Abstract
Background:
The symptoms of gastric outlet obstruction have traditionally been managed surgically or endoscopically. Enteral stenting (ES) is a less invasive endoscopic treatment strategy for this condition. Endoscopic ultrasound-guided gastroenterostomy (EUS-GE) has recently become a potential alternative technique.
Objectives:
We conducted a systematic review and meta-analysis of the effectiveness and safety profile of EUS-GE compared with ES.
Design:
Meta-analysis and systematic review.
Data sources and methods:
We searched multiple databases from inception to August 2023 to identify studies that reported the effectiveness and safety of EUS-GE compared with ES. The outcomes of technical success, clinical success, and adverse events (AEs) were evaluated. Pooled proportions were calculated using both fixed and random effects models.
Results:
We included 13 studies with 1762 patients in our final analysis. The pooled rates of technical success for EUS-GE were 95.59% [95% confidence interval (CI), 94.01–97.44, I2 = 32] and 97.96% (95% CI, 96.06–99.25, I2 = 63) for ES. The pooled rate of clinical success for EUS-GE was 93.62% (95% CI, 90.76–95.98, I2 = 54) while for ES it was lower at 85.57% (95% CI, 79.63–90.63, I2 = 81). The pooled odds ratio (OR) of clinical success was higher for EUS-GE compared to ES at 2.71 (95% CI, 1.87–3.93). The pooled OR of clinical success for EUS-GE was higher compared to ES at 2.72 (95% CI, 1.86–3.97, I2 = 0). The pooled rates of re-intervention for EUS-GE were lower at 3.77% (95% CI, 1.77–6.46, I2 = 44) compared with ES, which was 25.13% (95% CI, 18.96–31.85, I2 = 69). The pooled OR of the rate of re-intervention in the ES group was higher at 7.96 (95% CI, 4.41–14.38, I2 = 13). Overall, the pooled rate for AEs for EUS-GE was 8.97% (95% CI, 6.88–11.30, I2 = 15), whereas that for ES was 19.63% (95% CI, 11.75–28.94, I2 = 89).
Conclusion:
EUS-GE and ES are comparable in terms of their technical effectiveness. However, EUS-GE has demonstrated improved clinical effectiveness, a lower need for re-intervention, and a better safety profile compared to ES for palliation of gastric outlet obstruction.
Keywords
Introduction
Malignant gastric outlet obstruction (GOO) is a significant complication that can result from both malignant and benign conditions that affect the gastric antrum, duodenum, and pancreas, with symptoms including nausea, vomiting, dehydration, and inability to tolerate oral intake, thus reducing the quality of life. 1 Malignant GOO is associated with poor prognosis, with a median survival of 3–6 months. 2 The primary objectives for patients with GOO are to alleviate obstructive symptoms and administer palliative care. 3 GOO symptoms have traditionally been managed with enteral stenting (ES) using self-expandable metal stents (SEMS) or surgical gastrojejunostomy (SGJ).4,5 Palliation of symptoms is challenging in patients who are not eligible for surgical treatment. Surgical gastroenterostomy has high efficacy but is associated with significant morbidity and mortality. ES remains an alternative, less invasive endoscopic management strategy. Studies have shown good clinical success and lower morbidity with ES. 6 Nevertheless, its efficacy is limited in the long term, given recurrent obstruction due to stent migration or tumor infiltration. 7 EUS-GE is a novel, minimally invasive technique that involves the placement of a lumen-apposing metal stent (LAMS) between the gastric body and proximal jejunum, bypassing the obstruction under EUS and fluoroscopic guidance. 8 Previous studies have reported comparable effectiveness of EUS-GE to ES with lower rates of recurrence of symptoms given the lesser risk of tumor ingrowth or overgrowth. 3 The European Society of Gastrointestinal Endoscopy recommends EUS-GE as an alternative to ES or surgical gastroenterostomy when performed in an expert setting. 9 However, data remain limited when comparing the effectiveness and long-term outcomes of EUS-GE and ES. 10
We conducted a systematic review and updated meta-analysis to examine the effectiveness of EUS-GE in terms of technical and clinical success, rates of reintervention, and safety profile in comparison to ES. We hypothesized that EUS-GE and ES have comparable effectiveness and safety profiles. 11
Methods
The study was reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. 12
Search methodology
A literature search was conducted using multiple databases, including Cochrane, Ovid, MEDLINE, and Google Scholar, from inception to August 2023, to identify studies that reported the effectiveness and safety of EUS-GE compared to ES. Keywords used were, ‘Endoscopic ultrasound’, ‘endoscopic ultrasound-guided gastroenterostomy’, ‘EUS-GE’, ‘enteral stenting’, ‘ES’, ‘Duodenal self-expanding metal stents’, ‘D-SEMS’, ‘Gastric outlet obstruction’, ‘GOO’. The retrieved studies were carefully examined to exclude potential duplicate studies. Detailed search strategies are provided in Supplemental Appendix 1.
