Abstract
Background:
Patients with hematochezia often present with inadequate bowel preparation, which compromises the accuracy of colonoscopy. Despite this clinical challenge, evidence guiding optimal preparation strategies for this population is limited.
Objectives:
We directly compared the efficacy and safety of ultra-low-volume (1 L) polyethylene glycol/ascorbic acid (PEG/Asc) versus low-volume (2 L) PEG/Asc in patients undergoing colonoscopy for hematochezia.
Design:
In a prospective, randomized, multicenter non-inferiority trial, patients received either 1 or 2 L PEG/Asc using standard split-dose regimens.
Methods:
Bowel preparation quality was assessed using the Boston Bowel Preparation Scale (BBPS). Secondary outcomes included patient satisfaction, adverse events, colonic bleeding source identification rate, and adenoma detection rate (ADR).
Results:
In total, 184 patients were analyzed. Successful bowel cleansing was achieved in 78.3% of the 1 L PEG/Asc group and 76.1% of the 2 L PEG/Asc group, confirming non-inferiority. The mean BBPS score was higher with 1 L PEG/Asc (6.87 vs 6.30; p = 0.053). Patient satisfaction favored the 1 L PEG/Asc group, although the difference was not statistically significant. Colonic bleeding source identification was slightly higher in the 1 L PEG/Asc group than in the 2 L PEG/Asc group, but the difference did not reach statistical significance (82.4% vs 73.6%; p = 0.165). Completion rates, adverse events, and ADR were similar between groups.
Conclusion:
Ultra-low-volume (1 L) PEG/Asc is as effective and safe as 2 L PEG/Asc for bowel preparation in patients with hematochezia. Bowel cleansing and satisfaction were higher with the 1 L PEG/Asc regimen without statistically significant differences, supporting its use as a practical alternative.
Trial registration:
The study was registered with the Clinical Research Information Service, Republic of Korea, a primary registry in the WHO Registry Network (registration number: KCT0006124).
Plain language summary
Hematochezia, which means passing bright red blood from the bowel, is a common reason for people to seek medical care. When this happens, doctors usually recommend a colonoscopy to find the cause of bleeding and provide treatment. To perform a safe and accurate colonoscopy, the bowel must be properly cleaned. However, drinking large amounts of cleansing solution is often uncomfortable, especially for people who are already unwell. This study examined whether a smaller amount of bowel cleansing solution could work as well as a larger amount in patients with hematochezia. We compared two cleansing regimens: a 1-liter solution and a 2-liter solution. Our main goal was to learn whether the smaller amount would clean the bowel well enough for doctors to clearly see the inside of the colon. We also evaluated how patients felt while taking the solution and whether any side effects occurred. We conducted a randomized clinical study at two hospitals. Adults with hematochezia were assigned to drink either the 1-liter or the 2-liter solution before their colonoscopy. We measured bowel cleanliness, the ability of doctors to identify the bleeding source, patient comfort and willingness to repeat the same preparation, and any safety concerns. The study included 184 patients. Overall, the 1-liter solution cleaned the bowel just as well as the 2-liter solution. In some parts of the colon, especially the transverse and left segments, the 1-liter solution even performed slightly better. Both groups had similar success in identifying the source of bleeding, and no serious side effects were found. Many patients found the smaller-volume preparation easier and more comfortable to take. These results suggest that a 1-liter preparation is an effective and safe option for people with hematochezia who need a colonoscopy. A smaller amount may improve comfort without reducing the quality of the examination.
