Abstract
Background:
Infliximab (IFX) is widely used for the treatment of Crohn’s disease (CD), yet up to 40% patients experience primary non-response (PNR). Visceral adipose tissue (VAT) and intestinal ultrasound (IUS) could be used as noninvasive tools to predict treatment efficacy with limited value, respectively.
Objectives:
This study aims to investigate the value of IUS combined with VAT in predicting PNR in CD patients.
Design:
This was a single-center, prospective, and observational study.
Methods:
Consecutive patients with active CD initiating regular IFX therapy were prospectively included. Patients underwent IUS at baseline (T0), week 2–4 (T1), and week 14 (T2), and CT at T0 to collected visceral fat area (VFA). Multivariable analyses and logistic regression analysis were used to identify predictors based on the efficacy at T2, regardless of prior biologic exposure.
Results:
Of 100 patients, 31 (31%) experienced PNR. Multivariable analysis identified baseline bowel wall thickness [BWTT0; odds ratio (OR), 2.082; 95% confidence interval (CI), 1.038–4.176; p < 0.05], VFA (VFAT0; OR, 1.030; 95% CI, 1.008–1.052; p < 0.01), the decrease of BWT (△BWT; OR, 2.717; 95% CI, 1.349–5.472; p < 0.01) and the International Bowel Ultrasound Segmental Activity Score (△IBUS-SAS; OR, 1.082; 95% CI, 1.007–1.163; p < 0.05) from baseline to T1 were independent predictors of PNR. The △BWT combined with VFAT0 can accurately predict PNR with a receiver operating characteristic (ROC) of 0.841 (95% CI, 0.756–0.926), superior to BWTT0 combined with VFAT0 (ROC, 0.832; 95% CI, 0.754–0.909) and △IBUS-SAS combined with VFAT0 (ROC, 0.830; 95% CI, 0.753–0.917).
Conclusion:
Early change in BWT on IUS combined with baseline VFA is highly predictive of PNR to IFX in CD patients, allowing stratification of patients beneficial from treatment initiation and guiding individualized clinical practice.
Plain language summary
Infliximab (IFX) is widely used for the treatment of Crohn’s disease (CD), yet up to 40% patients experience primary non-response (PNR). Visceral adipose tissue (VAT) and intestinal ultrasound (IUS) could be used as noninvasive tools to predict treatment efficacy with limited value, respectively. This study explored whether IUS combined with VAT has better predictive performance. Among 100 patients, 31 experienced PNR. Further, our study showed that the decrease of bowel wall thickness (△BWT) combined with baseline visceral fat area (VFAT0) could predict PNR, superior to the baseline BWT (BWTT0) combined with VFAT0 and the decrease of International Bowel Ultrasound Segmental Activity Score (△IBUS-SAS) combined with VFAT0. The findings highlight early change in BWT combined with baseline VFA is highly predictive of PNR to IFX in CD patients. This allows for the stratification of patients who will benefit from treatment initiation and guides individualized clinical practice.
Keywords
Introduction
Crohn’s disease (CD) is a chronic nonspecific inflammatory disease of the intestines. Infliximab (IFX), an anti-TNF-α monoclonal antibody, is one of the most frequently biologic medications in CD and has led to major shifts in the therapeutic framework, with successful treatment leading to mucosal healing, reduced surgeries, and improvements in quality of life.1–3 Regrettably, however, 10%–40% patients experience primary non-response (PNR) with no significant improvement or even deterioration in symptoms, serology, imaging, and endoscopy within the induction period (the first 12–14 weeks for standard dose IFX). 4 The biology of non-response is complex, but being able to predict who will fail to IFX therapy could optimize the treatment plan, reduce unnecessary healthcare expenditures, and potentially improve long-term clinical outcomes by enabling timely transition to more effective therapies.
Multiple patients, diseases, biomarkers, and imaging indicators have been implicated in IFX treatment prediction, 5 but the availability of these predictive markers seems to be low, with limited value. Therefore, it is clinically important to screen patients sensitive to IFX therapy in advance and develop prognostic tools for IFX response.
Visceral adipose tissue (VAT) refers to the hormonally and metabolically active fat deposited around the viscera, which is involved in the pathogenesis of CD and increases the risk of bowel damage progression because it can release inflammatory mediators, such as TNF-α, IL-6. 6 Thus, VAT is appealing given the increasing drive for early predicting PNR to IFX. Our previous retrospective study found that Visceral fat area (VFA) could predict the efficacy of IFX for different sites of CD at 6 and 12 months, with high VFA values indicating poor efficacy, high serum TNF-α levels, and low IFX serum trough concentrations. 7 Previous studies have also shown that VFA is a predictor of anti-TNF-α efficacy, mucosal healing, and subsequent surgical risk, and patients with large VFA are more likely to have non-response.8–11 However, given that VFA is closely related to individual nutritional status and disease duration and is not suitable for frequent assessment due to the radiation exposure from computed tomography (CT) or computed tomography enterography (CTE), its utility in assessing early treatment response is limited in CD patients.
