Abstract
Background:
Three liters of polyethylene glycol administered in a split dose is a commonly recommended regimen for bowel preparation before colonoscopy.
Objectives:
The aim of this study was to compare the quality and tolerability of low-dose (2 L) polyethylene glycol combined with linaclotide (2 L+L) versus the 3 L polyethylene glycol (PEG) bowel preparation regimen.
Design:
A noninferiority, prospective, multicenter, randomized controlled trial.
Methods:
In this noninferiority, prospective, multicenter, randomized controlled study, patients scheduled for colonoscopy were enrolled and randomized to receive a 3 L PEG or 2L PEG+L regimen. The quality of bowel preparation and patients’ discomfort was assessed.
Results:
Over 12 months, 458 patients were randomized into 3 L PEG and 2 L+L groups. The primary endpoints showed that the 2 L+L regimen was superior to the 3L PEG regimen in overall bowel cleansing scores (Ottawa Bowel Preparation Scale: 3.3 ± 2.1 vs 3.7 ± 2.1, p = 0.021), but no significant difference in adequate bowel preparation rate between the two groups according to the OBPS score (97% vs 97.4%, p = 0.791). Before colonoscopy, patients in the 2 L +L regimen group had lower bloating scores (0.5 (0, 2) vs 1 (0, 3), p = 0.013), discomfort scores (1 (0, 2) vs 1 (0, 3), p = 0.006), and intolerability scores (1 (0, 3) vs 2 (0, 4), p = 0.016) than did those in the 3L PEG group.
Conclusion:
Two liters of polyethylene glycol combined with linaclotide may be an alternative regimen to 3 L of PEG taken in split doses for bowel preparation before colonoscopy.
Trial registration:
ChiCTR2100041992.
Plain language summary
The commonly recommended intestinal cleanser regimen is 3 L of polyethylene glycol, which achieves cleansing of the intestinal segments but may give discomfort to the patients. We have conducted a noninferiority, prospective, multicenter, randomized controlled trial to compare 2 L polyethylene glycol+ linaclotide and 3 L polyethylene glycol. The results showed that similar patients’ percentage had adequate bowel preparation rate in two groups according to the OBPS score. However, the 2 L+L regimen was superior to the 3 L PEG regimen in overall bowel cleansing scores and patients’ discomfort. Trial registration number: ChiCTR2100041992.
Introduction
Colorectal cancer is one of the most common malignant tumors worldwide, 1 endangering human health and life. Colonoscopy has proven to be the most effective method for detecting, screening, and diagnosing colorectal cancer. Detecting and removing colorectal polyps as soon as possible can help prevent tumors. However, colonoscopy efficacy is influenced by the quality of bowel preparation. It has been found that 18%–35% of patients have inadequate bowel preparation. 2 This leads to missed polyps, extended operating times, and unsuccessful cecal intubation. A modified split-dose regimen of 3 L of PEG is used in China to provide high-quality bowel preparation, while a 4 L polyethylene glycol (PEG) split-dose regimen is recommended for bowel cleaning preparation in Europe and America.3,4 However, ingesting a large volume of unpleasant-tasting liquid can lead to unpleasant side effects such as nausea and vomiting, which could lower patient compliance and tolerance and lead to an undesirable bowel preparation. 5 Since the quality of bowel preparation is closely related to patient tolerance, clinicians must consider improved bowel preparation protocols to enhance patient tolerance and compliance.
Linaclotide is a selective guanylate cyclase-C (GC-C) receptor agonist with visceral analgesic and pro-secretory effects 6 that can alleviate abdominal pain and constipation symptoms caused by irritable bowel syndrome. 7 Binding of linaclotide to GC-C increases intracellular cyclic guanosine monophosphate (cGMP), resulting in increased fluid secretion and an inhibited visceral nociceptive response. 8 The bowel preparation process causes unpleasant symptoms such as bloating, and linaclotide in combination with low-dose PEG may be a good bowel preparation regimen for improving patient tolerance. Compared with 4 L of PEG, linaclotide combined with low-dose (2 L) PEG is reported in some studies to be an effective and well-tolerated bowel preparation regimen.9,10 However, few studies have explored the quality and tolerability of low-dose (2 L) polyethylene glycol combined with linaclotide (2L+L) versus 3 L of PEG in China. Therefore, prospective, multicenter randomized controlled studies are needed to confirm the feasibility of low-dose (2 L) + linaclotide as an alternative to 3 L PEG. Considering patient compliance, 580 µg linaclotide (290 µg/day for 2 days) combined with a 2L PEG split dose was compared with a 3L PEG split dose in this study.