Introduction
Colonoscopy is a critical procedure for the diagnosis and treatment of colorectal diseases. Adequate bowel preparation is essential to ensure clear visualization of the intestinal mucosa, which directly impacts diagnostic accuracy and the effectiveness of therapeutic interventions.1,2 Poor bowel cleansing can obscure lesions, hinder the detection of pathology, prolong examination time, increase the risk of missed findings, and often necessitate early repeat procedures, ultimately compromising both diagnostic yield and procedural quality.3,4 Despite its central role in colonoscopy success, bowel preparation is frequently associated with patient discomfort, poor compliance, and, in rare cases, adverse reactions to the cleansing agents.4–7 To address these challenges and enhance patient adherence, newer preparation regimens have been developed, including low-volume polyethylene glycol (PEG) solutions supplemented with ascorbic acid and oral sulfate tablets. These formulations offer comparable cleansing efficacy to traditional methods while improving convenience and tolerability.8–11
Hematochezia is one of the most common gastrointestinal symptoms encountered in clinical practice. Due to the anatomical location of the bleeding, colonoscopy is recommended as the key diagnostic and therapeutic modality in most clinical guidelines. 12 In such patients, adequate bowel preparation is essential for effective lesion identification and treatment. Insufficient bowel preparation may impair visualization, which makes it difficult to accurately identify the source of bleeding or leads to failure of endoscopic treatment.12–14 A recent systematic review and meta-analysis reported that hematochezia itself is an independent risk factor for inadequate bowel preparation, in addition to previously known risk factors such as constipation, diabetes, and history of abdominal surgery. 15 Nevertheless, there are currently no clear guidelines for standard bowel preparation methods in patients with hematochezia, and some guidelines merely recommend administering PEG-based bowel preparation agents as quickly as possible.12,14,16 Recently, low-volume PEG preparations with improved convenience have been developed, and their clinical use is gradually expanding, showing good compliance and bowel preparation efficacy.11,17,18 However, no randomized studies have prospectively evaluated the efficacy and safety of such preparations in patients with hematochezia or directly compared formulations (e.g., ultra-low-volume (1 L) vs low-volume (2 L)). Therefore, this study evaluated the efficacy and safety of 1 L PEG/Asc in patients with hematochezia through a multi-center, prospective, randomized, non-inferiority trial comparing it with a 2 L PEG/Asc regimen.
Materials and methods
Study design and patients
We conducted a multicenter, prospective, randomized, single-blind (endoscopist-blinded) non-inferiority trial at two academic institutions (Wonju Severance Christian Hospital and Kangwon National University Hospital) from June 2020 to December 2021. This study was designed and reported in accordance with the Consolidated Standards of Reporting Trials (CONSORT) 2025 guidelines. 19 Prior to undergoing bowel preparation, all participants provided written informed consent after being thoroughly briefed on the study’s objectives, methodology, and potential side effects. The study was approved by the Institutional Review Board (IRB) of Wonju Severance Christian Hospital, Wonju, Republic of Korea (IRB No. CR320001, approved on April 14, 2020), and was registered with the Clinical Research Information Service, Republic of Korea, a primary registry in the WHO Registry Network (registration number: KCT0006124). All patient data were fully de-identified prior to analysis, and no personally identifiable information is included in the text, tables, or figures.
Consecutive eligible patients aged 20–84 years with hematochezia who were scheduled to undergo colonoscopy for diagnosis were enrolled. Hematochezia was defined as patient-reported rectal bleeding, subsequently confirmed by a physician through history taking and physical examination. The exclusion criteria were as follows: previous colectomy; congestive heart failure or acute myocardial infarction in the preceding 6 months; American Society of Anesthesiologists class ⩾III; uncompensated liver cirrhosis; ascites; severe renal insufficiency; inflammatory bowel disease; severe systematic inflammatory diseases; individuals with disabilities (e.g., cognitive impairment, intellectual disability) or pregnant women; ileus, intestinal obstruction, or toxic megacolon; and patients who refused to provide written informed consent. Enrolled subjects were randomly assigned to either the 1 or 2 L PEG/Asc group before bowel preparation. Randomization was performed using a computer-generated randomization program (https://www.randomizer.org) with a 1:1 allocation ratio. The allocation sequence was accessible only to the research coordinator who enrolled participants and assigned them to interventions, while the endoscopists remained blinded to the group assignments.
Bowel preparation
The principal investigator, colonoscopists, and endoscopy assistant nurses remained unaware of the assigned bowel preparation types throughout the study period. By contrast, both the patients and the clinical research coordinator responsible for administering the bowel cleansing preparations were aware of the assignments, as the preparation techniques differed between the groups. Bowel cleansing was performed using a split-dose regimen of PEG with ascorbic acid (PEG/Asc). In the 1 L PEG/Asc group, patients ingested 500 mL PEG/Asc followed by at least 500 mL water the evening before the colonoscopy, and an additional 500 mL PEG/Asc with 500 mL water on the morning of the procedure, completed within 3–6 h before colonoscopy. In the 2 L PEG/Asc group, patients ingested 1 L of PEG/Asc followed by 500 mL of water on the evening prior, and 1 L of PEG/Asc with 500 mL of water on the morning of the procedure. Participants were provided written and verbal instructions regarding the timing and method of intake, and adherence was confirmed by research coordinators at the time of colonoscopy. All colonoscopies were scheduled in the morning to ensure a uniform interval between bowel preparation and the procedure.
