Abstract
Background:
Up to every fourth woman with multiple sclerosis (MS) or neuromyelitis optica spectrum disorder (NMOSD) suffers a clinically relevant relapse during pregnancy. High doses of steroids bear some serious risks, especially within the first trimester of pregnancy. Immunoadsorption (IA) is an effective and more selective treatment option in disabling MS relapse than plasma exchange. Data on the use of IA during pregnancy and breastfeeding are scarce.
Methods:
In this retrospective multicenter study, we analyzed the safety and efficacy of IA treatment in acute relapses during pregnancy or breastfeeding. The primary outcome parameter - change of acute relapse-related disability after IA - was assessed using Expanded Disability Status Scale (EDSS) and visual acuity (VA) measurements for patients with optic neuritis (ON).
Results:
A total of 24 patients were analyzed, 23 with relapsing–remitting MS, and 1 with NMOSD. Twenty patients were treated with IA during pregnancy. Four patients received IA postnatally during the breastfeeding period. Treatment was started at a mean 22.5 [standard deviation (SD) 13.9] days after onset of relapse. Patients were treated with a series of 5.8 (mean, SD 0.7) IA treatments within 7–10 days. Sixteen patients received IA because of steroid-refractory relapse, eight were treated without preceding steroid pulse therapy. EDSS improved clinically relevant from 3.5 [median, interquartile range (IQR) 2] before IA to 2.5 (median, IQR 1.1) after IA, p < 0.001. In patients with ON, VA improved in four out of five patients. Altogether, in 83% of patients, a rapid and marked improvement of relapse-related symptoms was observed after IA with either a decrease of ⩾1 EDSS grade or improvement in VA ⩾20%. No clinically relevant side effect was reported in 138 IA treatments.
Conclusions:
Tryptophan-IA was found to be effective and well tolerated in MS/NMOSD relapses, both as an escalation option after insufficient response to steroid pulse therapy and as first-line relapse treatment during pregnancy and breastfeeding.
Keywords
Introduction
Multiple sclerosis (MS) is the most common acquired neurologic disorder of young adults with a female predominance. The prototypic patient is a young woman in reproductive age. Today, there are no medical concerns for women with MS planning to become pregnant. Results from several prospective studies on the effect of pregnancy on MS show no medical concerns. MS does not increase the risk of an adverse pregnancy outcome and uneventful pregnancy shows no negative long-term impact on MS progression.1–3 However, up to every fourth woman with MS experiences a clinically relevant relapse during pregnancy and nearly every third suffers from relapse during the first three months after birth. 2 Acute exacerbations in MS are generally associated with a high risk of incomplete remission, thus early and effective treatment is warranted. 4 Available disease-modifying therapies for MS (DMT) are either contraindicated for use in pregnancy or breastfeeding or applicable under risk–benefit analysis according to current guidelines.5,6
Neuromyelitis optica spectrum disorder (NMOSD) is a rare severe antibody-mediated inflammatory disorder of the central nervous system. The typical immunoglobulin G anti-aquaporin-4 autoantibodies (AQP-4 abs) play a key role in NMOSD pathogenesis and distinguish the disease from MS. NMOSD predominantly occurs in female patients.7,8 Different to MS, the NMOSD relapse rate is not reduced during pregnancy, but is also elevated 3 months postpartum. 6
Treatment of multiple sclerosis relapse
Standard treatment for MS relapse is the intravenous administration of high doses of steroids for 3–5 days, that is, steroid pulse therapy. High doses of steroids are not considered to be safe during the first trimester of pregnancy due to possible side effects for the embryo like teratogeni-city, higher rates of miscarriage, preterm births and lower birth weight.9–13 In addition, potential risks for the mother, like hypertension, neuropsychiatric symptoms and a diabetes-inducing effect, have to be considered, which are not limited to early pregnancy.14,15 After the first trimester, intravenous steroids may be used relatively safely during pregnancy. 16 In postpartum relapses treated with methylprednisolone, the amount in breast milk can be limited with a 2–4-h delay of breastfeeding after intravenous methylprednisolone infusion. 17
According to national and international guidelines, plasma exchange (PE) or immunoadsorption (IA) is recommended as escalation or second-line therapy in patients not efficiently responding to steroid pulses.10,18,19
After treatment with PE, functionally relevant clinical improvement was reported in 42–76% of patients.20–23 Recent studies evaluating the efficacy and tolerability of tryptophan IA as second-line therapy for MS relapses after one or more steroid pulses show response rates between 70% and almost 90% in more than 200 patients.24–28 In the largest study with 147 patients, good tolerability of tryptophan IA was reported in 98.9% of treatments. 27 But only little is known on the use, efficiency and safety of PE or IA during pregnancy or breastfeeding in patients with MS.