Study eligibility
Published studies were eligible if they reported a comparison of endoscopic ultrasound-guided gastroenterostomy (EUS-GS) versus ES. The studies included full manuscripts and published abstracts. Articles that did not meet the inclusion criteria were excluded. The exclusion criteria were as follows: (1) studies reporting ES or EUS-GE individually, (2) studies reporting a comparison of EUS-GE to surgical gastroenterostomy, (3) case reports and case series, (4) studies not published in English, (5) studies performed in pediatric populations, and (6) studies performed in animal models. The studies meeting the inclusion criteria were independently reviewed as full-text articles by two authors (M.A. and S.R.P.). Discrepancies were resolved through consensus.
Data extraction and quality assessment
The following data were independently abstracted by two authors (M.A. and S.R.P.) into a standardized form: study characteristics (primary author, period of study, type of study), study design, baseline characteristics of patients (total number of patients included, patient demographics), intervention details (successful procedure, indications of procedure), and outcomes [resolution of symptoms, need for re-intervention, adverse events (AEs)]. The Newcastle–Ottawa scale for cohort studies was used to assess the quality of studies. 13 This quality score consisted of eight questions, the details of which are provided in Supplemental Table 1.
Outcomes evaluated
We assessed the following outcomes in our meta-analysis: (1) pooled rates of technical success defined as proper stent positioning as reported by the authors, (2) pooled rates of clinical success defined by the ability to tolerate oral intake without vomiting, (3) pooled rates of re-intervention defined as the need to perform a repeat procedure, (4) pooled rates of overall AEs, and (5) pooled rates of major AE as defined by the ASGE (American Society for Gastrointestinal Endoscopy) lexicon for endoscopic AEs and AE subtypes.11,14 The AEs evaluated were perforation, stent migration, clinically relevant bleeding requiring intervention and/or blood transfusion, cholangitis, postprocedural pain, and death.
Statistical analysis
Microsoft Excel was used for data collection. Meta-analysis was performed by calculating the pooled proportions. Individual study proportions were transformed into quantities using the Freeman–Tukey variant of the arcsine square-root transformed proportion. The pooled proportion was calculated as the back-transform of the weighted mean of the transformed proportions, using inverse arcsine variance weights for the fixed-effects model and DerSimonian–Liard weights for the random-effects model. Forest plots were constructed to show the point estimates for each study in relation to the summary of the pooled estimates. The width of the point estimates in the forest plots indicates the weight assigned to that study. Heterogeneity among the studies was tested using I2 statistics and the Cochrane Q test based on inverse variance weights. I2 values of 0–39% were considered non-significant heterogeneity, 40–75% moderate heterogeneity, and 76–100% considerable heterogeneity. If the p value is >0.10, it rejects the null hypothesis that the studies are heterogeneous. The effect of publication and selection bias on the summary estimates was tested by both the Harbord–Egger bias 14 indicator and the Begg–Mazumdar bias indicator. 15 Funnel plots were constructed to assess potential publication bias.16,17 The agreement between the reviewers was calculated using Cohen’s k. Microsoft Excel 2019 (Microsoft, Redmond, WA, USA) was used to perform statistical analyses. 18
Results
The initial search identified 170 articles, of which 80 were selected and reviewed after title and abstract evaluation. Data were extracted from 13 studies (N = 1762) that met the inclusion criteria.3,19–30 A PRISMA flowchart describing the details of the review process is shown in Figure 1. The characteristics of the included studies are presented in Table 1. All included cohort studies had a retrospective design, except for one that was prospective. Six studies were published as full manuscripts,3,19–23 whereas seven studies were published as abstracts. Seven studies were multicenter studies and six were single-center studies. All pooled estimates given are calculated using the random-effects model. The agreement between reviewers was 1.0, as measured by Cohen’s k.

