Abstract
Background:
The efficacy of the 14-day esomeprazole–amoxicillin (EA) dual therapy in eradicating Helicobacter pylori (H. pylori) has been widely discussed previously. Vonoprazan, a novel potassium-competitive acid blocker, presents rapid, potent, and long-lasting acid inhibitory effects compared to esomeprazole. However, there is currently a scarcity of direct comparisons between the 10-day vonoprazan–amoxicillin (VA) and the 14-day EA dual therapy for H. pylori eradication.
Objectives:
This study aimed to compare the efficacy and safety of the 10-day VA and the 14-day EA dual therapy for H. pylori first-line eradication.
Design:
This study was a prospective, multicenter, open-label, randomized controlled trial.
Methods:
The study was conducted at 10 hospitals in China. In total, 570 newly diagnosed H. pylori-infected patients were recruited from April 2023 to February 2024. These patients were randomly assigned to either the 10-day VA group (vonoprazan 20 mg twice daily + amoxicillin 1000 mg three times daily) or the 14-day EA group (esomeprazole 20 mg four times daily + amoxicillin 750 mg four times daily). The primary outcome was the eradication rate, with secondary outcomes including adverse events and compliance.
Results:
The 10-day VA regimen outperformed the 14-day EA regimen in terms of eradication rates in intention-to-treat (ITT) analysis (85.4% vs 76.7%, p = 0.008), modified ITT analysis (90.7% vs 84.8%, p = 0.036), and per-protocol (PP) analysis (91.1% versus 85.5%, p = 0.047). The non-inferiority p-values in all three analyses were less than 0.001. No statistically significant difference was observed in the incidence of adverse events between the two groups (9.1% vs 11.7%, p = 0.308). The 10-day VA regimen demonstrated higher compliance compared to the 14-day EA regimen (p = 0.006).
Conclusion:
The 10-day VA dual therapy showed a satisfactory eradication rate of 91.1% (PP analysis), demonstrating good safety and better compliance compared to the 14-day EA dual therapy as the first-line eradication.
Trial registration:
This trial was registered in the Chinese Clinical Trial Registry (registration number: ChiCTR2300070475) on April 12, 2023.
Introduction
Helicobacter pylori (H. pylori) infection is a widespread public health problem that affects approximately 44% of adults and 35% of children and adolescents globally. 1 H. pylori infection is an infectious disease that can lead to chronic gastritis, as well as progress to severe stomach and duodenal conditions, such as peptic ulcers and gastric malignancies. 2 Eradicating H. pylori can lower the risk of gastric cancer, particularly before the onset of precancerous changes, and can also reduce the risk of metachronous gastric cancer after curative endoscopic resection of early gastric cancer.2,3
Currently, the antibiotic resistance of H. pylori (mainly to clarithromycin, metronidazole, and levofloxacin) has become a significant global issue, 3 posing a serious challenge to eradicate the bacteria. Bismuth-containing quadruple therapy (BQT), consisting of a proton pump inhibitor (PPI), bismuth, and two antibiotics, has been considered the first-line eradication treatment in regions with high clarithromycin resistance (>15%) for many years.4 –6 Although it achieves a relatively satisfactory eradication rate, there are still issues such as multiple types of medication, complex regimens, more adverse events, poor compliance, and high costs. Furthermore, the use of multiple antibiotics can disrupt the gut microbiota and increase secondary antibiotic resistance, reducing the options for subsequent remedial antibiotic treatments.7 –9 Therefore, it is urgent in clinical practice to explore eradication regimens with fewer antibiotics, fewer adverse events, and satisfactory eradication rates.
