Abstract
Objectives:
As with colonoscopy, adequate bowel cleansing is essential prior to colon capsule endoscopy (CCE). Because CCE requires that the capsule traverse the entire gastrointestinal tract during the examination, laxative ‘boosters’ are used. The objective of this prospective, single-center, single-arm study was to evaluate the safety of a bowel preparation consisting of polyethylene glycol (PEG) plus an oral sulfate solution.
Methods:
Subjects were healthy volunteers aged 50–75 years old with normal baseline serum chemistry. The bowel preparation consisted of 4 Senna tablets, 4 liters of PEG (split dose), 10 mg metoclopramide, 2 oral sulfate solution boosters (6 oz. and 3 oz.), and 10 mg bisacodyl. Serum chemistry was performed at baseline, following PEG intake, 24 hours after bisacodyl administration, and at 7 days post procedure (in subjects with abnormal 24 hour results). The primary endpoints were the percentage of subjects with a clinically significant change in serum chemistry at the last test and the adverse event (AE) rate.
Results:
A total of 25 subjects were enrolled. The serum chemistry was normal in all subjects at the final evaluation. One subject showed a slight elevation in creatinine (1.08 mg/dl 7 days post procedure from 0.84 mg/dl at baseline), deemed not clinically significant. Another subject had a transient elevation in serum creatinine (from 1.01 mg/dl at baseline to 1.45 mg/dl at 24 hours after the bowel preparation); values returned to near baseline at 7 days post procedure (1.06 mg/dl). There were no serious AEs, three moderate AEs related to the bowel preparation (nausea, headache, elevated creatinine) and two mild unrelated AEs (chills, abdominal cramping).
Conclusions:
A bowel cleansing regimen of PEG plus an oral sulfate solution can be used in healthy volunteers. These data provide support for the continued study of this regimen in future CCE clinical trials and in medical practice.
Keywords
Introduction
Screening and early detection can significantly reduce both the incidence and the associated mortality of colorectal cancer [Brenner et al. 2014]. While optical colonoscopy has traditionally been considered the gold standard, colon capsule endoscopy (CCE) is a minimally invasive technique that may increase patient compliance with screening recommendations and improve diagnostic yield following incomplete colonoscopy [Alarcon-Fernandez et al. 2013; Hagel et al. 2014; Triantafyllou et al. 2014; Spada et al. 2015].
Adequate bowel cleansing is critical prior to both traditional colonoscopy and CCE [Johnson et al. 2014; Rex, 2014a, 2014b; Singhal et al. 2014] and is often considered by patients the most disagreeable aspect of the procedure [Rex et al. 2014b]. Thus, many studies have been conducted to optimize the bowel preparation regimen in both colonoscopy and CCE [Spada et al. 2011; Mathus-Vliegen et al. 2013; Rex, 2014a, 2014b; Singhal et al. 2014].
Bowel preparation prior to CCE is particularly important because of the need to propel the capsule through the entire gastrointestinal tract during the examination. Thus, use of a booster agent during the capsule procedure is required to ensure that the capsule reaches the anal verge before the end of its battery life [Spada et al. 2012; Singhal et al. 2014]. Booster agents have included sodium phosphate, magnesium citrate, bisacodyl and polyethylene glycol (PEG) [Singhal et al. 2014].
An oral sulfate solution has also been assessed as a bowel cleansing agent for colonoscopy [Di Palma et al. 2009; Pelham et al. 2010; Rex et al. 2010, 2014a, 2014b] and may also be used as an effective booster agent for CCE.
The objective of this study was to evaluate the safety of a bowel preparation regimen consisting of sulfate-free PEG plus an oral sulfate solution as a booster agent in healthy adult subjects.
Methods
This study was conducted according to US Food and Drug Administration regulations, the International Conference on Harmonization E6 Guideline for Good Clinical Practice and the Declaration of Helsinki. The protocol was approved by Quorum Review IRB (20 January 2012) and all subjects provided written informed consent prior to participation. The study was registered on ClinicalTrials.gov as identifier NCT01575093 on 11 March 2012.
Objective
The objective of this prospective, single-center, single-arm study was to evaluate the safety of a bowel preparation regimen consisting of sulfate-free PEG electrolyte lavage solution (ELS) plus an oral sulfate solution as a booster in healthy adult subjects. No colon capsule examination was administered. The specific purpose was to optimize the bowel preparation regimen for future research on the PillCam® COLON 2 Capsule Endoscopy System (Covidien, Mansfield, MA, USA).