Advancements in cross-sectional imaging, particularly intestinal ultrasound (IUS), introduce new tools to monitor treatment response in CD. IUS is a noninvasive, point-of-care, radiation-free, cost-effective, and cross-sectional imaging modality. It is well-tolerated by patients, requires no fasting, bowel preparation, or sedation, and can be used to frequently assess colonic and small bowel inflammation—with diagnostic performance comparable to that of ileocolonoscopy, magnetic resonance enterography, and CTE.12–14 IUS parameters have been shown to accurately determine disease activity and correlate well with endoscopic mucosal activity in CD.14,15 Multicenter studies have indicated that most IUS parameters normalize following biologic therapy, and that bowel wall thickness (BWT) is highly correlated with treatment response.16,17 However, it has certain limitations, such as its moderate predictive value, its poor sensitivity in the assessment of VAT (especially mesenteric fat), and its inabillity to provide quantitative measurements of these fat tissues.12,18
The aim of this study is therefore to investigate the value of IUS combined with VAT in predicting PNR to IFX in patients with CD for the first time, with the expectation of appropriately selecting patients who benefit the most from early IFX treatment and guiding individualized clinical practice.
Materials and methods
Study design and patients
This prospective study was approved by the institutional ethics review board and written informed consent was obtained from all enrolled CD patients. From October 2023 to February 2025, consecutive patients with primary or recurrent active CD and scheduled to receive IFX were recruited at the Third Xiangya Hospital of Central South University, and standard induction therapy [IFX(Kexing Biopharm Co., Ltd., BioRay BioPharmaceutical Co., Ltd.), 5 mg/kg] was administered to eligible patients at week 0, 2, 6, and 14.
Inclusion criteria were as follows: (1) active CD patients [the Crohn’s Disease Activity Index (CDAI) ⩾150 or the Simple Endoscopic Score for Crohn’s disease (SES-CD ⩾3)] initiating IFX; (2) age ⩾18 and ⩽80 years; (3) availability of abdominal CT or CTE at baseline (T0); (4) consent for IUS at T0, week 2–4 (T1) and week 14 (T2); (5) consent for ileocolonoscopy at T0 and T2; (6) no history of ileocolonic surgery. The exclusion criteria were as follows: (1) contraindication to IFX treatment: allergy, active tuberculosis or other active infections, moderate-to-severe heart failure (NYHA III/IV), demyelinating lesions of the nervous system, live vaccines within the last 3 months; (2) pregnancy and breastfeeding; (3) a history of extensive intestinal resection or a recent intention to undergo a resection; and (4) prolonged use of steroids.
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of The Third Xiangya Hospital of Central South University (Approval Number: IRB Number R23770). The reporting of this study confirmed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 19
Sample size
A sample size of at least 60 patients (40 responders and 20 nonresponders) was required in this study based on the following input and assumption: power, 80%; two-sided significance level, 0.05; alternative hypothesis of the area under the curve (0.900); and an allocation ratio of sample sizes in the response group and the non-response groups of 2:1.
Study procedures
Demographic data were collected at baseline, including age, disease duration, gender, BMI, smoking history, CD location and behavior (Montreal), and previous treatment at enrollment. Serological markers (including C-reactive protein, leukocytes, platelets, hemoglobin, monocytes, lymphocytes, neutrophils, eosinophils, erythrocyte sedimentation rate, albumin) were collected, and the CDAI was calculated. Ileocolonoscopy was performed at T0 and T2. IUS was performed at T0, T1, and T2, within 1 week before or after ileocolonoscopy, but never on the same day to avoid any potential effects of bowel preparation. The Inflammatory Bowel Disease Questionnaire (IBDQ, scores ranging from 32 to 224, with higher scores indicating better health-related quality of life) and the Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F, scores ranging from 0 to 52, with higher scores indicating less fatigue) were also completed at T0 and T2.