Assessment of outcomes
The primary outcome was bowel cleansing quality, assessed using the Boston Bowel Preparation Scale (BBPS), a validated 9-point scoring system that evaluates three colonic segments (right, transverse, and left). Adequate preparation was defined as a total BBPS score ⩾6 with all segmental scores ⩾2. The Harefield Cleansing Scale (HCS) was also used as a secondary measure, with grades A or B indicating successful cleansing. Secondary outcomes included the colonic bleeding source identification rate, defined as the proportion of patients in whom the colonic bleeding source was clearly visualized during colonoscopy and used as a clinical indicator of mucosal visibility related to bowel preparation quality. This outcome was assessed only in patients whose bleeding etiology was potentially attributable to colonic sources; patients with confirmed small bowel or upper gastrointestinal bleeding were excluded from the denominator, as colonic mucosal visualization would not be relevant in these cases. Additional secondary outcomes included the adenoma detection rate (ADR); patient-reported tolerability and compliance based on standardized questionnaires; and safety outcomes such as adverse events (e.g., nausea, vomiting, abdominal discomfort, dizziness) and laboratory abnormalities (electrolyte disturbances and renal function changes).
Evaluation of tolerability, acceptability, and safety
Tolerability and acceptability were assessed using a structured, patient-reported questionnaire administered on the day of colonoscopy. Participants rated taste, ease of ingestion, satisfaction, and willingness to repeat the same regimen, which were used to evaluate both tolerability and acceptability. Compliance was confirmed by direct inquiry and verification of any remaining solution.
Safety was evaluated by monitoring adverse events and laboratory tests. Adverse events, including nausea, vomiting, abdominal pain, abdominal distension, sleep disturbance, headache, numbness and tingling, feeling of fatigue, thirstiness, hunger, seizure, drowsiness, fecal incontinence, and dizziness, were recorded and graded as mild, moderate, or severe. Laboratory assessments included tests for blood urea nitrogen, creatinine, sodium, potassium, chloride, magnesium, calcium, phosphate, bicarbonate, and aspartate aminotransferase, obtained before and after bowel preparation. Any clinically significant abnormalities or preparation-related complications were documented and reviewed.
Statistical analysis
The sample size was calculated based on an assumed adequate bowel preparation rate of 85% in both groups, a non-inferiority margin of 15%, a one-sided α of 0.025, and 80% power. The non-inferiority margin of 15% was determined based on prior randomized controlled trials evaluating 1 and 2 L PEG/Asc regimens, in which a 15% margin was consistently adopted.9,17,18,20 Given the anticipated adequate bowel preparation rate of approximately 85%, a difference within 15% was considered unlikely to result in a clinically meaningful deterioration in mucosal visualization. Based on these assumptions, 89 patients per group were required. Considering a 10% dropout rate, the final target sample size was 196 participants (98 per group).
The primary efficacy analysis was conducted in the per-protocol (PP) population, which included patients who completed bowel preparation and underwent evaluable colonoscopy. Given the non-inferiority design, PP analysis was selected as the primary analytic approach. A sensitivity analysis using the intention-to-treat (ITT) population, defined as all randomized patients, was additionally performed to assess the robustness of the results. In the ITT analysis, patients who withdrew consent or had incomplete colonoscopy were conservatively classified as having inadequate bowel preparation. There were no missing data for the primary endpoint in the PP population, and no additional imputation procedures were applied.
