Abstract
Background:
Effective Helicobacter pylori (H. pylori) eradication depends on maintaining intragastric pH >6 and overcoming antibiotic resistance. High-dose dual therapy (HDDT) with a proton pump inhibitor (PPI) and amoxicillin has shown promising results.
Objectives:
To compare the efficacy of 14-day vonoprazan-based (VA) and esomeprazole-based (EA) HDDT for H. pylori eradication and evaluate the impact of antibiotic resistance.
Design:
Randomized controlled trial (RCT).
Methods:
A total of 121 patients with confirmed H. pylori infection were randomized to receive either VA therapy (vonoprazan 20 mg twice daily plus amoxicillin 750 mg four times daily (QID)) or EA therapy (esomeprazole 40 mg three times daily plus amoxicillin 750 mg QID) for 14 days. Eradication was assessed by the ¹³C-urea breath test at week 8. Antibiotic susceptibility testing was performed on cultured isolates.
Results:
Baseline demographic and clinical characteristics were comparable between the VA and EA groups. In the intention-to-treat analysis, eradication rates were 86.9% (95% confidence interval (CI): 78.4%–95.4%) in the VA group and 81.7% (95% CI: 71.3%–89.4%) in the EA group (p = 0.430). Per-protocol (PP) analysis showed eradication rates of 93.0% (95% CI: 86.4%–99.6%) for VA and 84.5% (95% CI: 73.8%–92.1%) for EA (p = 0.150), indicating no statistically significant difference. Adverse events (AEs) were mild and similar between groups (5.3% in VA vs 5.2% in EA, p = 0.983), with constipation and diarrhea being the most reported. Both groups achieved 100% compliance. Antibiotic resistance patterns did not significantly affect outcomes.
Conclusion:
Both VA and EA-HDDT regimens demonstrated comparable efficacy, excellent compliance, and minimal AEs. Although VA therapy achieved a >90% eradication rate in the PP analysis, our study was underpowered to confirm superiority. Therefore, larger, adequately powered RCTs are warranted to validate the potential superiority of VA.
Trial registration:
ClinicalTrials.gov: NCT06811207.
Plain language summary
Helicobacter pylori (H. pylori) is a common type of bacteria that infects the stomach and can cause ulcers and even lead to stomach cancer if not treated. This study looked at two different treatments to get rid of H. pylori using high doses of acid-suppressing medicine and an antibiotic called amoxicillin. A total of 121 adults with H. pylori infection took part in the study. Each person was randomly assigned to one of the two treatment groups. They took their assigned medications for 14 days. Eight weeks later, a special breath test was used to see if the bacteria were gone. The results showed that both treatments worked well. The vonoprazan group had a slightly higher success rate, especially when only people who finished the treatment were included. Both groups had very few side effects, and almost everyone took all their medication as instructed. The study found that vonoprazan-based treatment may be slightly more effective, but the difference was not large enough to be considered statistically significant. Importantly, both treatments were well tolerated, with minimal side effects like mild constipation or diarrhea. This research suggests that both options are effective and safe for treating H. pylori, and that the newer medication, vonoprazan, may offer a slight benefit as a first-choice treatment.
