Abstract
Background:
High-volume polyethylene glycol (PEG) solutions are commonly used for bowel preparation but are often poorly tolerated, reducing patient compliance. Shouhui Tongbian Capsules (SHTBC), a traditional Chinese medicine known to promote gastrointestinal motility, may offer an alternative approach. However, its role in bowel preparation remains unclear.
Objectives:
To evaluate the efficacy, safety, and tolerability of a novel bowel preparation regimen combining SHTBC with low-volume PEG (2L) compared to conventional high-volume PEG (3L).
Design:
Multicenter, randomized, single-blind, parallel-controlled trial.
Methods:
A total of 404 participants scheduled for colonoscopy across 34 medical centers in China were randomized into two groups: the experimental group (SHTBC + 2L PEG, n = 202) and the control group (3L PEG, n = 202). Bowel preparation quality was assessed using the Boston Bowel Preparation Scale (BBPS), with successful cleansing defined as a total BBPS score ⩾6. The secondary outcomes included time to adequate bowel movement, number of bowel movements, patient tolerance, acceptance, and incidence of adverse drug reactions (ADRs).
Results:
The success rate of bowel preparation was comparable between groups (p = 0.7454). The experimental group had a slightly longer time to first adequate bowel movement on the day of colonoscopy (p = 0.0013) but experienced fewer bowel movements the day before (p < 0.0001). The experimental group reported significantly fewer ADRs (p = 0.0311) and better tolerance, including reduced bloating, nausea, and sleep disturbance (p < 0.01 for all). Patient acceptance was higher in the experimental group (92.89% vs 88.32%), although the difference was not statistically significant (p = 0.4170).
Conclusion:
SHTBC combined with 2L PEG is a safe, effective, and better-tolerated alternative to 3L PEG for bowel preparation before colonoscopy, offering a promising strategy to improve patient compliance.
Trial registration:
Chinese Clinical Trial Registry: ChiCTR2300069962.
Introduction
Colorectal cancer (CRC) is the third most common cancer globally. 1 In China, gastrointestinal tumors represent the most prevalent category of malignant tumors, accounting for half of all cancer cases. Among these, CRC ranks second in overall cancer incidence. 2 Colonoscopy is a cornerstone for CRC diagnosis and management, with adequate bowel preparation essential to ensure its efficacy, safety, and diagnostic accuracy. 3 Optimal bowel preparation should efficiently empty the intestines without altering colonic mucosal histology or disturbing water and electrolyte balance. The Chinese guidelines recommend 2–3 L of polyethylene glycol (PEG) administered in single or split doses, 4 whereas European and U.S. guidelines advocate for 4 L of PEG in divided doses.5,6 Both regimens are effective, but the substantial volume and unpleasant taste of PEG solutions often result in adverse effects, including nausea, vomiting, bloating, and abdominal pain. In addition, 15%–35% of patients experience inadequate bowel preparation.7,8 These drawbacks can reduce patient compliance and discourage future participation in CRC screening programs, emphasizing the need for more tolerable and patient-friendly alternatives.
Shouhui Tongbian Capsules (SHTBC) are widely recognized in China for treating functional constipation. A meta-analysis demonstrated that SHTBC significantly enhanced treatment efficacy, alleviated clinical symptoms, and reduced adverse reactions. 9 The formulation includes eight traditional Chinese medicines (TCM), Polygoni Multiflori Radix (Polygonum multiflorum Thunb.), aloe (Aloe barbadensis Mill.), Cassiae Semen (Cassia obtusifolia L.), Lycii Fructus (Lycium barbarum L.), Ginseng Radix et Rhizoma (Panax ginseng C. A. Mey.), Atractylodis Macrocephalae Rhizoma (Atractylodes macrocephala Koidz.), Aurantii Fructus Immaturus (Citrus aurantium L.), and Asini Corii Colla (Equus asinus L.). 10 These components contribute to the therapeutic efficacy of SHTBC. 10 Preclinical studies have shown that SHTBC promotes bowel motility by modulating the gut microbiota composition, enhancing serotonin (5-HT) release, and promoting enteric neuron differentiation.11–13 It has also been shown to improve the intestinal barrier function, increase fecal water content, and reduce inflammation.14,15 These mechanisms provide a theoretical basis for the adjunctive use of SHTBC in bowel preparation regimens.
Given the similar effects of SHTBC and PEG in promoting intestinal motility, we hypothesized that their combination could provide a novel approach for bowel preparation. By leveraging the complementary actions of SHTBC, this combination can reduce the required PEG dose, thereby minimizing its adverse effects. This study was designed to evaluate whether combining SHTBC with PEG can achieve effective bowel cleansing while enhancing patient tolerance and compliance. Recognizing the challenges associated with current bowel preparation protocols, this multicenter, randomized, single-blind, parallel-controlled trial aimed to assess the efficacy, safety, and patient tolerance of combining SHTBC with a low-volume PEG solution (2L) compared with the conventional high-volume PEG regimen (3L). By integrating a reduced-volume lavage solution with the therapeutic benefits of SHTBC, this approach offers a more patient-friendly alternative that has the potential to transform the standard bowel preparation protocols for colonoscopy.
Materials and methods
Study design and ethical issues
This multicenter, randomized, single-blind, parallel-controlled trial was conducted at 34 medical centers in China. The patients and the public were not involved in the design, conduct, reporting, or dissemination plans of this study. This clinical trial was registered in the Chinese Clinical Trial Registry (ChiCTR2300069962). The clinical trial protocol is available in the Supplemental Material. The study adhered to the principles of Good Clinical Practice, Declaration of Helsinki, and all relevant national regulatory requirements. The trial was reported following the Consolidated Standards of Reporting Trials (CONSORT) guidelines for reporting parallel group, randomized clinical trials. 16 All authors had access to the study data and reviewed and approved the final manuscript. All patient information was fully de-identified prior to data analysis and reporting to ensure participant confidentiality.
