Abstract
Aim
Several studies have reported a higher prevalence of migraine in patients with multiple sclerosis (MS) than in healthy controls (HCs). The aim of this study was to elucidate the headache prevalence in patients with MS or other neuroimmunological disorders, as well as to investigate the associations between headache characteristics and disease activity.
Methods
In this multicenter study in Japan, a headache questionnaire was distributed to patients with MS (n = 338), aquaporin-4 IgG-positive neuromyelitis optica spectrum disorder (NMOSD; n = 106), myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD; n = 51) or acetylcholine receptor antibody-positive myasthenia gravis (MG; n = 104), and healthy controls (HCs; n = 407). We included only participants aged 18–65 years in this study. The questionnaire responses were classified according to the International Classification of Headache Disorders, 3rd edition. The questionnaire was designed to systematically capture headache characteristics based on established diagnostic criteria.
Results
In total, any type of headache was found for 54.1% in MS, 63.2% in NMOSD, 43.1% in MOGAD, 43.3% in MG and 43.5% in HCs. After Holm–Bonferroni correction for comparisons among the five groups, the MS (p = 0.015) and NMOSD (p < 0.001) groups had significantly higher odds of any headache compared to the HC group. No difference was observed for migraine (MS 16.0%, NMOSD 16.0%, MOGAD 9.8%, MG 12.5%, HCs 17.0%). In contrast, patients with MS (24.0%) and NMOSD (37.7%) showed a significantly higher frequency of tension-type headache (TTH) than HCs (16.7%) even after adjusting for age and sex. Neither disease activity nor disease-modifying therapy displayed any association with headache severity in patients with MS, NMOSD, or MOGAD.
Conclusions
Our study showed no increased prevalence of migraine in MS and other neuroimmunological disorders, whereas TTH comorbidity was higher in MS and NMOSD than in controls.
This is a visual representation of the abstract.
Keywords
Introduction
Migraine is a common neurological disorder and its reported prevalence is 8.4% in the general population in Japan (1). Recent studies have suggested that chronic migraine can contribute to cognitive decline and reduced quality of life, whereas appropriate prophylactic treatments might help improve these outcomes (2). Additionally, structural abnormalities of gray matter have been observed in patients with migraine, with some evidence suggesting an association with cognitive impairment (3).
Conversely, patients with multiple sclerosis (MS), a neuroimmunological disease, have been reported to exhibit a markedly higher prevalence of migraine, ranging from 35.5% to 61.8% in studies conducted outside Japan (4,5). To our knowledge, only one study has investigated the prevalence of migraine among Japanese patients with MS, reporting a rate of 20.4% (6). The reasons for the increased prevalence of migraine in patients with MS remain unclear. Recent studies have suggested that migraine and MS may share common pathophysiological mechanisms, including neuroinflammatory processes and overlapping brain magnetic resonance imaging findings such as white matter lesions (7). Moreover, the relationship between disease activity and the frequency or severity of headaches in MS has not been fully elucidated.
If MS and migraine indeed co-occur at a high frequency, this could indicate the presence of shared pathophysiological mechanisms between the two diseases. As both diseases have many unresolved aspects in their pathophysiology, investigating their potential common mechanisms could provide valuable insights. Therefore, the present study aimed to determine whether patients with neuroimmunological disorders, including MS, have a higher prevalence of migraine than healthy controls (HCs) using International Classification of Headache Disorders, 3rd edition (ICHD-3) and explored the associations between headache frequency and disease activity in these disorders.
Methods
Patient enrollment
From February 2024 to January 2025, a headache questionnaire was distributed to patients diagnosed with MS, aquaporin-4-IgG-positive neuromyelitis optica spectrum disorder (NMOSD), myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) or acetylcholine receptor antibody-positive myasthenia gravis (MG).
Patients with MS, NMOSD and MOGAD were recruited from six collaborating centers: National Hospital Organization Hokkaido Medical Center, Tohoku Medical and Pharmaceutical University, Tohoku University Hospital, Osaka University Hospital, Keio University Hospital and Kyushu University Hospital, as well as Chiba University Hospital, and were required to meet established diagnostic criteria (8–10). At the collaborating centers other than Chiba University Hospital, approximately 60 consecutive cases were targeted for recruitment, whereas no predefined target number was set at Chiba University Hospital.