Introduction
Helicobacter pylori (H. pylori) infection is a major global health concern, affecting nearly half of the world’s population and playing a critical role in the development of peptic ulcer disease, gastric adenocarcinoma, and mucosa-associated lymphoid tissue lymphoma.1,2 Eradication of H. pylori is essential for preventing these severe complications and reducing the burden of gastric diseases. 3 However, successful eradication has become increasingly challenging due to the rising prevalence of antibiotic resistance, particularly clarithromycin resistance, which has significantly reduced the efficacy of traditional triple therapy regimens. 4 In regions where clarithromycin resistance exceeds 15%–20%, standard triple therapy often fails to achieve acceptable eradication rates, necessitating the development of alternative treatment strategies. 5
One promising strategy is high-dose dual therapy (HDDT), which consists of a high-dose proton pump inhibitor (PPI) and high-dose amoxicillin.6,7 This regimen is designed to enhance amoxicillin’s bactericidal activity by maintaining a consistently high intragastric pH, thereby reducing bacterial load and minimizing antibiotic resistance selection pressure. 8 Notably, H. pylori rarely develops resistance to amoxicillin, making HDDT an attractive alternative to multidrug regimens. 9 Studies have shown that maintaining an intragastric pH above 6 significantly improves amoxicillin’s minimum inhibitory concentration (MIC), optimizing its efficacy against H. pylori. 9
Given the importance of acid suppression, newer-generation acid inhibitors have been explored to improve treatment efficacy. Vonoprazan, a potassium-competitive acid blocker (P-CAB), provides stronger and more sustained acid suppression compared to traditional PPIs such as esomeprazole. 10 Unlike PPIs, vonoprazan does not require activation in an acidic environment, has a faster onset of action, and is less affected by CYP2C19 genetic polymorphisms, leading to more consistent acid suppression across different patient populations. 10 A recent systematic review and meta-analysis found that vonoprazan-based (VA) dual and triple therapies achieved higher eradication rates compared to PPI-based regimens, highlighting the potential advantage of potent acid suppression in optimizing treatment outcomes. 11
Despite promising clinical outcomes with HDDT, limited evidence exists regarding the efficacy of vonoprazan-based HDDT compared with esomeprazole-based (EA) HDDT, particularly in the first-line treatment of H. pylori infection. To our knowledge, few randomized controlled trials (RCTs) have directly compared these two regimens. This study addresses this critical gap by evaluating the efficacy, safety, and compliance of VA-HDDT versus EA-HDDT in a RCT. Accordingly, we aim to determine whether VA-HDDT offers a potential advantage as a first-line treatment option for H. pylori eradication.
Materials and methods
Ethical approval
This prospective, RCT was approved by the Institutional Review Board and Ethics Committee of Kaohsiung Chang Gung Memorial Hospital, Taiwan (IRB No. 202200416A3; approved on October 24, 2022). The study was conducted in accordance with the ethical principles of the Declaration of Helsinki (1975, as revised in 2013). Written informed consent was obtained from all participants prior to enrollment. The trial was registered at ClinicalTrials.gov (NCT06811207). Only adult participants were included, and no minors or children were enrolled.
Patient enrollment and data collection
Patient enrollment was conducted from December 1, 2022 to November 30, 2024. All follow-up assessments were completed within 8 weeks after treatment completion, as scheduled. The study duration included both the intervention and follow-up phases, allowing for evaluation of H. pylori eradication outcomes. Each participant underwent a structured questionnaire assessment, collecting demographic information and medical history, including age, sex, prior medical conditions, and lifestyle habits such as smoking and alcohol consumption. Smoking was defined as consuming at least one pack of cigarettes per week, while alcohol consumption was defined as drinking at least one cup per day.12,13
Study design and treatment protocol
This RCT enrolled 121 adult patients with confirmed H. pylori infection from Kaohsiung Chang Gung Memorial Hospital, and was conducted in accordance with the CONSORT 2010 guidelines. Participants were randomly assigned to one of two treatment groups using a computer-generated randomization sequence: VA Group (n = 61): Vonoprazan 20 mg twice daily (BID) + amoxicillin 750 mg four times daily (QID) for 14 days. EA Group (n = 60): Esomeprazole 40 mg three times daily (TID) + amoxicillin 750 mg QID for 14 days. During the study, four patients in the VA group and two in the EA group were lost to follow-up, leaving 57 patients in the VA group and 58 patients in the EA group who completed the study (Figure 1).