Intestinal ultrasound
IUS was performed by two ultrasound practitioners. They had received standard training to establish a consensus on the standardization of parameters based on the guidelines for IUS20,21 and were blinded to endoscopic, clinical, and biochemical data. The IUS was performed with the Siemens Healthineers device (Erlangen, Bavaria, Germany), using a convex array low-frequency probe (SC6-1U, 2.5–3.5 MHz) and subsequently a line array high-frequency probe (L14-5WU, 4.0–14.0 MHz). Before the examination, the patient was instructed to fast for 6–8 h and took the supine position for full abdominal exposure. The device settings, including depth, focus, gain, and color Doppler threshold, were adjusted based on the patient’s physical condition. Parameters were kept consistent for all examinations of the same patient. The key IUS parameters of the most severe segment were assessed and classified as detailed in Supplemental Table 1. International Bowel Ultrasound Segmental Activity Score (IBUS-SAS) was calculated for the same segment of bowel.
Ileocolonoscopy
Ileocolonoscopies were performed at T0 and T2 by experienced gastroenterologists. These procedural endoscopists, who performed the examinations, were blinded to all non-endoscopic data, including patients’ clinical symptoms, laboratory results, and IUS findings. Furthermore, they were not involved in the clinical management or treatment decisions of the enrolled patients. The SES-CD was assessed by a blinded central reader.
VAT measurements
CT or CTE images were obtained of the third lumbar (L3), and VAT was measured (Supplemental Figure 1). Each selected image was assessed by a researcher who was unaware of the clinical and biological data, using NIH Image J1.47 (Bethesda, MD, USA), free public domain software developed by the National Institutes of Health. A standard threshold of −150 to −50 Hounsfield units was applied to outline total fat area (TFA, cm2), VFA (cm2), subcutaneous fat area (SFA, cm2), and a mesenteric fat index (MFI) = VFA/SFA.
Outcomes
Active CD was defined as CDAI ⩾150, clinical response was defined as a decrease of at least 50% from baseline CDAI and clinical remission with a CDAI <150. 18 The SES-CD was used as a measurement of disease activity by ileocolonoscopies, active CD was defined as SES-CD ⩾3. The endoscopic response was defined as a decrease of at least 50% from baseline SES-CD, and endoscopic remission was defined as SES-CD ⩽2. Transmural healing was defined as bowel wall thickness ⩽3 mm and no Doppler flow signal assessed by IUS. 22 Of the overall assessment, patients who were classified as the primary response group (R group) showed clinical or endoscopic response at T2; otherwise, the patients were classified into the PNR group (NR group), including (1) received CD-related surgery, (2) required additional systemic corticosteroids, (3) increased IFX doses, (4) changed the medication regimen. In addition, patients were categorized into the biologic-naïve group and biologic-experienced group based on whether they were first-time biologics users. The study flowchart is presented in Figure 1.

The flowchart of the study.
Statistical analysis
Statistical analysis was performed by SPSS version 26.0 (IBM Inc., Armonk, NY, USA). The mean and standard deviation (SD) were used to describe normally distributed continuous data while the median value and inter-quartile range were used to describe abnormally distributed continuous variables. Categorical data are presented as frequencies and percentages. Differences in clinical, ultrasonographic parameters, and VAT between the response and non-response groups at baseline were compared by an independent sample test for normally distributed variables. Mann–Whitney U tests for continuous non-parametric variables and the Chi-square test or Fisher’s exact test (when expectation <5) for categorical variables. Logistic regression was used to identify predictors of IFX treatment effectiveness. Univariate analyses were first performed, with variables with p < 0.05 as candidates for predictors to be included in subsequent multivariate analyses. The diagnostic performance of baseline BWT, VFA, △BWT, and △IBUS-SAS was estimated by receiver operating characteristic (ROC) curves. The optimal cut-off values for ultrasound parameters and visceral fat in differentiating between response and non-response were determined by ROC curve analysis, followed by determination of the Youden index.
Results
Demographic and clinical data
A total of 100 patients were enrolled, including 84 men (84%) and 16 women (16%). Based on the efficacy at T2, 69 patients were categorized into the R group [57 men (82.6%), mean age 30.6 ± 10.9 years] and the remaining 31 patients classified as the NR group [27 men (87.1%), mean age 29.1 ± 10.2 years]. No significant differences were observed in demographics, clinical characteristics, biochemical parameters, and baseline CDAI and SES-CD scores between the R and NR groups (Table 1).
The demographic and disease characteristics between the R and NR groups.
