Abstract
Neuromyelitis optica spectrum disorder (NMOSD) is an autoimmune astrocytopathy characterized by autoantibodies against aquaporin-4 (AQP) that predominantly affecting women of childbearing potential. Unlike multiple sclerosis, NMOSD poses a higher risk of severe and irreversible postpartum relapse, making pregnancy a significant concern in affected women. Historically, in Japan, immunosuppressive treatments were contraindicated in women who could become pregnant, leading many to discontinue therapy before conception. This often resulted in relapse and sometimes required pregnancy termination or abandonment. At present, immunosuppressants such as azathioprine, tacrolimus, and cyclosporine are considered beneficial during pregnancy. Five monoclonal antibody therapies have now been approved and are covered by national insurance for the treatment of NMOSD. These developments have expanded treatment options, enabling safer pregnancies and allowing more women with NMOSD to consider childbirth. To ensure optimal outcomes, individualized, evidence-based treatment plans are essential. Shared decision-making between patients and healthcare providers is critical, particularly when evaluating the safety of monoclonal antibody therapies during pregnancy and their potential impact on fetal development. This review outlines recent insights into the impact of NMOSD on patients of childbearing age, including family planning, postpartum management, and breastfeeding. It emphasizes the importance of balancing disease control with reproductive goals through informed and collaborative care strategies that also encompass AQP4-antibody–negative NMOSD and myelin oligodendrocyte glycoprotein antibody-associated disease.
Introduction
Over the past decade, significant advances have been made in the management of pregnancy and childbirth in patients with multiple sclerosis (MS) and neuromyelitis optica spectrum disorder (NMOSD). Historically, MS was considered to be exacerbated by pregnancy due to the increased risk of disease relapse in the postpartum period. Consequently, pregnancy was often discouraged in women with MS. However, subsequent findings demonstrated a marked reduction in relapse rates during pregnancy, and the advent of disease-modifying drugs has enabled effective relapse prevention. These developments have contributed to safer pregnancy and delivery outcomes in patients with MS. Furthermore, pregnancy itself has been suggested to exert favorable long-term effects on disease prognosis, leading to an increasing trend in pregnancy rates among women with MS.
In contrast, NMOSD is associated with a substantially higher risk of postpartum relapse than MS (two- to three-fold higher than the pre-pregnancy period), with a single relapse often resulting in severe, irreversible neurological disability.1–4 NMOSD pregnancies are considered high-risk, with an increased likelihood of maternal and obstetric complications. Consequently, women with NMOSD have historically been hesitant to pursue pregnancy. 1
In Japan, immunosuppressive agents such as azathioprine, tacrolimus, and cyclosporine were previously contraindicated in women of childbearing age, compelling them to discontinue therapy before conception. This interruption frequently led to disease relapse, pregnancy termination, or spontaneous abortion. However, pregnancy contraindications for these immunosuppressive agents were removed. In addition, as of 2025, five monoclonal antibody (mAb) therapies have been approved for NMOSD.
These regulatory and therapeutic advancements have significantly improved the clinical management of pregnancy in patients with NMOSD, allowing more patients to achieve their reproductive goals. Further improvements in life events, including pregnancy, among patients with NMOSD will require continuous evaluation of the safety profiles of mAbs during pregnancy. Shared decision-making between clinicians and patients is essential to ensure optimal individualized care.