Preferred reporting items for Systematic Reviews and Meta-analysis flow diagram for detailing the review process.
Basic study characteristics.

A, ampullary; B, biliary; D, duodenum; EUS-GE, endoscopic ultrasound-guided gastroenterostomy; ES, enteral stenting; G, gastric; M; metastatic; NR, not reported; P, pancreatic; WEST, wireless EUS-GE simplified technique.
A total of 891 patients underwent EUS-GS and 871 patients underwent ES. Our study included 916 male patients and 846 female patients. Malignant GOO was the most common etiology requiring intervention, with benign etiology as the cause in a small proportion of patients.
The analysis showed that the overall pooled rate of technical success for EUS-GE was 95.59% (95% CI, 94.01–97.44, I2 = 32) compared to 97.96% (95% CI, 96.06–99.25, I2 = 63) for ES. The pooled OR of technical success for EUS-GE compared to ES was 0.65 (95% CI, 0.34–1.22, I2 = 0). Figure 2 shows the forest plot comparing the OR of overall technical success. The pooled rate of clinical success for EUS-GE was 93.62% (95% CI, 90.76–95.98, I2 = 54) while for ES it was 85.57% (95% CI, 79.63–90.63, I2 = 81). Pooled OR of clinical success for EUS-GE was higher compared to ES at 2.72 (95% CI, 1.86–3.97, I2 = 0). Figure 3 shows the forest plot comparing the OR of clinical success. Figure 4 represents the funnel plot to assess the publication bias.

Forest plot showing the OR of technical success for EUS-GE and ES.

Forest plot showing the OR of clinical success for EUS-GE and ES.
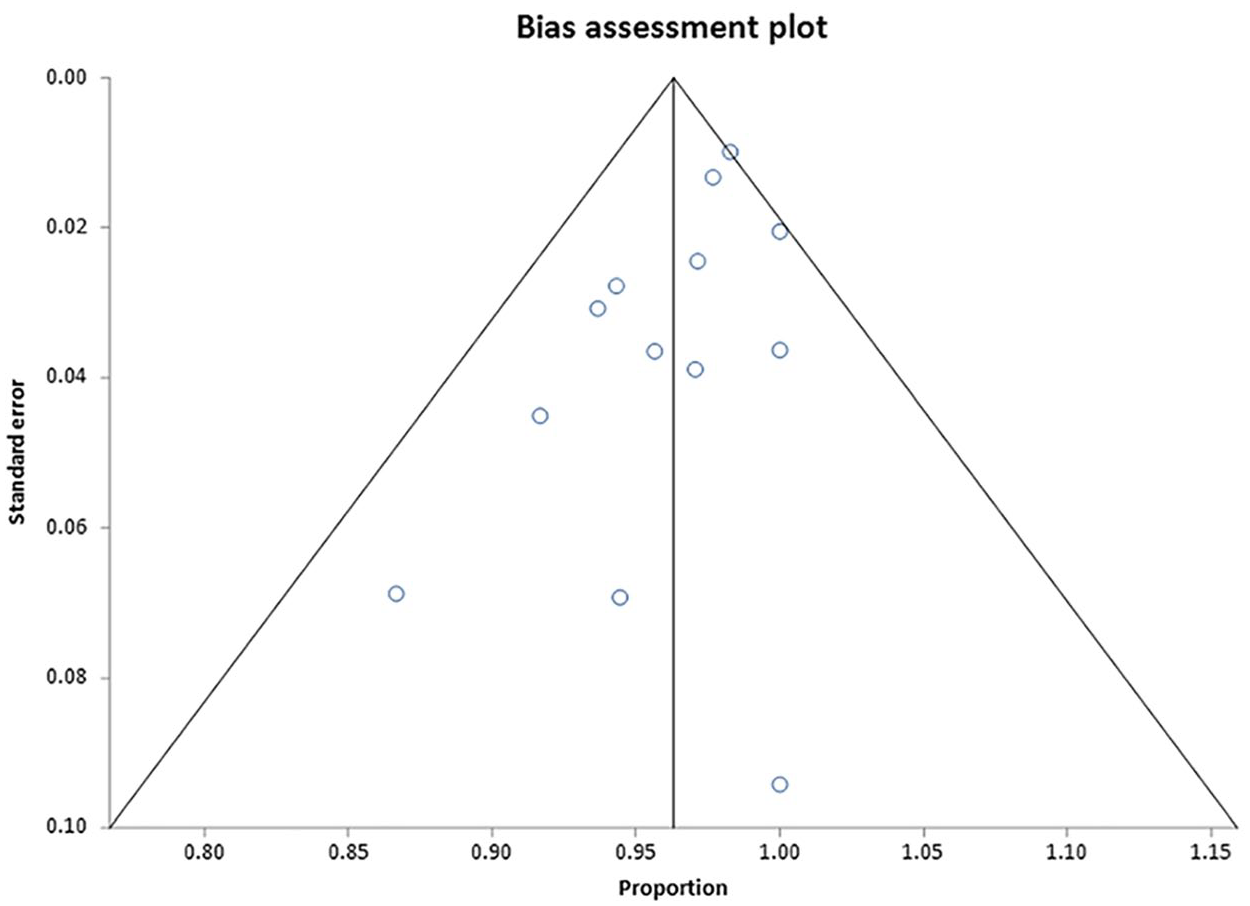
Funnel plot of publication bias on clinical success.