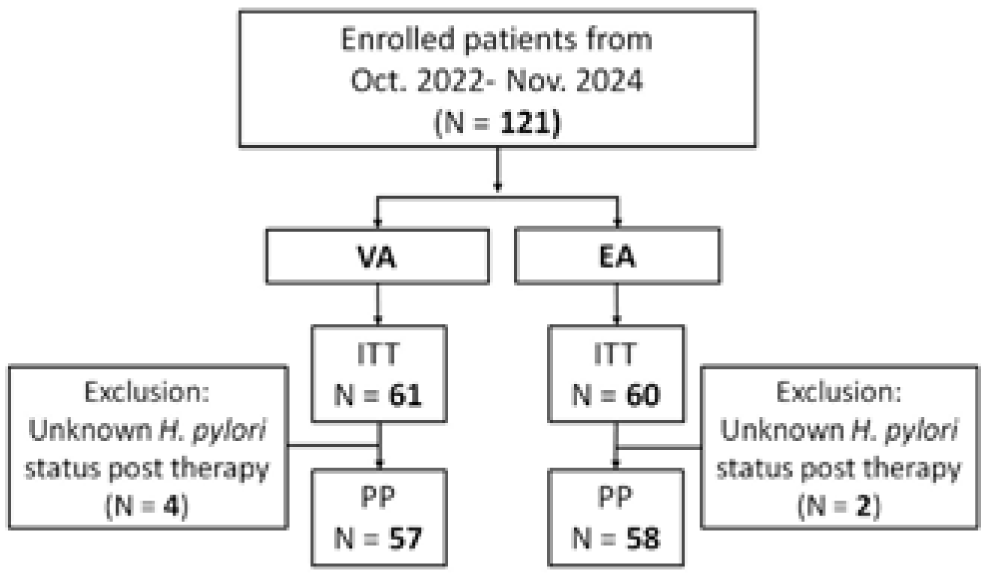
Patients’ deposition.
Diagnosis of H. pylori infection
All patients underwent esophagogastroduodenoscopy, during which gastric mucosal biopsies were collected from both the antrum and gastric body. H. pylori infection was confirmed if any of the following tests were positive: (1) Rapid urease test: biopsy samples were placed in 10% urea solution (pH 6.8) with phenol red and incubated at 37°C for up to 2 h. A color change from yellow to pink within this period was considered positive14,15; (2) Histology: tissue samples were Giemsa-stained and examined microscopically for H. pylori presence 16 ; (3) Bacterial culture: biopsy specimens were cultured on brain heart infusion agar with 7% horse blood at 37°C in a 12% CO2 incubator for bacterial isolation and susceptibility testing 15 ; (4) 13C-urea breath test ( 13 C-UBT): After a 4-h fasting period, patients consumed 75 mg of 13C-urea (Proto Pylori kit; Princeton International Industries, Taipei City, Taiwan). Breath samples were analyzed using an IRIS® Infrared Red Isotope Analyzer (Wagner Analysen Technik, located in Bremen, Germany). A delta over baseline value >4.8% was considered positive for H. pylori infection. 17
Exclusion criteria
Patients were excluded if they met any of the following conditions: (1) History of gastric surgery (e.g., partial gastrectomy); (2) recent antibiotic use (within the past 4 weeks); (3) frequent PPI or bismuth use (>3 times per week within 28 days prior to enrollment); (4) severe comorbidities, including congestive heart failure, stage IV/V chronic kidney disease, or decompensated liver cirrhosis; (5) previous H. pylori eradication therapy; (6) pregnancy or breastfeeding; (7) allergy to any study medications.
Assessment of treatment outcomes
The primary outcome of this study was the H. pylori eradication rate, evaluated using the 13C-UBT 8 weeks after treatment completion. Secondary outcomes included the incidence and severity of treatment-related adverse events (AEs) and patient drug compliance. Compliance: Assessed on Day 15 by checking the remaining medications. Patients who took <80% of prescribed medications were classified as having poor compliance. Eradication success: Defined by a negative 13C-UBT result performed 8 weeks after treatment completion. AEs: Patients reported side effects during therapy, categorized using a 4-point severity scale: None, Mild (discomfort but no impact on daily activities), Moderate (discomfort affecting daily life), and Severe (requiring discontinuation of therapy).12,13
Antibiotic susceptibility testing
Cultured H. pylori isolates were stored at −80°C in Brucella broth with 20% glycerol. MICs for amoxicillin, clarithromycin, levofloxacin, tetracycline, and metronidazole were determined using the agar dilution method and E-test (AB Biodisk, Solna, Sweden). 15 EUCAST breakpoints defined antibiotic resistance as follows 18 : Amoxicillin: MIC ⩾0.125 mg/L; Clarithromycin: MIC ⩾0.5 mg/L; Levofloxacin: MIC ⩾1 mg/L; Tetracycline: MIC ⩾1 mg/L; Metronidazole: MIC ⩾8 mg/L.