Continuous variables were compared using Student’s t test or the Mann–Whitney U test, as appropriate; the data are expressed as mean ± standard deviation or median (interquartile range). Categorical variables were compared using the Chi-square test or Fisher’s exact test; the data are presented as frequencies and percentages. Non-inferiority for the primary endpoint was concluded if the lower bound of the one-sided 97.5% confidence interval (CI) for the difference in adequate preparation rates was above the prespecified non-inferiority margin of −15%. A one-sided Miettinen–Nurminen approach was used to obtain the p value for non-inferiority of the risk difference with a prespecified margin of −15%, using a one-sided α of 0.025. Secondary outcomes, including BBPS and HCS scores, colonic bleeding source identification rate, ADR, and patient-reported tolerability and acceptability, were compared using appropriate parametric or non-parametric tests. Pre- and post-preparation laboratory parameters were analyzed using paired t tests or Wilcoxon signed-rank tests. Adverse events were summarized descriptively. All analyses were conducted using SPSS software (version 26.0; IBM Corp., Armonk, NY, USA), with p < 0.05 considered statistically significant.
Results
Baseline characteristics
The study flow is shown in Figure 1. In total, 196 patients were enrolled and randomized, of whom 12 were excluded (subject withdrawal: n = 5; incomplete colonoscopy: n = 7). Consequently, 184 patients completed the study and were included in the final analysis (1 L PEG/Asc group: n = 92; 2 L PEG/Asc group: n = 92). The baseline characteristics of the study population are summarized in Table 1. There were no significant differences between the two groups in age, sex, underlying comorbidities, or other variables, including the presence of overt hematochezia.

Flow of participants through the study, indicating ITT and PP analysis populations. A total of 200 patients were assessed for eligibility, and 196 were randomized (ITT population). After exclusions, 92 patients in each group completed bowel preparation and underwent evaluable colonoscopy, constituting the PP population (n = 184).
Baseline characteristics.

Unstable vital signs were defined as meeting any of the following within 24 h before colonoscopy: systolic blood pressure <90 mmHg, heart rate ⩾120 beats/min, hemoglobin <8 g/dL, or transfusion of ⩾2 units of blood.
If there is a large amount of active bleeding (150 mL or more) at least once within 24 h prior to colonoscopy.
Asc, ascorbic acid; BMI, body mass index; CKD, chronic kidney disease; DM, diabetes mellitus; HTN, hypertension; OPD, outpatient department; PEG, polyethylene glycol.
Efficacy of bowel cleansing
The 1 L PEG/Asc regimen was non-inferior to the 2 L PEG/Asc regimen for adequate bowel cleansing. The observed difference in adequate cleansing rates between the two groups was 2.2% (one-sided 97.5% CI: −10.1% to 14.4%), exceeding the prespecified non-inferiority margin of −15% (Table 2). Non-inferiority was also supported by the Miettinen–Nurminen test (one-sided p for non-inferiority = 0.0030). Sensitivity analysis using the ITT population demonstrated results consistent with the primary PP analysis, confirming the non-inferiority of the 1 L PEG/Asc regimen (Supplemental Table 1). The mean total BBPS score was higher in the 1 L PEG/Asc group, although the difference did not reach statistical significance. Segmental analysis also demonstrated consistently higher BBPS scores in the 1 L PEG/Asc group, with a significant difference observed in the transverse colon (Table 2). Segmental HCS scores were higher in the 1 L PEG/Asc group, with significant differences in the transverse, descending, and sigmoid colon but not in the cecum/ascending colon or rectum. The distribution of HCS grades (A–D) was significantly different between the two groups, with Grade A being more frequent in the 1 L PEG/Asc group (Figure 2).
Efficacy of bowel preparations as assessed by BBPS.

Asc, ascorbic acid; BBPS, Boston Bowel Preparation Scale; CI, confidence interval; PEG, polyethylene glycol.

Distribution of HCS grades between the two bowel preparation groups. The 1 L PEG/Asc group and the 2 L PEG/Asc group showed significantly different distributions of HCS grades (p = 0.037).
Clinical outcomes
Colonoscopy revealed no significant differences in the etiology of bleeding between the two groups. The colonic bleeding source identification rate, reflecting mucosal visibility, was higher in the 1 L PEG/Asc group than in the 2 L PEG/Asc group (82.4% vs 73.6%), although the difference was not statistically significant (p = 0.165). Among patients with an identified bleeding etiology, colon diverticulum was the most common cause, followed by colitis. Rebleeding within 2 weeks after colonoscopy occurred in one patient in each group. There was no significant difference in the ADR between the two groups (Table 3).
Clinical outcomes, including bleeding etiology, colonic bleeding source identification rate, rebleeding, and detection rates.
