Abstract
Background:
Rituximab (RTX) is a well-known effective treatment for neuromyelitis optica spectrum disorder (NMOSD).
Aims:
To investigate the effectiveness of RTX treatment in patients with NMOSD and compared medical expenses between RTX and other oral agents.
Methods:
Using data from the National Health Insurance System database (2010−2021), we compared the time to the first relapse and medical expenses after each medication between groups with RTX and oral agents. Also, we analyzed the association between the level of disability and the type of drugs.
Results:
A total of 899 patients were included, and they were divided into two groups according to the type of treatment. The group treated with RTX had a lower risk of relapse than those treated with other oral agents (hazard ratio, 0.479, p < 0.001). Regarding medical expenses, the increase in total medical costs was associated with the use of RTX and the number of relapses. The increase in medical costs was higher in cases with increased disability owing to NMOSD after adjustment for the number of relapses.
Conclusion:
In comparison to other oral agents, RTX showed a favorable treatment effect. It is also relatively cost-effective, considering the change in disability in a large-scale real-world nationwide study.
Keywords
Introduction
Neuromyelitis optica spectrum disorder (NMOSD) is a neuroinflammatory disease that affects central nervous system (CNS), typically the spinal cord or optic nerve. 1 The neurological disabilities from NMOSD can accumulate owing to recurring attacks throughout the course of NMOSD.2–4 To improve neurological outcomes in patients with NMOSD (pNMOSD), recurrent acute attacks should be prevented. Moreover, early aggressive treatment is an effective strategy to reduce the level of disability.5,6
Rituximab (RTX), which targets the B cell surface antigen CD20, is an aggressive and effective treatment for NMOSD. Recently, there have been reports on the effectiveness of treatments targeting B cells in controlling the course of NMOSD.7–11 Regarding the reduction in the relapse rate of NMOSD, RTX was considered better than or not different from other oral immunosuppressants (OI) such as azathioprine (AZA) or mycophenolate mofetil (MMF).7–12 However, RTX is usually applied for secondary therapy after taking oral agents owing to the restricted cost, limited access, or lack of experience of physicians.13,14 Furthermore, considering the medical expenses, RTX appears less cost-effective than other oral agents owing to its high price. However, the cost-effectiveness of RTX compared to other oral agents in pNMOSD associated with disease progression or relapse has not been investigated.
The National Health Insurance System (NHIS) is a uniform organization for claiming all types of medical expenses that are supported by the Korean government. Claims data may be used to collect clinical data to analyze the treatment effectiveness of specific drugs in the real world. Additionally, the medical expenses of pNMOSD can be compared based on claims data approved by the NHIS.
We aimed to study the treatment effectiveness of RTX for relapse in pNMOSD and compare the medical expenses of RTX and oral agents using data from the NHIS in Korea. We focused on investigating medical expenses according to the level of disability, considering that NMOSD is a neurological disease that causes neurological deficits following recurring relapses.
Methods
Data from the NHIS
In Korea, the NHIS, which maintains the national medical records of inpatient and outpatient visits, procedures, and prescriptions, is mandatory to claim payments to the government single-payer of this system. Inevitably, NHIS documents the healthcare data for each patient, from diagnosis to details of medical behavior. The diagnosis is usually represented by a diagnostic code in the International Classification of Diseases, 10th revision (ICD-10) entered by the physician. Additionally, the NHIS provides other medical information, such as treatment prescriptions or types of procedures supported by public health system in Korea, from each hospital. Therefore, the NHIS claims database takes advantage of providing nationwide healthcare information in a real-world setting with longitudinal follow-up.
Study population and design
We obtained medical information using the diagnostic code of NMOSD from the NHIS database from 2010 to 2021 and constructed a retrospective cohort aged ⩾18 years according to medical information obtained from patients diagnosed with NMOSD in the departments of neurology, ophthalmology, or rehabilitation between January 1, 2015, and December 31, 2020, with a washout period from 2010 to 2015. pNMOSD were defined for the diagnostic code G36.0, according to ICD-10, on admission or from outpatient medical records (n = 1455). To improve the accuracy of the diagnosis, we combined clinical data on prescriptions of medications for NMOSD, which included AZA, MMF, or RTX, except for oral steroids or any other maintenance therapy, after the registration of the NMOSD diagnosis. We excluded participants who had been diagnosed with NMOSD in the past 5 years from 2015 (n = 201), and those with all types of malignancy (n = 98). Considering the possibility of simultaneous use of both medications due to other underlying comorbidities, we also excluded patients who were prescribed AZA, MMF, or RTX concurrently after the diagnosis of NMOSD (n = 64) or who were prescribed any other immunosuppressant except AZA, MMF, or RTX (n = 492). Finally, 899 pNMOSD were included in this study. We assigned the patients to two groups according to the type of medication used to prevent the relapse of NMOSD: RTX (n = 155) and OI (n = 744; Figure 1).