Block randomization
Eligible patients were enrolled from the outpatient department according to predefined inclusion and exclusion criteria. All patient details were fully de-identified prior to analysis and reporting. Randomization was performed using block randomization with a fixed block size of 4, with each block containing two allocations to the vonoprazan-based group (VA) and two to the esomeprazole-based group (EA). Patients were assigned a sequential study number upon enrollment; those receiving code A were allocated to the VA group, and those with code B to the EA group, ensuring a 1:1 allocation ratio. To maintain allocation concealment, the randomization sequence was pre-generated and implemented using sequentially numbered, opaque, sealed envelopes prepared in advance by an independent coordinator not involved in patient enrollment or treatment.
Blinding
This was an open-label study. Neither participants nor care providers were blinded to treatment allocation due to the nature of the interventions, which differed in drug type and dosing schedule. Outcome assessors and data analysts were also not blinded.
Statistical analysis
The statistical plan was developed in consultation with the Biostatistics Center, Kaohsiung Chang Gung Memorial Hospital. Based on prior studies7,17 and assuming an expected eradication rate difference of 10% between groups, we calculated that a total sample size of 240 patients (120 per group) would be required to achieve 80% power at a two-sided significance level of 0.05. Due to constraints related to funding and recruitment feasibility, 121 patients were ultimately enrolled during the study period. Although this number was below the calculated target, the study was carried out as planned, and the limitation of statistical power is acknowledged in the “Discussion” section.
Eradication rates, AEs, and compliance were compared between the VA and EA groups using the Chi-square (χ2) test or Fisher’s exact test, as appropriate. Both intention-to-treat (ITT) and per-protocol (PP) analyses were performed. For ITT analysis, all randomized patients were included, with patients lost to follow-up or with missing data considered treatment failures. The PP analysis included only those who completed the treatment course and follow-up assessment, had ⩾80% compliance, and had no major protocol violations.
To explore predictors of eradication success, multivariate logistic regression was performed. Continuous variables were compared between groups using the independent-samples t test. A p-value <0.05 was considered statistically significant. Data were analyzed using IBM SPSS® Statistics version 22.0 (IBM Corp., Armonk, NY, USA).
Results
Demographic data and endoscopic findings
The baseline characteristics (Table 1) between the VA- and EA-HDDT groups were generally well balanced, with no significant differences in age (p = 0.265), gender distribution (p = 0.765), smoking (p = 0.734), or alcohol consumption (p = 0.448). However, coffee consumption was significantly higher in the VA group (10.5%) than in the EA group (1.7%; p = 0.048). Endoscopic findings showed that gastritis was the most common diagnosis in both groups (70.2% in VA vs 67.2% in EA, p = 0.734). Gastric ulcers were more frequent in the EA group (22.4%) than in the VA group (12.3%), but this difference was not statistically significant (p = 0.152). The distribution of duodenal ulcers and combined gastric and duodenal ulcers was also similar between groups.
Demographic data and endoscopic appearance of two patient groups.

EA, esomeprazole-based high-dose dual therapy; SD, standard deviation; VA, vonoprazan-based high-dose dual therapy.
Major outcomes
In the ITT analysis, eradication rates were 86.9% (95% confidence interval (CI): 78.4%–95.4%) in the VA group and 81.7% (95% CI: 71.3%–89.4%) in the EA group (p = 0.430). PP analysis showed eradication rates of 93.0% (95% CI: 86.4%–99.6%) for VA and 84.5% (95% CI: 73.8%–92.1%) for EA (p = 0.150), indicating no statistically significant difference. These findings suggest that VA-HDDT may provide a slight advantage in eradication efficacy over EA-HDDT, though the difference was not statistically significant. Compliance was excellent, with 100% adherence in both groups (Table 2).
The major outcomes of two patient groups.