Treatment of neuromyelitis optica spectrum disorder relapse
Recommended treatment options for NMOSD relapse are high-dose intravenous steroids, PE or IA. 29 Escalation of relapse therapy with PE or IA significantly improved outcome. Isolated myelitis responded even better to PE and IA as first treatment compared with high-dose steroids independently from AQP-4 antibody serostatus. 30 A recent study provided evidence for the efficacy of tryptophan IA as a valid treatment option for patients with NMOSD in the acute phase of the disease. 31
During PE, a patient’s plasma is discarded, including valuable proteins like coagulation factors and hormones, and is substituted using human plasma products. During IA, a patient’s plasma is reinfused after removal of antibodies and immune complexes, thus an essential advantage of IA compared with PE is its selectivity with no need for plasma substitution. 32 Protein replacement fluids, that are, albumin or fresh frozen plasma, involve the risk of allergic reactions, as well as, albeit rare, the risk of infection with viruses or with yet unknown pathogens.33,34
There is no general contraindication for therapeutic apheresis in pregnancy. 35 However, data on the use of IA in pregnancy are scarce, and specific guidelines for apheresis in pregnancy are lacking. The aim of our retrospective study was to increase evidence on the clinical use of IA for MS relapses during pregnancy and breastfeeding by analyzing for the first time a larger case series. IA treatment for NMOSD relapse during pregnancy was evaluated in one patient who is, to our knowledge, the first case described in the literature. Preliminary results of this study have been published in part elsewhere. 36
Methods and outcome measures
A retrospective multicenter analysis of patients treated with tryptophan IA during pregnancy or breastfeeding for MS or NMOSD relapse was performed. The study was approved by an international review board (No. 014/1756; International Ethics Commission, Freiburg, Germany), the Ethics Committee, University Bochum (No. 5395-15), and reported to an open-source online registry (No. DRKS00011770). Patients’ written informed consent was obtained. Patient information was anonymized before data entry. The main outcome parameter for efficacy was change of acute relapse-related disability. MS-related disability was assessed by the Expanded Disability Status Scale (EDSS). In patients with optic neuritis, best corrected visual acuity (VA) was used to monitor the clinical course. Marked improvement was defined as clinically significant improvement in function (decrease in EDSS by ⩾1 grade or improvement in VA ⩾20%). Clinical evaluation was performed before the first IA and after the last IA of the treatment series. IA treatments were performed using the single-use tryptophan adsorber TR-350 in combination with the OP-05W plasma separator (Asahi Kasei Medical, Tokyo, Japan) and the tubing system PA-420 together with the Octo Nova Technology (DIAMED, Cologne, Germany). For vascular access, double-lumen central venous catheters were used in 19 patients; peripheral veins were used in 5 patients. The treated plasma volume per IA was 2.031 ml (mean, SD 230 ml). Anticoagulation was performed with unfractionated heparin.
Statistics
The t test for paired samples was used to analyze changes in EDSS from baseline before IA to time after the IA series; p < 0.01 was considered significant. Descriptive statistics were provided as median with interquartile range (IQR) or mean with standard deviation (SD).
