Abstract
Background:
Although the Maastricht VI/Florence consensus report recommended high-dose proton pump inhibitor–amoxicillin dual therapy as possible rescue therapy for Helicobacter pylori infection, clinical evidence of its efficacy was lacking.
Objectives:
To compare the efficacy, safety, patient compliance, and cost between high-dose dual therapy (HDDT) and culture-based susceptibility-guided therapy (CB-SGT) as a rescue regimen for H. pylori infection.
Design:
A single-center, open-label, randomized controlled clinical trial.
Methods:
In all, 146 patients with a history of eradication failure were enrolled and randomly assigned to receive HDDT or CB-SGT. HDDT consisted of esomeprazole 20 mg and amoxicillin 750 mg, both given four times per day (qid). CB-SGT consisted of esomeprazole 20 mg twice daily (bid), amoxicillin 1000 mg bid plus clarithromycin 500 mg bid, metronidazole 400 mg bid, or levofloxacin 500 mg daily (qd) for sensitive patients, in that order. For patients with triple resistance, a bismuth-containing regimen with a high dose of metronidazole was chosen, including esomeprazole 20 mg bid, bismuth 220 mg bid, amoxicillin 1000 mg bid, and metronidazole 400 mg qid. All regimens were given for 14 days.
Results:
The eradication H. pylori rates achieved with HDDT in the intention-to-treat (ITT), per-protocol, and modified ITT analyses were all 84.9% [62/73, 95% confidence interval (CI): 76.5–93.9%], compared with 83.6% (61/73, 95% CI: 74.9–92.3%), 84.7% (61/72, 95% CI: 76.2–93.2%), and 84.7% (61/72, 95% CI: 76.2–93.2%) with CB-SGT, respectively. For patients with CYP2C19 polymorphisms of intermediate/poor metabolizers, the eradication rates of HDDT and CB-SGT were 90.70% (39/43, 95% CI: 77.86–97.41%) and 84.21% (32/38, 95% CI: 68.75–93.98%), respectively. The difference between groups was 6.49% (95% CI: −8.00% to 20.97%), and the non-inferiority p value was 0.0128. For patients with a treatment interval of more than 3 months, the eradication rates of the two regimens reached 88.71% (95% CI: 78.11–95.34%) and 71.97% (95% CI: 70.02–90.64%). The difference between groups was 6.74% (95% CI: −5.71% to 19.20%), with a non-inferiority p value of 0.0042. Patient adherence was high in both groups. The HDDT had a lower cost and rate of side effects (p < 0.001) compared with CB-SGT.
Conclusions:
HDDT can reach an eradication rate of 85% in treatment-experienced patients of H. pylori infection and 91% in patients with CYP2C19 polymorphisms of intermediate/poor metabolizers, with good compliance, lower side effects and costs, and less use of antibiotics. In conclusion, HDDT offers an effective rescue regimen for H. pylori infection.
Registration:
This clinical trial was registered at the Chinese Clinical Trail Registry (trail registration number: ChiCTR1900025044)
Introduction
Helicobacter pylori (H. pylori) infection is associated with various gastrointestinal diseases, including chronic gastritis, peptic ulcers, gastric mucosa-associated lymphoid tissue lymphoma, and gastric adenocarcinoma. Current guidelines recommend eradication of H. pylori regardless of clinical symptoms.1,2 However, the prevalence of H. pylori antibiotic resistance has been increased globally. 3 In China, pooled primary resistance rates of greater than 26% to clarithromycin, 60% to metronidazole, and 28% to levofloxacin were reported, with wide variation in these rates among different regions.4,5 These increased antibiotic resistance rates make eradication of H. pylori difficult, especially after primary treatment failure.
In consideration of the increasing rates of antibiotic resistance, it is essential to obtain individual antibiotic susceptibility data before treatment for H. pylori infection. The Maastricht V/Florence Consensus Report recommends culture-based susceptibility guided therapy (CB-SGT) as an evidence-based rescue treatment regimen whenever possible, especially in areas with high antibiotic resistance, and research has demonstrated the effectiveness of CB-SGT. 6 A single-arm interventional trial by Yu et al. showed that CB-SGT achieved an eradication rate of 94.5% in intention-to-treat (ITT) analysis. 7 Compared with empirical treatment, CB-SGT has the advantages of providing personalized treatment, reducing unnecessary antibiotic prescriptions, and allowing continued use of triple therapy for antibiotic-sensitive patients in areas with high antibiotic resistance. CB-SGT also has some limitations. For example, individual susceptibility testing is still not always available, despite considerable progress in recent years. In this case, clinicians commonly choose empiric therapy for H. pylori eradication based on local drug resistance data and guideline recommendations. Such therapies include concomitant or sequential non-bismuth quadruple therapy (BQT), BQT (containing metronidazole and tetracycline), and levofloxacin-containing triple therapy. However, these empirical treatment regimens also have shortcomings. For example, concomitant non-BQT, which contains three antibiotics, may lead to unnecessary antibiotic administration and multi-drug resistance. 8 Moreover, tetracycline and bismuth are commonly not available in many institutions. Thus, it is necessary to find a regimen that offers high effectiveness, good accessibility, few side effects, and a low cost.
