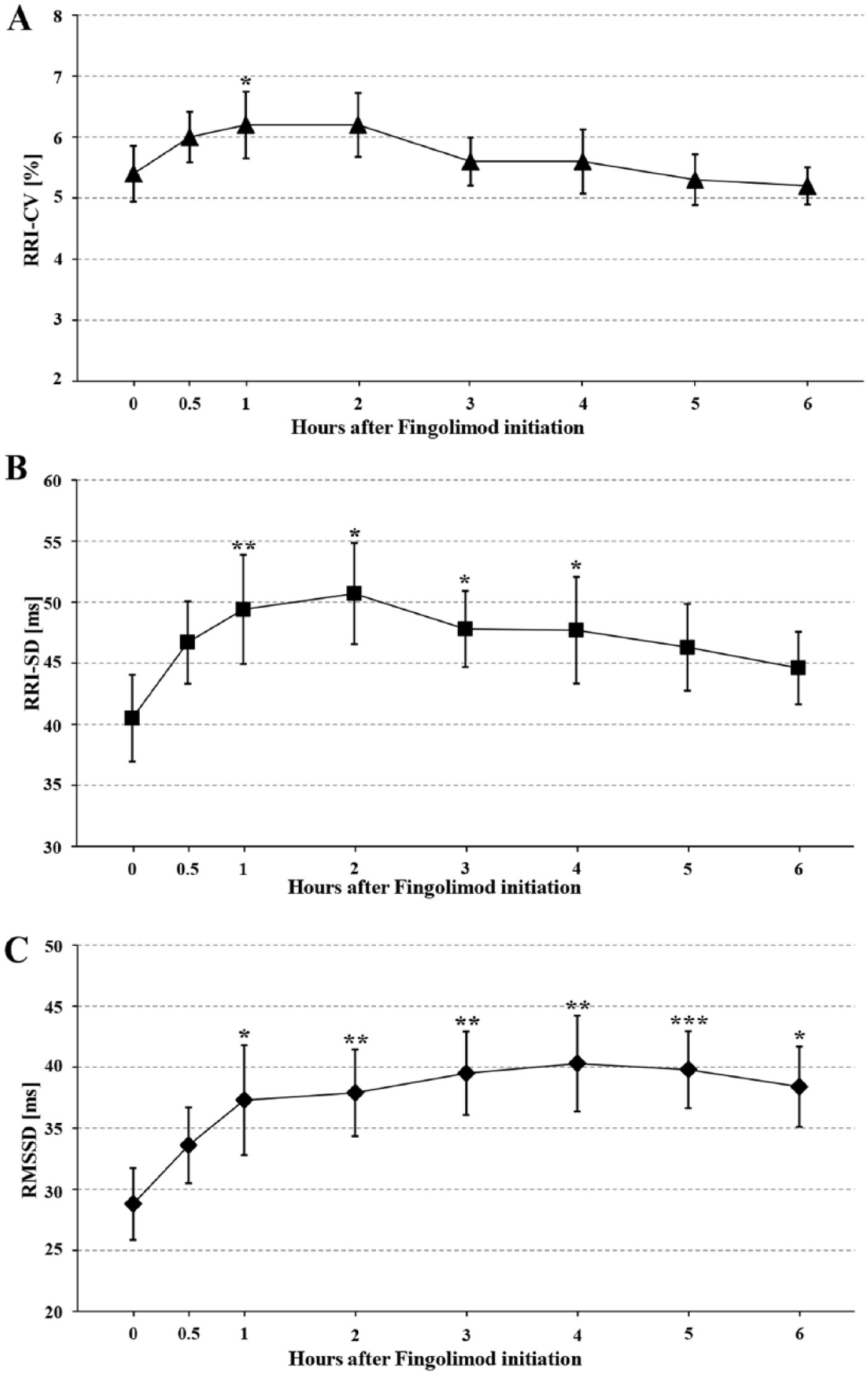
RRI-CV (panel A), RRI-SD (panel B) and RMSSD (panel C) in 21 patients with RRMS before and 0.5, 1, 2, 3, 4, 5, and 6 h after fingolimod initiation.
Frequency domain autonomic parameters at the eight time points of interest
Upon fingolimod initiation, parasympathetically-mediated RRI-HF powers had already slightly increased after 30 min and were significantly higher than baseline values after 1, 2, 3, 4, and 5 h, with a peak after 4 h and a subsequent decline.