Participants
Eligible participants were adults aged 18–75 years who were scheduled to undergo colonoscopy for medical reasons or routine screening. All participants were of Han Chinese ethnicity, the predominant ethnic group in China, accounting for over 91% of the national population. 17 Exclusion criteria included severe cardiovascular or cerebrovascular conditions, Parkinson’s disease, spinal cord injuries, or significant hepatic or renal dysfunction, defined as Child-Pugh class C or estimated glomerular filtration rate <30 mL/min/1.73 m2, respectively. Severe cardiac dysfunction was defined as New York Heart Association class III or IV. Individuals with intolerance or allergies to trial medications, prior colorectal surgery, or gastrointestinal tumors were excluded. Patients with intestinal obstruction, perforation, gastric retention, gastrointestinal bleeding, toxic enteritis, toxic megacolon, or Crohn’s disease were excluded. Female participants who were pregnant, planning pregnancy, or breastfed were also excluded. Additional exclusion criteria included psychiatric disorders affecting compliance, HIV, or syphilis positivity, recent use of medications affecting gastrointestinal motility (e.g., laxatives, tricyclic antidepressants, anesthetics) within 7 days, participation in other clinical trials in the past month, or any conditions considered unsuitable by investigators. Constipation status was determined based on self-reported symptoms captured in the baseline health questionnaire. Participants were considered to have constipation if they reported a reduced bowel movement frequency, straining, or sensation of incomplete evacuation.
Randomization and blinding
Eligible participants were enrolled consecutively at each center. Randomization was performed in ascending order based on screening numbers. Participants were randomly assigned in a 1:1 ratio to the intervention group (SHTBC combined with 2L PEG) or the control group (standard 3L PEG). The randomization sequence was computer-generated using block randomization to ensure a balanced group allocation of the study sites. Although blinding participants to treatment allocation was not feasible owing to the physical characteristics of bowel preparation regimens, the endoscopists performing the colonoscopies were blinded to maintain the objectivity of efficacy assessments.
Study procedures
Participants adhered to a pre-colonoscopy diet, which included a low-residue breakfast and lunch, followed by a liquid-only dinner the day before the procedure. Two bowel preparation regimens were tested: (1) the SHTBC + 2L PEG intervention and (2) the conventional 3L PEG regimen. The experimental group received SHTBC (0.35 g per capsule; Lunan Hope Pharmaceutical Co., Ltd., Linyi, Shandong Province, China) and a 2L PEG electrolyte solution (Shutaiqing®; Staidson (Beijing) Biopharmaceuticals Co., Ltd., Beijing, China). SHTBC was administered as three capsules after each meal the day before the colonoscopy, with the final dose taken by 18:00. On the day of the procedure, participants in this group consumed 2L of PEG solution 4–6 h before the procedure, prepared by dissolving 16 packets each of part A (13.125 g PEG 4000) and part B (containing 0.1785 g sodium bicarbonate, 0.3507 g sodium chloride, and 0.0466 g potassium chloride) in warm water. They drank 250 mL every 10–15 min, completing the solution within 2 h. The control group consumed a 1L PEG solution the evening before the colonoscopy, prepared with 8 packets each of parts A and B, following the same ingestion rate. On the day of the procedure, they consumed an additional 2L PEG solution following the intervention group’s protocol. Participants administered the preparations at home, with adherence monitored using a pill-counting method.
Efficacy assessment
The primary efficacy measure was bowel cleanliness, assessed using the Boston Bowel Preparation Scale (BBPS). 18 It evaluates three segments of the colon: the right side (cecum and ascending colon), the transverse colon, and the left side (descending colon, sigmoid colon, and rectum). Each segment is scored from 0 (unprepared colon) to 3 (perfectly clean colon), with a total score ranging from 0 to 9. Successful bowel preparation was defined as a BBPS score ⩾6, indicating adequate cleansing for colonoscopic visualization. Secondary outcomes included the time to adequate bowel movement (passage of clear, watery stool) and the total number of bowel movements experienced during the preparation period. Bowel movement frequency was self-reported using a standardized log. Bowel movement was defined as the active expulsion of a noticeable amount of stool (formed or watery), excluding minor events, such as gas or minimal discharge.
Safety evaluation
Safety assessments focused on the incidence and severity of adverse drug reactions (ADRs) recorded from the administration of the study medications until the completion of colonoscopy. ADRs were classified according to the Chinese guidelines. These guidelines categorize ADRs as definitively related, very likely related, possibly related, unlikely related, pending evaluation, or unassessable. The severity of ADRs was graded using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. 19 The CTCAE grades range from 1 (mild) to 5 (death). To complement ADR monitoring, serum electrolyte levels and renal function were assessed before and after preparation.
Patient tolerance and acceptance
Tolerance to bowel preparation regimens was the second primary outcome measure in this study. Tolerance was evaluated based on the severity of the discomfort symptoms, including abdominal pain, bloating, nausea, vomiting, sleep disturbance, abdominal cramps, and anal discomfort. Each symptom was rated on a scale of 0 (none) to 4 (very severe), and the total tolerance score was calculated as the sum of these items (range: 0–28), with lower scores indicating better overall tolerance. Patient acceptance was assessed by asking participants about their willingness to repeat the same regimen for future colonoscopies, categorized as “unacceptable,” “difficult to accept,” or “acceptable.”
Sample size calculation
The sample size calculation was based on the bowel preparation efficacy measured using the BBPS. Using our previous data, we anticipated mean BBPS scores of 8 ± 2.89 for the SHTBC + 2L PEG group and 7.09 ± 2.89 for the 3L PEG control group. 20 To detect a statistically significant difference with 80% power and a two-sided α level of 0.05, a total of 320 participants were required (160 participants per group). Accounting for a 20% dropout rate, the final sample size was 404 participants.
Statistical analysis
Efficacy analyses were conducted using the full analysis set (FAS) and per-protocol set (PPS). Safety analyses were based on the safety set (SS), which comprised participants who received at least one dose of the study medication. Continuous variables were summarized as means and standard deviations and compared using t tests. Categorical data are presented as frequencies and percentages, and comparisons were made using chi-square or Fisher’s exact tests, as appropriate. All statistical tests were two-sided, and statistical significance was set at p < 0.05. Analyses were performed using the SAS software (version 9.4, SAS Institute Inc., Cary, NC, USA).