Outline
Immunological mechanisms of pregnancy in NMOSD and preconception treatment
Clinical importance of pregnancy management 2.1 Preconception care 2.2 Timing of drug exposure and fetal impact
3. Treatment considerations during pregnancy and lactation 3.1 Management of immunosuppressants during pregnancy and lactation 3.2 Management of monoclonal antibodies (mAbs) during pregnancy and lactation
4. Management during delivery
5. Treatment of relapse during pregnancy
6. MRI during pregnancy and lactation
7. Assisted reproductive technology
8. myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) in pregnancy
9. Impact of NMOSD autoantibody positivity (aquaporin-4 (AQP4)-Ab, MOG-Ab, or double-negative) on pregnancy
10. Conclusion
Immunological mechanisms of pregnancy in NMOSD and preconception treatment strategy
The immunological mechanisms underlying pregnancy in NMOSD differ from those in MS. Pregnancy induces a shift toward Th2-dominant immunity, which leads to activation of humoral immune responses. Consequently, the production of aquaporin-4 (AQP4) antibodies (Abs) increases during pregnancy, leading to heightened disease activity. 5 As a result, the early postpartum period is associated with a high risk of relapse.2–4,6
During pregnancy, persistently elevated estrogen levels, enhanced immunoglobulin production, decreased apoptosis of autoreactive B cells, and increased B-cell activation have been observed. These factors are believed to contribute to the exacerbation of disease activity in patients with NMOSD. Furthermore, syncytiotrophoblasts, which form the interface between maternal and fetal circulation in the placenta, express high levels of AQP4 antigens. Maternal circulating AQP4-Abs can cross the placenta via the umbilical circulation and bind to placental AQP4 antigens, leading to complement activation and severe inflammation. In such cases, placental necrosis may occur, potentially resulting in miscarriage. 7
The hypothalamus plays a central role in regulating hormones during pregnancy. Hypothalamic lesions have been reported in patients with NMOSD. As the hypothalamus regulates sex hormones and autonomic functions, lesions in this area can cause various clinical manifestations, including endocrine abnormalities, thermoregulatory dysfunction, insomnia, and appetite disturbances. 8 Pittock et al. demonstrated high expression of AQP4 antigens in the hypothalamus, 9 identifying it as a target region in the central nervous system affected by AQP4-Abs in NMOSD. These findings indicate that achieving disease stabilization through appropriate preconception treatment is essential for managing pregnancies in patients with NMOSD. Maintaining low disease activity before conception is fundamental to ensuring favorable outcomes for both mother and fetus. 3
In women with NMOSD, reproductive health may be significantly compromised because of both immunological and hormonal disruptions. First, fecundity is potentially reduced as a result of AQP4-Ab targeting AQP4-rich uterine tissue, leading to astrocyte damage and cellular apoptosis. Additionally, dysregulation of the hypothalamic–pituitary–ovarian axis impairs hormonal homeostasis, whereas a decrease in anti-Müllerian hormone levels suggests a diminished ovarian reserve.
Second, the postpartum period, particularly the first 3 months, is associated with the highest risk of disease relapse, surpassing prepregnancy relapse rates. These relapses frequently involve the optic nerve and spinal cord, although other regions of the central nervous system may also be affected. 10
Third, pregnancy in women with NMOSD is linked to an increased incidence of fetal complications, including miscarriage, stillbirth, and preeclampsia. The underlying mechanisms are hypothesized to involve pathogenic AQP4-Ab targeting the placenta, elevated estrogen levels promoting a proinflammatory milieu, and a skewed Th17/Treg cell ratio, all of which may contribute to placental dysfunction and adverse pregnancy outcomes. 10
Pregnancy after the onset of NMOSD has been associated with an increased risk of miscarriage and hypertensive disorders of pregnancy. These potential risks should be explained to patients who desire to conceive. 11 However, a recent cohort study reported that these risks were not significantly higher than those in the general population. 12 Since AQP4-Abs can cross the placenta, neonatal serum AQP4-Ab levels may be transiently positive at birth; however, these antibodies typically disappear within a few months, and no adverse effects on infant growth or development have been observed. 5
Clinical importance of pregnancy management
Preconception care
NMOSD commonly occurs between the ages of 35 and 45. 13 This coincides with the reproductive years of many women. As such, treatment strategies for women of childbearing potential must account for both disease activity and the possibility of unintended pregnancies and their associated maternal and fetal risks.
Educating women with NMOSD, as well as their partners, families, and caregivers, that pregnancy is possible despite the diagnosis is crucial. NMOSD is not a hereditary disease; however, several research reports describe familial cases. Familial occurrence of NMOSD has been observed in approximately 3% of patients with a well-established diagnosis. 14 These cases typically involve one or two generations, with most reports describing two affected individuals. The most common kinship pattern was siblingship, reported in 21 families (52.5%). Initial syndromes were most often optic neuritis and transverse myelitis. Serum AQP4-Ab was positive in 79.7% of cases. Reported human leukocyte antigen (HLA) alleles shared between familial cases included HLA-A01 and HLA-DRB103. 15 Familial clustering of NMOSD appears more common than would be expected in the general population.