Similarly, RRI-LF powers had slightly increased after 30 min and significantly increased after 1 h while subsequent values steadily returned to baseline values (Table 2, Figure 3).
Time course of autonomic frequency domain parameters in patients with RRMS before and within 6 h after fingolimod initiation.

Data were expressed as mean ± SEM. Significant differences between before and after fingolimod initiation are highlighted as bold and italic numbers. The number of asterisks indicates the level of significance, with
ANOVA, analysis of variance; BPsys, systolic blood pressure; BRS, baroreflex sensitivity; HF, high frequency; LF, low frequency; nu, normalized unit; RRI, electrocardiographic RR interval; RRMS, relapsing-remitting multiple sclerosis; SEM, standard error of the mean; TP, total power.
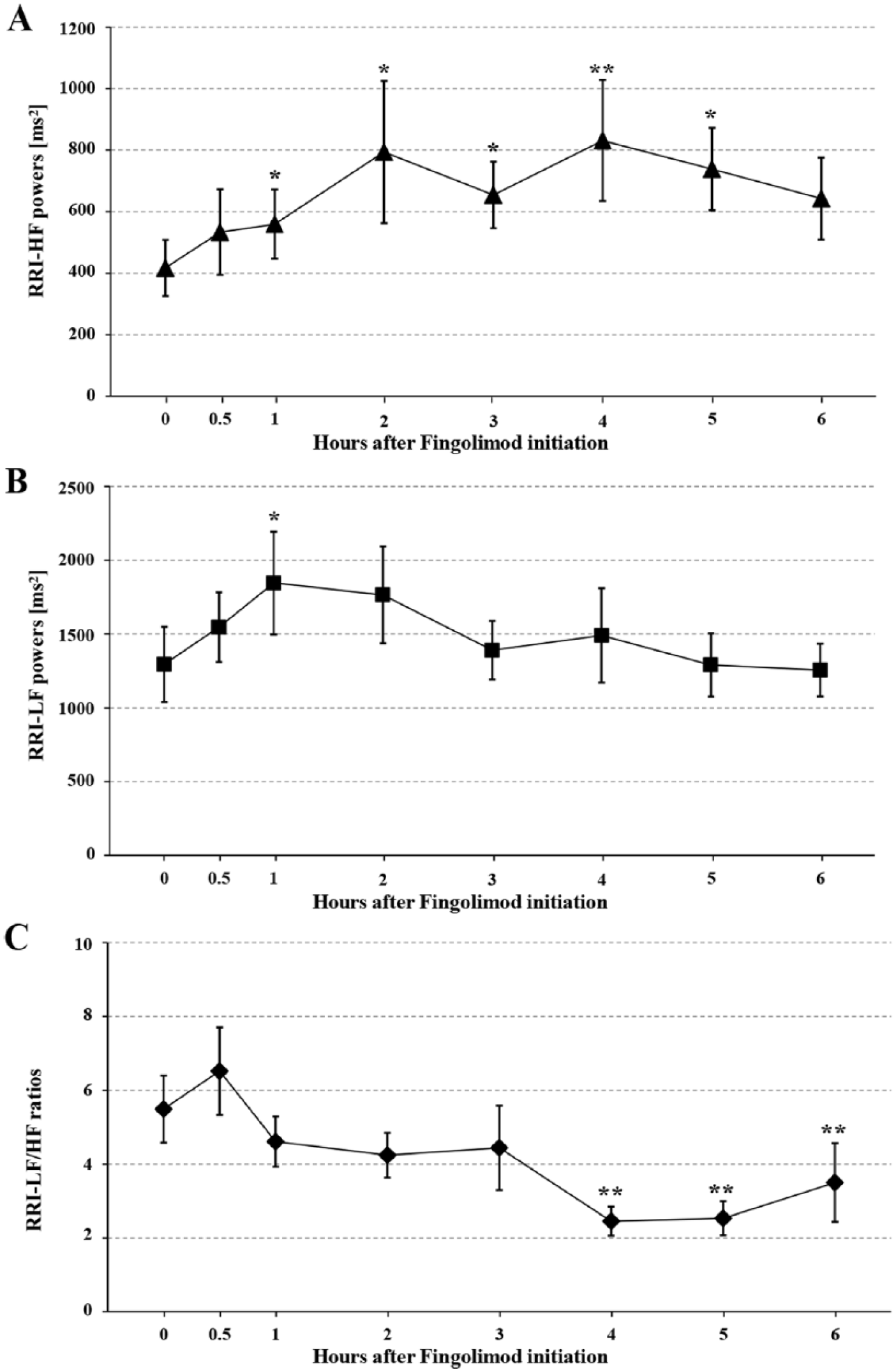
RRI-HF powers (panel A), RRI-LF powers (panel B), RRI-LF/HF ratios (panel C) in 21 patients with RRMS before and 0.5, 1, 2, 3, 4, 5, and 6 h after fingolimod initiation.