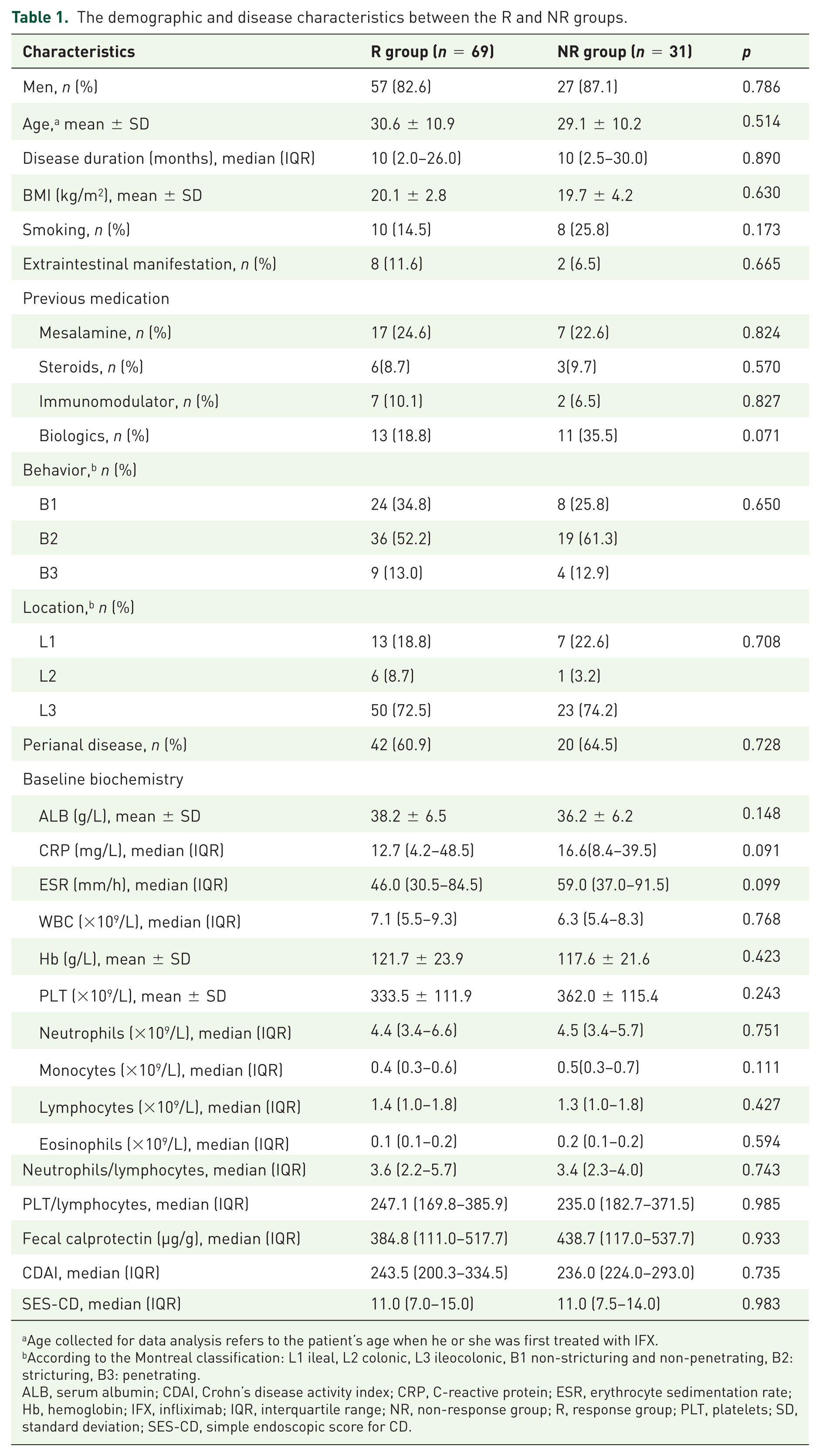
Age collected for data analysis refers to the patient’s age when he or she was first treated with IFX.
According to the Montreal classification: L1 ileal, L2 colonic, L3 ileocolonic, B1 non-stricturing and non-penetrating, B2: stricturing, B3: penetrating.
ALB, serum albumin; CDAI, Crohn’s disease activity index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; IFX, infliximab; IQR, interquartile range; NR, non-response group; R, response group; PLT, platelets; SD, standard deviation; SES-CD, simple endoscopic score for CD.
Only the disease duration was different between the biologic-naïve and biologic-experienced groups at T0 [5.0 (2.0–18.0) vs 24.5 (12.0–66.0) months, p < 0.001], and there were no statistically significant differences in baseline CDAI, SES-CD, or other demographic and disease characteristics (all p > 0.05). Clinical characteristics of patients enrolled are presented in Supplemental Table 2.