The primary objective of this study was to compare the bowel preparation quality of linaclotide combined with a low-dose (2 L) PEG split-dose regimen with that of the 3L PEG regimen.
Methods
Study design and participants
This multicenter prospective, randomized controlled study was conducted from February 1, 2021, to February 1, 2022, at Union Hospital, Tongji Medical College of Huazhong University of Science and Technology, the First Hospital of Wuhan, the Third People’s Hospital of Hubei Province, and the People’s Hospital of Liupanshui City. The study was approved by the Ethics Committee of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology (ethics number: 2020-0559-01) and was registered in the Chinese Clinical Trial Registry (ChiCTR2100041992), and in accordance with the Helsinki Declaration of 1975 as revised in 2013. The randomized trial in this study followed the CONSORT statement. 11 Patients were consecutively and randomly included in the study and signed an informed consent form before examination. Subsequently, all patient details were de-identified.
The study inclusion criteria were as follows: (1) age of 18–65 years; (2) scheduled for colonoscopy; (3) signed and dated informed consent form; and (4) commitment to comply with the study procedures and to cooperate with the implementation of the study. The exclusion criteria were as follows: (1) severe heart failure (New York Class III or IV); (2) recent acute myocardial infarction or unstable angina; (3) uncontrolled hypertension; (4) chronic kidney disease; (5) suspected intestinal obstruction or perforation; (6) scheduled for intestinal resection; (7) gastroparesis; (8) diarrhea more than three times per day; (9) cirrhosis of the liver or ascites; (10) pregnancy or nursing; (11) mental illness with an inability to cooperate with the examination; and (12) allergies to bowel preparation drugs.
Randomization and blinding
Computer-generated random numbers were sealed in envelopes, with odd numbers representing the 2L+L group and even numbers representing the 3L PEG. Eligibility screening for all patients was completed by a specialized internist and followed the principles of randomization and continuity. After eligibility screening, consent, and assessment, each patient was randomly assigned (1:1) to either the 2L+L group or the 3L PEG group by a dedicated nurse according to numerical numbers in envelopes. The examiners were unaware of the patient’s bowel preparation regimens. The investigators or endoscopists collecting data on the primary and secondary outcomes were unaware of the subject assignment.
Bowel preparation protocol and colonoscopy
Patients were randomized into a conventional bowel preparation group or a linaclotide-modified preparation group. For the conventional bowel preparation group, PEG (Beaufour Ipsen Pharma, Boulogne-Billancourt-France) was used as the bowel preparation agent (1 L of PEG bowel preparation solution ingested at 8:00 pm the day before the examination and 2 L of PEG bowel preparation solution ingested 4–6 h before the examination). For the linaclotide-modified group, on the day before the examination, 290 μg of linaclotide (AstraZeneca Pharmaceuticals Limited, Wuxi, China) was taken before the first meal, and 1 L of the PEG bowel preparation solution was ingested at 8:00 pm; subsequently, 290 μg of linaclotide and 1 L of the PEG bowel preparation solution were used 4–6 h before the examination. The linaclotide was dispensed directly to the patients by the investigator, and the patients did not make an additional payment. All patients were counseled on how to properly prepare their bowels and were placed on a low/no residue diet (i.e., no vegetables or fruits) for 3 days prior to the exam. A colonoscopy would be done when the bowel movements become watery, clear, and yellowish (checked by endoscopists). If the patient’s bowel preparation was poor, a colonoscopy would be performed without imposing an intervention. In this case, OBPS scoring and recording of polyp detection (without rinsing) were performed first, followed by rinsing through colonoscopy and repeat examination. Based on the quality of the patient’s final examination, a decision was made regarding the need for a short-term follow-up examination. All patients underwent colonoscopy while awake and filled out a questionnaire (including baseline information and feelings scores) before colonoscopy, then described the corresponding feeling scores during the colonoscopy procedure (recorded by nurses).