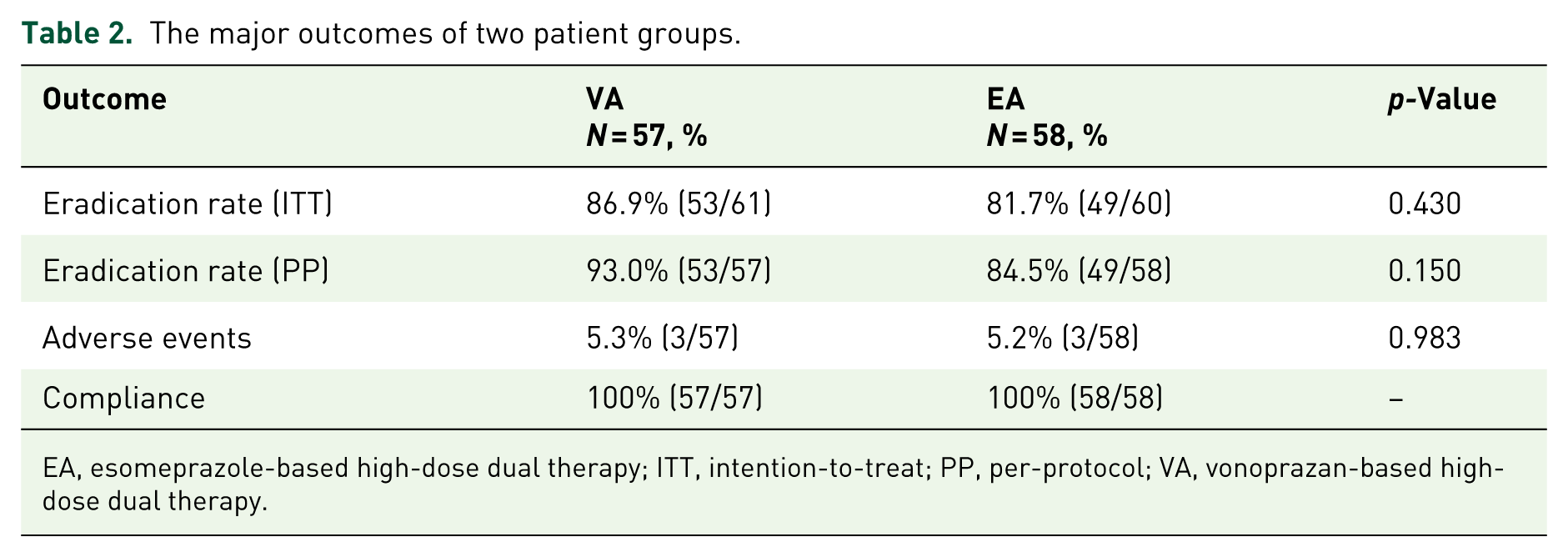
EA, esomeprazole-based high-dose dual therapy; ITT, intention-to-treat; PP, per-protocol; VA, vonoprazan-based high-dose dual therapy.
Adverse events
AEs were rare and comparable between the two groups (5.3% in VA vs 5.2% in EA, p = 0.983), suggesting good tolerability of both regimens (Table 2). Constipation and diarrhea were the only AEs observed in both groups, affecting a small proportion of patients (constipation: 1.8% in VA vs 1.7% in EA; diarrhea: 3.5% in VA vs 3.4% in EA). No patients reported abdominal pain, dizziness, headache, nausea/vomiting, or skin rash (Table 3).
Adverse events of two patient groups.

EA, esomeprazole-based high-dose dual therapy; VA, vonoprazan-based high-dose dual therapy.
Antibiotic resistance
Antibiotic susceptibility testing (n = 13) revealed no resistance to amoxicillin or tetracycline in either group. However, clarithromycin resistance was more prevalent in the VA group (33.3%) compared to the EA group (14.3%), although this difference was not statistically significant (p = 0.416). Metronidazole resistance was observed in 28.6% of the EA group but was absent in the VA group (p = 0.155). Levofloxacin resistance was also higher in the VA group (33.3%) compared to the EA group (14.3%), but this difference was not statistically significant (p = 0.416; Table 4).
Antibiotic resistance between two groups (culture number = 13).

EA, esomeprazole-based high-dose dual therapy; VA, vonoprazan-based high-dose dual therapy.