In recent years, high-dose dual therapy (HDDT) consisting of PPI and amoxicillin (3000 mg/day) has gained widespread attention due to its ability to achieve similar eradication rates as BQT, with fewer adverse events and lower costs.7,8,10,11 The classic HDDT regimen consists of either esomeprazole or rabeprazole (standard dose, qid) and amoxicillin (750 mg, qid), taken for 14 days. Increasing the dosage and frequency of PPI and amoxicillin can help maintain therapeutic blood levels of both drugs, creating a high pH environment in the stomach that is conducive to the growth of H. pylori. This, in turn, enhances the sensitivity of H. pylori to amoxicillin. 12 Vonoprazan, a novel potassium-competitive acid blocker, was approved for H. pylori eradication in Japan in 2015. Compared to PPIs, it offers faster, stronger, and longer-lasting acid inhibitory effects.13,14 Therefore, theoretically speaking, the combination of vonoprazan and amoxicillin could achieve a better eradication effect. Previous results from two randomized controlled trials (RCTs) in China indicated that the 7-day vonoprazan–amoxicillin (VA) dual therapy did not achieve satisfactory eradication rates.15,16 However, there is still controversy surrounding the 10-day VA dual therapy. A small sample single-center RCT 16 revealed that the 10-day VA dual therapy (vonoprazan 20 mg bid + amoxicillin 1000 mg tid) had an eradication rate of 81.1% for both intention-to-treat (ITT) and per-protocol (PP) analyses, indicating that it may not offer satisfactory eradication rates for the Chinese population. Conversely, a slightly larger single-center RCT 17 in China demonstrated that the 10-day VA dual therapy (vonoprazan 20 mg bid + amoxicillin 750 mg qid) yielded similar eradication rates to the 10-day BQT, with ITT analysis showing 91.2% versus 88.0% and PP analysis showing 93.4% versus 90.9%. Up to now, there is a lack of high-quality multicenter trial data directly comparing the efficacy of the 10-day VA dual therapy with the 14-day esomeprazole–amoxicillin (EA) dual therapy as a first-line treatment for H. pylori eradication.
Given the considerations mentioned above, we anticipate that the 10-day VA dual therapy may achieve the same eradication effect as the 14-day EA dual therapy. Therefore, to further optimize the current guidelines recommending 14-day high-dose PPI–amoxicillin dual therapy, we conducted a prospective multicenter RCT to directly compare the efficacy and safety of 10-day VA dual therapy (vonoprazan 20 mg bid + amoxicillin 1000 mg tid) versus 14-day EA dual therapy (esomeprazole 20 mg qid + amoxicillin 750 mg qid) in eradicating H. pylori, aiming to offer patients a highly efficient, convenient, and safe option.
Materials and methods
Study design and ethical statement
This trial was a prospective, multicenter, open-label, non-inferior, randomized controlled study. The study was approved by the Ethics Committee of Affiliated Hospital of Yangzhou University (2023-YKL03-30) and registered in the Chinese Clinical Trial Registry (registration number: ChiCTR2300070475). The trial was conducted in accordance with the Declaration of Helsinki and reported following the Consolidated Standards of Reporting Trials (CONSORT) statement.
Study participants
From April 2023 to February 2024, a total of 10 hospitals in China were selected for the recruitment of newly diagnosed H. pylori-infected patients. The detailed criteria for inclusion were as follows: (1) Adults aged between 18 and 70 years, without any sex restrictions; (2) H. pylori infection was diagnosed using either the 13C-urea breath test (UBT) or the 14C-UBT; (3) without a previous standardized history of H. pylori eradication treatment; and (4) provided informed consent prior to treatment and choose to participate in the study voluntarily.
Patients were excluded from the study if they met any of the following criteria: (1) allergy to the investigational drugs (vonoprazan, amoxicillin, or esomeprazole); (2) had taken antibiotics or bismuth in the past 4 weeks, acid inhibitor, such as PPI or P-CAB in the past 2 weeks; (3) had severe heart, lung, liver, kidney, or other diseases, or weakened immune systems; (4) had a history of acute upper gastrointestinal bleeding, gastrointestinal malignant tumors, Zollinger-Ellison syndrome, or gastric cancer surgery; (5) were pregnant or breastfeeding; and (6) had mental illness or cognitive impairment that would prevent them from cooperating in the study.
Randomization and intervention
Patients were randomly assigned to two groups in a 1:1 ratio using a computer-generated random allocation sequence. The sequence was generated by biostatistics professionals using SAS 9.4 software (SAS Institute Inc., Cary, NC, USA). One group received a 10-day VA dual therapy, while the other group received a 14-day EA dual therapy. The sequence was hidden from all researchers, but patients and researchers were not blinded to the allocation of treatment groups.