All the examinations were performed by experienced endoscopists using a high-resolution colonoscopy instrument (CF-HQ290I, Olympus, Tokyo, Japan); these endoscopists had performed at least 1000 colonoscopies. The examiners were trained on the Ottawa Bowel Preparation Scale (OBPS) prior to the examination and were asked to take high-quality pictures representing the level of bowel preparedness during colonoscopy. 12 The assessment was completed at the time of initial endoscopic passes. The OBPS was scored uniformly by two endoscopists for all subjects, and disagreements were confirmed by a third endoscopist after a comprehensive evaluation.
Outcome assessment
The primary outcome was to assess the quality of bowel preparation based on the total OBPS score. A total score of ⩽7 was considered adequate bowel preparation.
The secondary outcomes were each site’s bowel score, patients’ comfort level before and during the bowel examination; willingness to repeat the preparation in the future; and rates of cecal intubation, polyp detection, and adenoma detection. Each patient’s comfort level was measured on a 10-point scale for each of the following symptoms: abdominal pain, bloating, discomfort, and intolerability.
Statistical analysis
Sample size calculations were based on previously published data with a performance goal for optimal bowel preparation defined as ⩾85% of outpatients according to the Colonoscopy Quality Index. 13 Assuming an overall cleansing success rate of 85% in the linaclotide group, 85% in the 3L PEG group, a noninferiority margin of 12%, and an alpha level of 2.5%, a sample size of at least 374 patients (187 patients in each group) would be required to provide at least 90% efficacy to demonstrate noninferiority in each group. Considering a 10% dropout rate, a minimum of 410 patients needed to be included in this study. Continuous variables are described herein as the mean ± standard deviation or median (interquartile spacing) and were analyzed using Student’s t test or the Mann–Whitney U test. Categorical variables are expressed as frequencies and percentages and were analyzed using the Pearson χ2 test or Fisher’s exact test, as appropriate. Statistical analysis was performed using the SPSS 25.0 software package (IBM Corp., Armonk, NY, USA), and two-sided p < 0.05 was considered to indicate statistical significance.
Results
A total of 631 patients were screened for eligibility; 470 patients met the inclusion criteria and were randomly assigned to receive 3 L of PEG or 2L+L. Following the withdrawal of 12 patients (8 patients from the 3L group and 4 from the 2L+L group), 227 and 231 patients were included in the 3L PEG and 2L+L groups, respectively (Figure 1). Overall, the 2L+L group had a greater percentage of men than did the 3L PEG group (170 (73.6%) vs 146 (64.3%), p = 0.034), but there were no appreciable differences in age (42.0 ± 12.5 vs 42.0 ± 12.2, p = 0.103) or body mass index (23.0 ± 3.3 vs 23.5 ± 3.2, p = 0.511; Table 1). Other basic information such as hypertension (18 (7.8%) vs 22 (9.7%), p = 0.034), diabetes mellitus (2 (0.9%) vs 2 (0.9%), p > 0.999), medication history (42 (18.2%) vs 47 (20.7%), p = 0.555), history of abdominal surgery (34 (14.7%) vs 38 (16.7%), p = 0.608) and constipation (20 (8.7%) vs 28 (12.3%), p = 0.223) were not significantly different between the two groups.