However, the index of sympatho-vagal balance, the RRI-LF/HF ratio had slightly increased after 30 min, but then decreased and was significantly lower than at baseline after 4, 5, and 6 h, with its nadir at the fourth hour (Table 2, Figure 3).
RRI-TP had increased slightly after 30 min, and significantly after 1 and 2 h but then steadily decreased towards baseline values (Table 2, Figure 4A).

RRI-TP (panel A), RRI-HFnu powers (panel B), RRI-LFnu powers (panel C), in 21 patients with RRMS before and 0.5, 1, 2, 3, 4, 5, and 6 h after fingolimod initiation.
The relative values in RRI-HF powers (i.e. the RRI-HFnu powers), steadily increased after 0.5, 1, and 2 h, and were significantly higher than baseline values after 3, 4, 5, and 6 h, with peak values after 4 h (Table 2, Figure 4B). Changes in RRI-LFnu powers showed the mirror image of changes in RRI-HFnu powers, with a steady decrease reaching significance after 3, 4, 5, and 6 h and lowest values after 4 h (Table 2, Figure 4C).
Changes in sympathetically-mediated LF powers of BP modulation and mechanically-mediated HF powers of BP modulation were not significant upon fingolimod initiation. There was only a minor, insignificant increase in both parameters, 30 min after fingolimod intake (Table 2, Figure 5A and B).

BPsys-LF powers (A), BPsys-HF powers (B), and BRS (C) in 21 patients with RRMS before and 0.5, 1, 2, 3, 4, 5, and 6 h after fingolimod initiation.
In contrast, BRS had increased slightly after 30 min and significantly after 1, 2, 3, 4, 5, and 6 h, with peak values after 4 h (Table 2, Figure 5C).
Correlation between age, disease duration and BMI with cardiovascular autonomic parameters prior to fingolimod initiation
Patient age negatively correlated with RRI-SD (Pearson correlation coefficient = −0.497, p = 0.022), RRI-CV (Pearson’s correlation coefficient = −0.534, p = 0.013), RMSSD (Pearson’s correlation coefficient = −0.491, p = 0.024), RRI-HF powers (Spearman’s correlation coefficient = −0.566, p = 0.007), RRI-HFnu powers (Pearson’s correlation coefficient = −0.457, p = 0.037) and BRS (Spearman’s correlation coefficient = −0.438, p = 0.047), and positively correlated with RRI-LFnu powers (Pearson’s correlation coefficient = 0.457, p = 0.037). Disease duration positively correlated with RRI-LFnu powers (Pearson’s correlation coefficient = 0.473, p = 0.030) and negatively correlated with RRI-HFnu powers (Pearson’s correlation coefficient = −0.473, p = 0.030). BMI did not correlate with any of the cardiovascular autonomic parameters before fingolimod initiation in our MS patients.
Correlation between age, disease duration, and BMI with the maximum changes of biosignals and autonomic parameters recorded at their nadir or peaks during the first 6 h after fingolimod initiation
Only patient age correlated significantly with the decrease of RRI-LF/HF ratios seen 4 h after fingolimod initiation (Spearman’s correlation coefficient = 0.456, p = 0.038). Otherwise, age, disease duration, and BMI did not correlate significantly with any changes of biosignals and autonomic parameters recorded after fingolimod initiation.