Results
In total, data of 24 patients were analyzed. Twenty-three patients presented with a relapsing–remitting course of MS, 1 patient had at time of diagnosis AQP-4-abs-positive NMOSD, according to the new criteria. 7 All patients were treated from 2010 to 2017 with tryptophan IA for acute relapses in 13 different centers. Median age of patients at treatment was 30.5 years. Median duration of disease before pregnancy was 5.5 years. A total of 20 out of 24 patients had stopped DMT before pregnancy, the exact time interval is unknown. Three patients discontinued DMT in the first trimester and one patient continued azathioprine medication during pregnancy. Patient characteristics are summarized in Table 1. IA treatment was started at 22.5 days (mean, SD 13.9) after onset of relapse. Patients received a series of (mean) 5.8 (SD 0.7) single IA treatments within 7–10 days (Table 2).
Patient characteristics.

MS, multiple sclerosis; NMOSD, neuromyelitis optica spectrum disorder; RRMS, relapsing–remitting multiple sclerosis; AQP-4-abs, antiaquaporin-4 autoantibodies; IA, immunoadsorption; SD, standard deviation; DMT, disease-modifying therapy; GW, gestational week.
Treatment characteristics.

IA, immunoadsorption; SD, standard deviation.
Efficacy of immunoadsorption treatments
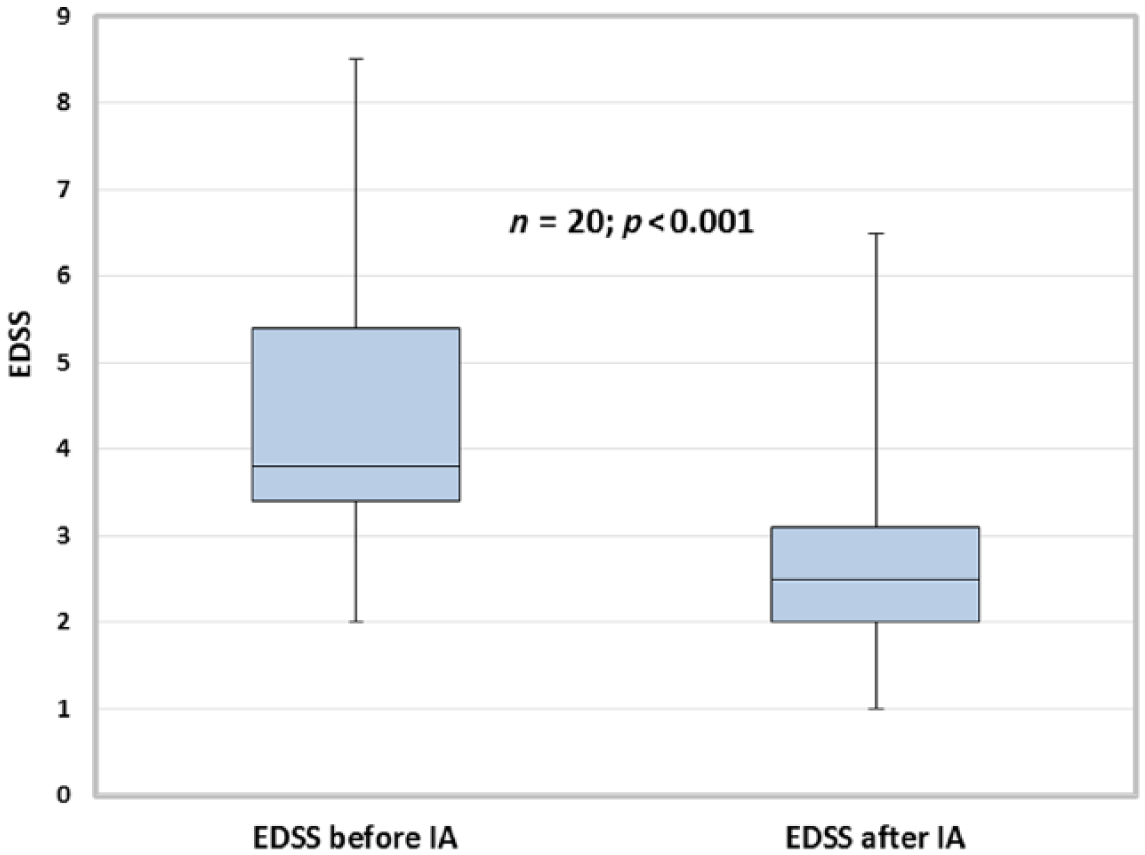
All 24 patients were analyzed for EDSS change at the time immediately after the IA series compared with baseline before the first IA. A total of 20 patients were treated with IA during pregnancy, and 4 patients during breastfeeding. EDSS improved significantly from 3.5 (median, IQR 2.0) to 2.5 (median, IQR 1.1), p < 0.001 (Figure 1). Time between onset of IA and first signs of improvement was (mean) 6.8 days (SD 4.2) in patients with response. The group of pregnant patients received IA in gestation week 16 (median, range 6–33). In this group, EDSS improved from 3.8 (median, IQR 2) to 2.5 (median, IQR 1.1), p < 0.001 (Figure 2).