The current evidence indicates that the condition does not adversely affect fetal development, including the risk of congenital anomalies, growth restriction, or neurodevelopmental delay. However, NMOSD is associated with a high risk of postpartum relapse, as well as a higher risk of miscarriage and preeclampsia.2–4,6,16 Therefore, early initiation of appropriate therapy before conception is critical for reducing relapse risk and maintaining disease remission throughout pregnancy.4,17,18
For women of childbearing age with NMOSD, counseling should follow the 2023 consensus of the French MS Society. 19 Patients should aim to remain relapse-free for at least 1 year before conception and undergo regular follow-up with both obstetricians and neurologists. Since NMOSD carries a high risk of relapse during pregnancy and the postpartum period, treatment discontinuation poses a risk of severe relapses and consequent disability. Therefore, the decision to continue or suspend therapy should be made by carefully weighing risks and benefits. Importantly, immunosuppressive and monoclonal antibody therapies administered before and during pregnancy may influence the risk of early postpartum relapses. 16
In clinical practice, a preconception checklist should be used to promote comprehensive care, including guidance on balanced nutrition, smoking cessation, and avoidance of alcohol. 20 It is imperative not to withhold necessary treatment out of concern for fetal exposure, as inadequate disease control may pose greater risks to both mother and child. When pharmacological treatment is considered in women who are planning a pregnancy or are already pregnant, clinicians must explain the necessity and safety profile of the treatment in detail and engage in shared decision-making to determine the most appropriate therapeutic approach.21,22 Optimal maternal and neonatal outcomes can be anticipated when disease remission is achieved and maintained before conception.
Timing of drug exposure and fetal impact
In cases where a patient experiences a miscarriage or delivers a child with congenital anomalies during or following pharmacological treatment—even in the absence of a causal association—the mother may experience guilt, and the treating physician may struggle with feelings of responsibility and regret. Therefore, it is essential to communicate to patients who are planning a pregnancy or who become pregnant while on medication that the risk of spontaneous miscarriage is approximately 15%, and the risk of congenital anomalies is approximately 3%. 21
The teratogenic risk of medication use during pregnancy varies depending on the timing of exposure. The first 4 weeks of gestation (from week 1 to the end of week 3) constitute the “all-or-none” period, during which teratogenic agents are thought to either result in embryonic loss or have no effect at all. Weeks 4–7 are critical for organogenesis and represent the peak window of teratogenic sensitivity. From weeks 8–11, the formation of the palate and genitalia continues, and medication use during this phase must be carefully considered. 22
Information on the safety of individual drugs during pregnancy and lactation should be obtained from official drug labeling as well as reliable databases such as LactMed®, 23 which provides evidence-based guidance on maternal drug exposure.
Treatment considerations during pregnancy and lactation
Management of immunosuppressants during pregnancy and lactation
Among the immunosuppressive agents used for relapse prevention in NMOSD, azathioprine, tacrolimus, and cyclophosphamide are generally considered acceptable during pregnancy, and their continued administration may help mitigate the increased risk of relapse associated with pregnancy and delivery. 18 According to the consensus of the French MS Society, azathioprine can be continued until the diagnosis of pregnancy and throughout pregnancy (Grade B), 19 and breastfeeding is also possible in women treated with it (Grade B). 19 International guidelines support the compatibility of all three agents with breastfeeding.
In contrast, methotrexate and mycophenolate mofetil are strongly contraindicated during pregnancy (Grade B), 19 and breastfeeding is also contraindicated (Grade C), 19 due to their high teratogenic potential and increased risk of miscarriage. 18
Management of mAbs during pregnancy and lactation
Eculizumab and ravulizumab
Eculizumab, a humanized mAb that inhibits terminal complement activation via C5 cleavage, has not accumulated substantial clinical data in pregnant or postpartum women with NMOSD (Tables 1 and 2). However, in other indications, such as paroxysmal nocturnal hemoglobinuria 24 and myasthenia gravis, 25 no serious maternal or fetal adverse events have been reported. Eculizumab is largely composed of human IgG2 and IgG4 subclasses. Since IgG2 exhibits minimal placental transfer, fetal exposure is expected to be limited. 26
Safety of therapeutic antibodies for NMOSD during pregnancy.

EMA, European Medicines Agency; FDA, Food and Drug Administration; NMOSD, neuromyelitis optica spectrum disorder; PMDA, Pharmaceuticals and Medical Devices Agency.