Results
Participant characteristics
From March 31, 2023, to November 23, 2023, a total of 426 participants were assessed for eligibility, with 22 excluded (17 not meeting criteria and 5 withdrawing consent). The remaining 404 participants were randomized equally into 2 groups: SHTBC + 2L PEG (n = 202) and standard 3L PEG (n = 202). The FAS included 201 and 198 participants, respectively, while the PPS included 185 and 195 participants, with exclusions due to missing data, medication non-adherence, or protocol violations. The details are shown in Figure 1. No significant differences between the two groups were observed in baseline demographic and clinical characteristics (Table 1).

Study flow diagram summarizing the enrollment, allocation, follow-up, and analysis processes.
Demographic data of participants in the full analysis set.
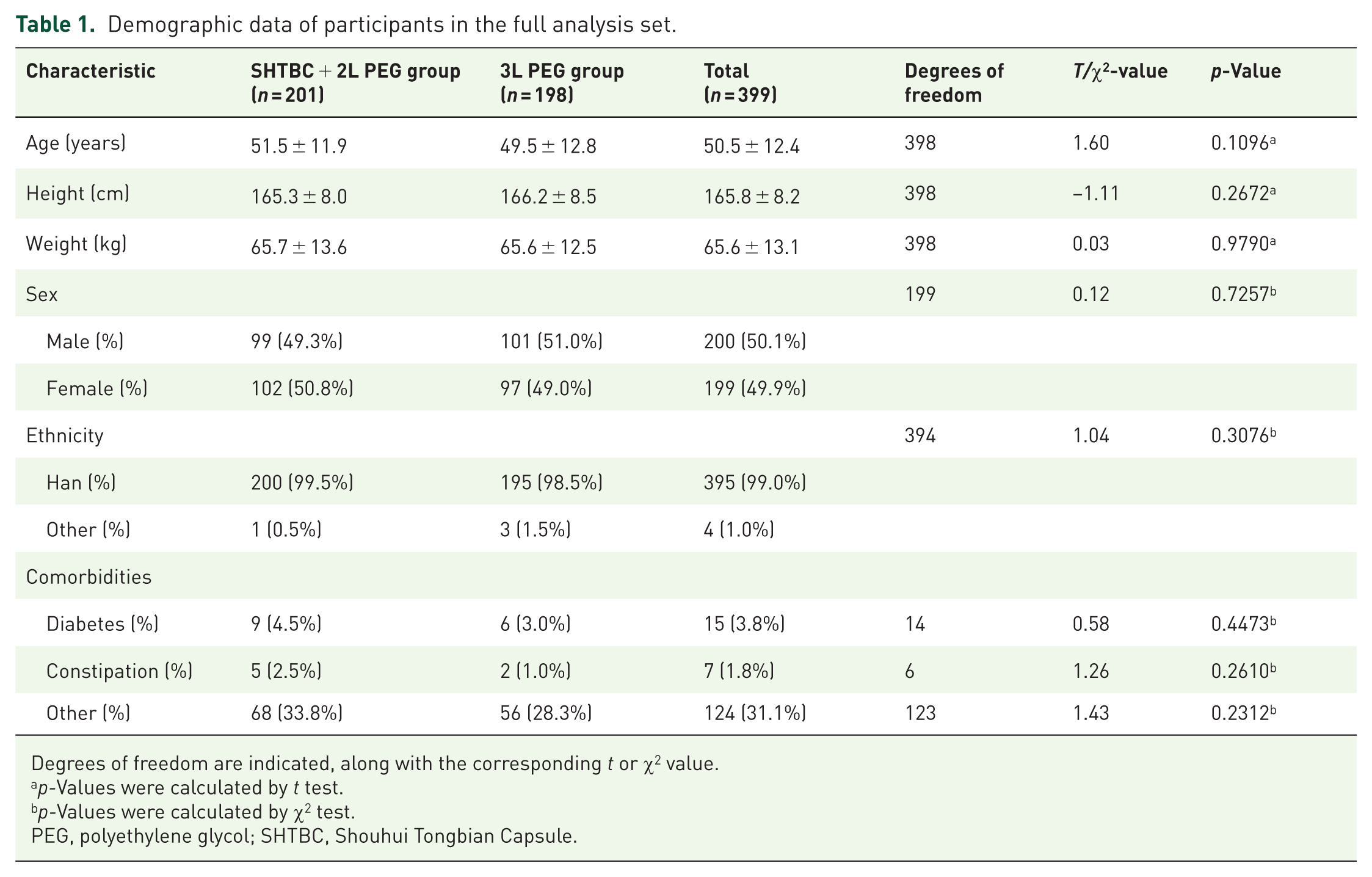
Degrees of freedom are indicated, along with the corresponding t or χ2 value.
p-Values were calculated by t test.
p-Values were calculated by χ2 test.
PEG, polyethylene glycol; SHTBC, Shouhui Tongbian Capsule.
BBPS scores and bowel preparation success rate
In FAS, the mean BBPS scores were similar between the two groups: 7.1 ± 1.39 in the SHTBC + 2L PEG group and 7.1 ± 1.24 in the 3L PEG group (p = 0.7454; Figure 2(a), Table 2). The bowel preparation success rates, as measured by the BBPS, were comparable between the SHTBC + 2L PEG group (87.88%, 174/198) and the 3L PEG group (90.91%, 180/198; mean difference: −3.03%, 95% confidence interval: −9.12% to 3.06%, χ2 = 0.96, p = 0.3275; Figure 2(b), Supplemental Table 1). PPS analysis confirmed the FAS analysis (Figure 2(a) and (b), Table 2, Supplemental Table 1).