Participants
This safety study enrolled healthy volunteers (per physician discretion) between the ages of 50 and 75 at a single center in the US. To be included in the study, all subjects must have had normal serum chemistry levels at baseline (per physician discretion). Exclusion criteria were: any allergy or other known contraindication to the medications used in the study; history or clinical evidence of renal disease or previous clinically significant laboratory abnormalities of renal function parameters; type I or type II diabetes; women who were pregnant or nursing at the time of screening; the presence of life-threatening conditions; current participation in another clinical study of an investigational drug or device; and inability to understand and comply with study instructions and provide informed consent.
Bowel preparation procedure
The detailed bowel preparation procedure is shown in Table 1 and consisted of:
(1) 2 Days prior to the procedure day: 4 Senna tablets at bedtime and 10 glasses of liquid during the day;
(2) Day before the procedure day: clear liquid diet after lunch and 2 liters of sulfate-free PEG-ELS in the evening
(3) Procedure day: clear liquid diet until procedure completion, 2 liters of PEG-ELS, 10 mg metoclopramide, 2 oral sulfate solution boosts (6 oz. and 3 oz.), prepared according to the package insert and followed by 1 quart (0.95 liter) of water, and a 10 mg bisacodyl suppository.
Bowel preparation procedure.

The oral sulfate solution used was SUPREP Bowel Prep Kit [Braintree Laboratories, 2013], an osmotic laxative indicated for cleansing of the colon as a preparation for colonoscopy in adults. It is a 6 oz. oral solution of sodium sulfate (17.5 g), potassium sulfate (3.13 g) and magnesium sulfate (1.6 g). Prior experience with this oral sulfate solution has demonstrated efficacy and safety in peer reviewed studies [Di Palma et al. 2009; Patel et al. 2009; Pelham et al. 2009, 2010; Rex et al. 2010, 2014]. For further details, refer to full prescribing information .
Study assessments
Serum chemistry tests were conducted on each subject at baseline (Test 1), following PEG intake on the procedure day (before metoclopramide administration) (Test 2), and at 24 hours after bisacodyl administration (Test 3). Any subject with a potentially clinically significant change in serum chemistry (relative to baseline; per physician discretion) on Test 3 was also required to undergo an additional serum chemistry test 7 days post procedure (Test 4). Serum chemistry tests included bicarbonate, blood urea nitrogen, calcium, chloride, creatinine, potassium and sodium.
Outcomes measures
The primary endpoints of this study were the percentage of subjects with a clinically significant change in serum chemistry at the latest blood test (i.e. 24 hours after suppository administration or 7 days after procedure) from normal baseline serum chemistry, as per physician discretion, and the adverse event (AE) rate.
Statistics
Analyses were performed using SQL Server8, SQL, and SPSS Version 20. Data were summarized by descriptive statistics (for continuous variables) or frequencies and percentages (for categorical variables). No formal sample size calculations were conducted for this single-arm safety study.
Results
Participants
A total of 25 healthy volunteers aged 51–70 were enrolled at a single site between March and June 2012.
Demographics and medical history
Subject demographics are listed in Table 2. Subjects were predominantly female (n = 19; 76%) with a mean age of 58.5 years and a mean body mass index of 30.5.
Subject demographics.

Data are shown as n/N (%) or mean ± standard deviation (minimum–maximum).
Procedural characteristics
All subjects completed the bowel preparation procedure as defined in the study protocol, with the exception of 2 subjects who were not able to drink the entire 2 liters of PEG during the morning of the procedure day (1.83 and 1.86 liters, respectively).
Change in serum chemistry
Serum chemistry results are shown in Table 3. Only 2 subjects out of the 25 subjects enrolled exhibited any notable change in serum chemistry. The first was a 69-year old woman with creatinine levels of 0.84 mg/dl at baseline and 1.03 mg/dl at 24 hours after bisacodyl administration. The subject’s creatinine level was 1.08 mg/dl 7 days after the bowel preparation procedure. Following that test, the subject had a colonoscopy performed at a different medical center (without informing the study site) and had taken an additional bowel cleansing solution (4 liters of GaviLyte). Creatinine levels after that second bowel cleansing were 1.03 mg/dl. A final laboratory test, 2 weeks later, revealed a creatinine level of 0.91 mg/dl. All other laboratory values were unchanged from baseline in this subject. This change was later deemed not clinically significant due to the fact that serum chemistry was not consistent with acute kidney injury and returned to baseline within 3 weeks. In the second subject, a 62-year old man, creatinine levels were 1.01 mg/dl at baseline, 1.17 mg/dl following PEG administration, 1.45 mg/dl 24 hours after the bowel preparation and 1.06 mg/dl at 7 days post procedure. Because creatinine levels returned to baseline values at 7 days post procedure, the results of this subject were not considered against the primary endpoint, but should be noted nonetheless.
