Abstract
Background:
Gastric outlet obstruction (GOO) caused by malignancy significantly impairs patient quality of life. Surgical gastrojejunostomy (SGJ), endoscopic stenting (ES), and endoscopic ultrasound-guided gastroenterostomy (EUS-GE) are the three main palliative treatments. However, the optimal approach remains unclear because of variations in study results and limited studies comparing treatment types.
Objectives:
To evaluate and compare the effectiveness and safety of EUS-GE, ES, and SGJ in the management of GOO.
Design:
Network meta-analysis.
Data sources and methods:
The PubMed, Cochrane Library, and Web of Science databases were systematically searched for full-length articles of randomized controlled trials and cohort studies in English comparing SGJ, ES, and EUS-GE in adult patients with malignant GOO. Studies with unbalanced baseline characteristics or technical variations within a single modality were excluded. The primary outcome was the reintervention rate for recurrent GOO. Secondary outcomes included clinical success and adverse event rates.
Results:
Fifty-four studies involving 6110 patients were analyzed (SGJ, n = 1974, ES, n = 3226, EUS-GE, n = 910). Compared with ES, both SGJ (odds ratio (OR): 0.32, 95% confidence interval (CI): 0.22–0.46) and EUS-GE (OR: 0.29, 95% CI: 0.17–0.51) significantly reduced the risk of reintervention. EUS-GE achieved a higher clinical success rate than ES (OR: 2.46, 95% CI: 1.50–4.04) and also lower adverse event rate than both SGJ (OR: 0.33, 95% CI: 0.19–0.56) and ES (OR: 0.61, 95% CI: 0.37–0.99). Heterogeneity was moderate across outcomes, with no major inconsistency or publication biases detected.
Conclusion:
Of the three treatments for GOO, EUS-GE demonstrated the most favorable profile in terms of efficacy, safety, and durability. SGJ remains a viable alternative, particularly in centers lacking expertise in advanced endoscopy. These findings may inform future clinical guidelines and support the broader adoption of EUS-GE in appropriate settings.
Keywords
Introduction
Gastric outlet obstruction (GOO) is a clinical condition characterized by mechanical blockage at the level of the gastric antrum, pylorus, or duodenum. 1 GOO is frequently associated with symptoms such as nausea, vomiting, dehydration, and an inability to tolerate oral intake, all of which contribute to a marked decline in quality of life. 2 GOO is most frequently attributable to malignant neoplasms originating from the distal stomach, duodenum, or pancreas, 3 although benign etiologies can cause it as well such as peptic ulcer disease, chronic pancreatitis, postoperative anastomotic strictures, Crohn’s disease, and corrosive injury can also lead to the obstruction. The primary therapeutic objectives are to alleviate obstructive symptoms and to provide suitable palliative care.
Surgical gastrojejunostomy (SGJ) has traditionally been the standard intervention to circumvent the site of obstruction. This method is generally effective, but is invasive and may be associated with a relatively high risk of adverse events. 4 Endoscopic stenting (ES) offers a minimally invasive alternative to treat GOO, with lower rates of adverse events and more favorable short-term outcomes, such as reduced length of hospitalization. 5 Compared with SGJ, however, ES is associated with a higher likelihood of reintervention due to the potential for stent occlusion. Endoscopic ultrasound-guided gastroenterostomy (EUS-GE) has recently emerged as an innovative technique that enabling endoscopic bypass of GOO result from both malignant and benign conditions5,6; in this method, a lumen-apposing metal stent (LAMS) is used to create a direct connection between the stomach and the small intestine distal to the obstructive lesion. This approach can minimize the risk of adverse events associated with surgical interventions while offering the sustained patency characteristic of a complete enteral bypass.1,7 While EUS-GE has increasingly been adopted worldwide in recent years, with accumulating evidence supporting its safety and efficacy; however, its implementation requires expertise and a learning curve, as highlighted in recent reports. 8
Although several studies have compared the efficacy and safety of SGJ, ES, and EUS-GE for GOO, few have included large numbers of patients. The present network meta-analysis (NMA) therefore evaluated and compared the effectiveness and safety of EUS-GE, ES, and SGJ in the management of GOO.
Methods
This NMA was conducted and reported in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement. 9 Studies were eligible for inclusion in this NMA if they were randomized controlled trials (RCTs) or prospective/retrospective cohort studies that compared at least two of the three treatment modalities, SGJ, ES, and EUS-GE, in patients with malignant GOO, and reported at least one of the following outcomes: technical success, clinical success, adverse event rate, reintervention rate, or survival time.