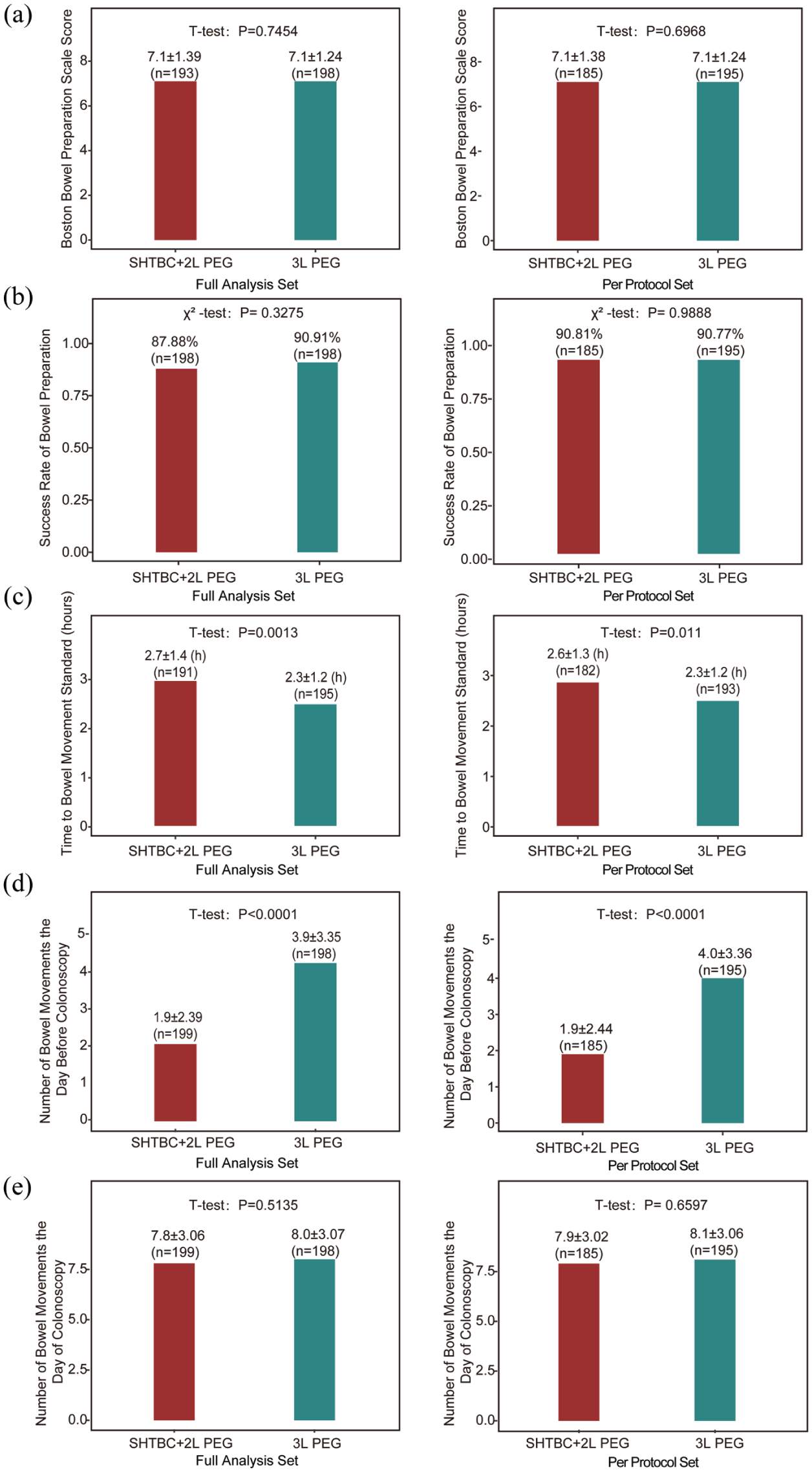
Efficacy evaluation of bowel preparation regimens between the SHTBC + 2L PEG group and the 3L PEG group in the full analysis set and per-protocol set. (a) BBPS scores for the SHTBC + 2L PEG group compared to the 3L PEG group. (b) Success rate of bowel preparation (BBPS ⩾6) for the SHTBC + 2L PEG group versus the 3L PEG group. (c) Time to achieve adequate bowel movement standards (hours) on the day of colonoscopy in the SHTBC + 2L PEG and 3L PEG groups. (d) Number of bowel movements on the day before the colonoscopy for the SHTBC + 2L PEG group compared to the 3L PEG group. (e) Number of bowel movements on the day of the colonoscopy for the SHTBC + 2L PEG and 3L PEG groups.
The Boston Bowel Preparation Scale Scores in the FAS and PPS analyses.