In recent years, high-dose dual therapy (HDDT), which contains a double dose of proton pump inhibitor (PPI) and at least 3 g of amoxicillin (both administered three times daily or more), has produced satisfactory eradication rates as first-line treatment for H. pylori infection. Study results have shown that compared with BQT, HDDT can achieve a comparable eradication rate with a lower rate of side effects. 9 Similarly, our previous study demonstrated that HDDT conforms to recommended treatment regimens and also confirmed that dosing four times daily can increase the effectiveness of HDDT (p = 0.030). 10
However, the evidence for HDDT as an empirical rescue treatment regimen is insufficient, even if recent guidelines recommend this regimen as a rescue treatment.2,11,12 First, few randomized controlled trials (RCTs) have evaluated HDDT as a rescue therapy regimen. Second, the efficacies of different dual therapies as tailored regimens are inconsistent and conflicting among relevant studies. For example, one RCT in Germany demonstrated that HDDT with omeprazole 120 mg and amoxicillin 3000 mg daily produced a less than 70% ITT eradication rate in patients in whom prior treatment had failed at least once. 13 In contrast, another study in Japan showed that 14-day HDDT with rabeprazole 10 mg and amoxicillin 500 mg, both administrated four times daily, accomplished a high ITT eradication rate exceeding 90% in patients after failure of 7-day triple therapy. 14 Therefore, it is essential to determine whether HDDT is as effective as CB-SGT as a rescue treatment regimen for H. pylori infection in different populations.
In the present trial, we compared the H. pylori eradication efficacy as well as safety, patient compliance, and cost between HDDT and CB-SGT in patients with a history of at least one previous H. pylori treatment failure. The findings of this study provide evidence for the use of HDDT as an empiric rescue regimen for eradicating H. pylori.
Materials and methods
Trial design and participants
This open-label RCT was conducted from August 2019 to July 2021 at Daping Hospital, Army Medical University. The study was approved by the Ethics Committee of Daping Hospital, Army Medical University, number (2019) 49. The study was registered at the Chinese Clinical Trial Registry (ChiCTR1900025044) and reported as recommended in the CONSORT statement.
From August 2019 to July 2021, patients aged between 18 and 70 years with chronic gastritis diagnosed by gastroscopy, positive H. pylori status by 13C-urea breath test (UBT), and a history of at least one standardized eradication failure were initially screened and enrolled in the Department of Gastroenterology, Daping Hospital, Army Medical University. The exclusion criteria included the following: currently suffering from other diseases (liver disease, etc.); use of a PPI, an H2-receptor antagonist, bismuth, an antibiotic, or a probiotic within the previous 4 weeks; an allergy to the study drug; pregnancy or breastfeeding; a history of gastric surgery; a history of H. pylori eradication with HDDT; and refusal to provide informed consent. A detailed flow diagram of the enrollment of the study population is presented in Figure 1.

Flow diagram of trial enrollment and analysis.
Randomization and blinding
All eligible patients were randomized to receive HDDT or CB-SGT at a ratio of 1:1 (Figure 1). An independent statistician provided the block randomization sequence by generating computerized random numbers with a block size of four. All investigators and participants were blinded to the randomization sequence. All random codes were placed in sealed opaque envelopes, which were kept by an independent research assistant in Daping Hospital. After each participant signed the informed consent form, the assistant opened the envelope with the treatment plan assigned to the participant and administered the corresponding H. pylori eradication treatment. The study was open-label, and the eradication protocol was not blinded to participants. The personnel involved in cultures, antibiotic resistance testing, and UBT were blinded to the eradication treatment allocation.