Comparison of IUS parameters and abdominal fat metrics at T0
The IUS parameters [BWT, color Doppler signal (CDS), bowel wall stratification (BWS), inflammatory mesenteric fat (i-fat), IBUS-SAS] of the worst bowel segment and abdominal fat metrics (TFA, VFA, SFA, MFI) at the L3 cone plane were compared between the R and NR groups at T0. Patients in the R group had lower BWT and IBUS-SAS than those in the NR group [5.5(4.7–6.9) vs 7.1(6.1–8.0) mm, p < 0.001; 64.8(37.8–75.5) vs 82.0(68.0–88.0), p < 0.001]. No statistically significant difference was observed in CDS between the two groups (p = 0.654). There was a statistically significant difference in BWS and i-fat (p = 0.013, p = 0.003, respectively). At T0, TFA, VFA, and SFA were lower in the R group than in the NR group (94.4 ± 57.8 vs 168.6 ± 96.2 cm2, p < 0.001; 39.1 ± 26.8 vs 84.9 ± 62.6 cm2, p < 0.001; 55.3 ± 38.3 vs 83.7 ± 43.4 cm2, p = 0.001). However, no statistically significant difference was observed in MFI (p = 0.074) between the two groups at T0 (Supplemental Table 3).
There was a statistically significant difference in CDS between the biologic-naïve and biologic-experienced groups at T0 (p = 0.023). No statistically significant difference was observed in BWT, BWS, i-fat, IBUS-SAS, and abdominal fat metrics (TFA, VFA, SFA, MFI) between the two groups (Supplemental Table 4).
Analysis of the efficacy after IFX treatment at T2
After completion of IFX induction therapy at T2, 65 (94.2%, 65/69) patients in the R group had achieved clinical remission, 49 (71.0%, 49/69) patients had achieved endoscopic response, 19 (27.5%, 19/69) patients had achieved endoscopic remission, and 24 (34.8%, 24/69) patients had transmural healing as assessed by IUS. However, none of the patients in the NR group had achieved endoscopic response, remission, or transmural healing. Supplemental Figures 2 and 3 illustrate the changes in IUS and endoscopy before and after IFX treatment.
Changes in IBDQ and FACIT-F
After 14 weeks of IFX treatment, both IBDQ and FACIT-F scores improved across all patients. At T0, there were no statistically significant differences in the scores of IBDQ (167.3 ± 19.5 vs 167.1 ± 25.8, p = 0.968) and FACIT-F (33.4 ± 6.2 vs 31.2 ± 8.1, p = 0.325) between the R and NR groups, either in each dimension between the two groups (all p > 0.05). However, statistical differences were observed in IBDQ (196.4 ± 16.9 vs 181.2 ± 25.6, p = 0.006) and FACIT-F (41.3 ± 4.4 vs 36.0 ± 7.4, p = 0.001) between the two groups at T2 (Supplemental Table 5, Figure 2). The R group showed a significant improvement in IBDQ (167.3 ± 19.5 vs 196.4 ± 16.9, p < 0.001) and FACIT-F (33.4 ± 6.2 vs 41.3 ± 4.4, p < 0.001) at T2 compared to baseline. In contrast, neither IBDQ nor FACIT-F showed significant improvement in the NR group.

Compraison of quality of life and fatigue scores before and after IFX treatment between the R and NR groups. (A) Changes in IBDQ Scores between T0 and T2. (B) Changes in FACIT-F Scores between T0 and T2 (T0, baseline; T2, Week 14 of IFX treatment).
There were no statistically significant differences in IBDQ, FACIT-F at T0 and T2 between the biologic-naïve and biologic-experienced groups (Supplemental Table 6 and Supplemental Figure 4).
Changes in IUS parameters
At T1 and T2, there were statistically significant differences in all IUS parameters between the R and NR groups (all p < 0.05, Table 2). For the changes of IUS quantitative parameters, BWT [5.5(4.7–6.9) vs 4.2 ± 1.5 vs 3.4(2.6–4.5) mm] and IBUS-SAS [64.8(37.8–75.5) vs 36.4(22.2–61.2) vs 23.8(10.8–44.1)] showed a significant decrease compared with T0 at both T1 and T2 (all p < 0.001, Supplemental Table 7) in the R group. The proportion of normal CDS, BWS, and i-fat was higher at T1 and T2 compared with T0. However, there was no significant change in the NR group. The change values of BWT and IBUS-SAS in the R group were significantly higher than those in the NR group (all p < 0.01; Figure 3).
Changes in IUS parameters before and after IFX treatment between the R and NR groups.

BWS, bowel wall stratification; BWT, bowel wall thickness; CDS, color Doppler signal; IBUS-SAS, International Bowel Ultrasound Segmental Activity Score; i-fat, inflammatory mesenteric fat; IFX, infliximab; IQR, interquartile range; IUS, intestinal ultrasound; NR, non-response group; R, response group.