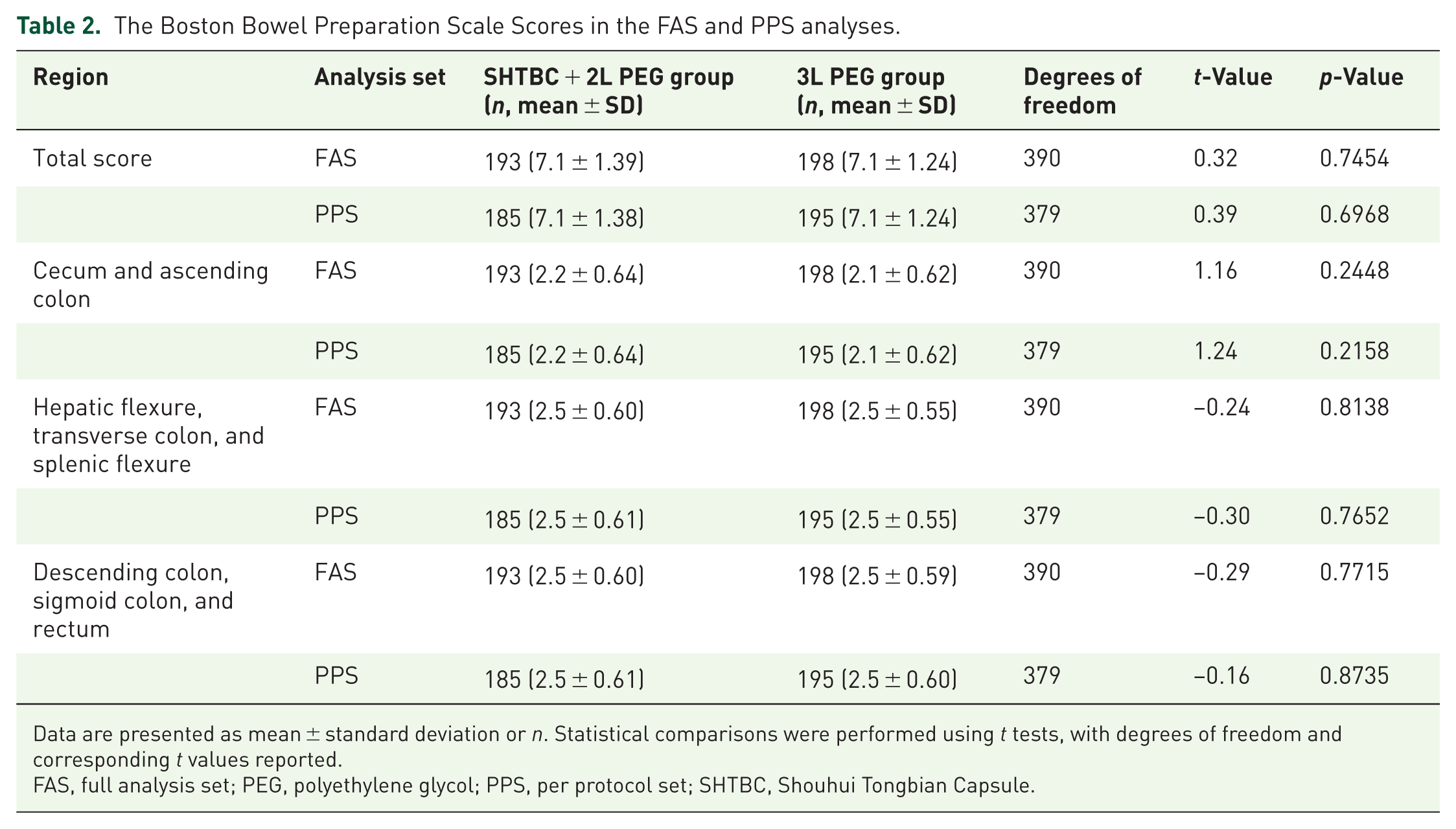
Data are presented as mean ± standard deviation or n. Statistical comparisons were performed using t tests, with degrees of freedom and corresponding t values reported.
FAS, full analysis set; PEG, polyethylene glycol; PPS, per protocol set; SHTBC, Shouhui Tongbian Capsule.
Time to adequate bowel movements after starting PEG solution on the day of the colonoscopy
In FAS, the mean time to achieve adequate bowel movement was slightly longer in the SHTBC + 2L PEG group (2.7 ± 1.42 h) than in the 3L PEG group (2.3 ± 1.19 h, p = 0.0013; Figure 2(c)). The PPS analysis yielded similar results (2.6 ± 1.32 h for the SHTBC + 2L PEG group, 2.3 ± 1.20 h for the 3L PEG group, p = 0.0110; Figure 2(c)).
Number of bowel movements during the preparation period
The FAS analysis revealed that the day before the colonoscopy, participants in the SHTBC + 2L PEG group had fewer bowel movements (1.9 ± 2.39) than those in the 3L PEG group (3.9 ± 3.35, p < 0.0001; Figure 2(d)). On the day of colonoscopy, the number of bowel movements was similar between the groups (SHTBC + 2L PEG: 7.8 ± 3.06; 3L PEG: 8.0 ± 3.07, p = 0.5135; Figure 2(e)). These results were consistent with the PPS analysis (Figure 2(d) and (e)).
Safety evaluation
The safety analysis conducted on the SS revealed a significantly lower incidence of ADRs in the SHTBC + 2L PEG group (36.82%) than in the 3L PEG group (47.47%; p = 0.0311). In the SHTBC + 2L PEG group, the most reported ADRs were nausea (16.92%), bloating (15.92%), and vomiting (13.43%). By contrast, the 3L PEG group had higher frequencies of bloating (31.31%), nausea (28.28%), poor sleep quality (15.06%), vomiting (12.12%), and anorectal discomfort (12.12%). All ADRs were classified as Grade 1 (mild). Notably, the SHTBC + 2L PEG group demonstrated significantly lower rates of nausea, bloating, and poor sleep quality than the 3L PEG group. The detailed results are summarized in Table 3. Laboratory indicators of renal function and electrolyte balance were evaluated before and after bowel preparation. Baseline values were comparable between groups (Supplemental Table 2), and the proportions of patients with normal clinical classifications were similar at baseline (Supplemental Table 4). No clinically meaningful changes were observed in blood urea nitrogen, serum creatinine, Na+, Cl−, or Ca²+. Although potassium levels decreased significantly in the SHTBC + 2L PEG group (p = 0.0021), they remained within normal limits (Supplemental Table 3). Post-preparation clinical classifications also showed no significant between-group differences (Supplemental Table 5). No serious procedure-related complications, such as perforation, gastrointestinal bleeding, or severe allergic reactions, were reported in either group during or within 24 h after colonoscopy.
Adverse drug reactions.

p Values were calculated by χ² test.
p Values were calculated by Fisher’s exact test.
PEG, polyethylene glycol; SHTBC, Shouhui Tongbian Capsule.