Flow chart of study.
Measurements
First, the primary outcome was suggestive relapse events (SREs) after starting maintenance treatment. SREs were administered as intravenous methylprednisolone for more than 3 days in series during the follow-up period with the prescription of treatment drugs. Secondary outcomes were set as the number of expired cases by all-cause mortality and the number of cases with plasmapheresis and re-treatment with steroid pulse within 14 days, which are usually performed for cases refractory to the first acute steroid pulse therapy in NMOSD. The severe disability group was identified using NHIS disability does, which classify patients with considerable restriction in walking or daily activities, or with visual ⩽0.06 with maximum corrective vision. Severe disability typically referred to the complete or partial paralysis of the upper or lower extremities, leading to marked functional impairment, such as inability to walk independently or perform activities of daily life. Additionally, the number of cases with adverse infection events according to the treatment groups was analyzed using diagnostic codes for pneumonia or urinary tract infection.
Second, we used information from the NHIS treatment database and the total approved medical care expenses, including claims for outpatient clinic visits and admissions, to calculate the medical expenses of each group according to the type of treatment for NMOSD. We compared the total medical expenses between the two groups and analyzed the factors associated with medical costs in all participants, especially the level of disability at the time of diagnosis of NMOSD, type of drug, type of residence, and economic status, which is representative of the health insurance fees paid by the Korean government.
Age, sex, residence type, and income level were obtained from an insurance eligibility database. The underlying diseases were defined as the presence of two or more ICD-10 codes on claims within 3 years prior to NMOSD diagnosis.
Statistical analysis
The baseline characteristics of the participants are represented by the average and standard deviation (SD) for continuous variables and the number or percentage for categorical variables. We used the t-test for continuous variables to compare the two groups, and χ2 and Fisher’s exact tests were used for categorical variables.
Participants contributed person-time of follow-up from the baseline date until the development of relapse, death, or the end of the study period (December 31, 2021), whichever first occurs. The study exposure was drug use, which was considered a time-varying variable. Participants who did not take drugs contributed to unexposed person-time, and participants who took drugs contributed to unexposed person-time while not taking them. The time to first relapse between the two groups was analyzed using Cox hazard regression models with time-varying and calculated hazard ratios (HRs) with 95% confidence intervals (CIs) for developing relapse after adjusting for age and sex for the year of the baseline as time-fixed variables.
Univariate and multivariate regression analyses were used to identify predictive factors for medical expenses. Medical expenses are all costs for claims with an NMOSD code from baseline to the date of death or the end of the study. Furthermore, we performed a multiple regression analysis, including an interaction term between drug type and change in disability, and compared medical expenses according to clinical factors such as age, drug type, change in disability level, and duration of diagnosis.
Statistical significance was set at p < 0.05 for all analyses. All statistical analyses were performed using the SAS Enterprise Guide 7.1 (SAS Institute Inc., Cary, NC, USA).
Data availability statement
Data can be shared on request by directly contacting the corresponding authors.
Ethics statement
The study was approved by the Korea University Institutional Review Board (IRB No. 2022GR0510). The need for informed consent was waived because anonymous and de-identified information was used in the analysis.
Results
Baseline characteristics
The mean age (SD) at the time of diagnosis of NMOSD in the RTX and OI groups was 47.7 (14.6) and 46.8 (11.9) years, respectively (p = 0.434). In the OI group, for which AZA or MMF was prescribed, approximately 78% of the patients were female, and in the RTX group, 90% of the patients were female (p = 0.001). The number of patients with severe disability was higher in the RTX group than in the OI group (p < 0.001). More patients in the RTX group had physical disabilities, brain lesions, and visual disabilities (p < 0.001). The number of patients living in urban areas was higher in the RTX group (p < 0.001). There was a significant difference in the household income of patients, especially those who belonged to Medicaid, or the highest group was higher in the group with RTX (p = 0.043; Table 1).