In addition, patients with MG were recruited exclusively at Chiba University Hospital to examine whether the findings observed in central nervous system (CNS) autoimmune diseases were specific to CNS autoimmunity or also applicable to peripheral immune-mediated disorders. Eligible participants were aged 18–65 years. The English version of the headache questionnaire is presented in the Supplementary material (Table S1). The questionnaire was not a formally validated diagnostic instrument. However, it was developed based on established headache diagnostic criteria and was reviewed and refined in consultation with an experienced headache specialist, comprising one of the investigators (MS), to ensure clinical relevance and clarity. The aim of the questionnaire was to systematically capture headache characteristics required for classification of headache types according to the ICHD-3.
At all participating facilities, physicians recorded clinical information on a standardized form, which, together with the completed questionnaires, was mailed to Chiba University for analysis. The standardized form recorded the following information: sex, age at the time of the questionnaire, diagnosis (MS, NMOSD or MOGAD), age at disease onset and use of treatments. Treatment data included whether the patient received disease-modifying therapies (DMTs) for MS and corticosteroids, immunosuppressive agents or biologics for NMOSD, as well as the age at initiation of these therapies. Additional data included the Neurostatus Expanded Disability Status Scale (EDSS) score at the time of questionnaire completion, the presence of oligoclonal bands (OCBs) and the number of attacks in the last one to two years preceding questionnaire completion (11, 12). Patients with a single attack, including first-attack cases, were included. The questionnaire also collected information on headache characteristics, including the use of acute headache therapies (e.g. non-steroidal anti-inflammatory drugs or triptans) and prophylactic headache medications. Headache days per month were obtained from the questionnaire. For responses reported as ranges (e.g. 1–2 days per month), the midpoint of the range was used for analysis. Missing responses were treated as missing and excluded from analyses of headache-day frequency. Information on symptomatic medication use was obtained from the questionnaire in both patients and HCs, whereas information on prophylactic treatments in patients was collected from the physicians. DMTs for patients with MS were classified into four categories based on their reported efficacy: high efficacy (ofatumumab and natalizumab), moderate efficacy (dimethyl fumarate, fingolimod and siponimod), low efficacy (glatiramer acetate and interferon-beta) and no treatment.
HC enrollment
At the cooperating facility (Chiba Foundation for Health Promotion and Disease Prevention, Chiba, Japan), the same headache questionnaire submitted to patients was distributed to staff members during their routine health examinations. The distribution methods included direct hand delivery via department heads, mailing to part-time staff with limited on-site workdays and notification via the intranet, allowing staff to print the questionnaire themselves.
Completed questionnaires were collected between 29 February 2024 and 25 March 2024, using two collection boxes placed at designated locations, where respondents were instructed to deposit their completed forms. The shorter collection period in HCs reflected the fixed schedule of annual health examinations.
Diagnostic criteria for patients
All patients with MS fulfilled the 2017 McDonald diagnostic criteria (8). All patients with NMOSD fulfilled the 2015 international consensus diagnostic criteria (9) and all were seropositive for anti–aquaporin-4 antibodies by cell-based assays. All patients with MOGAD tested for anti-myelin oligodendrocyte glycoprotein antibodies by cell-based assays and fulfilled the international diagnostic criteria (10). The MG diagnosis was based on typical clinical features of fatigable, fluctuating muscle weakness, as supported by electrophysiological evidence (repetitive nerve stimulation or single-fiber electromyography) or a positive ice-pack test, in accordance with international consensus guidance for MG management (MGFA, 2016; updated 2020) (13,14). Only patients with acetylcholine receptor antibody-positive MG were included.
Study size
This study was exploratory, and no formal sample size calculation was performed. The sample size was determined by the number of consecutive patients across the four diagnostic groups and HCs who met the inclusion criteria during the study period.