Serum chemistry results.
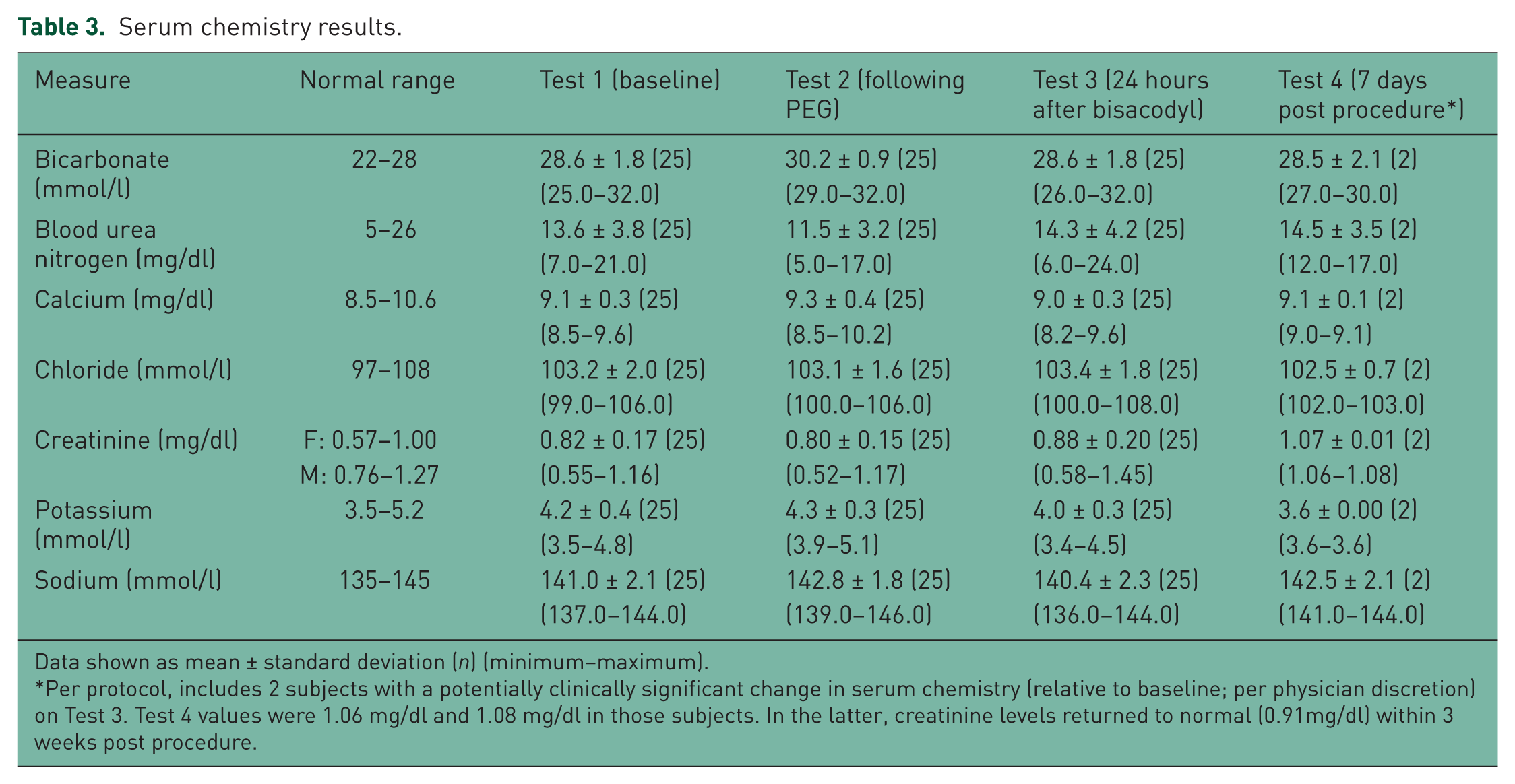
Data shown as mean ± standard deviation (n) (minimum–maximum).