Antimicrobial susceptibility testing
We selected the agar dilution method for antimicrobial susceptibility testing of H. pylori isolates. Different concentrations of antibiotics were applied to the agar medium. Growth under exposure to the selected antibiotic indicated resistance, while no growth was considered an indication of susceptibility among the isolates. According to the US Clinical and Laboratory Standards Institute guidelines, the minimum inhibitory concentrations (MICs) of the antibiotics were as follows: 1 μg/mL clarithromycin, 2 μg/mL amoxicillin, 2 μg/mL levofloxacin, 2 μg/mL furazolidone, 2 μg/mL tetracycline, and 8 μg/mL metronidazole. The standard strain ATCC 43504 (NCTC11637 H. pylori strain) was used as a control. Resistance was defined by MIC values greater than 1 μg/mL clarithromycin, 2 μg/mL amoxicillin, 2 μg/mL furazolidone, 2 μg/mL tetracycline, and 8 μg/mL metronidazole.
CYP2C19 genotyping
Genomic DNA was extracted from the gastric mucosal tissues of the study participants using a Genomic DNA Isolation Kit and used as a template for polymerase chain reaction (PCR) using primers as reported previously.15,16 CYP2C19 DNA sequencing was performed using the PCR products. Confronting two-pair forward and reverse primers were used to determine CYP2C19 polymorphisms separately. Sequencing Analysis 5.2 was employed for CYP2C19 polymorphism analysis. According to the genotypes of CYP2C19, the study participants were categorized into subgroups of extensive metabolizers, rapid metabolizers, intermediate metabolizers, and poor metabolizers.
Interventions
The 14-day HDDT group received esomeprazole (AstraZeneca China, Shanghai, China) 20 mg qid and amoxicillin (The United Laboratories, Hong Kong, China) 750 mg qid. Esomeprazole was administered 30 min before meals and 1 h before bedtime, and amoxicillin was administered 30 min after meals and before bedtime. The CB-SGT group was assigned a 14-day regimen according to the results of susceptibility testing (Figure 1). For clarithromycin-susceptible strains, esomeprazole 20 mg plus amoxicillin 1000 mg and clarithromycin (Abbott Pharmaceutical Co. Ltd., Shanghai, China) 500 mg (EAC) was administered, all twice daily. For clarithromycin-resistant but metronidazole-susceptible strains, esomeprazole 20 mg, amoxicillin 1000 mg, and metronidazole (Sichuan Kelun Pharmaceutical Co. Ltd., Sichuan, China) 400 mg bid (EAM2) were administered. For strains with clarithromycin and metronidazole resistance but susceptibility to levofloxacin, esomeprazole 20 mg bid combined with amoxicillin 100.0 mg bid and levofloxacin (Daiichi Sankyo Pharmaceutical Co. Ltd., Beijing, China) 500 mg qd were given (EAL). In addition, patients with triple-resistance received esomeprazole 20 mg, bismuth (Livzon Pharmaceutical Group, Zhuhai, China) 220 mg bid plus amoxicillin 1000 mg bid and metronidazole 400 mg qid (EBAM4). Esomeprazole and bismuth were given 30 min before breakfast and dinner; amoxicillin was given 30 min after meals; metronidazole doses were given 30 min after three meals and before bedtime.
All participants were educated about drug administration and adverse events before the start of treatment and were required to record any adverse events. In a telephone follow-up interview, patients were asked about adverse events, which were classified as ‘mild’ (feeling uncomfortable but not affecting daily life and work), ‘moderate’ (feeling uncomfortable and partially affecting life and work), or ‘severe’ (severe interference with daily life and work) based on their impact on daily life and work. Taking more than 80% of the total number of pills was defined as good adherence.
Outcomes
The primary outcome of this trial was the eradication rate achieved with HDDT compared with CB-SGT. At least 4 weeks after the treatment, H. pylori status was determined by 13C-UBT. Each patient took a capsule containing 13C-urea 75 mg (Zhonghe Headway Bio-Sci & Tech Co. Ltd, Shenzhen, China) orally. Baseline and 30-min breath samples were collected and assayed. Negative H. pylori status was defined when the detected value was less than the cutoff value of 2.4‰. Secondary outcomes included the incidence of adverse events, patient adherence, medication cost, rates of antibiotic resistance, and risk factors affecting eradication rates. The drug cost was calculated with reference to the 2020 Medication Pricing Catalogue in Chongqing. All prices were calculated in USD. According to the average exchange rate in 2020 from the China Foreign Exchange Trade System, 1.00 USD = 6.90 CNY.