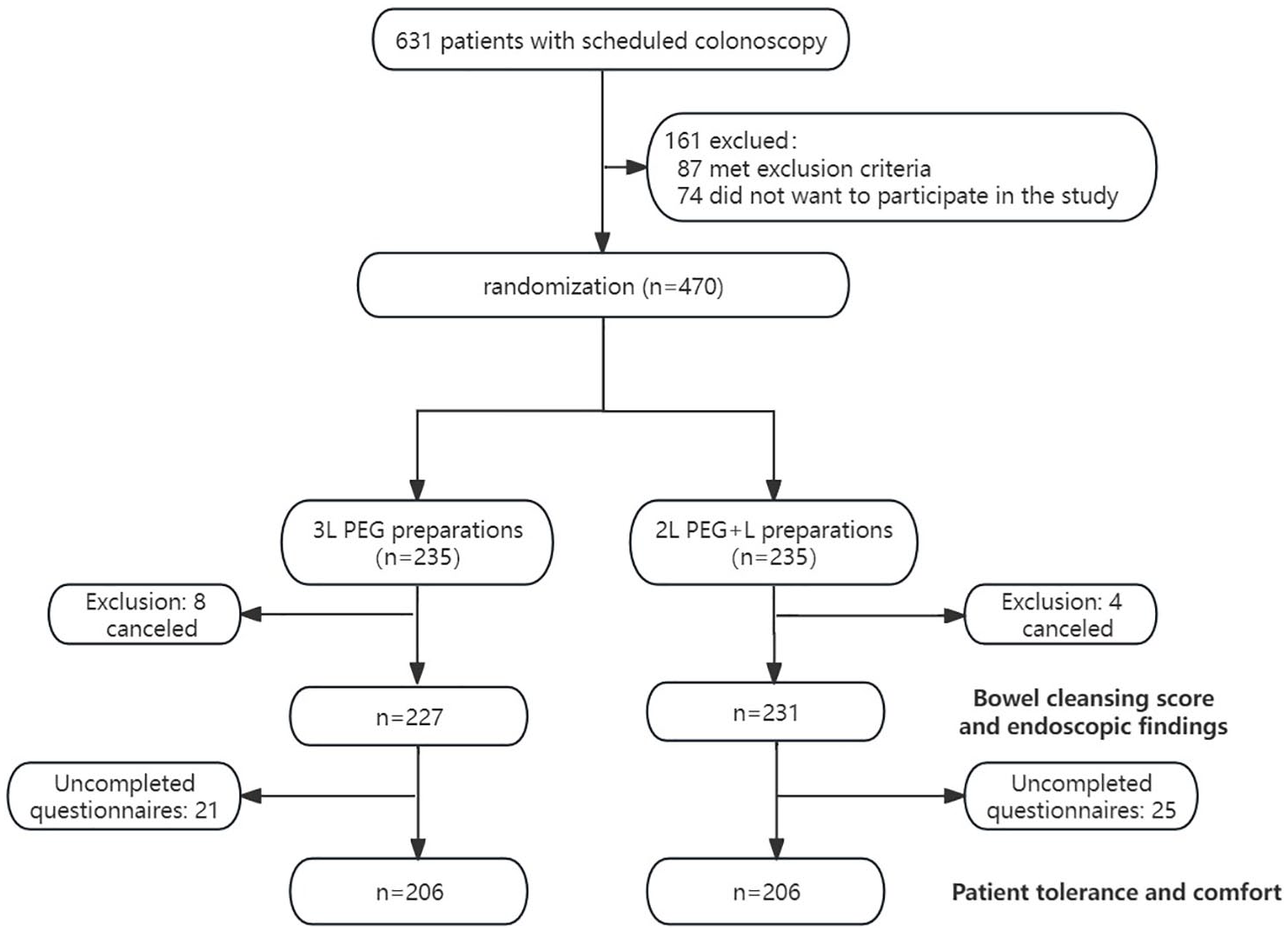
The flowchart of the research.
Baseline characteristics.

BMI, body mass index; PEG, polyethylene glycol.
Primary outcome
The results showed that similar patients’ percentages had adequate bowel preparation in two groups according to the OBPS score (97% vs 97.4%, p = 0.791). However, the 2L+L regimen was superior to the 3L PEG regimen in overall bowel cleansing results (3.3 ± 2.1 vs 3.7 ± 2.1, p = 0.021).