Discussion
Our data show that fingolimod-induced cardiovascular and autonomic changes go beyond the well-known HR-slowing with its nadir occurring approximately 4 h after fingolimod initiation [Kovarik et al. 2008; Dimarco et al. 2014; Gold et al. 2014]. Fingolimod-associated HR-slowing does not seem to be a primarily autonomic phenomenon. Rather, it may result from vagomimetic fingolimod effects [Camm et al. 2014; Gold et al. 2014] that start to wear off after approximately 4 h when the S1P receptor sensitivity towards fingolimod begins to decrease and the S1P receptor starts to internalize [Oo et al. 2007; Rossi et al. 2015]. Due to HR-slowing (i.e. increase in RRIs), values of RMSSDs and RRI-HF powers increased simultaneously because calculation of these parameters is based on the RRI variability [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Hilz et al. 2015, 2016]. Thus, changes in RMSSDs and RRI-HF powers closely follow the time course of RRI changes (see Table 2, Figures 1, 2 and 3). While RMSSDs and RRI-HF powers are commonly used to estimate parasympathetic HR modulation, the changes in RMSSDs and RRI-HF powers occurring upon fingolimod initiation do not seem to indicate changes in centrally-mediated cardiovagal output but reflect the increasing (and after 4 h, decreasing) vagomimetic effects of fingolimod [Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Camm et al. 2014; Simula et al. 2016].
Still, fingolimod not only modifies HR via its direct pharmacologic S1P1 receptor effects mimicking parasympathetic activity [Camm et al. 2014; Simula et al. 2016] but also modifies BP, overall cardiac autonomic modulation, and BRS.
While the subtle decrease in BPsys seen 3, 4, 5, and 6 h after fingolimod intake most likely results from the concomitant HR-slowing with subsequently lowered cardiac output [Hainsworth, 2000], the concurrent significant decreases in BPdia may be due to additional arterial vasodilation resulting from fingolimod’s ability to trigger endothelium-dependent nitric oxide release [Camm et al. 2014].
Whereas our results show a mild though significant decrease in BPdia already 3 h after fingolimod initiation, long-term fingolimod treatment (e.g. for 6 or 24 months) is associated with a mild BP increase [Cohen et al. 2010; Kappos et al. 2010]. The change from an initial BP decrease to an increase can be ascribed to changing effects of fingolimod on endothelial S1P1 receptors enhancing nitric oxide release, and on S1P3 receptors in vascular smooth muscle cells promoting vasoconstriction [Camm et al. 2014]. While initial fingolimod effects on endothelial S1P1 receptors outweigh effects on S1P3 receptors, S1P1 receptors are down-regulated with long-term fingolimod treatment resulting in a decreased production of endothelium nitric oxide synthase and less vasodilatation, and in a predominance of S1P2 and S1P3 receptors in vascular smooth muscle cells [Camm et al. 2014]. Thus, fingolimod effects on S1P3 receptors may contribute to the BP increase reported in patients on long-term therapy with fingolimod [Cohen et al. 2010; Kappos et al. 2010; Camm et al. 2014].
Our findings of an initial nonsignificant increase in BPdia and an almost significant increase in BPsys by 6.7 ± 3.3 mmHg (p = 0.053) within the first hour after fingolimod intake, likely reflect a baroreflex-mediated, central autonomic response to the steadily increasing vagomimetic fingolimod effects. Fingolimod-induced HR-slowing lowers cardiac output [Hainsworth, 2000] and may thus reduce baroreflex loading [Eckberg and Sleight, 1992], which in turn increases efferent baroreflex-mediated sympathetic vasomotor activity and subsequently raises BP [Hilz et al. 2016]. Evidently, the pharmacologic effects of fingolimod on HR override any baroreflex-mediated cardiovagal withdrawal and not only cause further HR-slowing but also lower BPsys and BPdia already 2 h after fingolimod intake (Table 1, Figure 1).
Yet, fingolimod still triggers an initial increase in sympathetic cardiac modulation as evidenced by the significant increase in the mainly sympathetically-mediated RRI-LF powers, 1 h after fingolimod intake, and also increases the overall cardiac autonomic modulation, evidenced by the significant increase in RRI-CV after 1 h, in RRI-SDs after 1, 2, 3, and 4 h, and in RRI-TP after 1 and 2 h upon fingolimod intake (Table 1, Figures 2 and 3).