The changes of BWT and IBUS-SAS at T0, T1, and T2 during IFX treatment. (A) Changes of BWT in the NR group. (B) Changes of IBUS-SAS in the NR group. (C) Changes of BWT in the R group. (D) Changes of IBUS-SAS in the R group. (E) The change values of BWT from the T0 between the R and NR groups. (F) The change values of IBUS-SAS from the T0 between the R and NR groups (**p < 0.01). Blue represents the individuals in the R group, and red represents the individuals in the NR group.
At T1 and T2, there were no statistically significant differences in all IUS parameters between the biologic-naïve and biologic-experienced groups (all p > 0.05, Supplemental Table 8).
Prediction of PNR to IFX from IUS parameters combined with VAT
Univariate analysis was performed for the following variables: BWT (BWTT0), IBUS-SAS (IBUS-SAST0), TFA (TFAT0), VFA (VFAT0), SFA (SFAT0), and MFI (MFIT0) at T0 and the decrease of BWT (△BWT) and IBUS-SAS (△IBUS-SAS) from baseline to T1. Then, the variables with p < 0.05 were included in the multivariate analysis. Results indicated that BWTT0 [odds ratio (OR), 2.082; 95% confidence interval (CI), 1.038–4.176; p < 0.05], △BWT (OR, 2.717; 95% CI, 1.349–5.472; p < 0.01), △IBUS-SAS (OR, 1.082; 95% CI, 1.007–1.163; p < 0.05), and VFAT0 (OR, 1.030; 95% CI, 1.008–1.052; p < 0.01) were independent predictors for PNR to IFX (Table 3). IUS parameters (BWTT0, △BWT, △IBUS-SAS) were analyzed, respectively, with VFAT0 and the results showed that BWTT0 + VFAT0 predicts PNR to IFX with 65.5% sensitivity, 96.8% specificity, and 0.832 AUC (95% CI, 0.754–0.909); △BWT + VFAT0 has 79.7% sensitivity, 77.4% specificity, and 0.841 AUC (95% CI, 0.756–0.926); and △IBUS-SAS + VFAT0 has 78.3% sensitivity, 77.4% specificity, and 0.830 AUC (95% CI, 0.753–0.917). The AUC of BWTT0 + VFAT0, △BWT + VFAT0, and △IBUS-SAS + VFAT0 in predicting PNR to IFX were significantly higher than that of a single index, with no significant difference among the three combinations (p > 0.05, as shown in Figure 4).
Logistic regression analysis showing IUS parameters and abdominal fat metrics associated with PNR to IFX at T2.

△, the decrease of BWT and IBUS-SAS at T1 compared with T0; BWT, bowel wall thickness; CI, confidence interval; IBUS-SAS, International Bowel Ultrasound Segmental Activity Score; IFX, Infliximab; IUS, intestinal ultrasound; MFI, mesenteric fat index; OR, odds ratio; PNR, primary non-response; SFA, subcutaneous fat area; TFA, total fat area; VFA, visceral fat area.

ROC curves for predicting PNR to IFX at T2 based on IUS parameters and abdominal fat metrics in patients with CD. (A) ROC curves of individual IUS parameters and abdominal fat metrics. (B) ROC curves of combinations of IUS parameter with VFA. (C) ROC curves of △BWT, alone and combined with VFA.
Sensitivity analysis regarding biologic exposure status
The sensitivity analysis was conducted to evaluate the robustness of the primary findings. Univariate analysis and ROC curve analysis in the biologic-naïve and biologic-experienced groups both demonstrated results consistent with the primary analysis. Notably, in the biologic-naïve group, BWTT0 (OR, 2.688; 95% CI, 1.530–4.723; p < 0.001), △BWT (OR, 4.755; 95% CI, 2.099–10.774; p < 0.001), and VFAT0 (OR, 1.030; 95% CI, 1.006–1.055; p = 0.015) were independent predictors for PNR to IFX. The AUC of △BWT + VFAT0 is 0.835 (95% CI, 0.737–0.933) in predicting PNR to IFX, significantly higher than that of a single index, with no significant difference among the three combinations (p > 0.05, as shown in Figure 5). In the biologic-experienced group, univariate logistic model showed that higher BWTT0 and VFAT0, and lower △BWT were associated with a higher rate of PNR. The AUC of △BWT + VFAT0 is 0.839 (95% CI, 0.667–0.974).

ROC curves for predicting PNR to IFX at T2 based on BWT and VFA in biologic-naïve (A) and biologic-experienced (B) patients with CD.
Discussion
In this prospective study of patients with active CD undergoing IFX treatment, our study first investigated the value of IUS combined with VAT in predicting PNR to IFX in CD patients. Our study found that 31% of patients exhibited PNR after 14 weeks of IFX treatment, consistent with previous study. 4 The decrease of BWT at week 2–4 after IFX treatment combined with baseline VFA may more accurately predict PNR to IFX with an AUC of 0.841, superior to individual metrics and other combinations, with the expectation of appropriately selecting patients who would benefit the most from early IFX treatment and guiding individualized clinical practice.