Per protocol, includes 2 subjects with a potentially clinically significant change in serum chemistry (relative to baseline; per physician discretion) on Test 3. Test 4 values were 1.06 mg/dl and 1.08 mg/dl in those subjects. In the latter, creatinine levels returned to normal (0.91mg/dl) within 3 weeks post procedure.
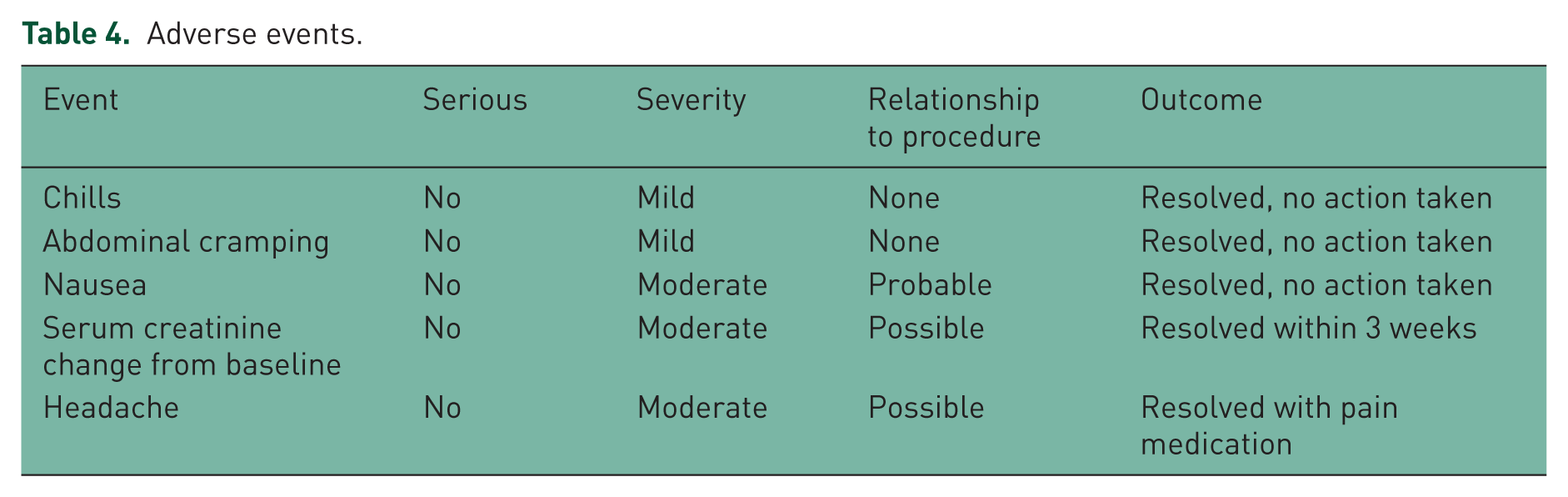
AEs
Details of AEs are shown in Table 4. No serious AEs were reported in any subject. A total of three moderate AEs were reported and considered related to the colon cleansing preparation. These included: one case of nausea, resolved the same day; one case of headache, resolved the same day after receiving pain medication; and one case of elevated serum creatinine, as described above. Two cases of mild AEs (chills and abdominal cramping) were also reported as unrelated to the procedure.
Adverse events.
Discussion
The objective of this study was to evaluate the safety of a bowel preparation regimen consist-ing of split dose PEG plus an oral sulfate solution as a booster agent in healthy adult subjects. Determination of the optimal bowel preparation method is critical for CCE, where rapid transit and adequate cleansing are paramount for a successful exam, and suction and irrigation cannot be used to improve the outcome. In the current study, no serious AEs and only three cases of moderate AEs were reported. Final serum chemistry tests were within normal ranges in all subjects; two subjects exhibited a transient elevation in serum creatinine.
In the 2014 Consensus Statement on Adequate Bowel Cleansing for Colonoscopy, the US Multi-Society Task Force on Colorectal Cancer recommended that bowel preparation should be sufficient to allow detection of polyps >5 mm in at least 85% of cases. Furthermore, the guidelines recommend a split dose bowel cleansing regimen in which approximately half of the bowel cleansing dose is given the day of the colonoscopy [Johnson et al. 2014].
Several studies have been conducted to optimize the bowel preparation regimen prior to CCE [Spada et al. 2011a, 2011b; Singhal et al. 2014]. While most studies use a high volume (3–4 liters) of PEG as the primary laxative, the incorporation of prokinetics and booster agents varies and includes combinations of sodium phosphate, magnesium citrate, bisacodyl, Gastrografin and Senna [Singhal et al. 2014; Spada et al. 2015]. Based on these studies, guidelines have recommended a split dose of PEG followed by booster preparations to improve capsule egestion rates and allow complete visualization of the entire colonic mucosa [Spada et al. 2012].