Classification of headache types
The questionnaire responses were classified according to ICHD-3. In line with the application of ICHD-3, a definite diagnosis was prioritized over a probable diagnosis. However, in cases in which the criteria for both probable migraine and probable tension-type headache (TTH) were met, the individual was classified as having probable migraine. Participants whose responses were insufficient to permit headache classification were categorized as “unclassifiable.” Headache subtype classification was performed by one of the investigators (HM) and subsequently reviewed by another (MS). Thus, the types of headaches were categorized as migraine, TTH, probable migraine, probable TTH, secondary trigeminal neuralgia, medication-overuse headache (MOH) and unclassifiable.
Statistical analysis
Statistical analyses were performed using SPSS, version 27.0 (IBM Corp., Armonk, NY, USA). For the analysis of m × n contingency tables, the chi-squared test was used. Continuous data were compared using the Mann–Whitney U-test. Spearman's rank correlation coefficient was used for correlation analysis. p < 0.05 (two-sided) was considered statistically significant.
In the primary analyses, we first examined the prevalence of any headache (yes/no) in each disease group versus HCs using logistic regression with adjustment for age and sex. Adjusted odds ratios (aORs) and 95% confidence intervals (CIs) were calculated. p-values were adjusted using Holm–Bonferroni correction.
Disease groups with a significant difference in overall headache prevalence versus HCs were then further analyzed for definite migraine and definite TTH. For each disease group, separate logistic regression models were fitted for migraine (yes/no) and TTH (yes/no) with adjustment for age and sex. Within each disease group, the two comparisons were corrected for multiple testing using the Holm–Bonferroni method (m = 2). Patients with probable migraine or other headache types were excluded from the primary regression analyses because of diagnostic uncertainty.
Sensitivity analyses were performed to assess the robustness of the findings. First, migraine was redefined to include both definite and probable cases, and the adjusted models were repeated. Second, multinomial logistic regression was conducted with “no headache” as the reference category and “migraine (definite + probable)” and “TTH (definite + probable)” as outcome categories. These sensitivity analyses were considered exploratory, and p-values were interpreted descriptively.
Results
Demographic and clinical profiles of patients and HCs
In total, we included 338 patients with MS, 106 patients with NMOSD, 51 patients with MOGAD and 104 patients with MG. Meanwhile, 796 questionnaires were distributed to HCs, of which 473 were returned, yielding a response rate of 59.4%. To match the age range of patients, only questionnaire responses from HCs aged 18–65 years were included. Consequently, 407 HCs were included in the analysis.
Table 1 presents the demographic and clinical profiles of patients and HCs, including headache days per month and details of acute and prophylactic headache medication use.
Demographic and clinical profiles of patients and HCs at questionnaire.

Data are presented as n (%) or median [interquartile range] (range). *p < 0.05.
**The data of a patient was not available.
***Multiple responses could be reported per patients.
****Percentages are shown as n/N (%), where N excludes patients with unknown medication status.
AN = attack number; DMF = dimethyl fumarate; ECU/RAV; eculizumab or ravulizumab; EDSS = neurostatus Kurtzke's Expanded Disability Status Scale; EFG = efgartigimod; FTY = fingolimod; GA = glatiramer acetate; HCs = healthy controls; IFNB = interferon-beta; INE = inebilizumab; MG = acetylcholine receptor antibody-positive myasthenia gravis; MG-ADL = Myasthenia Gravis Activities of Daily Living scale; MGC = Myasthenia Gravis Composite scale; MOGAD = myelin oligodendrocyte glycoprotein antibody-associated disease; NAT = natalizumab; MS = multiple sclerosis; NA = not available; NMOSD = aquaporin-4-IgG-positive neuromyelitis optica spectrum disorder; NSAIDs = non-steroidal anti-inflammatory drugs; OCB = oligoclonal band; OFA = ofatumumab; PSL = prednisolone; RIT = rituximab; ROZ = rozanolixizumab; SAT = satralizumab; SIP = siponimod.
The proportion of female participants and age at the questionnaires differed significantly among the five groups (p < 0.001).
Headache types in patients and HCs
Table 2 lists the headache subtypes in each disease group and HCs.
Headache prevalence and subtypes in patients and HCs.

Data are presented as n (%).
HCs = healthy controls; MG = acetylcholine receptor antibody-positive myasthenia gravis; MOH = medication-overuse headache; MOGAD = myelin oligodendrocyte glycoprotein antibody-associated disease; MS = multiple sclerosis; NMOSD = aquaporin-4-IgG-positive neuromyelitis optica spectrum disorder.