Sample size and statistical analysis
This trial was designed as a non-inferiority test. The sample size was calculated according to the results from previous studies.17,18 The eradication rates achieved with CB-SGT and HDDT were assumed to be 81.6% and 88.7%, respectively. With a non-inferiority margin delta of −0.1 (−10%), a power of 80%, and a one-sided alpha of 0.025, at least 66 participants were needed in each group to detect non-inferiority. Assuming a loss to follow-up rate of 10%, a total of 146 participants were required for this trial.
Eradication rates were evaluated using ITT, modified ITT (mITT), and per-protocol (PP) analyses. All participants were included in the ITT analysis. Participants without 13C-UBT results were considered as eradication failures in the ITT analysis and excluded from the mITT analysis. Participants with poor adherence were excluded from the PP analysis. The Clopper–Pearson method was chosen to calculate the confidence limits for eradication rates using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). Means with standard deviations and percentages were used to describe continuous variables and categorical variables, respectively. The Shapiro–Wilk test was used to evaluate the normal distribution of continuous variables. The t-test was used to evaluate differences between the two groups for continuous variables that conformed to the normal distribution, and the Mann–Whitney test was used to evaluate differences between continuous variables that were not normally distributed. The chi-square test or Fisher’s exact test was used to assess the differences in categorical variables between two groups. Statistical significance was considered when p < 0.05 (two-sided).
Results
Baseline demographic and clinical characteristics of study participants
Between August 2019 and July 2021, 357 H. pylori-positive treatment-experienced patients were assessed for eligibility, and 254 strains (71.1%) were successfully cultured. Of these, 146 patients were enrolled and randomly allocated to receive either HDDT or CB-SGT (Figure 1). All patients took more than 80% of the prescribed drugs (good adherence). One participant lacked the 13C-UBT result after eradication and was recorded as eradication failure and excluded from the mITT and PP analyses. The baseline demographic and clinical characteristics of the study participants in each treatment group are presented in Table 1. No significant differences in baseline and clinical characteristics were observed between the two groups, with the exception of a difference in gender (p = 0.006).
Baseline demographic and clinical characteristics of the study participants in each treatment group.

Data are expressed as mean ± SD; categorical data are presented as number of patients and percentage in parentheses.
Missing data.
CB-SGT, culture-based susceptibility-guided therapy; HDDT, high-dose dual therapy; SD, standard deviation.
Compared with the corpus mucosa, the antral mucosa exhibited a greater degree of inflammation (p < 0.001), more inflammatory activity (p < 0.001), a greater extent of atrophy (p = 0.043), a higher H. pylori load (p = 0.016), and more serious intestinal metaplasia (p < 0.001). The histology results for the antrum and corpus are presented in Supplemental Table 1.
H. pylori eradication rates
The H. pylori eradication rates in the ITT analysis were 84.9% [95% confidence interval (CI):76.5–93.9%] and 83.6% (95% CI: 74.9–92.3%) for HDDT and CB-SGT, respectively. The eradication rates determined in the mITT and PP analyses were 84.9% (76.5–93.9%) for HDDT and 84.7% (76.2–93.2%) for CB-SGT. As shown in Table 2, significant differences were not observed between the two groups in eradication rates (p = 0.820 from ITT analysis and p = 0.972 from PP and mITT analysis). For patients with CYP2C19 polymorphisms of intermediate/poor metabolizers, the eradication rates of HDDT and CB-SGT were 90.70% (95% CI: 77.86–97.41%) and 84.21% (95% CI: 68.75–93.98%), respectively. The difference between groups was 6.49% (95% CI: −8.00% to 20.97%), and the non-inferiority p value was 0.0128. For patients with a treatment interval of more than 3 months, the eradication rates of the two regimens reached 88.71% (95% CI: 78.11–95.34%) and 71.97% (95% CI: 70.02–90.64%). The difference between groups was 6.74% (95% CI: −5.71% to 19.20%), with a non-inferiority p value of 0.0042 (Table 3).
H. pylori eradication rates achieved with HDDT and CB-SGT.

p values were obtained from one-sided test comparisons of non-inferiority between the HDDT and CB-SGT groups.
p values were from two-sided comparisons of differences between the HDDT and CB-SGT groups.
CB-SGT, culture-based susceptibility-guided therapy; CI, confidence interval; HDDT, high-dose dual therapy; ITT, intention-to-treat; mITT, modified ITT; PP, per-protocol.
Subgroup analyses in H. pylori eradication rates achieved with HDDT and CB-SGT.