In this analysis, the pooled rates of re-intervention were lower for EUS-GE at 3.77% (95% CI, 1.77–6.46, I2 = 44) compared to ES at 25.13% (95% CI, 18.96–31.85, I2 = 69). The pooled OR of the rate of re-intervention for ES was higher than that for EUS-GE at 7.96 (95% CI, 4.41–14.38, I2 = 13). Figure 5 shows the forest plot of the pooled OR for reintervention.
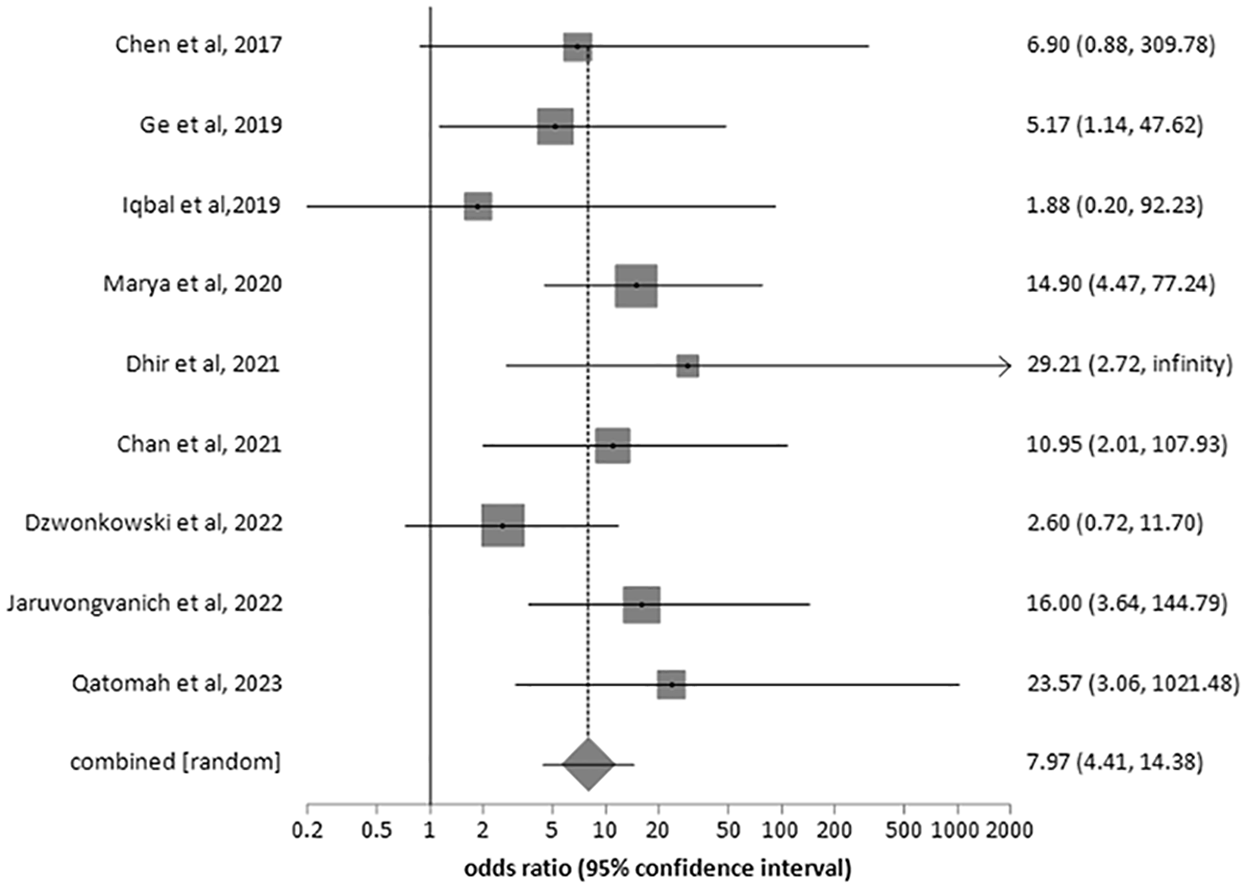
Forest plot showing OR of reintervention for ES and EUS-GE.