Bowel preparation tolerance
On the day before the colonoscopy, the FAS analysis showed that participants in the SHTBC + 2L PEG group reported significantly lower tolerance scores (0.4 ± 0.96) compared to those in the 3L PEG group (1.31 ± 1.95), indicating better tolerance (p < 0.0001; Figure 3(a)). As shown in Figure 4, the SHTBC + 2L PEG group also had significantly lower scores for bloating (0.1 ± 0.37 vs 0.3 ± 0.56, p < 0.0001), nausea (0.1 ± 0.34 vs 0.3 ± 0.60, p < 0.0001), vomiting (0.0 ± 0.16 vs 0.1 ± 0.47, p = 0.0017), sleep disturbance (0.0 ± 0.20 vs 0.2 ± 0.61, p = 0.0002), and anal discomfort (0.1 ± 0.25 vs 0.1 ± 0.38, p = 0.0425). The scores for abdominal pain and cramping were similar between the two groups (p > 0.05). On the colonoscopy day, the two groups had similar tolerance scores (0.7 ± 1.42 for the SHTBC + 2L PEG group vs 0.6 ± 1.36 for the 3L PEG group, p = 0.7906; Figure 3(b)).

Tolerance and acceptance evaluation of bowel preparation regimens between the SHTBC + 2L PEG group and the 3L PEG group in the full analysis set and per-protocol set. (a) Bowel preparation tolerance scores on the day before the colonoscopy. (b) Bowel preparation tolerance scores on the day of the colonoscopy. (c) Overall bowel preparation tolerance scores. (d) Acceptance rates of the bowel preparation regimen.

Comparison of discomfort symptoms on the day before the colonoscopy between the SHTBC + 2L PEG group and the 3L PEG group. Symptoms evaluated include bloating, nausea, vomiting, sleep disturbance, and anal discomfort in the FAS (a) and PPS (b). Scores represent mean ± standard deviation. Statistical comparisons were performed using t tests.
The overall tolerance score, combining the day before and the day of colonoscopy, was significantly better in the SHTBC + 2L PEG group (1.1 ± 1.86) than in the 3L PEG group (1.9 ± 2.89, p = 0.0007; Figure 3(c)). The SHTBC + 2L PEG group also had lower scores for bloating (0.2 ± 0.56 vs 0.5 ± 0.80, p = 0.0005), nausea (0.3 ± 0.69 vs 0.5 ± 0.99, p = 0.0063), and sleep disturbance (0.1 ± 0.30 vs 0.3 ± 0.83, p = 0.0011). Scores for abdominal pain, vomiting, and cramps were similar between the groups (p > 0.05).
The PPS analysis confirmed these findings (Figure 3(a)–(c)). Supplemental Table 6 provides additional details.
Acceptance of the bowel preparation regimen
In the FAS analysis, 183 participants (92.89%) in the SHTBC + 2L PEG group found the bowel preparation regimen acceptable, 11 participants (5.58%) found it difficult to accept, and 3 participants (1.52%) could not accept it. In the 3L PEG group, 174 participants (88.32%) found the regimen acceptable, 20 participants (10.15%) found it difficult to accept, and 3 participants (1.52%) could not accept it. Although the acceptance rate was higher in the SHTBC + 2L PEG group than in the 3L PEG group, the difference was not statistically significant (χ2 = 2.84, p = 0.4170; Figure 3(d), Supplemental Table 7). The PPS analysis yielded similar results (Figure 3(d) and Supplemental Table 7).
Discussion
Inadequate bowel preparation significantly increases the risk of missed lesions during colonoscopy, compromising diagnostic accuracy and clinical outcomes. 21 It also imposes additional burdens on patients, often requiring repeat procedures, 22 which leads to inconvenience, increased healthcare costs, and higher resource utilization. Known risk factors for inadequate bowel preparation include the absence of split-dose regimens and poor patient tolerance to current protocols. 23 This highlights the critical need for strategies that ensure effective cleansing while minimizing adverse effects and improving compliance.
The selection of 3L PEG as the control regimen aligns with the current clinical practice in China. According to the 2023 Consensus on Bowel Preparation for Colonoscopy (Guangzhou), 3–4 L PEG in split doses is standard, 4 particularly for patients at risk of inadequate preparation. This approach also reflects international guidelines, including those from Europe and the United States, which recommend 4L PEG regimens in divided doses.5,6 Recent updates from the U.S. The Multi-Society Task Force on Colorectal Cancer has emphasized the feasibility of lower-volume regimens in selected patients. 24
Although 2L PEG is considered sufficient in some Asian populations, evidence indicates that it may be inadequate in certain subgroups, such as older adults or individuals with slow colonic transit. Studies have reported that 15%–35% of patients may still experience inadequate cleansing with standard PEG doses.7,8 In our trial, only 2% of participants had a history of constipation, suggesting preserved bowel motility in the study population. This may have attenuated the observable pro-motility effect of SHTBC and partially contributed to the cleansing efficacy of the 2L PEG regimen alone. Nevertheless, the combination of SHTBC and 2L PEG achieved a cleansing efficacy comparable to that of 3L PEG, while significantly improving patient tolerance and reducing adverse effects such as bloating, nausea, vomiting, and sleep disturbances. These findings support the potential of SHTBC as an effective adjunct for improving the quality and acceptability of bowel preparation.
This multicenter randomized trial showed that SHTBC combined with 2L PEG achieved cleansing efficacy comparable to 3L PEG, based on similar BBPS scores. In addition, the SHTBC + 2L PEG group reported fewer adverse effects, including bloating, nausea, vomiting, and sleep disturbances. Improved tolerability may enhance adherence to bowel preparation protocols, improve colonoscopy success rates, and facilitate the early detection of colorectal abnormalities.
Several preclinical studies have provided mechanistic support for the gastrointestinal benefits of SHTBC. Bai et al. 11 demonstrated that SHTBC modulates the gut microbiota–5-HT–intestinal motility axis to alleviate constipation, while Lin et al. 12 showed that it enhances short-chain fatty acid production and flora diversity in loperamide-induced models. These findings support the potential of SHTBC to promote evacuation and improve bowel preparation. Our clinical results align with these mechanistic insights, validating the translational relevance of SHTBC in bowel preparation regimens.
The sample size was determined based on the anticipated differences in BBPS scores. A total of 404 participants (202 per group) met the predefined power requirement for detecting statistically meaningful group differences. This finding supports the reliability of the efficacy and safety conclusions derived from this study.