p values were obtained from one-sided test comparisons of non-inferiority between the HDDT and CB-SGT groups.
p values were from two-sided comparisons of differences between the HDDT and CB-SGT groups.
CB-SGT, culture-based susceptibility-guided therapy; CI, confidence interval; HDDT, high-dose dual therapy; ITT, intention-to-treat.
The efficacy of each CB-SGT regimen was also evaluated by subgroup analysis. The eradication rates from the ITT analysis were 87.0% (20/23, 95% CI: 66.4–97.2%), 95% (21/22, 95% CI: 77.2–99.9%), 100% (2/2, 95% CI: 100–100%), and 69.2% (18/26, 95% CI: 48.2–85.7%) with EAC, EAL, EAM2, and EBAM4, respectively, and those from the PP analysis were 90.9% (20/22, 95% CI: 70.8–98.9%), 95.5% (21/22, 95% CI: 77.2–99.9%), 100% (2/2, 95% CI: 100–100%), and 69.2% (18/26, 95% CI: 48.2–85.7%), respectively. For the rapid metabolism, intermediate metabolism, and poor metabolism groups, the eradication rates were 78.5% (51/65), 86.7% (52/60), and 95.2% (20/21), respectively (p = 0.132).
Antibiotic resistance rates
The overall antibiotic resistance rate among the study participants were 72.6% (106/146) for clarithromycin, 94.5% (138/146) for metronidazole, 50.0% (73/146) for levofloxacin, 0.7% (1/146) for amoxicillin, and 0.7% (1/146) for tetracycline. All participants were found to be susceptible to furazolidone. The rates of dual resistance were 70.5% (103/146) for clarithromycin and metronidazole, 42.5% (62/146) for clarithromycin and levofloxacin, and 47.3% (69/146) for metronidazole and levofloxacin. In addition, the rate of triple resistance was 41.8% (61/146). No significant differences in antibiotic resistance rates were detected between the HDDT and CB-SGT groups (Table 1). In patients who had received prior treatment with regimens containing clarithromycin, metronidazole, and levofloxacin, the resistance rates were 80.7% (88/109), 91.9% (80/87), and 100% (7/7) to these antibiotics, respectively, among our study patients.
Adverse events, compliance, and medication cost
The prevalence rates of adverse events were 13.7% (10/73) in the HDDT group and 41.7% (31/73) in the CB-SGT group (p < 0.001). No severe side effects were reported, and all adverse events were resolved after the conclusion of treatment (Table 4). Significant differences were observed in the occurrence of taste distortion (p = 0.001) and tongue discoloration/darkened stool (p = 0.006) between the groups. Adherence was good for all patients, and no one withdrew from the study due to side effects.