Apart from the direct cardiovascular effects of fingolimod, there might also have been autonomic cardiovascular effects due to emotional excitement related to the initiation of a new treatment option. Particularly the significant increase in sympathetic cardiac modulation seen only 1 h after fingolimod initiation could still reflect possible emotional effects [Pagani et al. 1989; Malliani et al. 1991; Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996].
In contrast, the effect of fingolimod on vasomotor sympathetic activation is minute and yields an only small and nonsignificant increase in sympathetically-mediated LF-BP modulation after 0.5 and 1 h (Table 1, Figure 5 A and B). Moreover, RRI-LF/HF ratios increased only slightly after 30 min, showing a minor shift of HR modulation towards more sympathetic than parasympathetic activity. After 30 min, the index of sympathetic-parasympathetic HR balance continuously decreased and showed significantly more parasympathetic than sympathetic cardiac modulation after 4, 5, and 6 h. Thus, the vagomimetic effect of fingolimod outweighs any centrally-mediated increase in sympathetic activity.
While the overall cardiac autonomic modulation increased 1, 2, 3, and 4 h after fingolimod initiation, the measures of relative changes in cardiac autonomic modulation, the normalized RRI-LF and RRI-HF powers, showed that vagomimetic effects steadily increased and were maximal after 4 h while sympathetic cardiac modulation decreased to its lowest values after 4 hours.
The findings suggest that fingolimod initiation has several beneficial cardiovascular effects including a mild though significant decrease in BP, particularly in BPdia, indicating reduced peripheral resistance due to fingolimod-induced nitric oxide release [Camm et al. 2014], and augmented overall cardiac autonomic modulation and BRS, both resulting from the shift towards less sympathetic and more parasympathetic, or rather vagomimetic, cardiac modulation [Eckberg and Sleight, 1992; Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996; Goldberger et al. 2001; Hilz et al. 2011].
Our data also confirm that our MS patients had the typical age-related changes in autonomic cardiovascular modulation with an increase in sympathetic and a decrease in parasympathetic cardiac activity, as previously described in healthy persons [Fluckiger et al. 1999; Brown et al. 2003; Hilz et al. 2014]. However, the positive correlation of disease duration with sympathetically-mediated RRI-LFnu powers and the negative correlation with parasympathetic RRI-HFnu powers suggest that the duration of MS itself, shifts central autonomic modulation towards higher sympathetic and less parasympathetic influences. This age-related increase in sympathetic modulation might also account for the more prominent decrease in RRI-LF/HF ratios with increasing age (i.e. for a more pronounced vagomimetic effect of fingolimod in those patients who have a higher sympathetic modulation already prior to fingolimod initiation).
Although we only included patients in whom we had tried our best to rule out possible effects on the autonomic nervous system due to other diseases, medications or MS itself, we cannot completely exclude that there were effects of the MS on the central autonomic network and on autonomic modulation already prior to fingolimod treatment and also after fingolimod initiation. Several studies have shown that there may be subtle confounding effects of MS-related central nervous system lesions on cardiovascular modulation [Hengstman and Kusters, 2011; Razazian et al. 2014; Racosta et al. 2015; Hilz, 2016]. Moreover, we did not compare the results of our patients to those of a healthy control group as we considered it not ethically justified to expose healthy volunteers to the intake of fingolimod in an exploratory study. However, vagomimetic effects of fingolimod with HR-slowing have been repeatedly shown in healthy volunteers [Kovar et al. 2004; Schmouder et al. 2006; Kovarik et al. 2008; Schmouder et al. 2012]. These vagomimetic effects account for the increase in total cardiac autonomic modulation and in BRS seen in the 21 MS patients enrolled in this study.
Therefore, we assume that the cardiovascular autonomic effects seen during the first 6 h after fingolimod initiation are not primarily due to MS specific changes in autonomic modulation but might be equally detectable in healthy persons.
Our findings may have some clinical relevance, particularly regarding the decrease in BP seen with fingolimod initiation. This BP decrease should be taken into account in MS patients with low BP or orthostatic hypotension (e.g. due to MS) [Kaplan et al. 2015]. Moreover, any medication associated with a BP decrease might have to be adjusted to avoid a possible inadvertent potentiation of the fingolimod-related BP decrease. In particular, antihypertensive drugs, dopamine receptor agonists, phosphodiesterase type V inhibitors or tricyclic antidepressants [Goldstein and Sharabi, 2009] should be carefully considered in patients who are about to be initiated on fingolimod.