Safety of therapeutic antibodies for NMOSD during lactation.
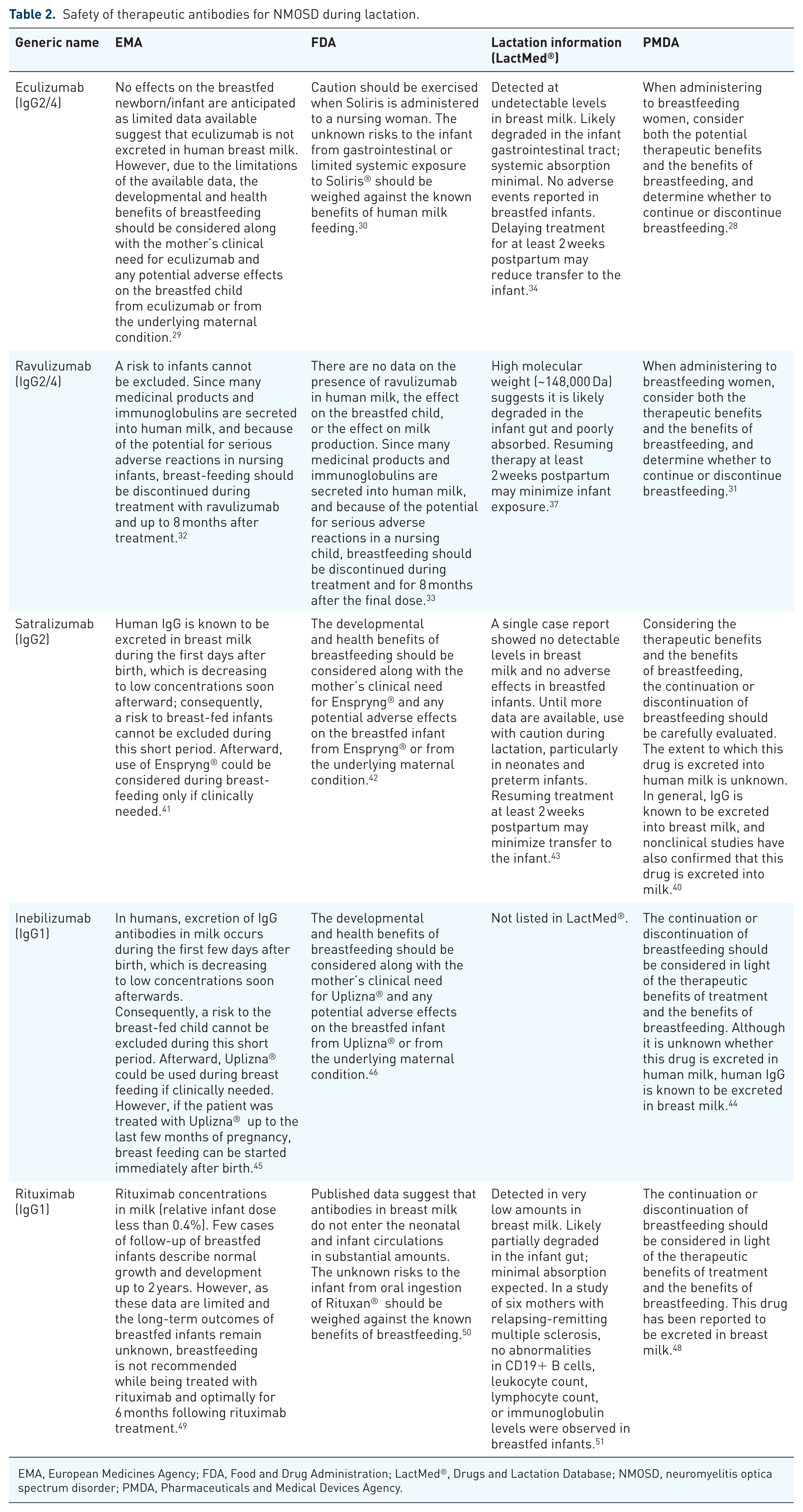
EMA, European Medicines Agency; FDA, Food and Drug Administration; LactMed®, Drugs and Lactation Database; NMOSD, neuromyelitis optica spectrum disorder; PMDA, Pharmaceuticals and Medical Devices Agency.