MOH comorbidity refers to cases with an underlying headache disorder. These patients are also included in the above primary headache categories.
A definite diagnosis was prioritized over a probable diagnosis. In cases where criteria for both probable migraine and probable TTH were met, the individual was classified as probable migraine.
In Figure 1, Migraine and TTH subtypes are presented as stacked bar graphs for each group. To improve visual clarity, secondary trigeminal neuralgia, unclassifiable and participants without headache (“none”) are not presented. However, the percentages represent the proportion of each headache type among all participants, including those without headache.
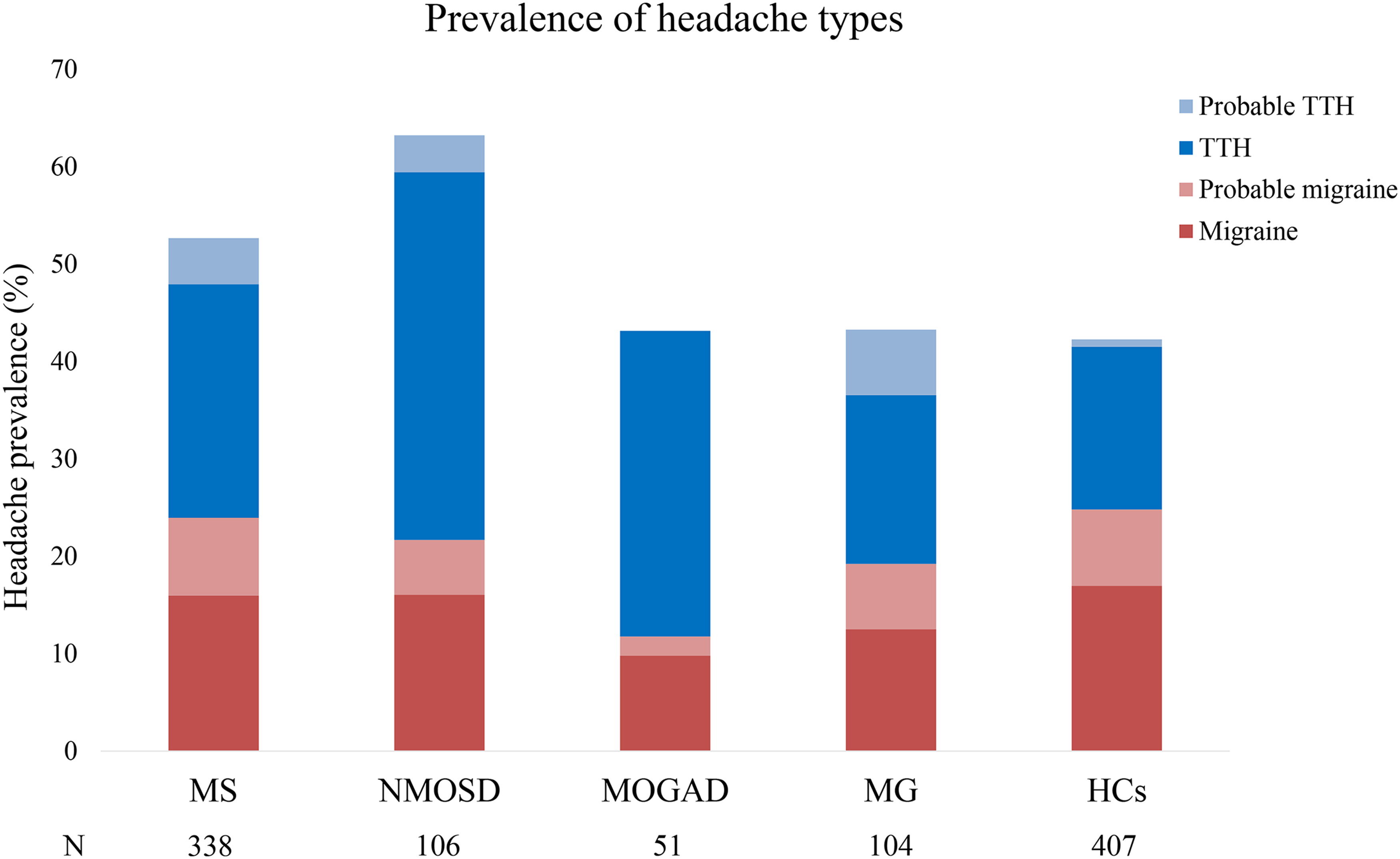
Prevalence of migraine and tension-type headache (TTH) in patients and HCs. Proportion of patients with different headache types across disease groups. The bar graph illustrates the relative frequency of four headache subtypes (migraine, probable migraine, TTH and probable TTH) in each of the five groups (multiple sclerosis (MS), aquaporin-4 IgG-positive neuromyelitis optica spectrum disorder (NMOSD), myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD), acetylcholine receptor antibody-positive myasthenia gravis (MG) and healthy controls (HCs)). For clarity, patients with other headaches or without headache are not presented.