Sodium phosphate has not been recommended for bowel cleansing during small bowel capsule endoscopy due to the potential for renal failure and other AEs, and warnings have been provided regarding use during CCE in patients with potential toxicity [Spada et al. 2012, Mathus-Vliegen et al. 2013].
The oral sulfate solution used in the current study has been assessed as a bowel cleansing agent for colonoscopy [Di Palma et al. 2009; Pelham et al. 2010; Rex et al. 2010; Rex et al. 2014a, 2014b]. Rex and colleagues compared this oral sulfate solution to a standard 4-liter PEG solution in patients undergoing colonoscopy. In that study, the oral sulfate solution resulted in a higher percentage of successful bowel preparation (98.4% versus 89.6%) and a higher rate of ‘excellent’ cleansing levels (71.4% versus 34.3%). The authors also reported no clinically significant changes in serum chemistry, although statistically significant changes were seen relative to baseline in magnesium levels. In addition, 2 patients in that analysis had increases in serum phosphate levels following administration of the oral sulfate solution (from 3.7 mg/dl to 9.2 mg/dl in one subject and from 3.7 mg/dl to 5.7 mg/dl in a second subject); redraw levels returned to baseline and the authors concluded that these changes were not clinically significant [Rex et al. 2010]. The current analysis unfortunately did not assess magnesium or serum phosphate, so no comparison can be made to the analysis by Rex and colleagues.
Two unpublished pilot studies have assessed the oral sulfate solution with PEG as a preparation for CCE. In the first study [ClinicalTrials.gov identifier: NCT01185002], 10 hour excretion rates were higher with the use of the oral sulfate solution boost (90% with a 6 oz. first boost and 85% with a 3 oz. first boost) compared with a magnesium citrate boost (75%). Good or excellent cleansing levels were observed in 86% and 82% of patients administered the 6 oz. and 3 oz. oral sulfate boosts, respectively, compared with 74% who received a magnesium citrate boost. A total of 10 mild AEs (nausea, headache, vomiting and hemorrhoid flare up) and one moderate AE (headache) were reported in the 61 patients who received the oral sulfate solution. In a second pilot study [ClinicalTrials.gov identifier: NCT01269372], which also utilized the oral sulfate solution booster in 30 patients, there were 7 mild AEs related to the bowel preparation regimen [abdominal pain, abdominal cramping, diarrhea, headache, nausea (2 events) and vomiting], all of which revolved within 24 hours. The present study did not include a CCE examination and thus did not assess bowel cleansing levels; future research will be needed to assess this cleansing regimen in patients undergoing CCE.
In a multicenter study of 884 subjects undergoing capsule endoscopy using a bowel preparation procedure consisting of an oral sulfate solution plus PEG, Rex and colleagues reported 128 nonserious AEs (vomiting, nausea, headache, dizziness, gagging, diarrhea, hemorrhoidal bleeding, bloating, rash, abdominal pain, syncope, tinnitus, chest burning, chills, flu-like symptoms) related to the bowel preparation procedure [Rex et al. 2015]. Thus, there were no unanticipated side effects related to oral sulfate solution intake.
Limitations
This study is a single-arm, single-center analysis in healthy volunteers, designed as a safety study. The results may not be generalizable to all patient populations undergoing CCE (who would likely have a greater incidence of comorbidities than the healthy volunteers assessed in this study) and real world practice patterns. No CCE exam was conducted and thus the efficacy of this bowel preparation method in bowel cleansing was not assessed.
Conclusion
In summary, a safe and effective bowel preparation regimen is important for the success of a CCE exam, where rapid bowel transit and robust cleansing are essential, and suction and irrigation cannot be used to improve the outcome. A bowel cleansing method consisting of PEG plus an oral sulfate solution can be used in healthy volunteers aged 50–75 years old, with no long-term clinically significant changes in serum chemistry. These data provide support for the continued study of this regimen in future CCE clinical trials and in medical practice.
Footnotes
Funding
The study was sponsored and entirely funded by Covidien (Duluth, GA, USA), a manufacturer of colon capsule devices.
Conflict of interest statement
P.K.K. received research support from Covidien to conduct this study. R.P. is a full-time employee of Covidien. Medical writing and editorial support was provided by a professional medical writer (Kristin L. Hood, PhD) employed by Covidien.