In conditions other than NMOSD, eculizumab has not been detected in breast milk, and no adverse effects have been reported in breastfed infants. 26 In a case report describing two patients with AQP4-Ab-positive NMOSD who received eculizumab throughout pregnancy, no adverse events were observed in either the mother or infant. 27
Eculizumab and ravulizumab should not be initiated without prior vaccination against Neisseria meningitidis and should be administered at least 2 weeks before the first dose, except in urgent clinical situations.28–33
Eculizumab is a large protein molecule with a molecular weight of approximately 148,000 Da and is therefore presumed to be degraded in the infant gastrointestinal tract and not absorbed systemically. To date, eculizumab has not been detected in the breast milk of mothers undergoing treatment, and no adverse events attributable to eculizumab have been reported in breastfed infants. Based on these findings, breastfeeding is considered compatible with eculizumab therapy.34–37
To date, there have been no epidemiological studies or case reports on the use of ravulizumab during breastfeeding. Given that ravulizumab is a large protein with a molecular weight of approximately 148,000 Da, it is degraded in the infant gastrointestinal tract, making systemic absorption unlikely. Delaying the resumption of treatment until at least 2 weeks postpartum may help minimize potential transfer to the infant. 37
Satralizumab
Satralizumab, a pH-dependent humanized mAb targeting the interleukin-6 receptor, has been reported in case studies involving pregnant and lactating women. In one patient, satralizumab was undetectable in the infant’s serum after 10 months of breastfeeding. 38 The infant experienced no serious infections, demonstrated normal growth and development, and the mother did not experience postpartum relapse.38,39
In the report by Yoshida et al., 38 satralizumab concentrations in both breast milk and the infant’s serum were below the detection limit (0.002 μg/mL). Based on these findings, breastfeeding appears to be compatible with satralizumab. Nakashima et al. also described the use of satralizumab during breastfeeding in patients with NMOSD, and no adverse events were observed in infants. 39
Data on pregnancy outcomes with satralizumab remain limited. In women with NMOSD who are pregnant or planning pregnancy, continuation of satralizumab therapy during pregnancy may be considered if the benefits clearly outweigh the risks.40–42
Although the transfer of satralizumab into human breast milk remains unknown, product labeling advises that its administration during pregnancy or in women who may become pregnant should be based on a benefit–risk assessment. The decision to continue or discontinue breastfeeding during treatment should be made with careful clinical judgment.40–43
Inebilizumab
Inebilizumab, a humanized anti-CD19 mAb, is contraindicated during pregnancy and for at least 6 months after the final dose, as specified in the prescribing information, due to the requirement for effective contraception during this period.44–46 During lactation, the decision to continue or discontinue breastfeeding should be based on a careful assessment of the therapeutic benefits for the mother and the advantages of breastfeeding for the infant.44–46 Neonates exposed to inebilizumab in utero may have an increased risk of infection; therefore, caution is advised when administering live or live-attenuated vaccines to these infants.44–46
Rituximab
Reports from Western countries on the use of rituximab, an anti-CD20 mAb, in pregnant and lactating women with NMOSD have not identified any serious adverse events. 47 According to Japanese prescribing information, 48 the EMA, 49 and the FDA, 50 rituximab may be administered during pregnancy only when the potential benefits outweigh the risks. The decision to continue or discontinue breastfeeding should be based on a careful assessment of both the therapeutic benefits to the mother and the benefits of breastfeeding to the infant.
The LactMed® database, 51 EMA, 49 FDA, 50 and Japanese labeling 48 support the compatibility of rituximab with breastfeeding but advise caution when nursing neonates or preterm infants.
Pregnancy and delivery outcomes following rituximab treatment were assessed in eight patients with NMOSD. Four patients delivered at term. Two patients underwent elective pregnancy termination upon medical recommendation, and one experienced intrauterine fetal death (IUFD). Rituximab was administered during pregnancy in two patients, and one patient was still pregnant at the time of reporting. Congenital anomalies or neonatal infections were observed. 52
To date, few studies have evaluated the safety and efficacy of rituximab in patients with NMOSD who desire pregnancy.