Secondary outcomes
As for segment scoring, the left colon and transverse colon in the 2L+L regimen outperformed the 3L PEG regimen (left colon: 0.9 ± 0.7 vs 1.1 ± 0.7, p = 0.016; transverse colon: 0.8 ± 0.7 vs 1.9 ± 0.7, p = 0.018) (Table 2 and Figure 2). The cecal intubation rate of the 2L PEG+L group was not inferior to that of the 3L PEG group (99.1% vs 98.2%, p = 0.446).
Bowel preparation outcome scores and colonic intubation rate.
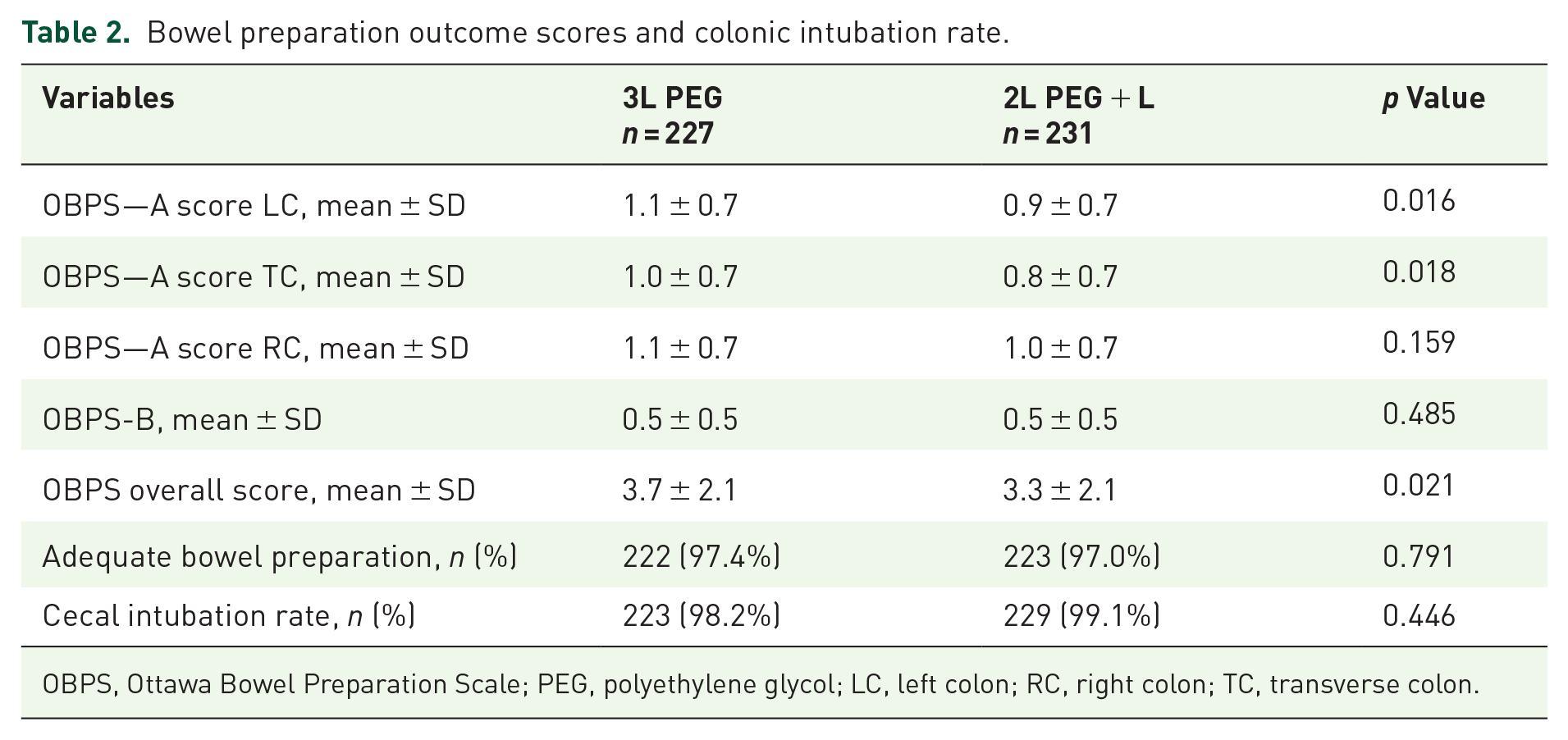
OBPS, Ottawa Bowel Preparation Scale; PEG, polyethylene glycol; LC, left colon; RC, right colon; TC, transverse colon.