Univariate analysis of factors influencing eradication efficacy
Several clinical factors were analyzed to determine their impact on H. pylori eradication rates. In both groups, age (⩾60 vs <60 years) did not significantly influence eradication rates (VA: 93.8% vs 93.0%, p = 0.797; EA: 87.5% vs 80.8%, p = 0.481). Female patients in both groups had numerically higher eradication rates than males (VA: 97.1% vs 87.0%, p = 0.143; EA: 90.0% vs 76.0%, p = 0.120), though these differences were not statistically significant. Smoking and alcohol use did not negatively impact eradication rates, with all smokers and alcohol users achieving 100% eradication in both groups. Coffee and tea consumption lifestyle factors did not significantly affect eradication rates, with all coffee and tea consumers in the VA group achieving 100% eradication.
Clarithromycin resistance appeared to impact eradication in the EA group (100% eradication in sensitive cases vs 33.3% in resistant cases, p = 0.143), but not in the VA group (75.0% in sensitive vs 100% in resistant cases, p = 0.667). Metronidazole resistance may have contributed to slightly lower eradication rates in the EA group (80.0% for sensitive cases vs 50.0% for resistant cases, p = 0.524), though this was not statistically significant. Levofloxacin resistance appeared to impact eradication rates in the EA group (83.3% in sensitive cases vs 0% in resistant cases, p = 0.286), while it had no negative impact in the VA group (75.0% in sensitive vs 100% in resistant cases, p = 0.667; Table 5).
Univariate analysis of the clinical factors influencing the efficacy of H. pylori eradication in each therapy.
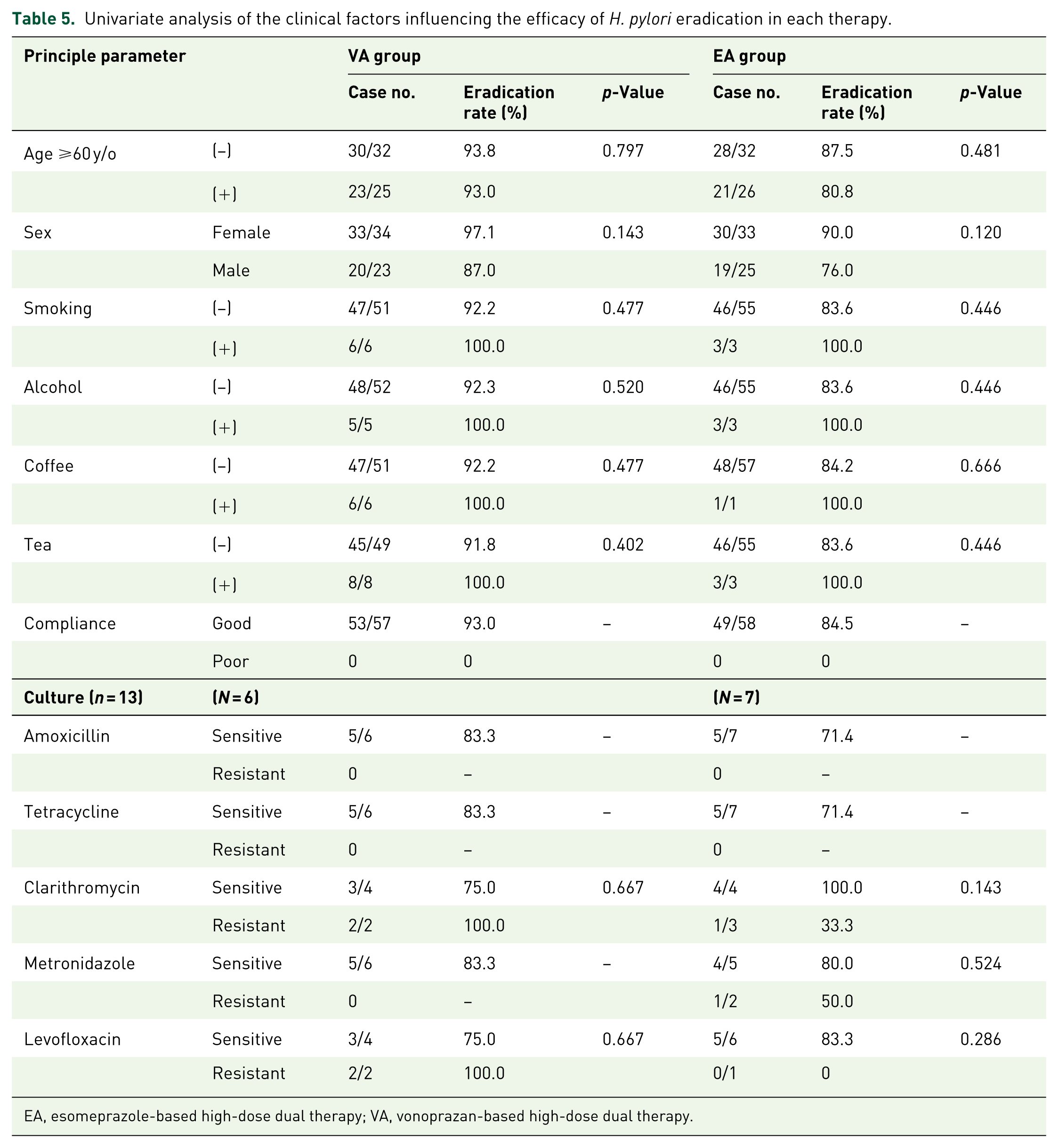
EA, esomeprazole-based high-dose dual therapy; VA, vonoprazan-based high-dose dual therapy.
Discussion