Management during delivery
Managing anesthesia during childbirth in patients with NMOSD requires careful consideration to provide effective analgesia while minimizing the risk of neurological deterioration. Two reports53,54 suggested that spinal anesthesia may be associated with the onset of the disease, raising concerns about a possible causal relationship between neuraxial anesthesia with bupivacaine and NMOSD onset. 54 Since then, anesthesiologists have often avoided regional anesthesia in patients with NMOSD and have preferred general anesthesia for cesarean section, given the potential risk of neurological deterioration and disease recurrence after neuraxial anesthesia. 55
However, several case reports describe successful regional anesthesia in women with NMOSD. Gunaydin et al. performed successful epidural anesthesia using a combination of bupivacaine and fentanyl in a paraplegic woman with NMOSD scheduled for cesarean section. 56 Chang et al. reported successful epidural anesthesia in a patient with NMOSD who underwent labor induction with ropivacaine and fentanyl for IUFD without neurological deterioration. 57 Greene et al. performed intrathecal anesthesia in a primigravida who underwent cesarean section at 32 weeks of gestation due to acute exacerbation of NMOSD, with no anesthesia-related adverse events. 58
Successful use of epidural or intrathecal anesthesia in pregnant women with NMOSD has also been reported in other cases.56–58 Tympa Grigoriadou et al. described an unplanned cesarean section with effective epidural supplementation in a pregnant woman with NMOSD in whom labor epidural analgesia had been selected. 59
These findings align with recent recommendations from the French Multiple Sclerosis Society, which encourage anesthesiologists to manage anesthesia in patients with NMOSD in the same way as in the general population. 19 Cesarean section and epidural anesthesia have been safely performed in patients with NMOSD without adverse events reported in the literature.56–59 Therefore, these procedures can be performed in the same manner as in the general obstetric population. 19
Treatment of relapse during pregnancy
In patients who experience relapse during pregnancy, high-dose intravenous methylprednisolone pulse therapy (IVMP) should be considered (Grade B). 19 Previous reports of pregnant patients with NMOSD who received corticosteroid treatment during pregnancy have been published,56,60–63 and the delivery of healthy infants was reported. Shang et al. 63 concluded that corticosteroid treatment is appropriate for NMOSD during pregnancy, particularly in the first trimester.
However, administration of IVMP during the first trimester should be avoided if possible, due to potential risks of miscarriage, congenital anomalies, and cleft palate. 64 In addition, a case of intrauterine growth restriction resulting in premature birth and cesarean section was reported in a pregnant patient with NMOSD who had been receiving continuous prednisolone and tacrolimus before and during pregnancy. 4 Other studies have reported an association between IVMP use during pregnancy and reduced birth weight. 65
Therefore, when considering IVMP for relapse during pregnancy in patients with NMOSD, the risks and benefits should be carefully weighed, and treatment should proceed only after obtaining informed consent from the patient and her family.
Plasma exchange is another therapeutic option, 66 and intravenous immunoglobulin (IVIg) may also be considered67,68 (Grade B), 19 particularly in patients with relapse of optic neuritis. These interventions should only be undertaken when the anticipated benefits outweigh the potential risks. In all cases, thorough counseling regarding risks and benefits must be provided, and informed consent obtained from the patient.