Baseline characteristics.

AZA, azathioprine; MMF, mycophenolate mofetil; RTX, rituximab.
Comparison of relapses between the OI and RTX groups
Overall, we found 269 cases of relapse after treatment initiation in both groups. The majority of the OI and RTX groups (67.2% and 83.9%, respectively) did not show relapse. Nevertheless, the OI group experienced a substantially higher number of relapses compared to the RTX group (p = 0.001; Table 2). The RTX group experienced a longer time to first relapse, whereas the OI group exhibited a notably higher incidence rate. During the follow-up period of 2616.4 person-years, 244 and 25 first relapses were observed, and the incidence of first relapse per 100 person-years was 11.7 and 4.6 in the OI and RTX groups, respectively. The HR for relapse in the RTX group was 0.474 (CI, 0.295−0.675) after adjustment for age and sex (Table 3).
Primary and secondary outcomes between groups with oral immunosuppressants and rituximab.

AZA, azathioprine; MMF, mycophenolate mofetil; RTX, rituximab.
Hazard ratio for relapse according to treatments.
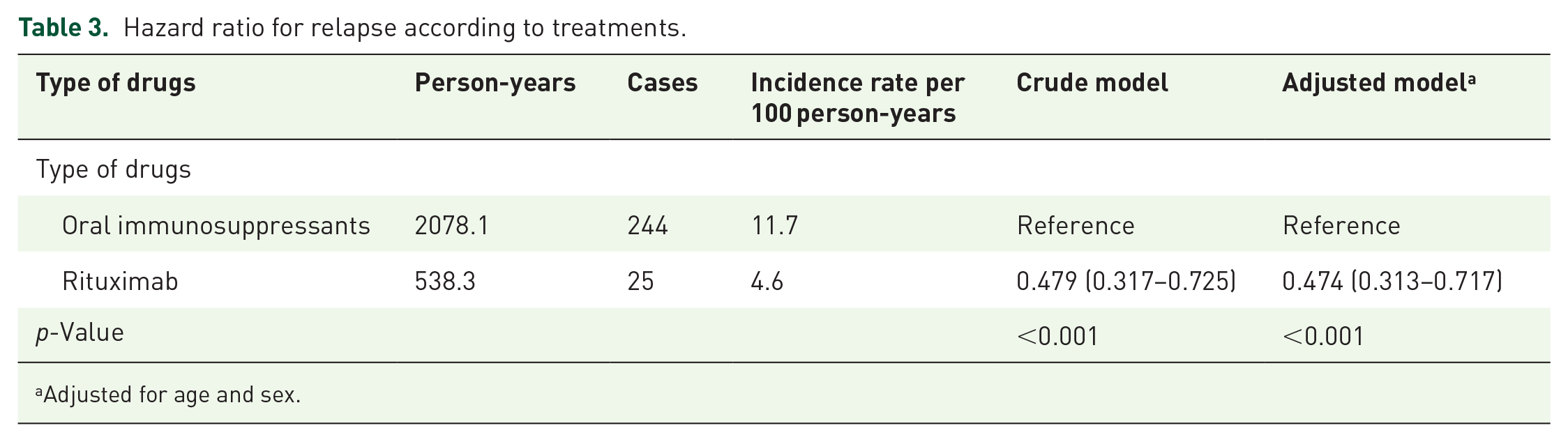
Adjusted for age and sex.
Comparison of refractory cases between the OI and RTX groups
We selected cases for re-treatment with steroid pulse therapy within 14 days of the first steroid pulse therapy. Seven patients were selected, and the average duration of re-treatment was 7.8 (±3.2) days after the first steroid pulse therapy. There was no difference in the number of cases of re-treatment with steroid pulse therapy between the two groups. We also selected and compared patients who underwent plasmapheresis. The number of patients who underwent plasmapheresis after steroid pulse therapy did not differ significantly between the two groups (Table 2).