Relapse during pregnancy or breastfeeding: Expanded Disability Status Scale (median, interquatile range) in multiple sclerosis patients and one neuromyelitis optica spectrum disorder case before and immediately after a series of 5.8 (mean) tryptophan immunoadsorption treatments.
Relapse during pregnancy: Expanded Disability Status Scale (median, interquatile range) in multiple sclerosis patients and one neuromyelitis optica spectrum disorder case with relapse before and immediately after a series of 5.7 (mean) tryptophan immunoadsorption treatments.
Sixteen patients received IA because of a steroid-refractory relapse. The mean cumulative steroid dose before IA was 6.0 g (SD 4.1). These patients improved in EDSS from 3.5 (median, IQR 1.6) to 2.5 (median, IQR 1.1), p < 0.001 (Figure 3a). Eight patients, including the patient with NMOSD, were treated without preceding steroid pulse therapy. Five of them were in the first trimester and two in the second. EDSS improved from 4.0 (median, IQR 3) to 2.0 (median, IQR 1.8), p < 0.01 (Figure 3b). Optic neuritis was the main relapse symptom in six patients, including the patient with NMOSD. In five patients, VA improved after IA; in one patient, the visual field defect persisted (Table 3).
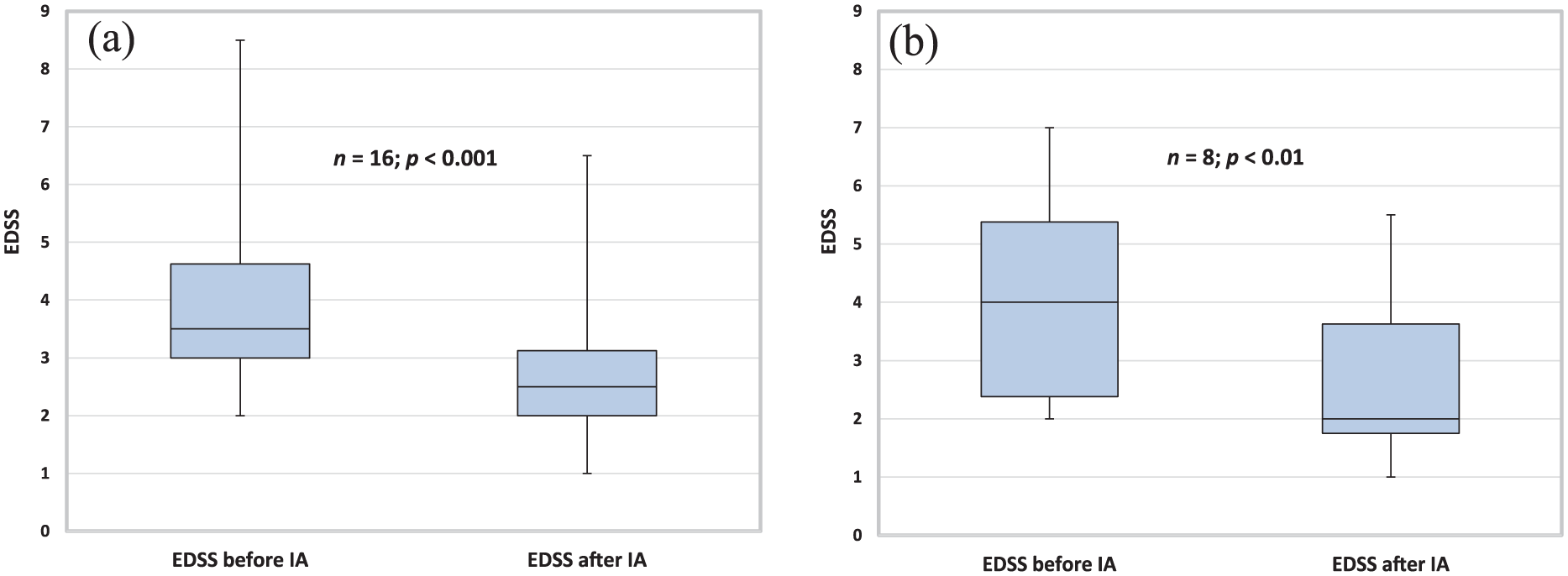
Patients with multiple sclerosis relapse during pregnancy or breastfeeding treated with (a) and without (b) a steroid pulse before immunoadsorption.
Characteristics of patients with optic neuritis.

ID, identification; GW, gestation week; IVIG, intravenous immunoglobulin; NMOSD, neuromyelitis optica spectrum disorder; ON, optic neuritis; PP, postpartum; VA, visual acuity; IA, immunoadsorption.
The clinical outcome of patients with ⩽20 days between relapse onset and IA treatment, and patients with >20 days between relapse onset and IA treatment, was analyzed separately. The mean difference in EDSS before and after the IA series was 1.6 (p = 0.001) in the group with early start of therapy and 1.1 (p < 0.001) in the group with therapy starting more than 20 days after onset of relapse. Taken together, 20 out of 24 patients (83%) showed a marked improvement of relapse symptoms after IA, with either a decrease of ⩾1 point in EDSS, or improvement in VA ⩾20%.
Tolerability and safety