MRI during pregnancy and lactation
MRI during pregnancy should be avoided in the first trimester, as this period corresponds to the critical phase of fetal organogenesis. 69 An MRI can be performed during pregnancy if clinically warranted (Grade B). 19
Gadolinium-based contrast agents should not be used during pregnancy because they can cross the placenta, enter the amniotic fluid, and potentially be swallowed by the fetus, leading to systemic fetal exposure. 69
Although gadolinium-enhanced MRI is considered safe during lactation, if complete avoidance of infant exposure is desired, breastfeeding should be withheld for 24 hours following contrast administration. 70
Assisted reproductive technology
Assisted reproductive technology (ART) cannot be routinely recommended for women with NMOSD due to limited evidence. However, collaboration with ART specialists and consultation with neurologists are advised when considering its use. As with natural conception in women with NMOSD, it is important to ensure disease stability before attempting conception in order to optimize maternal and fetal outcomes. 19
Myelin oligodendrocyte glycoprotein antibody-related disease (MOGAD) in pregnancy
Close to 40% of patients suspected of having NMOSD who test negative for AQP4-Ab in serum are positive for myelin oligodendrocyte glycoprotein (MOG)-Ab using advanced cell-based assays (CBA). 71
MOG-Ab-associated disease (MOGAD) can complicate pregnancy. MOGAD is an autoimmune inflammatory demyelinating disease of the central nervous system. Pathogenic autoantibodies (MOG-Abs) bind to the myelin sheath of nerve fibers, causing demyelination. MOGAD leads to inflammation of the optic nerve, spinal cord, and brain. Symptoms include visual disturbance, sensory disturbance, motor disturbance, and urinary dysfunction. 72
In recent years, pregnancies complicated by MOG-Ab-positive disease have been reported. Similar to MS and NMOSD, cases of MOGAD have shown an increased frequency of relapses after delivery compared with the prepregnancy period.73,74 Immunosuppressive therapy has been shown to reduce relapse risk. 23
Leite et al. reviewed 107 relevant publications, including 65 clinical studies. 1 Collated pregnancy outcome data show clear risks in AQP4-Ab-positive NMOSD, including reduced rates of healthy delivery and an increased annualized relapse rate postpartum, often requiring adaptation of treatment regimens. For MOG-Ab-positive disease, as with MS and NMOSD, preconception care is important and may help prevent relapse after delivery.
Disease activity in MOGAD appears to be attenuated during pregnancy, but with an increased risk of relapse in the postpartum months. However, strong conclusions cannot yet be made due to the limited number of available cases. 74 Karalius et al. described two patients who presented with optic neuritis during pregnancy, one of whom was diagnosed with MS and the other with MOGAD. 75
For MOGAD, standard treatments outside of pregnancy include low-dose prednisolone and monthly intravenous immunoglobulin (IVIg). 75 Prednisolone is not contraindicated and may be continued during pregnancy. Regular IVIg carries potential risks of maternal hypovolemia and hyperviscosity, which may increase the risk of thromboembolism and therefore require monitoring. Nonetheless, IVIg is generally considered safe based on its use in pregnancy for other conditions.76,77
The diagnostic criteria for MOGAD have only recently been published, 72 and further accumulation of case data on pregnancies complicated by MOG-Ab-positive disease is awaited.
Impact of NMOSD autoantibody positivity (AQP4-Ab, MOG-Ab, or double-negative) on pregnancy
Collongues et al. investigated whether NMOSD autoantibody status (AQP4-Ab, MOG-Ab, or double-negative) affects pregnancy outcomes and examined the role of immunosuppressive therapy (IST) administered before or during pregnancy in reducing postpartum recurrence risk. 12
The results of this study showed a statistically significant decrease in the annual relapse rate (ARR) during pregnancy across all antibody subgroups (p < 0.001). However, a significant rebound in ARR was observed in the first postpartum trimester (PP1) compared with the first trimester, regardless of autoantibody type.
Pregnancies in AQP4-Ab-positive, MOG-Ab-positive, and double-negative NMOSD patients demonstrated similar patterns: a reduced risk of recurrence during pregnancy, followed by a rebound early postpartum. Compared with pre-pregnancy levels, this rebound was associated with a significant increase in ARR in AQP4-Ab-positive patients. In MOG-Ab-positive patients, the rebound tended to be less pronounced, although the difference was not statistically significant. Furthermore, administration of IST immediately before or during pregnancy was associated with a reduced risk of postpartum recurrence in all antibody subgroups.
Conclusion
Recent advances in relapse-prevention therapies have significantly reduced the risk of early postpartum relapse in NMOSD, allowing for improved disease control and enabling patients to pursue pregnancy and childbirth according to their reproductive goals. The increasing availability of mAb treatments has ushered in a new era of therapeutic management. Given the high efficacy of mAb therapy, the number of women of childbearing age with NMOSD receiving such treatments is expected to increase.
Although none of these mAbs are formally contraindicated during pregnancy or lactation, their long-term safety profiles remain to be fully established. In addition, careful consideration must be given to the timing of neonatal vaccination following in utero or postpartum exposure to mAbs.
To ensure maternal, fetal, and neonatal safety during treatment with novel therapeutics, shared decision-making tailored to individual patient circumstances is essential when determining management strategies for women of childbearing potential. Ongoing accumulation of clinical evidence and continuous updating of knowledge are required to provide optimal care during pregnancy, delivery, and breastfeeding in this patient population.