Bowel-preparation outcome scores for the two groups.
There were 206 patients who completed the questionnaires on tolerance and comfort scores in the two groups (Table 3). Before colonoscopy, patients in the 2L+L regimen group had lower bloating scores (0.5(0, 2) vs 1(0, 3), p = 0.013), discomfort scores (1(0, 2) vs 1(0, 3), p = 0.006), and intolerability scores (1(0, 3) vs 2(0, 4), p = 0.016) than did those in the 3L PEG group (Figure 3), but the willingness to undergo the same cleaning regimen in the future was similar (85.4% vs 83.0%, p = 0.589). Interestingly, patients who received the 2L+L regimen were more willing during the colonoscopy to repeat the bowel preparation regimen than those were in the 3 L PEG group (86.4% vs 74.8%, p = 0.004). Additionally, patients in the 2L+L group scored lower than those in the 3 L PEG group on abdominal pain, abdominal distension, and discomfort scores, although there was no statistically significant difference (abdominal pain score: 2.8 ± 2.4 vs 3.1 ± 2.3, p = 0.324; abdominal bloating score: 3.6 ± 2.4 vs 3.8 ± 2.5, p = 0.557; abdominal discomfort score: 3.3 ± 2.3 vs 3.6 ± 2.5, p = 0.557).
Comparison of patient adverse events and tolerance between the two groups.

PEG, polyethylene glycol.

Precolonoscopy tolerance scores for the two groups.
Endoscopic findings in two bowel cleaning regimens
The results showed that endoscopic findings in the 2L+L group were not inferior to those in the 3L PEG group (Table 4). There was no significant difference in the overall polyp detection rate, adenoma detection rate, or advanced adenoma detection rate between the two groups, but the number of polyps per person in the 2L+L regimen group was greater than that in the 3L PEG group (0.7 vs 0.6, p = 0.003). The polyp detection rate and adenoma detection rate of each bowel segment were also similar in the two bowel preparation protocols.
Comparison of polyp detection between the two groups.
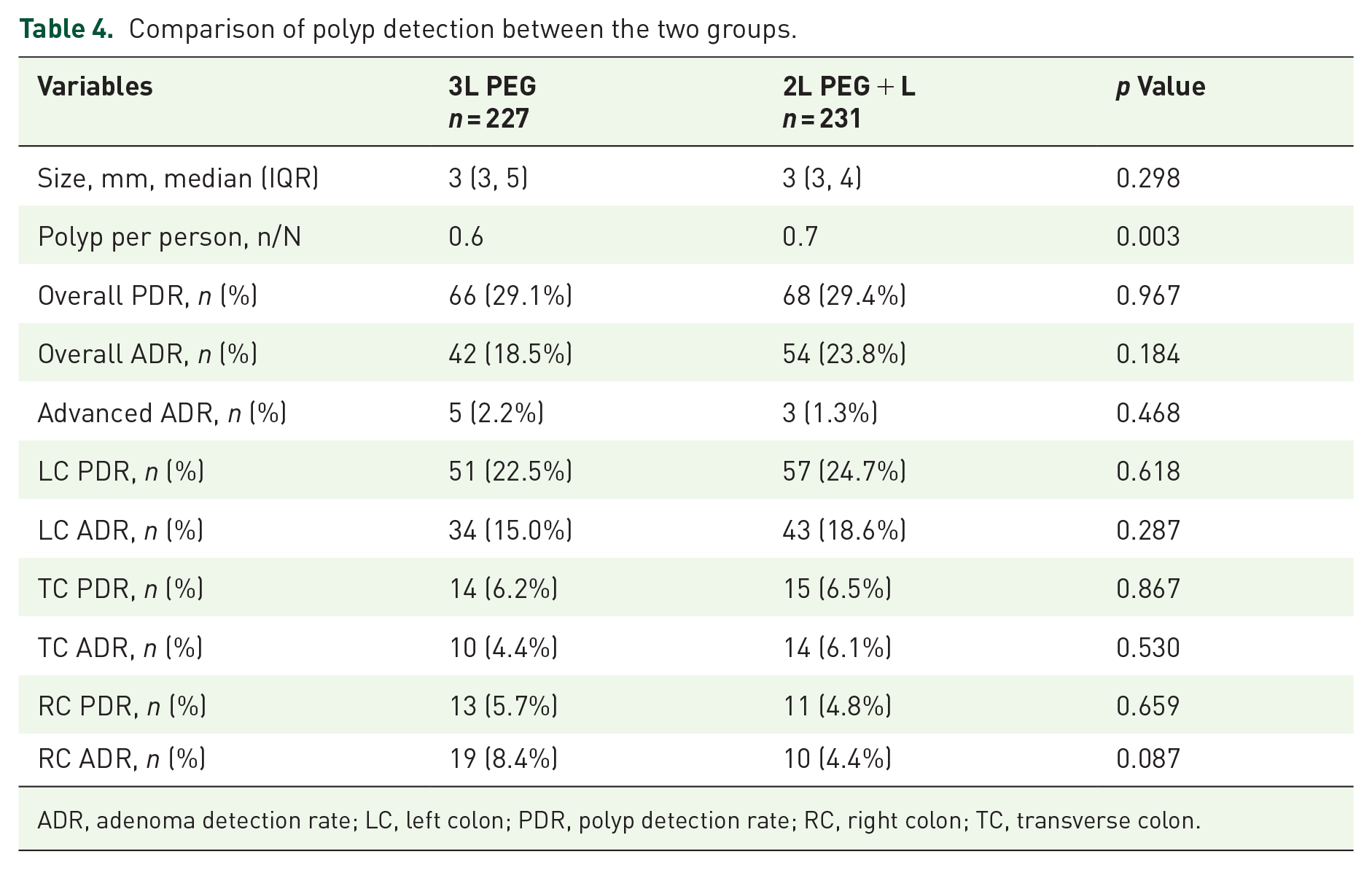
ADR, adenoma detection rate; LC, left colon; PDR, polyp detection rate; RC, right colon; TC, transverse colon.
Adverse events
In total, 303 (73.5%) patients presented with mild adverse events that were mostly associated with bowel preparation, including slight bloating, abdominal pain, discomfort, intolerability, without the most frequent treatment-emergent adverse events (Table 2). Interestingly, the 2L+L group had a lower adverse event rate than the 3L PEG group (68.9% vs 78.2%, p = 0.044).
Discussion