In the VA dual regimen, patients were instructed to take 20 mg of vonoprazan twice daily and 1000 mg of amoxicillin three times daily for 10 days. Vonoprazan was to be taken 30 min after breakfast and supper, and amoxicillin was to be taken 30 min after three meals. In the EA dual regimen, patients were instructed to take 20 mg of esomeprazole four times daily and 750 mg of amoxicillin four times daily for 14 days. Esomeprazole was to be taken 30 min before three meals and before sleep, and amoxicillin was to be taken 30 min after three meals and before sleep.
Study procedures and outcomes
After passing the initial screening, eligible participants who agreed to take part in the study signed an informed consent form and completed an electronic questionnaire regarding their demographic information and basic clinical details. They were then randomly assigned to either a 10-day VA group or a 14-day EA group. Patients were verbally introduced to their treatment regimen and the importance of adhering to their medication schedule. Throughout the treatment period, patients were asked to keep a daily record of any adverse events experienced and their medication usage. For mild to moderate adverse events, patients were advised to continue taking their medication while being monitored, with the option of receiving symptomatic treatment if necessary. In cases of serious adverse events, participants were instructed to stop taking the medication and seek medical attention. Patients were advised to undergo a 13C/14C-UBT 4–8 weeks after completing the medication course.
The primary outcome was the H. pylori eradication rate. Successful eradication was determined by a negative result on the 13C/14C-UBT at least 4 weeks after the end of treatment. The staff conducting the 13C/14C-UBT assessment were kept blind to the treatment group allocation. The secondary outcomes were the incidence of adverse events and medication compliance. Follow-up staff contacted patients 1–3 days after completing medication to assess adverse events and medication compliance. An electronic questionnaire was filled out by accompanying staff to document any adverse events and medication compliance. Adverse events were categorized based on their impact on daily activities as either “mild” (temporary and easily managed, with no significant impact on daily activities), “moderate” (resulting in some discomfort and partially disrupting daily routines), or “severe” (causing severe discomfort and significantly interfering with daily activities). Medication compliance was considered good if patients took at least 80% of the total medication.
Sample size calculation and statistical analysis
The sample size calculation was based on the H. pylori eradication rate using PASS 15 software (NCSS, LLC. Kaysville, Utah, USA). Referring to previous studies, the H. pylori eradication rate of the 10-day VA dual therapy was taken as the average of 86.2% in two RCTs,16,17 and the H. pylori eradication rate of the 14-day EA dual therapy was taken as the average of three large-scale RCTs8,10,11 in China, which was 87.3%. A non-inferiority threshold of −10% was set, with a significance level of 0.025 (one-sided) and a statistical power of 80%. To demonstrate non-inferiority between the two therapies, a sample size of at least 228 participants per group would be required. Considering a potential 20% loss to follow-up, the planned sample size for each group should be at least 285 participants, with a total sample size of 570 participants.
The H. pylori eradication rate was assessed based on ITT, modified ITT (MITT), and PP analyses. All patients who were randomized were included in the ITT analysis. Patients who received at least one dose of medication and had UBT results were included in the MITT analysis. Patients who took at least 80% of the total medication with UBT results were included in the PP analysis. The analyses of secondary outcomes were conducted with the ITT analysis. The safety set (SS) included all randomly assigned patients with at least one follow-up observation. The analysis of factors influencing the eradication rate was conducted in the MITT dataset.