In our study, most IUS parameters, especially BWT normalized within the first 14 weeks of IFX treatment. Baseline BWT predicted PNR at week 14 with an AUC of 0.741 and a cutoff of 6.3 mm, consistent with previous reports. 23 The early change in BWT (△BWT) after 2–4 weeks was also predictive (AUC 0.797), and its value exceeded that of baseline BWT alone, indicating that early improvement in BWT may be more informative than the baseline value for short-term outcome prediction. Of note, the observed decrease in BWT (14.3%) was slightly lower than that reported in other studies (18.0%), 16 which may relate to differences in outcome definitions and timing of assessment.
Elevated BWT, as measured by IUS, directly reflects the severity of transmural inflammation. Patients with higher baseline BWT likely harbor a greater inflammatory burden, characterized by increased local production of pro-inflammatory cytokines, especially TNF-α, the target of IFX. This heightened “TNF-α load” may exceed the neutralizing capacity of standard IFX induction dosing. 24 Furthermore, severe and chronic inflammation can promote fibrotic remodeling, a process less responsive to anti-TNF-α therapy. 25 Thus, more severe baseline inflammation raises the threshold for achieving an early response to standard IFX regimens, explaining why these patients are over-represented in the PNR group and may require treatment intensification, such as dose optimization, interval shortening, or earlier switch to alternative therapies.
Several ultrasonography-based activity scores have been developed to assist in managing patients with IBD, monitor disease activity, and treatment response effectively. Among these scores, IBUS-SAS demonstrated good performance in the evaluation of response and therapeutic efficacy.26–28 Huang et al. 29 demonstrated that IBUS-SAS was responsive to the treatment of IFX, showing a significant decrease after 14–26 and 44–56 weeks. However, there are few studies on predicting PNR to IFX in the early treatment of CD, and our study partially addresses this gap to some extent. We found that baseline IBUS-SAS can predict PNR to IFX at week 14 with an AUC of 0.758, higher than the predictive efficacy of baseline BWT and the 20.6% decrease of IBUS-SAS (0.758 vs 0.741 vs 0.731). We consider this reasonable, because the IBUS-SAS score is a comprehensive scoring system, and BWT is only a part of it, which may provide a richer understanding of bowel lesions at baseline, while cannot directly and simply reflect the degree of improvement after treatment, especially when predicting the short-term efficacy of drugs.
CT appears more specific to identify visceral and subcutaneous adipose tissue. Thus, the use of single slice at L3 in CT appears to be the most robust technique for estimating abdominal fat parameters in patients with CD. Grillot et al. 30 and Sehgal et al. 11 found that visceral obesity was associated with adverse consequences and recurrence in patients with CD, and could independently predict the recurrence time of CD. Our previous study also demonstrated that VFA could predict the efficacy of IFX in CD patients at 6 and 12 months. 7 In this study, we found that patients with high VFA at baseline were more likely to have a loss of response. VFA was an independent predictor of early IFX efficacy in CD patients with an AUC of 0.749. This may be because patients with high VFA produce increased TNF-α without a corresponding increase in the dose of IFX administered, resulting in poor therapeutic efficacy. Studies have found that the increase of SFA or MFI was associated with poor efficacy of early induction therapy,9,11 but this phenomenon was not observed in our study, which might be due to the difference in the included population and our small sample size.
Although both IUS parameters and VFA could predict the efficacy of IFX in CD patients, the AUC was around 0.700. Therefore, we firstly investigated their complementary to predict PNR to IFX in CD patients and found that △BWT integrated with baseline VFA achieved the highest AUC of 0.841 (95% CI, 0.756–0.926), which was marginally superior to △BWT alone (AUC 0.797) and to other combinations such as BWTT0 combined with VFAT0 (ROC, 0.832; 95% CI, 0.754–0.909) and △IBUS-SAS combined with VFAT0 (ROC, 0.830; 95% CI, 0.753–0.917). These results suggest the improvement of bowel wall thickness in the early stage of treatment may have more predictive value than baseline measurements alone. There was no statistical difference among the three combinations, which might be related to our smaller sample size, underscoring the need for validation in larger cohorts.