We performed a prospective, multicenter, randomized controlled trial in China that compared bowel cleansing efficacy and preexamination and intra-examination tolerability between a 2L+L group and a 3L PEG group. The results showed that the 2L+L group was superior to the 3L PEG group in colon cleansing scores. The degree of bowel cleansing is critical to the quality of colonoscopy. Small-volume detergents in combination with other medications may improve patient compliance and achieve similar bowel cleansing results relative to high-dose detergents. Two studies demonstrated that 2L+L provided an intestinal cleansing efficacy similar to that of 4 L of PEG,9,10 and 2L+L was also suggested as an intestinal cleansing regimen that could be used instead of 4 L of PEG. However, there is no direct comparison between 2L+L and 3 L of PEG bowel-cleansing regimens, and the feasibility of introducing 2L+L in China is uncertain. This multicenter prospective, randomized controlled study demonstrated that 2L+L was superior to 3 L of PEG for overall bowel cleansing scores. Meanwhile, the adequate bowel cleansing rates and cecum intubation rates were not significantly different between the two groups. A previous study also showed that the adequate bowel cleansing rate and cecum intubation rate of 2L+L were comparable to those of 4L PEG or other drugs combined with 2L PEG. 9
Relative to 3L PEG, the 2L+L regimen improved patients’ pre-examination discomfort. Polyethylene glycol, a macromolecular polymer that is rarely absorbed in the intestinal tract, reduces exogenous water absorption, thereby increasing the water content of the feces, softening the feces, and facilitating elimination, and generally does not affect the balance of water and electrolyte exchange. 14 However, high doses of polyethylene glycol are poorly tolerated in patients, which influences the effectiveness of cleansing. In patients in poor physical condition, short-term and large doses of laxatives may cause water-electrolyte disorders. Therefore, a bowel-cleansing regimen that reduces patient discomfort at a low dose of PEG without compromising the cleansing effect should be explored by clinicians. Previous studies have indicated that 2L+L or 2L PEG combined with other drugs (e.g., citrates and simethicone) can reduce patient discomfort and increase their willingness to repeat bowel-cleansing regimens relative to 4L PEG.9,15,16 On this basis, the present study divided tolerance into two time points, before and during colonoscopy, confirming that 2L+L improved patients’ discomfort mainly before colonoscopy, while there was no significant difference from the 3L regimen during the examination. The present results suggest that medication improves symptoms mainly during bowel preparation, and it is difficult to have an impact on the sensation experienced during colonoscopy, which may need more research for future improvement.