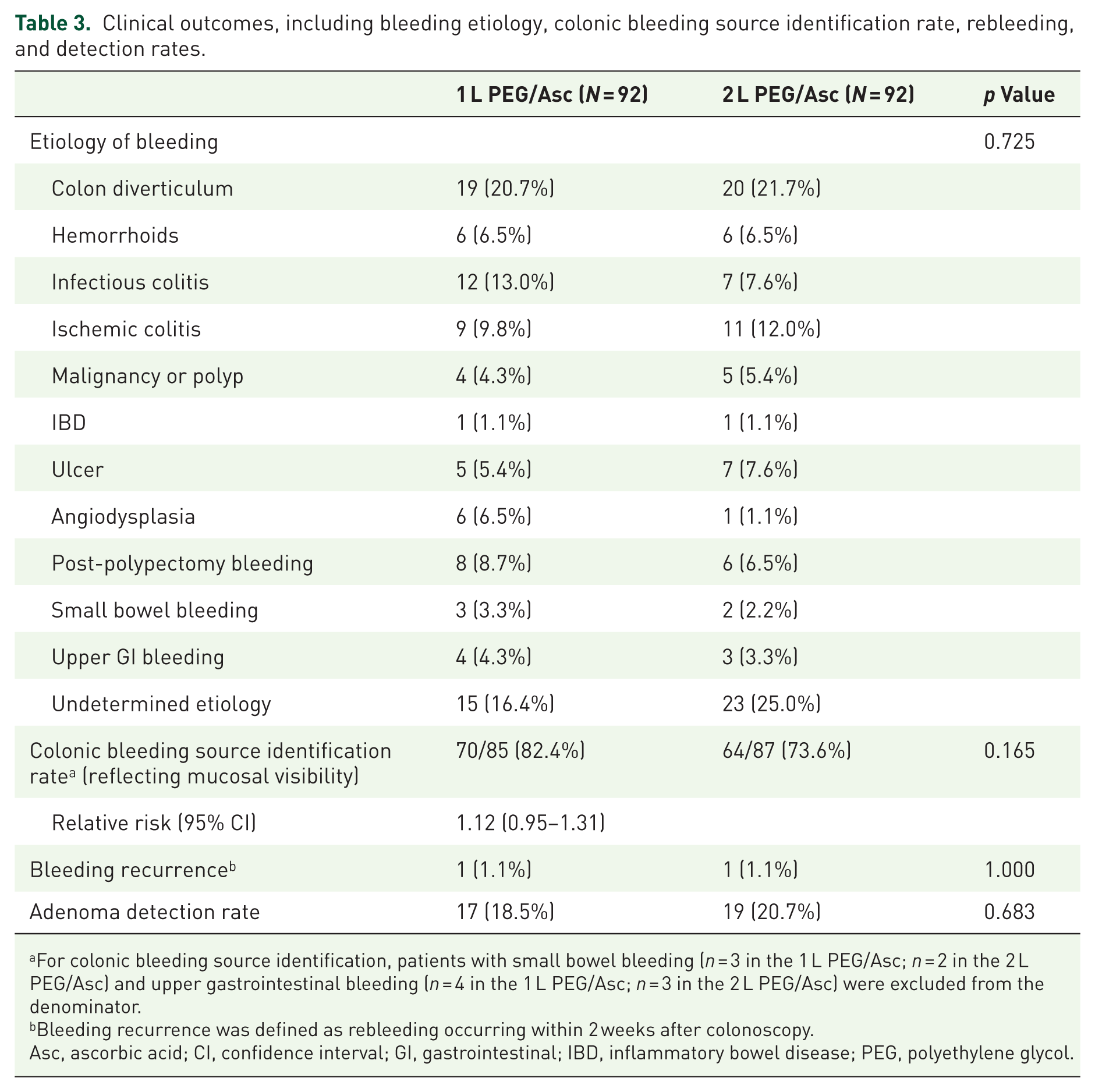
For colonic bleeding source identification, patients with small bowel bleeding (n = 3 in the 1 L PEG/Asc; n = 2 in the 2 L PEG/Asc) and upper gastrointestinal bleeding (n = 4 in the 1 L PEG/Asc; n = 3 in the 2 L PEG/Asc) were excluded from the denominator.