Statistical analyses were conducted using SPSS 29.0 (IBM Corp., Armonk, NY, USA) and SAS 9.4 (SAS Institute Inc., Cary, NC, USA). Continuous variables that followed a normal distribution were presented as mean ± standard deviation and compared using Student’s t-test. Non-normally distributed measurement data were presented as median (quartile range) and compared using nonparametric tests, such as the Mann–Whitney U-test. Categorical variables were presented as frequency and percentage and compared using the Chi-squared test or Fisher’s exact test. For the H. pylori eradication rate, a non-inferiority Z-test was used to assess the comparison of non-inferiority between the two groups. The two groups were deemed non-inferior at a significance level of p < 0.025, and the lower limit of the 95% CI for the difference in eradication rates exceeded −10%. If the 95% CI for the difference in eradication rates is not only entirely above −10% but also completely falls on the right side of the value 0, and the p-value for the difference is less than 0.05, it can be inferred that the eradication rate of the 10-day VA group is better than that of the 14-day EA group. In addition, factors influencing the eradication rate were determined using both univariate and multivariate logistic regression models. All factors with a p-value of less than 0.1 in the univariate analysis were included in the multivariate logistic regression analysis. All p-values, except for the non-inferiority test, p < 0.05 indicated statistical significance.
Results
Patient enrollment and baseline characteristics
A total of 648 individuals underwent an eligibility assessment, with 78 individuals who did not meet the criteria being excluded. Ultimately, 570 patients were randomly assigned to either the 10-day VA group (n = 287) or the 14-day EA group (n = 283). Figure 1 illustrates the flowchart for patient enrollment. No statistically significant differences were found in the baseline demographic and clinical characteristics between the two groups (all p > 0.05), as shown in Table 1.
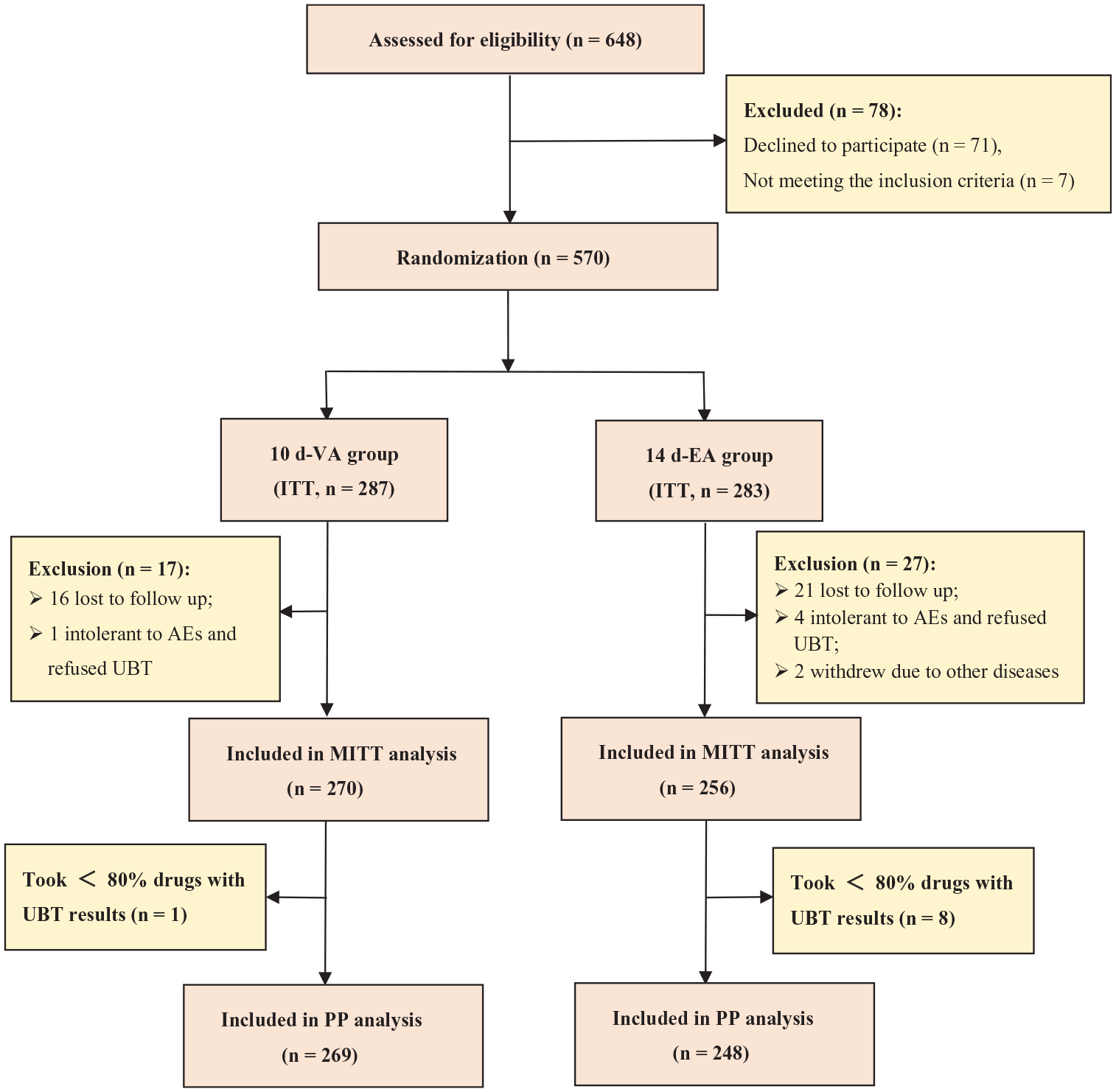
Flow diagram of patient enrollment.