Studies that focused solely on benign GOO and those that did not provide extractable data specific to malignant GOO were excluded. Studies were also excluded if they lacked balanced baseline characteristics or appropriate statistical adjustments. Specifically, studies with significantly unbalanced baseline characteristics between groups (e.g., age, performance status, disease stage, comorbidities) without appropriate statistical adjustments such as propensity score matching, were excluded to minimize confounding. In addition, studies were excluded if they did not compare different palliative treatment modalities, if they compared technical variations (e.g., surgical techniques, anastomosis methods, or stent types) within a single treatment modality, if full texts were unavailable or if results were published only as conference abstracts.
Search strategy
Two investigators (T.T. and Y.Y.) independently and comprehensively searched the PubMed, Cochrane Library, and Web of Science databases using the search terms (“gastric outlet obstruction” OR “pyloric stenosis” OR “duodenal obstruction” OR “duodenal obstructions”) AND (“gastroenterostomy” OR “gastroenterostomies” OR “gastric bypass” OR “surgical gastrojejunostomy” OR “endoscopic ultrasound guided gastroenterostomy” OR “endoscopic duodenal stent placement” OR “duodenal stent placement” OR “duodenal stent” OR “endoscopic ultrasound” OR “endoscopic ultrasonography”). The search was limited to full-length articles in English describing studies of adult human subjects. The systematically search of the PubMed, Cochrane Library, and Web of Science databases was with the final search completed in April, 2025. References cited in the original articles and prior systematic reviews were also screened to identify additional relevant studies.
Outcome variables and data extraction
The primary outcome of this NMA was the reintervention rate due to recurrent GOO after treatment for GOO. The secondary outcomes included clinical success rate and early adverse event rate after GOO treatment. Discrepancies or uncertainties between the two investigators (T.T. and Y.Y.) were resolved by consensus decision after group discussions. Study characteristics and statistical data related to the outcomes of interest were recorded.
Statistical analysis and network outcomes
Effect sizes were measured using odds ratios (ORs) with 95% confidence intervals (CIs) for dichotomous outcomes, and mean differences with 95% CIs for continuous outcome. The NMA was conducted to integrate the direct and indirect comparison. Heterogeneity among studies was assessed using I2, with values greater than 50% indicating substantial heterogeneity. A random-effect model was employed to obtain conservative estimates, allowing for any heterogeneity among studies. The NMA was employed in frequentist framework and conducted using the netmeta package (version 2.1–0) in R software (version 4.2.0; R Foundation for Statistical Computing, Vienna, Austria). Results showing value of p < 0.05 were considered statistically significant. 10
The results of the NMA are presented in forest plots and league tables. Treatment effectiveness was ranked using the p score (ranging from 0 to 1), which quantifies the mean extent of certainty that a treatment is better than other treatments; higher p scores correspond to more favorable treatments. For outcomes such as complication rate and reintervention rate, effect sizes were defined in the negative direction, indicating that lower values correspond to better treatment efficacy. Publication bias was assessed using funnel plot. The statistical methods have been double-checked T.T. and K.W. and remain appropriate.
Results
Figure 1 outlines the selection process. The initial database search identified 2419 records; of these, 54 studies met the inclusion criteria, comprising of 4 RCT11–14 and 50 non-RCTs published between 2002 and 2024,2,4,7,15–60 met the inclusion criteria. The characteristics of these studies are presented in Table 1 and Supplemental Table 1. The analyzed studies included a total of 6110 patients, of whom 1974 underwent SGJ, 3226 underwent ES, and 910 underwent EUS-GE.

PRISMA flow diagram showing the study selection process. A total of 2419 records were identified through database searching. After removing 834 duplicates, 1585 studies were screened by title and abstract, and 96 full-text articles were assessed for eligibility. Of these, 42 were excluded because they were review articles or they evaluated only one treatment modality. Ultimately, 54 studies (4 RCTs and 50 non-RCTs) were included in the network meta-analysis.
Summary of 52 studies included in the network meta-analysis.