In addition to BP changes, the changes of autonomic modulation might have clinical relevance. While a decrease in cardiovascular autonomic modulation is a risk factor for cardiovascular complications and increased mortality [La Rovere et al. 1998; Thayer et al. 2010; Hilz et al. 2016], the observed augmented cardiovascular autonomic modulation is associated with improved cardiovascular prognosis [Iellamo et al. 2000; La Rovere et al. 2002; Jurca et al. 2004]. In acute emergency patients with stroke, respiratory distress, acute coronary syndrome, severe trauma, or intoxication, Yperzeele and colleagues showed that a decrease in cardiac autonomic modulation during the prehospital phase yields an unfavorable outcome such as the need for intensive care unit treatment with prolonged hospitalization beyond 30 days or death during hospitalization; while higher HR variability is associated with a favorable outcome [Yperzeele et al. 2016]. Moreover, long-term epidemiological studies confirmed an association between overall HR variability or BRS and the cardiovascular prognosis [Dekker et al. 1997; La Rovere et al. 1998]. In a 30-year follow-up study comprising >2300 clinically disease-free participants, Dekker and colleagues confirmed that HR variability and BRS inversely correlate with the incidence of cardiac events and mortality [Dekker et al. 1997]. Similarly, augmented BRS has cardioprotective effects and is associated with improved cardiovascular prognosis [Eckberg and Sleight, 1992; La Rovere et al. 2002; Hilz et al. 2016], while reduced BRS correlates with an increased risk of cardiac complications and mortality [La Rovere et al. 1998; Lanfranchi and Somers, 2002; Ormezzano et al. 2008; Kiviniemi et al. 2014] including sudden death [Ormezzano et al. 2008]. In addition, the vagomimetic fingolimod effects themselves have cardioprotective potential [Thayer and Lane, 2007; De Ferrari et al. 2011]. Epidemiologic studies confirmed that augmented parasympathetic modulation and lowered HR are associated with a reduced risk of sudden cardiac death [Jouven et al. 2005] and all-cause mortality [Fox et al. 2007a; Thayer and Lane, 2007].
In summary, most MS patients might benefit from the cardioprotective potential of fingolimod-associated increases in cardiovagal and total cardiac autonomic modulation and in BRS [Dekker et al. 1997; La Rovere et al. 2002; Yperzeele et al. 2016], while only 0.5–2.4% of MS patients are at risk of clinically relevant bradycardia or arrhythmias [Pelletier and Hafler, 2012; Dimarco et al. 2014; Gold et al. 2014]. However, there is evidence that patients at risk of cardiac side effects might be identified prior to fingolimod treatment [Hilz et al. 2015; Rossi et al. 2015]. Using noninvasive autonomic testing of HR and BP modulation in 55 MS patients prior to fingolimod initiation, Rossi and colleagues found correlations between the lowest HR values upon fingolimod initiation and the extent of parasympathetic responses to autonomic challenge maneuvers, such as a Valsalva maneuver or deep metronomic breathing, assessed prior to fingolimod treatment [Rossi et al. 2015]. Among 21 MS patients of a previously reported study, we also found altered responses to cardiovascular autonomic challenge, prior to fingolimod initiation, in those 7 patients who had prolonged HR-slowing beyond the first 6 h after fingolimod initiation [Hilz et al. 2015]. Compared with healthy persons, these seven MS patients showed higher resting BP and increased BP overshoot during a handgrip exercise, increased parasympathetically-mediated RRI-HF powers, and a 4-times higher baroreflex-mediated cardiovagal HR-slowing after strain release of a Valsalva maneuver [Hilz et al. 2015]. We concluded that MS-related lesions interfering with the central autonomic network might account for inadequate cardiovascular responses to autonomic challenge [Hilz et al. 2015]. Thus, readily available autonomic tests might preselect MS patients who require intensified and prolonged cardiovascular monitoring when starting fingolimod therapy.