Baseline characteristics of the study patients.

BMI, body mass index, was calculated as follows: BMI, body mass (kg)/(height (m))2; BSA, body surface area, was calculated as follows: BSA (m2) = 0.0061 × height (cm) + 0.0128 × body weight (kg) − 0.1529; EA, esomeprazole and amoxicillin dual therapy; H. pylori, Helicobacter pylori; VA, vonoprazan and amoxicillin dual therapy.
H. pylori eradication rates
The comparison results of H. pylori eradication rates between the two groups, based on ITT, MITT, and PP analyses, are summarized in Table 2. In the ITT analysis, the eradication rates for the 10-day VA group and the 14-day EA group were 85.4% (245/287; 95% CI 81.3%–89.5%) and 76.7% (217/283; 95% CI 71.7%–81.6%), respectively. In the MITT analysis, the eradication rates for the 10-day VA group and the 14-day EA group were 90.7% (245/270; 95% CI 87.3%–94.2%) and 84.8% (217/256; 95% CI 80.3%–89.2%), respectively. In the PP analysis, the eradication rates for the 10-day VA group and the 14-day EA group were 91.1% (245/269; 95% CI 87.6%–94.5%) and 85.5% (212/248; 95% CI 81.1%–89.9%), respectively. The difference in eradication rates between the two groups was statistically significant (p = 0.008, 0.036, and 0.047 in the ITT, MITT, and PP analyses, respectively).
Helicobacter pylori eradication rates of each group.

CI, confidence interval; ITT, intention-to-treat; MITT, modified intention-to-treat; PP, per-protocol; VA, vonoprazan and amoxicillin dual therapy; EA, esomeprazole and amoxicillin dual therapy.
The p values were calculated from one-sided test comparisons of non-inferiority between the 10-day VA and 14-day EA group.
The p values were calculated from two-sided comparisons of difference between the 10-day VA and 14-day EA group.
When comparing the eradication rates between the 10-day VA group and the 14-day EA group in the non-inferiority test, the differences in eradication rates between the two groups were 8.69% (95% CI: 2.28%–15.09%) in the ITT analysis, 5.98% (95% CI: 0.38%–11.57%) in the MITT analysis, and 5.59% (95% CI: 0.04%–11.15%) in the PP analysis. The lower bounds of the 95% CI for the eradication rates in all three analyses were higher than the pre-specified non-inferiority margin (−10%), and the non-inferiority p-values in all three analyses were less than 0.001, indicating that the eradication rate in the 10-day VA group was not inferior to that in the 14-day EA group. Furthermore, in the ITT, MITT, and PP analyses, the 95% CI for the difference in eradication rates fell completely on the right side of the value 0, and the p-values for the differences were all <0.05, suggesting that the eradication rate in the 10-day VA group was superior to that in the 14-day EA group.