Our study demonstrated that both VA- and EA-HDDT regimens achieved high eradication rates with excellent safety and compliance. Notably, VA-HDDT attained a numerically higher PP eradication rate (93.0%) compared with EA-HDDT (84.5%), although this difference did not reach statistical significance, likely due to the limited sample size. Importantly, VA-HDDT consistently achieved eradication rates exceeding 90%, the benchmark threshold for an effective H. pylori therapy. To our knowledge, this trial is among the first randomized controlled studies directly comparing VA- and EA-based HDDT regimens, thereby providing novel evidence that VA-HDDT may represent a promising first-line treatment option. Recent RCTs support the efficacy of vonoprazan-based dual therapy. A prospective RCT 19 demonstrated that 14-day vonoprazan–amoxicillin (VA) dual therapy achieved eradication rates of 94.1%–95.9% (PP analysis), significantly improving outcomes compared to shorter treatment durations. Additionally, a multicenter RCT 20 comparing 10-day VA therapy with 14-day rabeprazole-amoxicillin therapy found that VA achieved higher eradication rates (91.4% vs 86.6% PP, p = 0.047), supporting its non-inferiority and potential superiority over PPI-based dual therapy. A recent multicenter RCT 21 compared a 10-day VA regimen (vonoprazan 20 mg BID + amoxicillin 1000 mg TID) with a 14-day EA regimen (esomeprazole 20 mg QID + amoxicillin 750 mg QID) for first-line H. pylori eradication. The study reported significantly higher eradication rates in the VA group compared to the EA group (91.1% vs 85.5% PP, p = 0.047), along with better patient compliance (p = 0.006). These findings further support the efficacy, safety, and tolerability of vonoprazan-based high-dose dual therapy as a promising first-line treatment option.
In contrast, the efficacy of PPI-based HDDT has been highly variable, with eradication rates ranging from 52.0% to 96.4% depending on trial design, treatment duration, and patient characteristics.22,23 This variability is influenced by several factors, including CYP2C19 polymorphisms, regional antibiotic resistance patterns, and dosing regimens.