In total 138 IA treatments were performed in 24 patients, with a mean of 5.8 (SD 0.7) per patient. The overall tolerability was good. No clinically relevant side effects were reported. One patient experienced a light malaise that resolved with infusion of 500 ml saline. In one patient, vascular access had to be changed after the first IA, from peripheral to central, because of poor veins. In one patient, the IA series was limited to four treatments because of a catheter-related complication (hematoma) (Table 2).
The further course of pregnancy after onset of IA was uncomplicated in 11 of 15 patients. Four patients experienced another relapse during pregnancy. Time between treatment with IA and onset of the second relapse during pregnancy was 4, 5, 7 and 15 weeks. In one patient, relapse symptoms were mild and improved spontaneously without treatment; in three patients, relapse symptoms improved after steroid pulse therapy. No patient received a second series of IA. Seven women gave birth spontaneously, eight by Cesarean section. All newborns were healthy (n = 15). One patient was lost to follow up before delivery; four pregnancies were ongoing at the time of submission (Table 4).
Further course of pregnancy and delivery.

IA, immunoadsorption.
Discussion
To our knowledge, this is the largest study retrospectively analyzing the use of tryptophan IA in pregnancy and breastfeeding in 24 patients. In general, acute exacerbations in MS and NMOSD are associated with a high risk of incomplete remission.4,30 Hence, effective relapse treatment may be decisive for long-term outcome and disability. In recent studies in nonpregnant patients, tryptophan IA reduced the level of disability and was well tolerated as second-line therapy for MS and NMOSD relapses.24–28,30,31 These studies and the given risks of high steroid doses have been our rationale for also applying IA in pregnant and breastfeeding patients. We observed functional improvement of acute relapse-related symptoms in 83% (20/24) of patients shortly after the IA series. Start of IA therapy was a mean of 22.5 days after onset of relapse. An early start of IA treatment is thought to have a positive influence on the response rate. In our study, patients with start of IA therapy less than 20 days after onset of relapse showed a slightly better response than patients with IA treatment later than 20 days after onset of relapse. However, the sample size was too small for a statistically robust subgroup analysis.