Adverse events and medication compliance
Table 3 contains a summary of the adverse events experienced by all patients. There was no statistically significant difference in the overall incidence of adverse events between the 10-day VA group and the 14-day EA group (9.1% vs 11.7%, p = 0.308). Common adverse events during the eradication process included diarrhea, nausea/vomiting, abdominal distension, abdominal pain, epigastric discomfort, skin rash, and itching. The majority of adverse events were mild to moderate and gradually disappeared after completing the medication. In the 10-day VA group, one participant had to discontinue medication treatment due to severe diarrhea intolerance, and this symptom disappeared after discontinuing the medication. In the 14-day EA group, one patient had to stop medication treatment due to severe diarrhea intolerance, another patient stopped medication treatment on their own due to bloating after taking the medication, and three patients discontinued drug therapy due to allergic reactions (skin rash and itching). These symptoms disappeared after terminating treatment and providing symptomatic treatment, without any serious sequelae. There was no statistically significant difference in the occurrence rates of all specific adverse events reported between the two groups. Regarding medication compliance, the 10-day VA group demonstrated higher compliance compared to the 14-day EA group, with a statistically significant difference (98.95% vs 95.05%, p = 0.006).
Adverse events and medication compliance between the two groups.

AE, adverse events; EA, esomeprazole and amoxicillin dual therapy; VA, vonoprazan and amoxicillin dual therapy.
Factors influencing the eradication rates
As summarized in Table 4, medication compliance (p = 0.008) and eradication regimen (p = 0.038) were factors influencing the eradication rates of H. pylori in univariate logistic analyses. Further multivariate logistic regression analysis indicated that only good compliance can improve the H. pylori eradication rates (odds ratio (OR] = 5.128, 95% CI: 1.303–20.175, p = 0.019), while poor compliance can increase the risk of eradication failure.
Univariate and multivariate analyses of factors associated with H. pylori eradication.

BMI, body mass index; BSA, body surface area; CI, confidence interval; OR, odds ratio; EA, esomeprazole and amoxicillin dual therapy; H. pylori, Helicobacter pylori; VA, vonoprazan and amoxicillin dual therapy.
Discussion
As far as we know, this is the first head-to-head multicenter RCT that compares the efficacy and safety of the 10-day VA with the 14-day EA dual therapy in eradicating H. pylori. The findings indicated that the 10-day VA dual therapy achieved a satisfactory eradication rate of 91.1% according to PP analysis and had superior eradication rates in ITT analysis, MITT analysis, and PP analysis compared to the 14-day EA dual therapy. No statistically significant difference was observed in the incidence of adverse events between the two groups (9.1% versus 11.7%). In addition, it was observed that the medication compliance of the 10-day VA dual therapy was higher than that of the 14-day EA dual therapy. Multivariate logistic regression analysis revealed that compliance was a factor affecting the eradication rate, with good compliance leading to higher H. pylori eradication rates.
There are several reasons why the 10-day VA dual therapy can achieve a high eradication rate as a first-line treatment. First, the resistance rate of H. pylori to amoxicillin is relatively low, with the latest research showing it to be around 3% in China, 18 laying the foundation for the eradication of H. pylori with dual therapy. Second, amoxicillin has time-dependent characteristics and a short half-life. The effectiveness of its bactericidal action relies on the percentage of time that the blood plasma concentration exceeds the minimum inhibitory concentration. After 6–8 h of administration, a significant portion of amoxicillin is eliminated from the body. Therefore, giving amoxicillin every 6 or 8 h (either 3 or 4 times a day) is preferable for sustaining adequate blood drug concentrations over an extended period of time.10,19 In our study, the total daily dose of amoxicillin was 3000 mg. In the 10-day VA group, amoxicillin was administered three times a day (1000 mg each time, three times a day), which was less frequent compared to the classic regimen (750 mg each time, four times a day), making it more convenient and potentially improving patient compliance with medication. Third, sufficient acid suppression is a crucial factor in effectively eliminating H. pylori. When the stomach pH exceeds 6, H. pylori switches to a replicative state and becomes susceptible to amoxicillin, making it more vulnerable to eradication. Moreover, the bactericidal action of amoxicillin is influenced by stomach pH levels, with higher stomach pH levels providing greater stability for amoxicillin to work effectively. 19 Vonoprazan inhibits H+, K+-ATPase competitively and reversibly, providing faster and more sustained acid suppression compared to esomeprazole 20 mg or rabeprazole 10 mg when administered at a dose of 20 mg. 20 Prior studies have indicated that healthy participants in Japan who were given vonoprazan 20 mg twice daily had a gastric pH of ⩾4 and ⩾5 maintained for 100% and 99% of the time on the seventh day. 21 In addition, unlike PPIs, vonoprazan is primarily metabolized by CYP3A4, meaning it is not affected by variations between individuals related to CYP2C19 polymorphisms. As a result, it can effectively prevent differences in acid suppression effects caused by the CYP2C19 rapid metabolizer genotype. 22 In summary, vonoprazan exhibits a stronger and more durable acid suppression effect than PPI, leading to a quicker induction of H. pylori replication. This also increases the sensitivity and antibacterial activity of amoxicillin, thereby improving the eradication efficacy of H. pylori.