Importantly, while the addition of VFA slightly enhanced predictive performance, the absolute gain in AUC was modest. In clinical practice, △BWT obtainable from routine, non-invasive IUS, remains a robust and radiation-free predictor for treatment response. 31 The incorporation of VFA should thus be viewed as an adjunctive tool, particularly relevant for patients who have already undergone CT imaging for other clinical indications. We do not advocate additional CT scans solely for VAT assessment, given the associated radiation exposure. Instead, our findings highlight that in settings where CT data are already available, VFA may offer incremental prognostic information, particularly for risk stratification in patients with high visceral adiposity.
Previous studies have indicated that the efficacy of IFX treatment in patients failed other biological agents has diminished due to alterations in the immune system, the presence of anti-drug antibodies, other adaptive changes, or the complexity of the disease.32,33 Therefore, in our study, patients were also categorized into the biologic-naïve and biologic-experienced groups. We found after 14 weeks of IFX treatment, the biologic-naïve group exhibited a greater response rate than the biologic-experienced group, albeit the difference lacked statistical significance. And the degree of improvement in BWT and IBUS-SAS for the biologic-naïve group was significantly higher than that of the biologic-experienced group, indicating that the biologic-naïve patients exhibited heightened sensitivity to IFX and demonstrated superior enhancement in IUS parameters along with a more favorable therapeutic outcome trajectory, similar to previous studies.34,35 Nonetheless, the limited sample size of our study, particularly the scant number of biologic-experienced patients, necessitates a larger cohort in future research to evaluate the efficacy of IUS in patients initiating biologic therapy and those with prior biologic exposure.
We additionally studied the quality of life and fatigue of patients before and after IFX treatment. CD exerts considerable psychological and physiological impacts. Approximately 35% of patients experience anxiety, 22% depression, and up to 50% fatigue. 36 In our study, the scores of IBDQ and FACIT-F in the response group were considerably superior to those in the non-response group after 14 weeks, regardless of prior biologic exposure, indicating that there was a significant improvement of quality of life and fatigue along with the clinical response.
The study presented several limitations. The analysis was conducted in a relatively small population at a single center and only included patients who received IFX treatment. Furthermore, the predominantly male composition of our cohort (84% male) may limit the generalizability of our findings to female patients. Future studies with balanced sex representation are warranted. In addition, this single-center study did not employ centralized reading for IUS, or CT-based visceral fat measurements, which could potentially introduce inter-observer variability despite our internal quality control procedures. Future studies with balanced sex representation, larger cohorts, multicenter designs, and centralized imaging review are warranted to validate and generalize our findings, including their applicability to other therapies.
Additionally, our study did not assess the role of IUS in predicting the long-term effect of IFX treatment over a period of 6 months to 1 year, and our group will conduct further investigations in the future. Furthermore, the outcomes of the IUS examination depend on the operator’s expertise and the patient’s intestinal conditions, including factors such as obesity, which may result in variability in the examination results.
Conclusion
The early change in BWT assessed by IUS serves as a robust and radiation-free predictor for identifying PNR to infliximab in patients with CD. While the combination of △BWT with baseline VFA yielded the numerically highest AUC, the addition of baseline VFA provides only marginal improvement in predictive accuracy and should be regarded as an adjunctive tool due to its radiation exposure. These findings support the use of early change in BWT on IUS, alone or in combination with existing VFA measurements, for allowing stratification of patients likely beneficial from infliximab therapy and guiding individualized clinical practice, without recommending additional CT imaging solely for adipose tissue assessment.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251413366 – Supplemental material for Does intestinal ultrasound combined with visceral adipose tissue more accurately predict primary non-response to infliximab in patients with Crohn’s disease? A prospective, observational study
Supplemental material, sj-docx-1-tag-10.1177_17562848251413366 for Does intestinal ultrasound combined with visceral adipose tissue more accurately predict primary non-response to infliximab in patients with Crohn’s disease? A prospective, observational study by Mingmei Ye, Panpan Zhao, Feng Gao, Ying Mei, Rujing Xiong, Yuanyuan Huang, Xiayu Mao, Tianjing Meng and Li Tian in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pdf-2-tag-10.1177_17562848251413366 – Supplemental material for Does intestinal ultrasound combined with visceral adipose tissue more accurately predict primary non-response to infliximab in patients with Crohn’s disease? A prospective, observational study
Supplemental material, sj-pdf-2-tag-10.1177_17562848251413366 for Does intestinal ultrasound combined with visceral adipose tissue more accurately predict primary non-response to infliximab in patients with Crohn’s disease? A prospective, observational study by Mingmei Ye, Panpan Zhao, Feng Gao, Ying Mei, Rujing Xiong, Yuanyuan Huang, Xiayu Mao, Tianjing Meng and Li Tian in Therapeutic Advances in Gastroenterology
Footnotes
Appendix
Acknowledgements
We thank all patients who provided data for this study and approved the publication of its findings.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