Bleeding recurrence was defined as rebleeding occurring within 2 weeks after colonoscopy.
Asc, ascorbic acid; CI, confidence interval; GI, gastrointestinal; IBD, inflammatory bowel disease; PEG, polyethylene glycol.
Tolerability and acceptability of bowel cleansing solution
Patient tolerability and acceptability of the bowel preparations are summarized in Table 4. Intake of all solution rates for the bowel preparation were 92.4% in the 1 L PEG/Asc group and 91.3% in the 2 L PEG/Asc group. Willingness to repeat the same preparation was reported by 68.5% and 62.0% of patients in the 1 L PEG/Asc and 2 L PEG/Asc groups, respectively. Both overall experience and taste ratings were more favorable in the 1 L PEG/Asc group, although the differences were not statistically significant.
Tolerability and acceptability outcomes of bowel preparation.
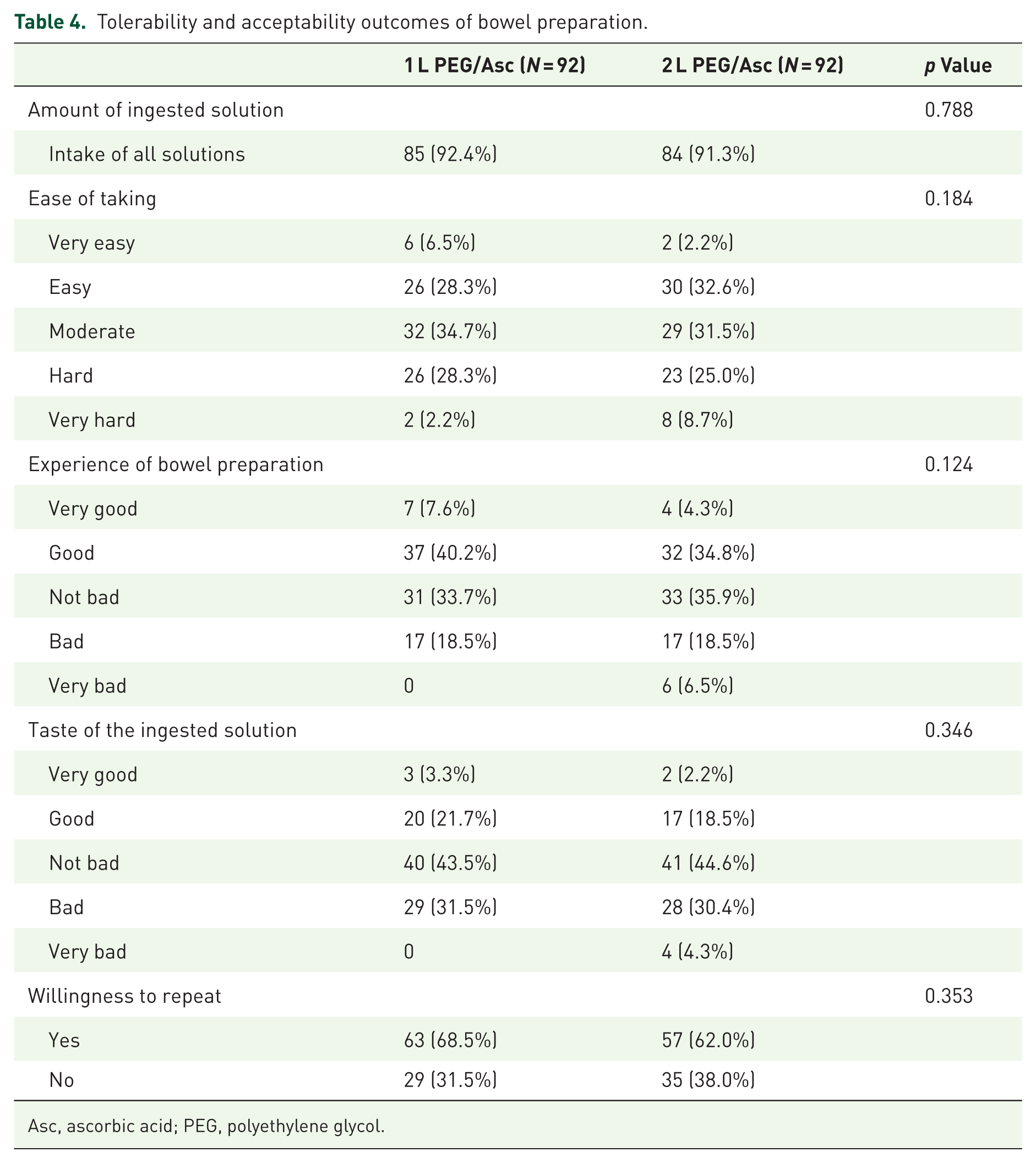
Asc, ascorbic acid; PEG, polyethylene glycol.