Recently, there were several multicenter studies on the optimization of the 10-day VA dual therapy and its comparison with other regimens. A multicenter RCT (n = 516) from China, conducted by Peng et al., showed that both the 10-day and 14-day courses of vonoprazan (20 mg bid) combined with high-dose amoxicillin (750 mg qid) provided satisfactory eradication rates (ITT analysis: 86.6% vs 89.5%; PP analysis 90.9% vs 94.5%, respectively). The 10-day regimen was not inferior to the 14-day regimen. However, the eradication effect of 10 days of vonoprazan (20 mg bid) combined with low-dose amoxicillin (1000 mg bid) was not satisfactory, with ITT and PP analysis showing rates of 79.7% and 82.0%, respectively. 23 In another RCT (n = 314) comparing the efficacy of the 10-day dual therapy (vonoprazan 20 mg bid and amoxicillin 1000 mg tid) versus the 14-day BQT, it was found that in the ITT analysis (86.0% vs 89.2%), MITT (88.2% vs 91.5%), and PP (90.8% vs 91.3%), the 10-day VA dual therapy was not inferior to the 14-day BQT. 24 A multicenter RCT with a larger sample size (n = 690) compared the efficacy of a 10-day VA dual therapy (vonoprazan 20 mg bid + amoxicillin 1000 mg tid) to a 14-day RA dual therapy (rabeprazole 10 mg tid + amoxicillin 1000 mg tid) for H. pylori eradication. The results showed that the 10-day VA therapy was non-inferior to the 14-day RA therapy in ITT analysis (89.3% vs 84.9%), MITT analysis (90.6% vs 85.9%), and PP analysis (91.4% vs 86.6%). 25 In our study, the eradication rates of the 10-day VA dual therapy in ITT, MITT, and PP analyses were generally consistent with the results of the three recent studies mentioned above.
During the eradication process of H. pylori, adverse events often occur. In this study, the incidence rates of adverse events in the 10-day VA regimen and 14-day EA regimen were 9.1% and 11.7%, respectively, with no statistically significant difference between the two groups, which was similar to previous studies.17,25 This indicates that the 10-day VA dual therapy for H. pylori eradication has good safety, providing a new safe option for first-line treatment.
The medication compliance has a significant impact on eradicating H. pylori. Poor compliance may not only lead to treatment failure but also secondary antibiotic resistance due to inadequate drug dosage.26,27 In this study, we found that the medication compliance of the 10-day VA regimen was higher than the 14-day EA regimen. Furthermore, the results of multivariate logistic regression analysis showed that poor compliance can increase the risk of eradication failure, which was consistent with recent research findings.25,28 It is worth noting that in our multicenter study, the ITT eradication rate of the 14-day EA was only 76.7%, lower than the ITT eradication rates of several previous RCTs.8,10,11,29 This discrepancy may be partly due to the fact that our study included participants from a wider range of regions compared to previous studies, and regional, demographic, and antibiotic resistance differences may influence the eradication rates. Another potentially more significant reason is that our study had slightly lower compliance compared to previous studies, with a higher proportion of participants lost to follow-up and not rechecked with UBT, leading to a lower eradication rate in ITT analysis. In the VA group, patients took vonoprazan twice and amoxicillin three times daily after meals, while in the EA group, patients took amoxicillin four times daily after meals and esomeprazole four times daily before meals. This difference in dosing regimens made the EA regimen more complex and may potentially impact patient compliance, thereby affecting the eradication outcome. Compared to the 14-day EA regimen, the 10-day VA regimen offers a lower frequency of dosing, a shorter treatment period, and a more convenient administration method (amoxicillin and vonoprazan were both taken after meals), resulting in better compliance.