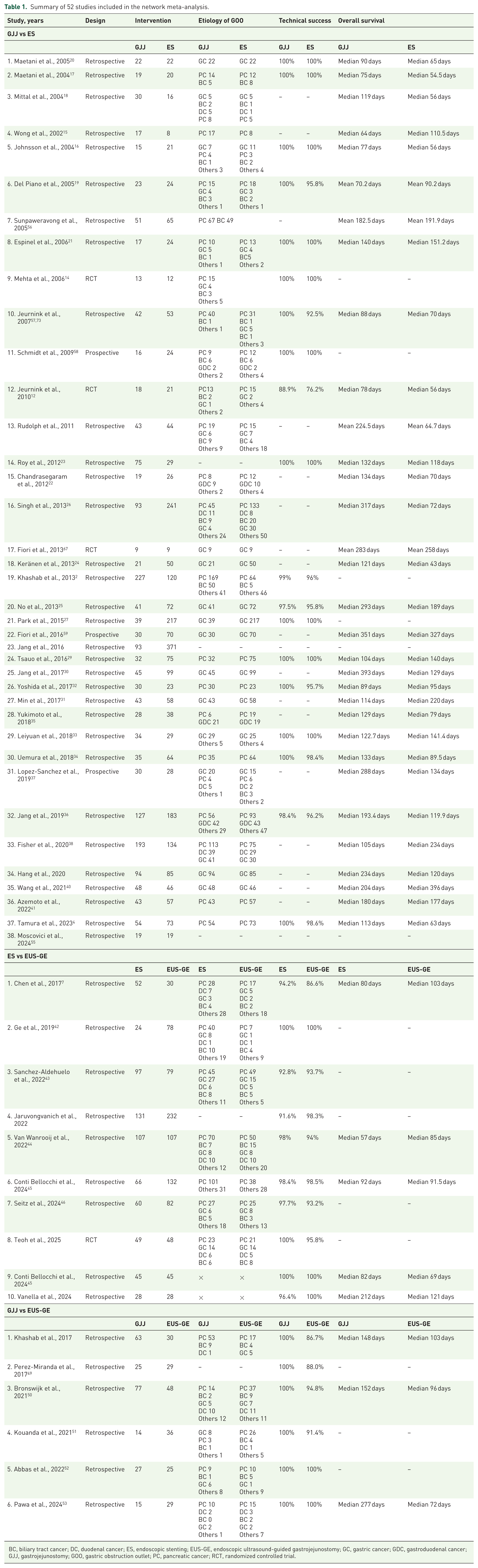
BC, biliary tract cancer; DC, duodenal cancer; ES, endoscopic stenting; EUS-GE, endoscopic ultrasound-guided gastrojejunostomy; GC, gastric cancer; GDC, gastroduodenal cancer; GJJ, gastrojejunostomy; GOO, gastric obstruction outlet; PC, pancreatic cancer; RCT, randomized controlled trial.
Primary outcomes
The risk of reintervention for recurrent GOO was significantly lower in patients who underwent SGJ (OR: 0.32; 95% CI: 0.22–0.46) and EUS-GE (OR: 0.29; 95% CI: 0.17–0.51) than those who underwent ES, although the certainty of evidence was low to very low (Figure 2(b) and (c)). Based on reintervention rates, EUS-GE (p = 0.80) and EUS-GE (p = 0.69) had higher probabilities of being the optimal treatment, with ES having the lowest probability of preventing reintervention (p = 0.00; Figure 2(d)). Heterogeneity within this network was moderate (τ2 = 0.428; I2 = 52.4%; Figure 2(b)). No inconsistency was observed between direct and indirect comparisons, and no publication bias was detected in the network for this outcome (Figure 2(e)). The results of standard pairwise meta-analyses were consistent with those of the NMA.

Network meta-analysis of reintervention rates for recurrent GOO. (a) Network plot of comparisons between treatment modalities. (b) Forest plot showing ORs and 95% CIs for each comparison. (c) League table with pairwise comparisons of reintervention rates. (d) p-Score ranking of the treatment modalities for preventing reintervention. (e) Funnel plot assessing publication bias. EUS-GE and SGJ were associated with significantly lower reintervention rates than ES.
Secondary outcomes
The clinical success rate was significantly higher in patients who underwent EUS-GE than in those who underwent ES (OR: 2.46; 95% CI: 1.50–4.04) and SGJ (OR: 1.96; 95% CI: 1.16–3.31; Figure 3(b) and (c)). EUS-GE had the highest probability of achieving clinical success (p = 0.99), followed by SGJ (p = 0.45), with ES having the lowest probability (p = 0.05; Figure 3(d)). The heterogeneity in this network was moderate (τ2 = 0.305; I2 = 44.1%; Figure 3(b)). No significant inconsistency or publication bias was detected. The findings of standard pairwise meta-analyses were consistent with those of the NMA (Figure 3(e)).