Linaclotide, a guanylate cyclase-C (GC-C) agonist that increases intestinal chloride and fluid secretion by activating the guanosine cyclic phosphate (cGMP) cascade reaction 17 for the treatment of irritable bowel syndrome (IBS) and visceral pain, 7 has proven to be safe in clinical trials.3,18 A study noted that linaclotide combined with PEG was superior to PEG alone in patients with chronic constipation in terms of both an adequate bowel preparation rate and the Boston Bowel Preparation Scale (BBPS) score. 19 Linaclotide also exerted a positive effect on intestinal cleansing regimen in low-risk individuals that without the following associated risk factors: constipation, age older than 70 years, body mass index (BMI) over 25 kg/m2, diabetes, Parkinson’s disease, prior surgery of gastrectomy or colorectal resection, history of stroke or spinal cord injury, and use of narcotics or tricyclic antidepressants. In the low-risk population, the ultralow-volume regimen (1 L of PEG+L) was not inferior to the low-dose regimen (2 L of PEG) and resulted in better tolerability in patients, as well as their willingness to repeat the bowel preparation. 20 In brief, linaclotide has promising applications in bowel-cleansing regimens.
The endoscopic findings in the 2L+L group were comparable to those in the 3 L PEG group. The adenoma detection rate (ADR) is strongly correlated with postcolonoscopy colorectal cancer incidence, CRC-related deaths, and serrated lesion detection, and it has been shown to be the gold standard for colonoscopy quality. 21 Few studies have further examined the changes in the adenoma detection rate and polyp detection rate with the 2L+L regimen, and our study confirmed that this regimen was not inferior to the high-dose cleansing regimen in terms of overall adenoma detection, polyp detection, advanced-adenoma detection, and adenomas detected in various parts of the bowel segments. Therefore, 2L+L achieved a similarly adequate bowel preparation rate but was superior to the 3L PEG regimen in overall bowel cleansing results and improved patient tolerance, and this cleansing regimen could be applicable not only in Europe and the United States but also in China.
Limitations
Nonetheless, our study has limitations. First, there were not enough indicators to evaluate patient tolerance, and more symptoms (e.g., nausea, vomiting, and sleep quality) could have been included. Second, other baseline information such as specific drug classifications (like TCA, laxative, and opioids), bowel habits and colorectal diverticula were missing and may have had a biased effect on the results. Third, we did not add an additional group undergoing the 4 L PEG cleansing regimen but instead used the 3L PEG regimen alone, which is commonly used in China. Finally, although the multicenter bowel-cleansing regimen research was conducted, the total number of people included in the present study was still relatively small, and more studies may be needed in the future to investigate the effects of the combination of two drugs on bowel preparation.
Conclusion
In conclusion, 2 L PEG combined with 580 μg linaclotide (290 µg/day for 2 days) yielded better intestinal cleanliness and patient tolerability than the 3 L PEG regimen, and the adenoma detection rate and polyp detection rate were not inferior to those with the 3 L PEG regimen. The addition of linaclotide to low-volume (2 L) PEG may be an alternative to 3 L PEG taken in split doses.