The estimated pooled rate for overall AEs for EUS-GE was 8.97% (95% CI, 6.88–11.30, I2 = 15), while for ES it was 19.63% (95% CI, 11.75–28.94, I2 = 89). Similarly, the pooled OR of AEs for ES was higher in comparison to EUS-GE at 2.60 (95% CI, 1.53–4.41, I2 = 58). The forest plot showing the odds ratio (OR) of AEs is shown in Figure 6.

Forest plot showing OR of AEs for ES and EUS-GE.
The estimated pooled rates of major AEs were similar for EUS-GE at 3.62% (95% CI, 2.16–5.43, I2 = 7) and ES at 3.62% (95% CI, 1.40–6.84, I2 = 67). The pooled OR of major AEs for EUS-GE compared to ES was 1.08 (95% CI, 0.46–2.54, I2 = 32). The Begg–Mazumdar bias indicator yielded Kendall’s tau b value of 0.44 (p = 0.11), suggesting no publication bias. Figure 7 shows the forest plot of the OR of major AEs.

Forest plot showing OR of major adverse events for ES and EUS-GE.
Discussion
Endoscopic placement of a self-expanding metal stent was first described in 1992 31 and has been widely used as an alternative approach for the management of malignant GOO, especially in poor surgical candidates. SGJ is an effective treatment modality but is associated with high morbidity and AEs. EUS-GE and ES are preferred techniques over SGJ due to their less invasive nature and short survival associated with malignant GOO. Previous studies evaluating ES have shown good outcomes, but efficacy is limited due to tumor ingrowth or migration, in contrast to EUS-GE.7,32 EUS-GE is a technically challenging procedure that requires special expertise in the unique use of echoendoscopy for the creation of an iatrogenic perforation between the gastric wall and small bowel wall. Technical failure of EUS-GE results due to misdeployment or maldeployment of the stent. Our analysis revealed that the technical effectiveness of both the ES and EUS-GE groups was comparable. In the ES group, through the scope stents of varying sizes including (22 mm × 6 cm, 22 mm × 9 cm, or 22 mm × 12 cm) were placed under fluoroscopic guidance using standard gastroscope. Technical failures in duodenal stenting were reported to be due to failure to pass the guidewire or deployment of the stent, given the significant tumor burden. EUS-GE group studies reported using a 15 × 10 or 20 × 10 mm HOT-AXIOS LAMS depending on the availability. Sanchez-Aldehuelo et al. reported two cases of misdeployment in the peritoneum and one in the colon. Ge et al. reported two cases of EUS-GE mal-deployment with resultant perforation but both cases were managed endoscopically with the successful deployment of LAMS in the same session without the need of surgical intervention. 19 Multiple salvage techniques have been reported in the literature in cases of EUS-GE misdeployment, such as placement of over the scope clip for closure of the gastric wall defect, coaxial LAMS, or placement of SEMS through the tract.33–35 Recently, Rizzo et al. reported the use of natural orifice transluminal endoscopic surgery to retrieve maldeployed LAMS in the peritoneum by performing transgastric peritoneoscopy. 36 Technical success was comparable for EUS-GE and ES in most studies (>95%).