Comparison of other secondary outcomes between the OI and RTX groups
There were no significant differences between the groups in terms of number; both groups had no differences. The number of visits to the outpatient clinic was higher in the group with RTX (38.7 ± 39.2) than in the group with OI (31.1 ± 43.3; p = 0.046). The duration of admission did not differ between the two groups. The number of cases of urinary tract infections, pneumonia, and other infectious diseases did not differ between the groups. However, the number of deaths was higher in the OI group (p = 0.011; Table 2).
Comparison of total medical expenses
The total medical expenses per person in pNMOSD were 12,416.0 (SD, 21,085.9) USD in the OI group and 20,124.0 (SD, 26,439.5) USD in the RTX group, which was significantly higher in the RTX group (p = 0.001).
Factors associated with medical costs
Univariate regression analysis revealed that total medical expenses were associated with increased age (p = 0.014), use of RTX (p < 0.001), number of SREs (p < 0.001), and level of disability (p = 0.005; Table 4). Medical expenses increased the most as the number of SREs increased (β = 7795.3). Subsequently, the use of RTX and severe disability were associated with increased medical expenses (β = 7708.7 and 7610.6, respectively). Age was the least associated with an increase in medical expenses (β = 128.6).
Factors associated with medical expenses (unit, USD).

AZA, azathioprine; CI, confidence interval; MMF, mycophenolate mofetil; RTX, rituximab.
In the multivariable regression model, an increase in age (p = 0.001), use of RTX (p < 0.001), and number of SREs (p < 0.001) were still significantly related to medical costs per patient. Among them, medical expenses for pNMOSD increased the most with the use of RTX (β = 9175.5). Subsequently, the number of relapses was associated with increased medical expenses (β = 8248.1).
Medical expenses considering the interaction between treatment and disability
To confirm the different effects of the type of drug and the level of disability, we performed an interaction analysis between treatment and disability. Medical expenses increased with the use of RTX (β = 7006.9, p < 0.001), aggravation of disability level (β = 18116.0, p ⩽ 0.001), and concomitant condition of RTX and aggravation (β = 43,712.9, p = 0.001) after adjustment for age, sex, and number of SREs (Table 5). In a subgroup analysis, when the level of disability aggravated to severe disability, medical expenses increased approximately three times in both treatment groups (OI, p < 0.001; RTX, p = 0.003; Figure 2). However, in patients without an aggravation of disability level, there were no significant differences between the OI and RTX groups (Table 6).
The interaction analysis for medical expenses according to the effects of treatments and disability aggravation.

SE, standard error.

Comparison of medical expenses between the two groups according to treatment and change in disability.
Comparison of medical expenses between the two groups according to treatment and change in disability (unit, USD).

AZA, azathioprine; MMF, mycophenolate mofetil; RTX, rituximab.
Also, we performed a subgroup analysis after excluding cases with severe disability. In the subgroup analysis, medical expenses were significantly higher in patients who were older, received RTX, or had a higher number of SRE. This trend was consistent with the findings from analysis of whole group (Supplemental Table 1).
Discussion
We conducted a longitudinal nationwide cohort study to investigate the treatment effects and medical expenses by treatment groups. Using NHIS claim data, we compared the time to the first relapse after starting the medication between oral agents, AZA or MMF, and intravenous treatment targeting RTX B cells. Additionally, we compared the medical expenses between the two groups. Although the time to SREs in the RTX group was significantly longer than in the OI group, the total medical expense was higher in the RTX group than in the OI group. However, in the interaction analysis considering the aggravation of disabilities by drug use after adjustment for other risk factors, medical expenses in the RTX group were not higher than those in the OI group.
The results of this study on the treatment effect of RTX were comparable to those of previous studies, confirming the efficacy of RTX for NMOSD.7–9,11,15 Traditionally, AZA and MMF have been used as effective therapies for NMOSD.16–18 Recently, RTX has been reported to significantly extend the relapse-free time in NMOSD. Furthermore, several studies have shown similar trends in that RTX has superior treatment effects over other oral drugs, compared to the relapse rate among the groups treated with AZA, MMR, or RTX.19,20 The HR for relapses in the RTX group was lower than in the OI group (HR = 0.474) after adjustment for age and sex in this study. Also, the mortality rate was lower in the RTX group than in the OI group.