Table 3 presents the logistic regression results for headache (any type) in the disease groups versus HCs following adjustment for age and sex and correction for multiple testing using the Holm–Bonferroni method.
Logistic regression results for headache (any type) in disease groups versus HCs, with Holm–Bonferroni correction.

CI = confidence interval; HCs = healthy controls; MG = acetylcholine receptor antibody-positive myasthenia gravis; MOH = medication-overuse headache; MOGAD = myelin oligodendrocyte glycoprotein antibody-associated disease; MS = multiple sclerosis; NMOSD = aquaporin-4-IgG-positive neuromyelitis optica spectrum disorder; OR = odds ratio.
After Holm–Bonferroni correction for four primary comparisons, patients with NMOSD (p < 0.001, below the adjusted threshold of 0.0125) and MS (p = 0.015, below the adjusted threshold of 0.0167) displayed significantly higher odds of headache than HCs, whereas no difference was noted for patients with MG and MOGAD.
There was no significant difference in migraine prevalence between patients with MS and HCs (p = 0.74). For TTH, patients with MS had a higher prevalence than HCs, and this association remained significant after Holm–Bonferroni correction (aOR = 1.6, 95% CI = 1.11–2.41, p = 0.013, below the adjusted threshold of 0.025).
Migraine prevalence did not differ between patients with NMOSD and HCs (p = 0.24). By contrast, TTH was significantly more frequent in patients with NMOSD, and this difference remained significant after Holm–Bonferroni correction (aOR = 3.6, 95% CI = 2.13–6.15, p < 0.001).
In sensitivity analyses including probable cases, the migraine prevalence in both patients with MS (aOR = 1.12, 95% CI = 0.77–1.60, p = 0.56) and NMOSD (aOR=1.34, 95% CI = 0.75–2.39, p = 0.33) did not differ from that in HCs. Conversely, TTH remained significantly more frequent in both disease groups (MS: aOR = 1.92, 95% CI = 1.32–2.78, p < 0.001; NMOSD: aOR = 3.58, 95% CI = 2.13–6.04, p < 0.001).
An overview of the associations between headache types and disease groups is summarized in Table 4.
Overview of associations between headache types and disease groups.