Safety and adverse events
No serious preparation-related adverse events or deaths occurred during the study period. Pre- and post-colonoscopy laboratory tests revealed no clinically significant changes and no evidence of nephrotoxicity or electrolyte disturbances (Supplemental Table 2). Details of clinical adverse events are presented in Supplemental Table 3. The most common adverse event was nausea, with no significant difference between the groups. Abdominal distension was significantly more frequent in the 2 L PEG/Asc group, whereas headache was more common in the 1 L PEG/Asc group. No other adverse events significantly differed between the groups. All adverse events resolved by the end of the study, and none led to discontinuation of the trial.
Discussion
In this multi-center, prospective, randomized, non-inferiority trial, 1 L PEG/Asc was non-inferior to 2 L PEG/Asc for bowel preparation in patients with hematochezia. The rate of adequate bowel preparation, defined as a total BBPS score ⩾6 with all segmental scores ⩾2, was comparable between the two groups and met the prespecified non-inferiority margin. Furthermore, the 1 L PEG/Asc group showed higher mean total BBPS and HCS scores than the 2 L PEG/Asc group, with statistically significant differences observed in certain segments, including the transverse, descending, and sigmoid colon. The colonic bleeding source identification rate, reflecting mucosal visibility, was also higher in the 1 L PEG/Asc group than in the 2 L PEG/Asc group (82.4% vs 73.6%), although the difference was not statistically significant (p = 0.165). This observation suggests that enhanced bowel cleanliness with the 1 L PEG/Asc regimen may facilitate clearer visualization and identification of bleeding sources during colonoscopy. These findings suggest that 1 L PEG/Asc not only provides bowel cleansing efficacy equivalent to the standard 2 L PEG/Asc regimen but may also offer superior cleansing in specific segments, supporting its role as an effective alternative for bowel preparation prior to colonoscopy in patients with hematochezia.
The ultra-low-volume preparation, 1 L PEG/Asc, may offer significant clinical benefits in patients with hematochezia. These patients often require prompt colonoscopy; thus, reducing the volume of bowel preparation can improve patient compliance, shorten preparation time, and minimize discomfort associated with large-volume intake, thereby enabling faster and more effective examinations.12,13 Indeed, Sun et al. 21 found that, in hospitalized patients, the use of low-volume bowel preparation maintained cleansing efficacy while reducing preparation time and contributing to shorter hospital stays compared with high-volume regimens. Furthermore, hematochezia is recognized as an independent risk factor for inadequate bowel preparation, highlighting the need for low-volume regimens that can ensure both cleansing efficacy and patient tolerability in this population. 15 Previous studies on acute lower gastrointestinal bleeding have primarily focused on the timing of colonoscopy or traditional high-volume PEG-based preparations, with limited direct evidence supporting low-volume regimens.22–24 This study is the first prospective, randomized controlled trial to directly compare 1 and 2 L PEG/Asc in patients with hematochezia, demonstrating the practicality and clinical value of ultra-low-volume bowel preparation with 1 L PEG/Asc and providing important evidence to support its future clinical application.
The 1 L PEG/Asc regimen demonstrated significantly higher bowel cleansing scores in the transverse and left colon compared to the 2 L PEG/Asc regimen. This may be attributable to the characteristics of the 1 L PEG/Asc formulation, as suggested by previous studies.9,18 First, the lower volume is associated with better patient compliance and reduced discomfort or gastrointestinal distress during ingestion, which may facilitate smoother colonic transit. Second, to maintain equivalent cleansing efficacy, 1 L PEG/Asc contains a higher concentration of ascorbic acid compared to the 2 L PEG/Asc formulation. This higher ascorbic acid concentration enhances osmotic activity, synergizing with PEG to draw more water into the bowel lumen, thereby softening stool and improving cleansing efficacy. The left colon is particularly challenging to cleanse due to slower motility and prolonged stool stasis, and thus, the enhanced osmotic effect of ascorbic acid may be particularly beneficial in this segment. The improved scores in the transverse colon may similarly be attributable to the increased compliance and reduced transit time associated with the lower-volume regimen. These results suggest that 1 L PEG/Asc may be advantageous for achieving effective bowel cleansing in patients with hematochezia.