Although reported adverse events of a steroid pulse in acute relapses are mostly mild, albeit frequent, some possible short-term severe adverse events have to be mentioned, for example, hypertension, hepatotoxicity, anaphylaxis and ventricular arrhythmias. 15 During pregnancy, further serious risks are discussed, especially within the first trimester, for example, orofacial cleft and miscarriage, thus alternative treatment options are warranted.9–13 In 39%, IA was used as first-line relapse treatment. Both groups of patients, with or without preceding steroid pulse, improved significantly in EDSS, including the patient with NMOSD. In contrast to MS patients, women with NMOSD have an elevated rate of pregnancy complications, including miscarriage and preeclampsia. 37 Experimental and clinical data revealed that AQP-4 is present in human placenta and maximally expressed during midgestation. There is growing evidence that an AQP-4-abs-mediated placental inflammation can cause fetal death.37,38 The NMOSD patient in our study was positive for AQP-4 abs until onset of azathioprine intake 5 years earlier. She continued azathioprine during pregnancy, experienced no further relapse after IA and gave birth to a healthy newborn. These results suggest that IA might be an effective drug-free and safe first-line treatment option for MS and NMOSD relapse during pregnancy.
Typically, a continuous short-term reduction of MS relapse rate by 70–80% occurs in the course of pregnancy, followed by increased relapse rate after delivery, yet about a quarter of women with MS suffer relapses during pregnancy.2,39 The protective effects of pregnancy on MS disease activity seem to be at least in part mediated by immunomodulatory effects of pregnancy hormones. Respectively, only 15% of the patients in our study presented with a relapse during the third trimester. Numerous hormones undergo pronounced shifts in their plasma levels during pregnancy, including estrogens, glucocorticoids, vitamin D and progesterone. 39 This supports the use of selective IA during pregnancy to preserve protective plasma proteins instead of discarding them with PE.
Recent studies revealed that women who breastfed exclusively had significantly lower postpartum disease activity during the first 3 months as compared with women who did not breastfeed exclusively or not at all.2,40 None of the oral DMTs are recommended for use while breastfeeding, intravenous methylprednisolone infusion is considered as acceptable if applied in delayed time to lactation. 17 Four of our patients experienced an MS relapse during breastfeeding; in three of them, relapse symptoms significantly improved after IA. The nonresponding patient was treated with IA very late after onset of relapse symptoms which might have impaired possible response. Two preceding steroid pulse therapies and three intravenous immunoglobulin infusions were also ineffective in this patient. Pregnancy in this patient was largely uncomplicated, 10 relapses in 7 years before pregnancy were responsive to steroid pulse therapy.
The mechanism of action of IA for MS in not yet fully understood. In addition to the immediate reduction of plasma antibodies and immune complexes after IA, a redistribution of antibodies from the extravascular space is induced, together with immunomodulatory changes.41,42 An important role of humoral autoimmunity, B cells and antibodies is increasingly accepted in the pathogenesis of MS.43,44
In clinical practice in Germany, IA is increasingly used to treat autoimmune neurological diseases replacing unselective PE because of the advantage of better tolerability and safety profile. In patients with myasthenic crisis, IA proved to have significantly fewer side effects than PE; patients treated with IA had a shorter stay in hospital and a better score at discharge.32,45 Protein replacement fluids involve the risk of allergic reactions, as well as a potential risk of viral infections.33,34 The very special situations of pregnancy and breastfeeding deserve particular attention. The elimination of coagulation factors during a series of PE treatments could lead to an increased risk of bleeding in the perinatal period as well as to an increased thrombotic risk by reduction of antithrombin III. 46 In contrast to PE, most coagulation factors remain unaffected by IA, except fibrinogen. 47 Fibrinogen substitution was not necessary in our study. Variations of the oncotic pressure or concentration of electrolytes during PE can cause fluctuations in the blood pressure interfering with the flow of placental blood. 46 In our study, no relevant side effects were reported during IA. Side effects related to vascular access with central lines are not specifically attributable to IA.