However, it is unclear whether the possible beneficial autonomic changes prevail with long-term fingolimod therapy. Limited data exist regarding the cardiovascular autonomic changes after long-term fingolimod therapy. In 27 MS patients, Simula and colleagues recently showed a shift towards predominantly sympathetic HR modulation after 3 months of fingolimod treatment [Simula et al. 2016]. Racca and colleagues reported a decrease in left ventricular systolic function after 1 and 6 months of fingolimod treatment but also saw a partial recovery of left ventricular ejection fraction after 12 months of fingolimod therapy [Racca et al. 2016]. While some preclinical animal studies suggested that fingolimod might be beneficial in minimizing inflammation [Zhang et al. 2016] or preserving cardiac function in pre-existing myocardial infarction or diabetic cardiomyopathy [Abdullah et al. 2016; Santos-Gallego et al. 2016; Zhang et al. 2016], clinical studies by Gold and colleagues show that patients with pre-existing cardiovascular conditions such as recurrent symptomatic bradycardia, resting pulse rate of 45–54 beats per minute, history of a positive tilt test for vasovagal syncope, and history or presence of Mobitz type I second-degree AV block on the screening or baseline electrocardiogram, are at increased risk of developing adverse cardiac events [Gold et al. 2014]. The US Food and Drug Administration even defined cardiac conditions, including myocardial infarction or unstable angina in the last 6 months, decompensated heart failure, history of Mobitz type II second-degree or third-degree AV block or sick sinus syndrome, as contraindications for fingolimod treatment [Kaplan et al. 2015]. Yet, we only included patients in our study who had no pre-existing cardiac conditions. Our data suggest that fingolimod initiation might have beneficial effects on the cardiovascular system in MS patients without these pre-existing cardiac conditions.
However, studies of fingolimod effects after a longer treatment period such as 2 years must show whether the initial, possibly beneficial cardiovascular changes prevail or only last during the onset of fingolimod treatment [Pelletier and Hafler, 2012; Simula et al. 2016; Racca et al. 2016].
While the sympathetic and parasympathetic changes observed in our 21 MS patients 6 h after fingolimod initiation may still be of limited clinical relevance for a practicing neurologist, our results support the need for further studies in larger patient populations and after long-term application of fingolimod.
It also might be worthwhile to evaluate whether a lower starting dose or dosing fingolimod every other day might attenuate the risk of clinically relevant bradycardia or arrhythmias, which did not occur in any of our 21 patients, but may afflict 0.5–2.4% of MS patients upon fingolimod initiation [Pelletier and Hafler, 2012; Dimarco et al. 2014; Gold et al. 2014], and to test whether such reduced dosing would also mitigate any potential cardiac benefits due to the autonomic changes seen in this study.
Clinical perspective of our findings
Although our data reflect results from a small patient group, the theoretical benefits on the cardiovascular system stimulate the question of whether fingolimod might have any therapeutic cardiovascular effects not only in MS patients but perhaps also in patients with autonomic dysfunction such as autonomic neuropathies. So far, there is insufficient knowledge about the direct effects of fingolimod on the sympathetic nervous system [Nishimura et al. 2010], or possibly even on the central autonomic network [Camm et al. 2014]. Yet, large-scale studies in healthy volunteers, and then perhaps in patients with mild though not cardiologically-endangering autonomic neuropathies might further elucidate this issue.
Footnotes
Acknowledgements
We thank Mrs Beate Beck and Mrs Barbara Kraus for technical assistance and data collection. MJH and RW contributed equally to this work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, or publication of this article: this study received partial financial support from Novartis Pharma, Germany.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, or publication of this article:
MJH has received personal compensation for activities from Genzyme, A Sanofi Company (Germany). MJH has received research support from the Rolf and Hubertine Schiffbauer Foundation, Bayer Health Care (Germany) and Novartis Pharma GmbH, Germany.
CRL has received travel grants from Genzyme, A Sanofi Company.
FC has received travel grants from Genzyme, A Sanofi Company.
DHL has received travel grants or speaker honoraria from Bayer Health Care Pharmaceuticals (NJ, USA), Biogen Idec (Cambridge, MA, USA), Merck Serono (Germany), Novartis Pharma GmbH, and TEVA Pharmaceutical Industries (Germany).
RAL has received travel grants or speaker honoraria from Bayer Health Care Pharmaceuticals, Biogen Idec, Merck Serono, Novartis Pharma GmbH, Roche (Switzerland), and TEVA Pharmaceutical Industries Ltd. RAL has received research support from Novartis Pharma GmbH, Biogen Idec, and Merck Serono.
RW, ML, SR, KHM and KW declare that there is no conflict of interest.