This prospective randomized trial systematically evaluated the safety and adverse event profiles of 1 and 2 L PEG/Asc in patients with hematochezia. No serious adverse events were observed in either group, and there were no clinically significant changes in key laboratory parameters, including electrolyte levels or renal function. Although there are concerns about bowel preparation in patients with hematochezia, including discomfort during ingestion and the potential risk of rebleeding, both regimens demonstrated high completion rates and good tolerability in this study, with most adverse events being mild and fully reversible. These findings indicate that both the 1 and 2 L PEG/Asc regimens can provide not only convenience but also excellent safety in patients with hematochezia, offering important evidence to support the clinical applicability and safety of ultra-low-volume and low-volume bowel preparations in this population, where data have been limited.
There are several strengths to this study. First, this is the first multi-center, prospective, randomized controlled trial to evaluate the efficacy and safety of an ultra-low-volume 1 L PEG/Asc regimen in patients with hematochezia. Unlike previous studies on low- and ultra-low-volume PEG preparations, which have primarily been conducted in asymptomatic patients undergoing elective colonoscopy, this study demonstrated the efficacy and safety of ultra-low-volume bowel preparation specifically in patients presenting with hematochezia, underscoring its novelty. Second, bowel cleansing was rigorously assessed using both the BBPS and HCS, and we comprehensively evaluated completion rates, patient acceptability, adverse events, and laboratory changes, providing clinically applicable evidence for real-world practice.
However, several limitations should also be noted. First, the study was conducted at two centers in Korea, which may limit the generalizability of its findings to populations with different dietary habits or ethnic characteristics. Second, the study included predominantly hemodynamically stable patients with hematochezia (approximately 83% stable vs 17% unstable), which may limit the extrapolation of the results to those with massive bleeding or significant hemodynamic instability. Caution should therefore be exercised when generalizing these findings to patients presenting with active massive bleeding. Third, although the study employed a single-blind (endoscopist-blinded) design, patients were aware of their assigned bowel preparation regimen because of differences in preparation volume. This may have introduced reporting bias in patient-reported outcomes, such as taste and satisfaction, which should be taken into account when interpreting these findings.
Conclusion
In conclusion, this study is the first prospective, multi-center, randomized trial to demonstrate that ultra-low-volume 1 L PEG/Asc is non-inferior to low-volume 2 L PEG/Asc in terms of bowel cleansing efficacy among patients with hematochezia, with superior cleansing in certain segments, as well as high compliance and favorable safety. These findings suggest that 1 L PEG/Asc may serve as an effective and safe alternative for bowel preparation prior to colonoscopy in patients with hematochezia and could provide an important basis for the development of future clinical guidelines on bowel preparation in this population.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261438238 – Supplemental material for 1 versus 2 L PEG/Asc for bowel preparation in patients with hematochezia: a multicenter, prospective, randomized, non-inferiority study
Supplemental material, sj-docx-1-tag-10.1177_17562848261438238 for 1 versus 2 L PEG/Asc for bowel preparation in patients with hematochezia: a multicenter, prospective, randomized, non-inferiority study by Su Young Kim, Sang Hoon Lee, Jundeok Lee, Hyunil Kim, Hong Jun Park and Hyun-Soo Kim in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848261438238 – Supplemental material for 1 versus 2 L PEG/Asc for bowel preparation in patients with hematochezia: a multicenter, prospective, randomized, non-inferiority study
Supplemental material, sj-docx-2-tag-10.1177_17562848261438238 for 1 versus 2 L PEG/Asc for bowel preparation in patients with hematochezia: a multicenter, prospective, randomized, non-inferiority study by Su Young Kim, Sang Hoon Lee, Jundeok Lee, Hyunil Kim, Hong Jun Park and Hyun-Soo Kim in Therapeutic Advances in Gastroenterology