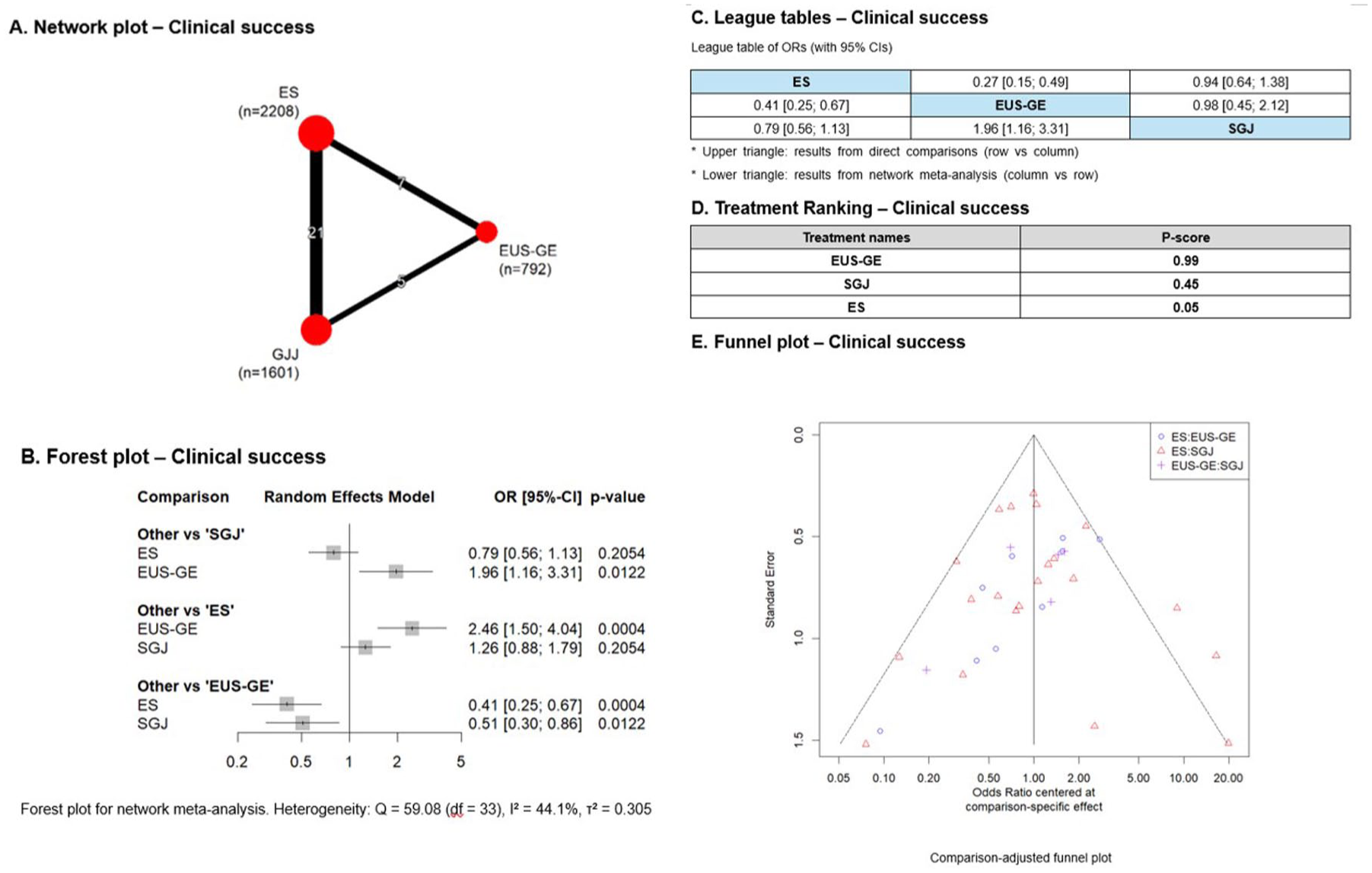
Network meta-analysis of clinical success rates. (a) Network plot of included studies. (b) Forest plot comparing clinical success rates across treatments. (c) League table of clinical success. (d) p-Score ranking based on clinical success. (e) Funnel plot for publication bias. EUS-GE showed the highest probability of achieving clinical success.