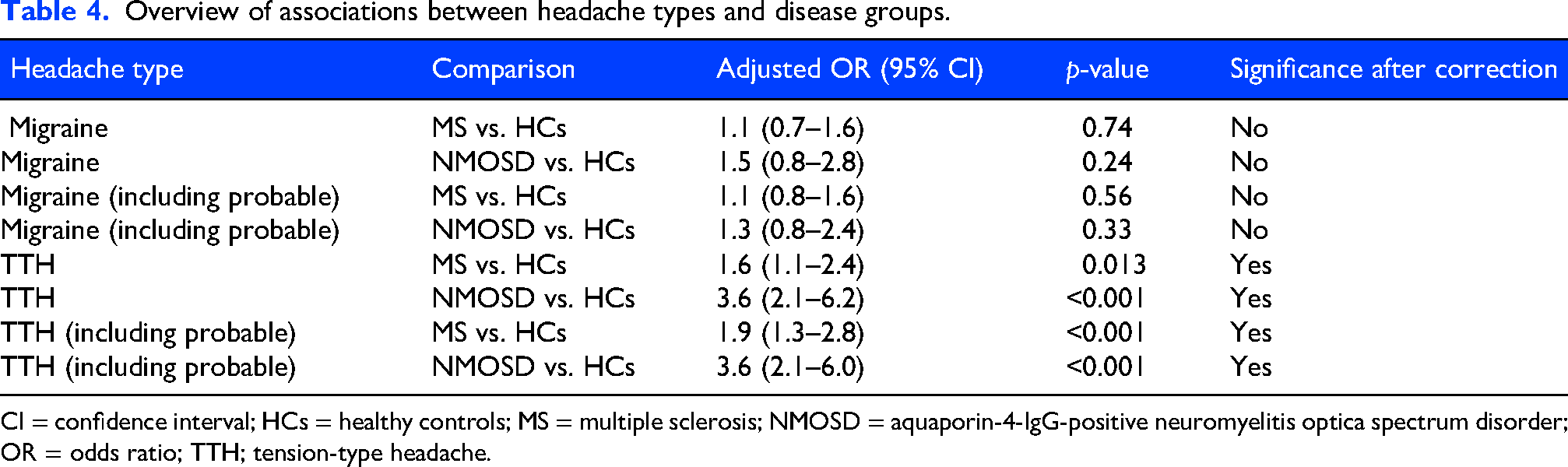
CI = confidence interval; HCs = healthy controls; MS = multiple sclerosis; NMOSD = aquaporin-4-IgG-positive neuromyelitis optica spectrum disorder; OR = odds ratio; TTH; tension-type headache.
Associations between headache and disease activity or treatment
In patients with MS, there was no correlation between the number of relapses in the past one or two years and the number of headache days, regardless of whether patients had any type of headache, migraine or TTH. Similarly, no correlation was found between relapse frequency and headache days in patients with NMOSD or MOGAD. There was no significant association between the DMT classification (low efficacy, moderate efficacy, high efficacy or none) and the presence of headache (p > 0.2). EDSS revealed no associations with headache frequency in MS, NMOSD and MOGAD.
Temporal relationship between headache and disease onset
In patients with MS and NMOSD, headache was significantly more likely to precede disease onset (p < 0.001 and p = 0.006, respectively). A similar trend was observed in patients with MOGAD (p = 0.065). Conversely, no temporal relationship weas observed between disease onset and headache onset in patients with MG (p = 0.14).
Association between OCB positivity and headache
No significant associations were observed between OCB positivity and the presence or absence of migraine, migraine plus probable migraine, TTH, or TTH plus probable TTH in patients with MS, NMOSD or MOGAD.
Discussion
In the present study, patients with MS and NMOSD had a higher prevalence of any headache than HCs, even after adjusting for age and sex, whereas the prevalence of headache did not differ between patients with MOGAD or MG and HCs. TTH was more frequent in patients with MS and NMOSD than in HCs, and the difference remained significant after adjustment for age and sex. Headache was not associated with disease activity in patients with MS, NMOSD or MOGAD. The prevalence of headache did not differ among patients with MS according to the DMT classification. In both the MS and NMOSD groups, headache often preceded disease onset.
Many studies employing the ICHD classification for headache diagnosis have reported that the prevalence of migraine is higher in patients with MS than in the general population (6,15–18). At the same time, several studies have found that TTH is the most frequent headache type in MS, followed by migraine (6,18). In the present study, the prevalence of migraine did not differ between patients with MS and HCs. However, the prevalence of migraine in patients with MS was 16%, and this value increased to 24.8% when including probable migraine, which is largely consistent with previous reports. Meanwhile, the prevalence of migraine was relatively high at 17% in HCs, which could explain the lack of between-group differences. A large-scale Japanese study previously reported a migraine prevalence of 8.4%, but almost 10% of participants in that study were aged 15–19 or >70 years (1). The same study also revealed that the prevalence of migraine was lower in these age groups than in the 20–69-year-old group. In our study, the age of HCs was restricted to 18–65 years to match that in the disease cohorts, particularly MS. This might have contributed to the relatively higher prevalence of migraine in HCs in our study. Therefore, caution is required when comparing prevalence across studies with different age structures.