This trial provides the groundwork for future investigation. Subsequent studies should target high-risk populations, such as those with chronic constipation or impaired bowel motility, and explore the use of SHTBC with PEG volumes below 2L to evaluate its role in ultralow-volume regimens. Additional study is needed to determine the optimal SHTBC dose and to assess outcomes in repeat colonoscopy settings. Due to the small number of constipated patients enrolled in each group (three in the 3L PEG group and three in the SHTBC + 2L PEG group), stratified analysis based on constipation status was not performed, as it would have introduced statistical bias. This limits the ability to evaluate the differential effects of SHTBC in patients with impaired bowel motility. Future studies should predefine subgroup analyses in larger samples enriched for constipated or bowel preparation-challenged populations to better assess the adjunctive benefits of SHTBC.
Strengths include the multicenter design, enhancing generalizability, and the use of both FAS and PPS analyses, which strengthen the outcome validity. However, this study has the following limitations: (1) a single-blind design may have biased the tolerance reporting; (2) the exclusion of patients with significant gastrointestinal comorbidities and specific comorbidities limits their external validity. Future studies should include patients with complex gastrointestinal conditions to better evaluate the efficacy and safety of SHTBC; (3) the population consisted entirely of Han Chinese outpatients, most of whom were relatively healthy, potentially restricting the generalizability to other ethnic or older populations, and patients with chronic conditions; (4) constipation was self-reported without standardized tools (e.g., Rome IV, 25 Patient Assessment of Constipation Symptoms (PAC-SYM), 26 or Bristol Stool Form Scale 27 ), potentially leading to misclassification. Standardized definitions and validated tools should be used in future research to more accurately characterize the baseline bowel function and its impact on preparation quality. Likewise, stool frequency data relied on patient self-reporting, which may have introduced recall or interpretive bias, particularly during frequent defecation episodes. Although laxative use was restricted to within 7 days before enrollment, detailed histories of chronic use were not collected. Future trials should apply validated tools (e.g., the Bristol Stool Form Scale) and document long-term laxative use and analyze subgroups with functional constipation or laxative dependence to better characterize baseline bowel function and account for real-world variability and confounding factors; (5) colonoscopy performance metrics, such as insertion and withdrawal times and adenoma detection rates, were not uniformly collected, limiting insights into procedural quality. Future trials should incorporate these endpoints to provide a more comprehensive evaluation; and (6) although the split-dose PEG regimen aligns with global standards, SHTBC lacks international regulatory approval, which may limit its adoption outside China.
Conclusion
This study demonstrates that combining the TCM SHTBC with a reduced-volume PEG solution is an effective, safe, and well-tolerated alternative to the conventional high-volume PEG regimen for bowel preparation before colonoscopy. The improved patient tolerance and reduced incidence of adverse reactions associated with SHTBC highlight its potential to enhance patient compliance with colonoscopy screening protocols. These findings provide valuable insights for developing innovative bowel preparation strategies in clinical practice, incorporating low-volume PEG regimens supplemented with adjunctive medications such as SHTBC.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251378043 – Supplemental material for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial
Supplemental material, sj-docx-1-tag-10.1177_17562848251378043 for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial by Feng Ye, Ni Guo, Yaping Wang, Baochun Wang, Chunyan Niu, Zujin Ji, Ming Guo, Yilin Hou, Yi Jian, Jianhua Cui, Dazhi Chen, Shuhui Li, Dongxia Wang, Hongping Guo, Junlei Jiu, Chunlei Qiu, Hui Cao, Ying He, Ming Chen, Hua Li, Xuezhi Xin, Rongjuan Zheng, Wei Chen, Dongya Chen, Lingyun Niu, Zhenqin Cui, Zhanquan Zou, Bin Jia, Leyao Zhang, Yinming Bai, Wanyu Li, Xiaojun Yang, Min Hua, Shaoming Long, Guoliang Cheng, Zhiyan Huang, Enhua Yang, Jinjin Shi and Guoxin Zhang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251378043 – Supplemental material for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial
Supplemental material, sj-docx-2-tag-10.1177_17562848251378043 for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial by Feng Ye, Ni Guo, Yaping Wang, Baochun Wang, Chunyan Niu, Zujin Ji, Ming Guo, Yilin Hou, Yi Jian, Jianhua Cui, Dazhi Chen, Shuhui Li, Dongxia Wang, Hongping Guo, Junlei Jiu, Chunlei Qiu, Hui Cao, Ying He, Ming Chen, Hua Li, Xuezhi Xin, Rongjuan Zheng, Wei Chen, Dongya Chen, Lingyun Niu, Zhenqin Cui, Zhanquan Zou, Bin Jia, Leyao Zhang, Yinming Bai, Wanyu Li, Xiaojun Yang, Min Hua, Shaoming Long, Guoliang Cheng, Zhiyan Huang, Enhua Yang, Jinjin Shi and Guoxin Zhang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848251378043 – Supplemental material for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial
Supplemental material, sj-docx-3-tag-10.1177_17562848251378043 for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial by Feng Ye, Ni Guo, Yaping Wang, Baochun Wang, Chunyan Niu, Zujin Ji, Ming Guo, Yilin Hou, Yi Jian, Jianhua Cui, Dazhi Chen, Shuhui Li, Dongxia Wang, Hongping Guo, Junlei Jiu, Chunlei Qiu, Hui Cao, Ying He, Ming Chen, Hua Li, Xuezhi Xin, Rongjuan Zheng, Wei Chen, Dongya Chen, Lingyun Niu, Zhenqin Cui, Zhanquan Zou, Bin Jia, Leyao Zhang, Yinming Bai, Wanyu Li, Xiaojun Yang, Min Hua, Shaoming Long, Guoliang Cheng, Zhiyan Huang, Enhua Yang, Jinjin Shi and Guoxin Zhang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-4-tag-10.1177_17562848251378043 – Supplemental material for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial
Supplemental material, sj-docx-4-tag-10.1177_17562848251378043 for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial by Feng Ye, Ni Guo, Yaping Wang, Baochun Wang, Chunyan Niu, Zujin Ji, Ming Guo, Yilin Hou, Yi Jian, Jianhua Cui, Dazhi Chen, Shuhui Li, Dongxia Wang, Hongping Guo, Junlei Jiu, Chunlei Qiu, Hui Cao, Ying He, Ming Chen, Hua Li, Xuezhi Xin, Rongjuan Zheng, Wei Chen, Dongya Chen, Lingyun Niu, Zhenqin Cui, Zhanquan Zou, Bin Jia, Leyao Zhang, Yinming Bai, Wanyu Li, Xiaojun Yang, Min Hua, Shaoming Long, Guoliang Cheng, Zhiyan Huang, Enhua Yang, Jinjin Shi and Guoxin Zhang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-5-tag-10.1177_17562848251378043 – Supplemental material for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial
Supplemental material, sj-docx-5-tag-10.1177_17562848251378043 for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial by Feng Ye, Ni Guo, Yaping Wang, Baochun Wang, Chunyan Niu, Zujin Ji, Ming Guo, Yilin Hou, Yi Jian, Jianhua Cui, Dazhi Chen, Shuhui Li, Dongxia Wang, Hongping Guo, Junlei Jiu, Chunlei Qiu, Hui Cao, Ying He, Ming Chen, Hua Li, Xuezhi Xin, Rongjuan Zheng, Wei Chen, Dongya Chen, Lingyun Niu, Zhenqin Cui, Zhanquan Zou, Bin Jia, Leyao Zhang, Yinming Bai, Wanyu Li, Xiaojun Yang, Min Hua, Shaoming Long, Guoliang Cheng, Zhiyan Huang, Enhua Yang, Jinjin Shi and Guoxin Zhang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-6-tag-10.1177_17562848251378043 – Supplemental material for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial
Supplemental material, sj-docx-6-tag-10.1177_17562848251378043 for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial by Feng Ye, Ni Guo, Yaping Wang, Baochun Wang, Chunyan Niu, Zujin Ji, Ming Guo, Yilin Hou, Yi Jian, Jianhua Cui, Dazhi Chen, Shuhui Li, Dongxia Wang, Hongping Guo, Junlei Jiu, Chunlei Qiu, Hui Cao, Ying He, Ming Chen, Hua Li, Xuezhi Xin, Rongjuan Zheng, Wei Chen, Dongya Chen, Lingyun Niu, Zhenqin Cui, Zhanquan Zou, Bin Jia, Leyao Zhang, Yinming Bai, Wanyu Li, Xiaojun Yang, Min Hua, Shaoming Long, Guoliang Cheng, Zhiyan Huang, Enhua Yang, Jinjin Shi and Guoxin Zhang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-7-tag-10.1177_17562848251378043 – Supplemental material for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial
Supplemental material, sj-docx-7-tag-10.1177_17562848251378043 for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial by Feng Ye, Ni Guo, Yaping Wang, Baochun Wang, Chunyan Niu, Zujin Ji, Ming Guo, Yilin Hou, Yi Jian, Jianhua Cui, Dazhi Chen, Shuhui Li, Dongxia Wang, Hongping Guo, Junlei Jiu, Chunlei Qiu, Hui Cao, Ying He, Ming Chen, Hua Li, Xuezhi Xin, Rongjuan Zheng, Wei Chen, Dongya Chen, Lingyun Niu, Zhenqin Cui, Zhanquan Zou, Bin Jia, Leyao Zhang, Yinming Bai, Wanyu Li, Xiaojun Yang, Min Hua, Shaoming Long, Guoliang Cheng, Zhiyan Huang, Enhua Yang, Jinjin Shi and Guoxin Zhang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-8-tag-10.1177_17562848251378043 – Supplemental material for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial
Supplemental material, sj-docx-8-tag-10.1177_17562848251378043 for Efficacy and safety of Shouhui Tongbian Capsules, a traditional Chinese medicine, combined with 2L polyethylene glycol for bowel preparation before colonoscopy: a multicenter, randomized, single-blind, parallel-controlled clinical trial by Feng Ye, Ni Guo, Yaping Wang, Baochun Wang, Chunyan Niu, Zujin Ji, Ming Guo, Yilin Hou, Yi Jian, Jianhua Cui, Dazhi Chen, Shuhui Li, Dongxia Wang, Hongping Guo, Junlei Jiu, Chunlei Qiu, Hui Cao, Ying He, Ming Chen, Hua Li, Xuezhi Xin, Rongjuan Zheng, Wei Chen, Dongya Chen, Lingyun Niu, Zhenqin Cui, Zhanquan Zou, Bin Jia, Leyao Zhang, Yinming Bai, Wanyu Li, Xiaojun Yang, Min Hua, Shaoming Long, Guoliang Cheng, Zhiyan Huang, Enhua Yang, Jinjin Shi and Guoxin Zhang in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors thank the following collaborators for their valuable support: Dr Xi Wang (Department of Oncology, The 903rd Hospital of PLA Joint Logistic Support Force, China), Dr Jian Wang (Department of Gastroenterology, Shandong Health Zibo Hospital, China), Dr Yan Liu (Department of Anorectal Surgery, Changxing People’s Hospital, China), Dr Ke Zhang (Department of Gastroenterology, Wujin Hospital of TCM, China), Dr Xianbo Zhao (Department of Gastroenterology, People’s Hospital of Yinan, China), Dr Bin Xu (Department of Gastroenterology, The First Hospital of Anhui University of Science and Technology, China), Dr Yong Yang (Department of Gastroenterology, The First People’s Hospital of Wuhu, China), Dr Chenxi Cao (Department of Gastroenterology, The Second Hospital of Jiaxing, China), Dr Xingsheng Wang (Department of Gastroenterology, The First People’s Hospital of Baiyin, China), and Dr Xu Ai (Department of Anorectal, Jingmen Central Hospital, China).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