The risk of adverse events was significantly lower in patients who underwent EUS-GE than in those who underwent SGJ (OR: 0.33; 95% CI: 0.19–0.56) and ES (OR: 0.61; 95% CI: 0.37–0.99; Figure 4(b) and (c)), and was lower in patients who underwent ES than SGJ (OR: 0.54; 95% CI: 0.37–0.79). EUS-GE had the highest probability of minimizing adverse events (p = 0.98), followed by ES (p = 0.51), with SGJ having the lowest probability (p = 0.00; Figure 4(d)). The heterogeneity in this network was moderate (τ2 = 0.465; I2 = 50.6%; Figure 4(b)). No significant inconsistency or publication bias was detected (Figure 4(e)). The findings of standard pairwise meta-analyses were consistent with those of the NMA.

Network meta-analysis of adverse event rates. (a) Network plot of study comparisons. (b) Forest plot showing ORs and 95% CIs for adverse events. (c) League table showing pairwise comparisons. (d) p-Score ranking of safety profiles. (e) Funnel plot assessing publication bias. EUS-GE was associated with the lowest risk of adverse events, followed by ES and SGJ.
The present NMA attempted to compare the technical success rates between EUS-GE, ES, and SGJ. All three procedures, however, demonstrated uniformly high success rates, resulting in insufficient variability to allow for a meaningful network comparison. Although attempts were made to compare post-procedural overall survival between the three groups, the substantial heterogeneity in patient populations, study designs, and subsequent treatments precluded meaningful comparisons.
Discussion
This systematic review and NMA provided a comprehensive comparison of three current treatment strategies for GOO; SGJ, ES, and EUS-GE. Parameters compared included rates of reintervention rates for recurrence of GOO, along with rates of adverse events and clinical success. EUS-GE demonstrated the most favorable outcomes across all three endpoints, with a significantly higher probability of achieving clinical success (p = 0.99) than either SGJ (p = 0.45) or ES (p = 0.06). EUS-GE was associated with lower adverse event rates than either ES or SGJ. The reintervention rate for recurrence of GOO was comparable between patients who underwent EUS-GE and SGJ, but lower than in those who underwent ES. These findings suggest that EUS-GE represents the most balanced and effective treatment option for GOO. Contrasting with previous meta-analyses, which were limited to pairwise comparisons, this NMA approach enabled a comprehensive evaluation across all three treatment modalities.1,5,61–80
The major limitations of EUS-GE are its technical complexity and limited availability. 81 EUS-GE is performed in a stepwise manner by advancing a nasobiliary or feeding tube beyond the gastric or duodenal stenosis to infuse saline, thereby enabling EUS visualization of the distended proximal jejunal loop. A cautery-enhanced LAMS is subsequently deployed, with the distal flange first inserted into the jejunum, and the proximal flange into the stomach, to create a gastroenteric anastomosis. 44 Leakage or perforation may occur, however, due to the mobility of the jejunal loop. Performing EUS-GE requires not only expertise in interventional EUS techniques but also specialized equipment, restricting its use to high-volume centers with experienced endoscopists. Therefore, the favorable outcomes associated with EUS-GE in the present meta-analysis may thus reflect results from expert centers, limiting the generalizability of these findings. The EUS-guided double-balloon occluded gastrojejunostomy bypass (EPASS) technique, which involves occlusion of the proximal small bowel loop with a double-balloon catheter, has enabled the safe and consistent performance of this procedure. A recent global multicenter RCT demonstrated that EUS-GE using EPASS technique offers the advantage of stabilizing the jejunal loop, thereby reducing the risk of stent misdeployment, fistula, or perforation. 13
Of the three treatments evaluated in the present study, ES showed the lowest clinical success rate for GOO and the highest rate of reintervention rate for recurrent GOO. ES is a generally accepted, minimally invasive, and technically simple procedure with advantages that include early resumption of oral intake and shorter duration of hospitalization. 4 The long-term utility of ES, however, is limited by the high risk of stent obstruction due to tumor ingrowth or migration, resulting in frequent reinterventions for GOO.
SGJ, a conventional surgical approach, demonstrated a moderate clinical success rate and the highest incidence of adverse events among the three treatments. Despite its invasive nature of the treatment and the association with longer recovery times, SGJ is a well-established procedure that offering long-term patency with a relatively low risk of reintervention. SGJ, therefore, remains a viable treatment option, particularly for patients with good performance status and a high expected long-term survival rate. Compared with traditional open surgery, the use of laparoscopic techniques has reduced the invasiveness of SGJ, improving postoperative recovery while maintaining durability.