It has been reported that interferon-beta treatment can worsen headache in patients with MS (6,19). In the present study, we divided DMTs into four types according to efficacy to compare headache prevalence, but no clear differences emerged. In contrast, previous studies have suggested that patients with untreated MS have a higher frequency of headache than their counterparts receiving any DMT (20). In our study, headache prevalence did not differ according to DMT categories. This could be related to the recent decline in interferon-beta use and the current trend toward initiating some form of DMT at an earlier disease stage.
Primary headache has not been well characterized in NMOSD (21). One study employing the International Classification of Diseases, 10th revision for headache classification reported that the risk of migraine was increased in MS but not in NMOSD (22). Another study found that patients with AQP4 antibody positivity had a higher prevalence of migraine compared their antibody-negative counterparts (6). In our analysis, the prevalence of migraine or TTH did not differ significantly from that in HCs. Further investigation is required to confirm this finding.
Regarding MOGAD, extremely few studies have investigated the prevalence of primary headache. Reports have mainly described ocular pain during optic neuritis or migraine-like headache in the acute phase of optic neuritis (23,24). In our study, the prevalence of migraine or TTH in patients with MOGAD during remission was similar to that in HCs. However, caution is needed in interpreting this result because the prevalence of migraine in HCs in our study might have been higher than that generally expected in the Japanese population (1).
The increased prevalence of “any headache” observed in MS and NMOSD in the present study appears to be primarily driven by a higher frequency of TTH rather than migraine. The mechanisms linking headache to the immunopathogenesis of MS, NMOSD and MOGAD are likely heterogeneous. In MS, cortical pathology and meningeal inflammation, including ectopic B-cell follicle formation, have been proposed as potential contributors to headache (25). In NMOSD, headache may reflect disruption of central pain-processing pathways due to spinal cord and brainstem involvement and astrocyte dysfunction, rather than focal cortical pathology (21). In MOGAD, headache may be related to cortical and/or meningeal inflammation in specific clinical phenotypes. In addition to these biological factors, psychological comorbidities may also contribute, particularly for TTH, as depression and anxiety have been associated with the frequency and severity of TTH attacks (26–28). In NMOSD, a higher prevalence of pain and depressive symptoms has been reported and these factors have been shown to be interrelated (29–31). Recent studies have suggested that acupuncture treatment for TTH may improve not only headache symptoms, but also pain and depressive symptoms (32). It is therefore possible that the higher frequency of TTH observed in patients with NMOSD in the present study was influenced, at least in part, by underlying depression or anxiety. However, depressive symptoms were not assessed in the present study. Future studies should include systematic evaluation of depression and anxiety and examine changes before and after therapeutic interventions.
Headache is often reported by patients with MG (33), but studies specifically investigating headache in MG are scarce. One study found that, among 184 patients with MG, 71 (38.6%) had TTH, whereas nine (4.9%) reported migraine, suggesting that TTH is relatively common (34). In the same study, 13.6% of patients developed new-onset or worsening headache after MG onset, and the odds of headache was increased in the presence of ocular symptoms. In our analysis, there were no particular features, such as a higher prevalence of migraine or TTH, in patients with MG, nor was there a specific association between headache and disease onset. It should be noted that all of our patients were acetylcholine receptor-positive, whereas, in the previous study, only 72.8% were antibody-positive, which might reflect differences in patient background.
This study had several limitations. First, HCs were recruited on a voluntary basis during routine health check-ups, and it is possible that individuals experiencing headache were more likely to participate, introducing a potential selection bias. Second, even when based on the ICHD criteria, self-reported questionnaires can permit misclassification, particularly between migraine and TTH or in “probable” categories. Third, a small number of patients had partially incomplete questionnaire responses, making it unclear whether they met the diagnostic criteria for MOH. Because we did not confirm the missing information by phone or other means, diagnoses were made only within the limits of the available data. In addition, cluster headache may have been underrecognized due to reliance on patient-reported symptoms. Beyond these methodological limitations, the potential influence of ongoing immunotherapies, including interleukin-6-targeted therapies, on headache-related outcomes could not be specifically evaluated in this study. Previous studies have also reported that pain occurs more frequently in NMOSD than in MS (31,35), and it is possible that patients with NMOSD were taking more analgesic medications. These factors could have contributed to the frequency of headaches in patients with NMOSD. Finally, recall of past headache frequency and onset timing might also have introduced errors, which could differ between patient groups and HCs.