The CYP2C19 enzyme system plays a central role in metabolizing first-generation PPIs such as omeprazole and lansoprazole. Genetic polymorphisms divide individuals into poor metabolizers (PMs), intermediate metabolizers, and extensive metabolizers (EMs), with PMs exhibiting higher drug exposure and more potent acid suppression.24,25 Meta-analyses have shown that eradication rates tend to follow a gene-dosage effect: PMs achieve 85%–87% success compared to 77%–80% in EMs. However, increasing PPI doses can neutralize this genetic effect, as double-dose PPI regimens have reported uniform eradication rates (~85.2%) across all genotypes.25,26 P-CABs, such as vonoprazan, are less affected by CYP2C19 metabolism. Vonoprazan-based regimens consistently achieve >90% eradication rates irrespective of CYP2C19 status, making them a promising solution for overcoming genetic variability.10,27
Current guidelines recommend bismuth-based quadruple therapy (BQT) or concomitant therapy as alternative first-line treatments, particularly in regions with high clarithromycin resistance. 28 However, these multidrug regimens are associated with increased side effects, leading to poor compliance and treatment discontinuation. Reported adverse effects include nausea, diarrhea, abdominal discomfort, and metallic taste, with an overall AE rate exceeding 30% in some studies.29,30
In contrast, HDDT offers a simplified regimen with fewer side effects, improving patient adherence. Our study confirmed this advantage, as both VA and EA regimens had minimal side effects (overall AE rate ~5%). The most reported AEs were mild gastrointestinal symptoms, such as constipation and diarrhea, occurring at similar rates in both groups. These findings are consistent with previous studies, where PPI-based HDDT showed superior tolerability compared to BQT. While BQT is associated with AE rates of 26.6%–68%, HDDT generally has lower rates (10.6%–30%), particularly for gastrointestinal symptoms like diarrhea (9%–20%), nausea (5%–11%), and abdominal pain (4%–7%). 31 Notably, severe AEs requiring treatment discontinuation remain rare (<3% incidence) across all regimens. In our study, no patients discontinued treatment due to side effects, and compliance was 100%, further supporting HDDT’s safety and practicality as a first-line therapy.
Interestingly, our study found that female patients in both groups had numerically higher eradication rates than males, although the difference was not statistically significant. This trend aligns with previous studies suggesting a potential sex-based difference in H. pylori eradication rates. 32 However, other studies have reported no significant differences between sexes.6,7,17 A large-scale cross-sectional study 33 involving 81,754 Chinese adults found that older women had a higher H. pylori infection rate, lower eradication rate, and higher recurrence rate. Possible explanations for sex-based differences in eradication outcomes include variations in gastric acid secretion, drug metabolism, hormonal influences, and gut microbiota composition.34,35 However, further research with larger sample sizes and multicenter cohorts is needed to establish a definitive relationship between sex and treatment outcomes.
Besides efficacy and safety, cost is also an important consideration in selecting eradication regimens. Recent cost-analysis data demonstrated that the 14-day EA dual therapy cost approximately 82.8 USD per patient, which was 10.1% higher than BQT, whereas the 14-day VA dual therapy cost only 40.1 USD, representing a substantial 17.3% reduction per patient compared with vonoprazan–amoxicillin–clarithromycin triple therapy. 36 Similarly, Zhang et al. 37 reported that high-dose PPI–amoxicillin dual therapy significantly reduced treatment expenses compared with quadruple therapy, while maintaining comparable efficacy and safety. These findings indicate that VA-based dual therapy may offer not only clinical but also economic advantages, particularly in regions with increasing concerns about healthcare costs.
Our study has several limitations. First, based on our original sample size calculation, 120 participants per group were required to achieve adequate statistical power. However, only 121 patients were enrolled in total, resulting in an underpowered study that may not detect statistically significant differences between treatment groups. Second, although antibiotic susceptibility testing was performed, it was limited to a small subset of patients, which restricts the generalizability of our findings regarding resistance patterns and their impact on treatment efficacy. Third, CYP2C19 genotyping was not conducted, so we could not assess the potential influence of genetic polymorphisms on esomeprazole metabolism, which may have affected eradication rates in the EA group. In addition, although all smokers and alcohol users in our cohort achieved successful eradication, the number of patients in these subgroups was small. As such, these findings should be interpreted with caution, and larger studies are needed to better understand the role of lifestyle factors in treatment outcomes.
Conclusion
Both VA- and EA-HDDT demonstrated good efficacy and excellent tolerability as first-line treatments for H. pylori infection. However, VA-HDDT consistently achieved eradication rates exceeding 90%, reinforcing its potential as a superior first-line option, particularly in populations with high clarithromycin resistance.