When comparing medical expenses between the RTX and OI groups, the total medical expenses were higher in the RTX group. As the cost of RTX is much higher, patients treated with RTX pay more for the drug itself. In terms of monthly cost, the cost of using RTX is estimated to be approximately 10 times or more than the cost of using AZA or MMF. Moreover, the increase in medical costs was more associated with the use of RTX (β = 9175.5 USD, p < 0.001) in multiple regression analysis. It also suggests that, for these reasons, extending the interval dosing of RTX may be an efficient way to increase cost-effectiveness.
Otherwise, because NMOSD is a neurological disorder with high morbidity, the progression of disability and relapsing attacks in NMOSD should be considered when calculating medical expenses. The number of relapses was also associated with increased medical expenses (β = 8248.1 USD, p < 0.001). In terms of disability, although the association with the level of overall disability was not clear (p = 0.055), severe disability showed the possibility of an association with increased medical expenses (p = 0.016). Therefore, we analyzed the correlation with medical expenses considering the change in disability and type of drug after adjusting for age and number of relapses. In the interaction analysis, the group using OI had the lowest medical costs and no deterioration of disability. However, even if RTX was used, the medical cost when there was no deterioration in disability (7006.9 USD) was lower than when OI was used and disability worsened (18,116.0 USD).
As recurrent relapses can result in greater disability in NMOSD, this increase in medical expenses based on the number of relapses was also believed to reflect not only the increasing cost of acute therapy for relapses but also the cumulative cost of the progression of disability owing to recurring attacks in NMOSD. Furthermore, since medical expenses were calculated by summing individual expenditures and government-supported costs in this study, the prevention of severe disability will be related to individual medical expenses for pNMOSD, as well as providing greater socioeconomic benefits for the entire fiscal system for health care. Recent economic evaluations have tended to analyze the incremental cost-effectiveness ratio (ICER), expressed as the cost per quality-adjusted life-year (QALY).21–23 In the future, ICER analysis should be considered to assume the cost per QALY gained if it is possible to obtain information on the quality of life in pNMOSD.
This study has the strength of investigating the treatment effects of RTX in a real-world, large-scale nationwide cohort. To the best of our knowledge, this is the first comparative study of treatment effects with a large number of participants (n = 899) with NMOSD using national healthcare data. However, this study has some limitations. First, given that we used diagnostic codes based on NHIS claims data, there was the possibility of including misdiagnosed cases, such as other CNS demyelinating diseases or vice versa. This is a limitation of all studies that use claim data. Second, we used indirect evidence of relapse using data based on the prescription of intravenous steroid pulse therapy. High-dose steroids may be used for other diseases, such as rheumatoid diseases. However, most rheumatoid diseases are not treated with intravenous steroids in series for several days, even in the acute state. 24 Third, we were unable to provide details for each patient, including the cause of death. Therefore, the interpretation of the results of this study is limited.
According to recent guidelines, OI, such as AZA or MMF, and RTX are recommended for the prevention of relapse in pNMOSD. We confirmed the good treatment effect of RTX in NMOSD compared to AZA or MMF. Furthermore, the medical expense of using RTX is suggested to be relatively cost-effective, considering the change in disability. Based on these results, we may consider RTX as the first treatment of choice for NMOSD. Currently, many therapeutic options such as tocilizumab, eculizumab, and satralizumab have been developed for pNMOSD.25–27 In the future, a study to evaluate the cost-effectiveness of these new treatments for NMOSD will be considered.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251314020 – Supplemental material for Comparison of treatment efficacy and cost-effectiveness of rituximab and oral agents among patients with neuromyelitis optica spectrum disorders: a population-based cohort study
Supplemental material, sj-docx-1-tan-10.1177_17562864251314020 for Comparison of treatment efficacy and cost-effectiveness of rituximab and oral agents among patients with neuromyelitis optica spectrum disorders: a population-based cohort study by Hye Lim Lee, Minwoong Kang, Jin Myoung Seok, Byung-Jo Kim and Byoung Joon Kim in Therapeutic Advances in Neurological Disorders
Footnotes
Appendix
Acknowledgements
This was supported by the Korea Disease Control and Prevention Agency (No. 2020-ER6901-02/2023-ER0505-00) and clinical data management were performed using the internet-based clinical research and trial management system (iCReaT No. C140020).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