Enteral stents are associated with a shorter time to oral intake than surgical gastroenterostomy in previous comparative studies. Our meta-analysis showed that the pooled OR of clinical success for EUS-GE was significantly higher than that for ES. Sanchez-Aldehuelo et al. reported a clinical success rate of 92% for EUS-GE and 83% for ES. Similarly, van Wanrooij et al. reported clinical success rates of 90% for EUS-GE and 77% for duodenal stenting. Our results also show that EUS-GE is associated with lower rates of recurrence of obstruction and the need for re-intervention. ES is associated with a significant risk of recurrence of GOO symptoms secondary to stent dysfunction, and Sanchez-Aldehuedo et al. reported 25% ES dysfunction compared to 9% EUS-GE dysfunction. Enteral stent dysfunction rates requiring re-intervention were consistent with those reported in previous studies. 11 EUS-GE bypasses the tumor location with a theoretically longer patency of the EUS-GE. Longer patency is associated with lower rates of dysfunction and re-intervention. Ge et al. reported one case of mesh dysfunction requiring replacement of EUS-GE. 19
EUS-GE was also shown to have a better safety profile than ES with fewer AEs. Van Wanrooij et al. reported 10% of AEs in the EUS-GE group patients and 20% of patients in the ES group, the difference was not statistically significant. The major side effects were infectious complications, such as aspiration pneumonia (1% in the EUS-GE and 4% in the ES group) and cholangitis (3% in the EUS-GE group and 4% in the ES group). Maldeployment of LAMS occurred in three patients requiring salvage surgery and post-procedural bleeding in one patient. However, in the ES group, there was one case of stent migration and three cases of postprocedural bleeding. Sanchez-Aldehuelo et al. reported 10% of AEs in the EUS-GE group and 10% of patients in the ES group. Four patients who underwent ES presented with AEs related to pancreatic or biliary obstruction (pancreatitis, cholangitis), but no patient in the EUS-GE group had such complications. The nature of EUS-GE stenting preserves access to the pancreatic duct and bile duct for future interventions, if necessary. The pooled rate of major AEs for EUS-GE in our meta-analysis was higher than that for ES, but the rate of overall AEs was lower.
Our meta-analysis concluded that EUS-GE and ES are comparable in terms of technical effectiveness, but EUS-GE has significantly higher clinical effectiveness than ES compared to clinical success reported in previous studies. 11 Our results also indicate that EUS-GE is associated with lower rates of recurrence of obstruction and need for re-intervention. In addition, EUS-GE was shown to have a better safety profile than ES with fewer AEs. EUC-GE is a relatively recent endoscopic technique with limited experience, but our study has shown better outcomes of EUS-GE compared to ES and should be the preferred modality for the treatment of symptomatic GOO.
Our meta-analysis had several strengths. We used clear and specific inclusion and exclusion criteria and thoroughly reviewed the literature to ensure that we included all relevant studies comparing EUS-GE to ES. Our analysis included a large cohort of 1762 patients. There are some limitations to our analysis as well. Seven of the studies in our analysis were published as abstracts, with details of patient characteristics and procedures not clearly mentioned, which can lead to inevitable bias. In addition, analysis of other important treatment-related parameters such as length of hospital stay, re-obstruction rates, in-hospital mortality, and long-term survival benefits could also be not performed. Most studies were conducted at tertiary care centers by expert endoscopists; therefore, the technical success and AE rates cannot be generalized. We found no statistically significant difference between EUS-GE and ES in terms of technical effectiveness and utilized confidence intervals to quantify the uncertainty of any differences in success and failure rates.
Conclusion
EUS-GE and ES are comparable in terms of their technical effectiveness. However, EUS-GE has improved clinical effectiveness and a lower re-intervention rate and safety profile than ES for the palliation of GOO. EUS-GE is the preferred method for treating GOO in centers with expertise.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241248219 – Supplemental material for Endoscopic ultrasound-guided gastroenterostomy versus enteral stenting for gastric outlet obstruction: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848241248219 for Endoscopic ultrasound-guided gastroenterostomy versus enteral stenting for gastric outlet obstruction: a systematic review and meta-analysis by Muhammad Asghar, David Forcione and Srinivas Reddy Puli in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848241248219 – Supplemental material for Endoscopic ultrasound-guided gastroenterostomy versus enteral stenting for gastric outlet obstruction: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tag-10.1177_17562848241248219 for Endoscopic ultrasound-guided gastroenterostomy versus enteral stenting for gastric outlet obstruction: a systematic review and meta-analysis by Muhammad Asghar, David Forcione and Srinivas Reddy Puli in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