Current treatment recommendations for malignant GOO vary internationally. The National Comprehensive Cancer Network and the National Institute for Health and Care Excellence guidelines recommend SGJ for patients with an expected survival exceeding 3 months. Similarly, Japanese clinical practice guidelines for pancreatic cancer recommend surgical SGJ in patients with duodenal obstruction who are expected to show relatively good long-term prognosis.4,5,82 On the other hand, the European Society for Medical Oncology and Korean guidelines emphasize the short-term benefits of ES. Recently updated guidelines from the European Society for Gastrointestinal Endoscopy recommended EUS-GE as an alternative to ES or surgical SGJ in expert settings. 11 Similarly, the present study provides strong evidence supporting the clinical utility of EUS-GE. A recent decision tree analysis of procedure selection demonstrated that SGJ resulted in worse oral intake at 1 month than ES and EUS-GE, mainly due to postoperative gastroparesis after SGJ. However, oral intake after ES declined over time because of stent dysfunction, becoming inferior to EUS-GE and SGJ from 3 months onward. By 5–6 months, SGJ and EUS-GE achieved better sustained oral intake than ES, with EUS-GE showing similar mid-term efficacy to SGJ and better early tolerance. 64
None of the prospective RCTs in the present NMA compared EUS-GE and SGJ. Prospective studies directly comparing these two treatment modalities thus appear warranted. Studies are also needed to evaluate long-term outcomes, including patient quality of life, nutritional status, and feasibility of chemotherapy. The broader adoption of EUS-GE will require the establishment of structured training programs and institutional support for equipment.
This study showed several inherent limitations. The study was not registered in PROSPERO or any other protocol registry before implementation, which may have affected the transparency and reproducibility. First, while the inclusion of non-RCTs expanded the number of patients included, their inclusion may have introduced potential bias due to residual confounding, despite the exclusion of studies with significantly unbalanced baseline characteristics. Second, the evidence base for EUS-GE remains limited, as only one RCT was available at the time of analysis, and most included data were derived from retrospective cohorts. Third, technological advances and procedural refinements over time likely influenced outcomes; and fully adjusting for the impact of evolving techniques was impossible. In addition, the secondary outcomes showed moderate to substantial heterogeneity despite stratification. The effects of anastomosis configuration, stent type, and procedural approach (e.g., laparoscopic vs open SGJ or different EUS-GE techniques) could not be evaluated because of insufficient information in previous reports. Cost-effectiveness and patency duration, as clinically meaningful endpoints, also could not be assessed. Lastly, definitions and grading systems for complications differ in the surgical and endoscopic literature, making direct comparisons challenging. However, data were synthesized across both modalities to ensure a comprehensive assessment. In addition, operator experience and center volume may affect EUS-GE outcomes. As the procedure is technically demanding, greater expertise and higher institutional volume are likely associated with improved success and safety. Future studies should evaluate learning curves to optimize outcomes. The search strategy was restricted to full-length articles published in English, which may have introduced language bias and excluded potentially relevant studies from non-English-speaking regions.
Conclusion
In summary, this NMA provides the most comprehensive comparison to date of the three primary palliative interventions for malignant GOO: ES, SGJ, and EUS-GE. EUS-GE demonstrated superior clinical success and safety profiles, along with a low reintervention rate, suggesting its potential role as a preferred approach. SGJ remains a valid option, particularly in institutions lacking advanced endoscopic capabilities. As EUS-GE continues to evolve, well-designed prospective studies comparing this method with surgical approaches in various clinical settings will be critical to inform future guidelines and improve patient outcomes.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251410800 – Supplemental material for A network meta-analysis on the optimal treatment of malignant gastric outlet obstruction
Supplemental material, sj-docx-1-tag-10.1177_17562848251410800 for A network meta-analysis on the optimal treatment of malignant gastric outlet obstruction by Takashi Tamura, Ke Wan, Reiko Ashida, Yasunobu Yamashita, Yuki Kawaji, Masahiro Itonaga and Masayuki Kitano in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251410800 – Supplemental material for A network meta-analysis on the optimal treatment of malignant gastric outlet obstruction
Supplemental material, sj-docx-2-tag-10.1177_17562848251410800 for A network meta-analysis on the optimal treatment of malignant gastric outlet obstruction by Takashi Tamura, Ke Wan, Reiko Ashida, Yasunobu Yamashita, Yuki Kawaji, Masahiro Itonaga and Masayuki Kitano in Therapeutic Advances in Gastroenterology
Footnotes
Appendix
Acknowledgements
The authors thank all the members of the Second Department of Internal Medicine and the Clinical Study Support Center at Wakayama Medical University.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