In summary, migraine prevalence was not elevated in MS or other neuroimmunological disorders, whereas TTH was more common in MS and NMOSD compared to controls. Headache was not related to disease activity or DMT classification, but, in both MS and NMOSD, headache often preceded disease onset. These findings suggest that headache, particularly TTH, is a characteristic comorbidity in MS and NMOSD. However, the relatively high prevalence of migraine in our control group should be considered when interpreting the results. Larger studies, ideally population-based, are warranted to confirm these findings and further clarify the relationship between headache and neuroimmunological disorders.
Key findings
Patients with MS and NMOSD had a higher overall headache prevalence compared to healthy controls. Migraine prevalence did not increase in MS, NMOSD, MOGAD or MG compared to controls. Tension-type headache was more common in MS and NMOSD, and headache severity was not associated with disease activity or treatment.
Supplemental Material
sj-docx-1-cep-10.1177_03331024261429158 - Supplemental material for Headache prevalence and types in multiple sclerosis and related neuroimmunological diseases: A multicenter study
Supplemental material, sj-docx-1-cep-10.1177_03331024261429158 for Headache prevalence and types in multiple sclerosis and related neuroimmunological diseases: A multicenter study by Hiroki Masuda, Akiyuki Uzawa, Mamoru Shibata, Masahiro Mori, Yusei Miyazaki, Masaaki Niino, Juichi Fujimori, Ichiro Nakashima, Yoshiki Takai, Naoya Yamazaki, Tatsuro Misu, Makoto Kinoshita, Tatsusada Okuno, Koji Sekiguchi, Jin Nakahara, Hajime Takeuchi, Keisuke Mizutani, Ayano Matsuyoshi, Yuu-ichi Kira, Kazunori Iwao, Eizo Tanaka, Koji Shinoda, Mitsuru Watanabe, Katsuhisa Masaki, Noriko Isobe, Mayumi Muto, Ryohei Ohtani, Shinji Aoyama, Sonoko Misawa, Kazumoto Shibuya, Tomoki Suichi, Manato Yasuda, Yuya Aotsuka, Marie Morooka, Ryo Otani, Yosuke Onishi, Hiroyuki Akamine, Hideo Handa, Etsuko Ogaya, Moeko Ogushi, Kentaro Kurumada and Satoshi Kuwabara in Cephalalgia
Footnotes
Acknowledgments
We thank Dr Takehiko Fujisawa, Mr Kenji Ishii and Ms. Fumika Kumahara from the Chiba Foundation for Health Promotion and Disease Prevention for their kind participation in the headache questionnaire survey.
ORCID iDs
Ethical considerations
Ethical approval for this multicenter study was obtained through centralized review by the ethics committee of Chiba University Hospital, which provided approval for all participating institutions (No. HK202306-02). The methods used in this study complied with the Declaration of Helsinki and its subsequent amendments, and all procedures were performed in accordance with relevant guidelines and regulations.
Consent to participate
All patients provided their written informed consent.
Author contributions
HM drafted the first manuscript. AU, MS, MM (Mori), MN, JF, IN, TM, TO, KS (Sekiguchi), JN, KS (Shinoda), NI and SK revised the manuscript and approved the final version submitted for publication. HM, MM (Mori), SH and SK contributed to the conception and design of the study. HM drafted the text and prepared the figures. The lead investigators at each participating center (AU, MM (Mori), MN, JF, TM, TO, KS (Sekiguchi) and KS (Shinoda)) contributed to study coordination, participated in video conferences and provided important input on the study direction. Investigators at the participating centers (HM, AU, MM (Mori), YM, MN, JF, IN, YT, NY, TM, MK, TO, KS (Sekiguchi), JN, HT, KM (Mizutani), AM, YK, KI, ET, KS (Shinoda), MW, KM (Masaki), NI, MM (Muto), RO (Ohtani), SA, SM, KS (Shibuya), TS, MY, YA, EM, RO (Otani), YO, HA, HH, EO, MO and KK) contributed to patient recruitment and data collection. HM, AU, MM and KS (Shibuya) designed the data collection form for patient clinical information used across centers.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability
Data are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