As a limitation of our study, it has to be mentioned that the use of IA is limited in some countries due to the lack of medical device approval, while PE is in use worldwide. There is no approval of tryptophan IA in North America, and despite approval in all European and several Asian countries, including Japan, different national reimbursement regulations have an impact on the actual clinical use. In Germany, both PE as well as IA are implemented in the coding system for hospital reimbursement which represents a rather unique situation from an international perspective.
Our study is also limited by the observational and retrospective design without a control group. However, enrolling pregnant women in a prospective controlled trial may rise ethical concerns. Results regarding NMOSD are based on the experience with a single case, and require confirmation with increased patient numbers. Prospective collection of data in a national or international registry may be appropriate to resolve both issues.
Conclusion
Tryptophan IA was found to be effective, safe and well tolerated. For patients with MS relapses during pregnancy and breastfeeding, IA represents a drug-free therapeutic option with rapid response both in first and second-line relapse treatment. The patient with NMOSD relapse also improved after IA, yet further studies with more patients are warranted.
Footnotes
Conflict of interest statement
F Hoffmann has received speaker honoraria and grant support from Bayer Vital, Biogen, DIAMED Medizintechnik, Merz, Genzyme, Grifols, Ipsen, Novartis and Teva. A Kraft has received grant support from Bayer Vital, Boehringer, Ipsen, Novartis and Pfizer. R Klingel and C Fassbender received research grants from Asahi Kasei Medical and DIAMED Medizintechnik. F Heigl received lecture fees from B Braun, Melsungen, DIAMED Medizintechnik and Fresenius Medical Care. J Koehler has reported a professional or personal relationship to Almirall, Bayer, Biogen Idec, Fresenius, Genzyme, Merck Serono, Novartis Pharma, Roche Pharma, Sanofi-Aventis and Teva. L Harms received grant support from Bayer Vital, Biogen, DIAMED Medizintechnik, Genzyme, Merz, Novartis, Roche and Teva. T Kümpfel has received travel expenses and speaker honoraria from Bayer Healthcare, Teva Pharma, Merck, Novartis Pharma, Sanofi-Aventis/Genzyme, CLB Behring, Roche Pharma and Biogen, as well as grant support from Bayer-Schering AG, Novartis and Chugai Pharma. W. Köhler has received speaker honoraria and grant support from Bayer Vital, Biogen, DIAMED Medizintechnik, Merck Serono, Genzyme, Grifols, Ipsen, Novartis, Roche and Teva. A Bayas received personal compensation from Merck, Biogen, Bayer Vital, Novartis, Teva, Roche and Sanofi/Genzyme, and grants for congress trips and participation from Biogen, Teva, Novartis, Sanofi/Genzyme and Merck. G Ellrichmann has received grant support from Biogen, Novartis and Teva. T Slowinski received speaker fees and research grants from DIAMED Medizintechnik. K-H Henn received speaker honoraria from Genzyme, Merck Serono, Beratung Genzyme, Merck Serono, Novartis, Teva, and grant support from Teva, Biogen, Bayer, Merck Serono and Boehringer Ingelheim. S Schimrigk has received grant support and speaker honoraria from Bayer Vital, Bionorica research GmbH, Biogen, DIAMED Medizintechnik, Genzyme, Novartis, Pfizer and Teva.
The following authors declare that there is no conflict of interest: J Weinmann-Menke, I Ayzenberg, E Mauch, H Weihprecht, S Ehrlich, C Beuker, H Fritz, M Brand, T Stiegler and J Galle.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