Limitations
Several limitations in our research should not be ignored. First, this study did not conduct testing for amoxicillin resistance in H. pylori; therefore, the impact of amoxicillin resistance on the eradication of H. pylori cannot be evaluated. Without resistance testing, it is difficult to fully assess the efficacy of the treatment regimens, especially considering the increasing concern about antibiotic resistance in H. pylori treatment. However, the general resistance to amoxicillin in China was quite low (approximately 3%), 18 so antibiotic resistance testing may not have a significant impact on the results. Second, this regimen was not suitable for patients with allergies to penicillin or amoxicillin, as it contained amoxicillin. This limits the clinical application of the findings. It is worth noting that Gao et al. 30 recently developed a new dual therapy regimen—vonoprazan–tetracycline dual therapy (vonoprazan 20 mg bid + tetracycline 500 mg tid, 14 days)—which achieved a satisfactory eradication rate of 92%. Therefore, for patients allergic to amoxicillin, vonoprazan–tetracycline dual therapy or bismuth quadruple therapy without amoxicillin can be used. Third, the genetic background of patients, especially involving enzymes such as CYP2C19 and CYP3A4, can significantly impact the pharmacokinetics of esomeprazole and vonoprazan. CYP2C19 polymorphism can affect the metabolism of esomeprazole, leading to reduced efficacy in rapid metabolizers due to faster drug clearance. While vonoprazan is minimally affected by CYP2C19 variability, its metabolism primarily relies on CYP3A4, 21 and genetic variations in CYP3A4 may still influence the efficacy of vonoprazan. This study did not detect CYP2C19 and CYP3A4 gene polymorphisms, so the potential impact on eradication rates cannot be ruled out. Fourth, this study was an open-label trial, potentially affecting the evaluation of the subjective outcome (adverse events). Finally, the conclusions of this study may only apply to the Chinese population. Further research may be needed to optimize treatment regimens, dosages, and dosing frequencies for patients in other regions.
Conclusion
In conclusion, the 10-day VA dual therapy showed a satisfactory eradication rate of 91.1% (PP analysis), demonstrating good safety and better compliance compared to the 14-day EA dual therapy. Considering the satisfactory eradication rate of over 90% with the 10-day dual therapy, along with a lower frequency of dosing, a shorter treatment duration, and more convenient administration, it should be prioritized as the first-line eradication regimen.
Supplemental Material
sj-doc-1-tag-10.1177_17562848241309870 – Supplemental material for Ten-day vonoprazan–amoxicillin dual therapy versus 14-day esomeprazole–amoxicillin dual therapy for first-line Helicobacter pylori eradication: a prospective multicenter randomized controlled trial
Supplemental material, sj-doc-1-tag-10.1177_17562848241309870 for Ten-day vonoprazan–amoxicillin dual therapy versus 14-day esomeprazole–amoxicillin dual therapy for first-line Helicobacter pylori eradication: a prospective multicenter randomized controlled trial by Ben-Gang Zhou, Ming-Wen Guo, Li-Juan Zhang, Zhi-Dong Liu, Chun-Hua Liu, Xue-Feng Li, Shun-Song Li, Peng Xiao, Bing Bao, Yao-Wei Ai and Yan-Bing Ding in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We express our heartfelt gratitude to Yi-Juan Guo, Yao-Jia Guo, Meng-Jin Wang, Min Wang, Xin-Tong Zhang, Lu Xie, Qiang She, Yao-Sheng Chen, and Zhan-Lou Hua for their dedication to patient follow-up and generous help. We are also very grateful to Dr. Meng-Jie Lu for providing us with random sequence numbers.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