Drug-induced adverse events and patient adherence in HDDT and CB-SGT groups.
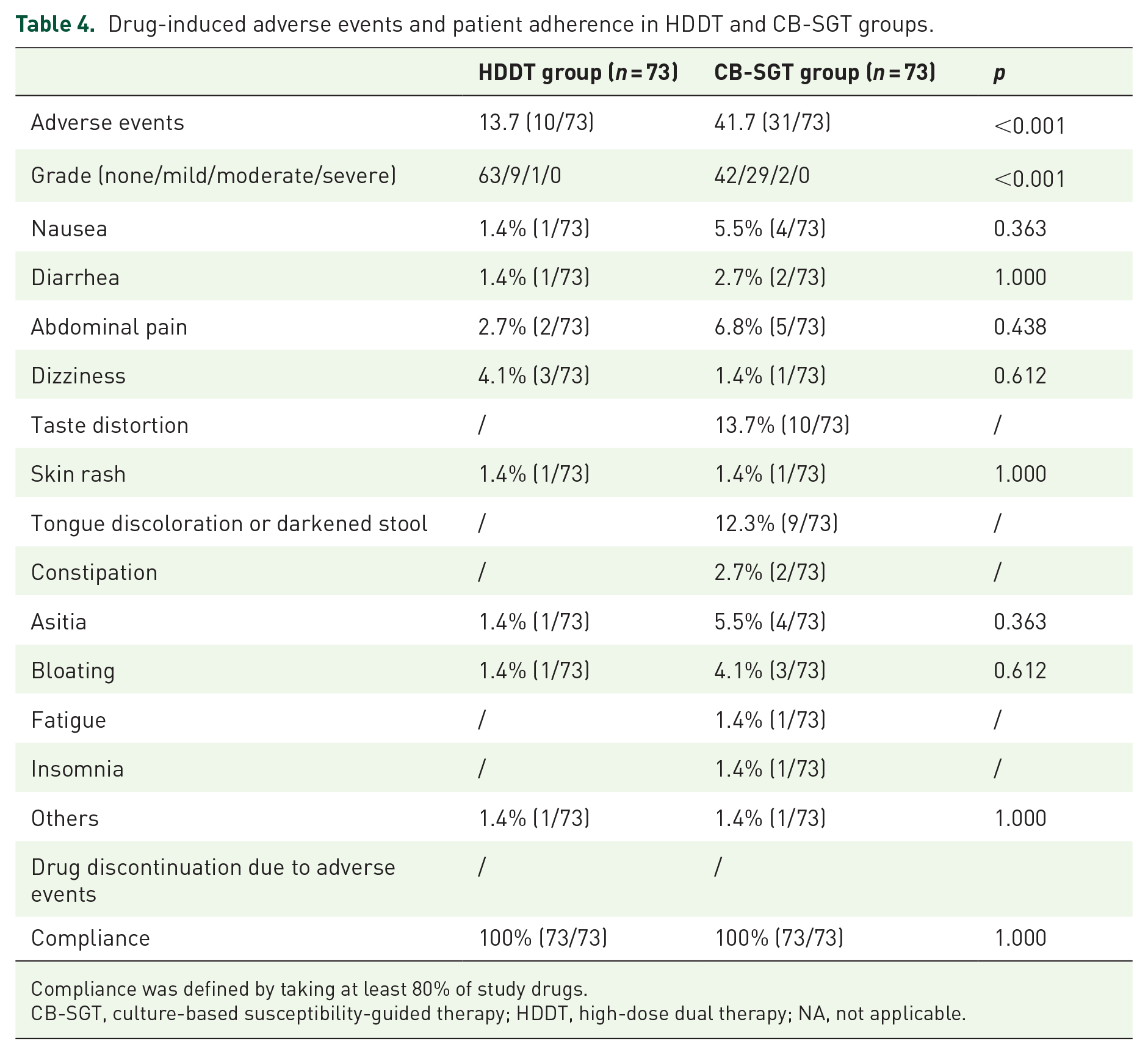
Compliance was defined by taking at least 80% of study drugs.
CB-SGT, culture-based susceptibility-guided therapy; HDDT, high-dose dual therapy; NA, not applicable.
The total drug cost for HDDT was $81.71 ($66.49 for 56 tablets of esomeprazole and $15.22 for 168 capsules of amoxicillin). The total drug costs for the different CB-SGT regimens were $98.17 ($33.25 for 28 tablets of esomeprazole plus $10.14 for 112 capsules of amoxicillin plus $54.78 for 56 pills of clarithromycin) for EAC, $60.19 ($33.25 for 28 tablets of esomeprazole and $10.14 for 112 capsules of amoxicillin plus $16.80 for 14 capsules of levofloxacin) for EAL, $44.10 ($33.25 for 28 tablets of esomeprazole plus $10.14 for 112 capsules of amoxicillin and $0.71 for 56 pills of metronidazole) for EAM2, and $52.50 ($33.25 for 28 tablets of esomeprazole plus $7.69 for 56 capsules of bismuth plus $10.14 for 112 capsules of amoxicillin plus $1.42 for 112 capsules of metronidazole) for EBAM4. Additional costs for CB-SGT included $101.68 for the susceptibility test performance and endoscopy and $2.17 for an additional registration fee.
Factors affecting the efficacy of HDDT and CB-SGT
To determine the risk factors affecting the H. pylori eradication rate, univariate and multivariate analyses were performed, including gender, age, body mass index (BMI), cigarette smoking, alcohol drinking, history of H. pylori eradication, time interval since last treatment, atrophy, intestinal metaplasia, CYP2C19 genotype, clarithromycin resistance, metronidazole resistance, levofloxacin resistance, and triple resistance. Among the total study population, cigarette smoking and BMI >23 kg/m2 were identified as risk factors for treatment failure in both univariate and multiple logistic regression analyses (Supplemental Table 2), and subgroup analysis found that these two factors mainly influenced the eradication rate of HDDT (p = 0.001 versus p = 0.558 for cigarette smoking and p = 0.002 versus p = 0.127 for BMI). Resistance to levofloxacin [odds ratio (OR): 4.068, 95% CI: 1.412–11.722, p = 0.009] and triple resistance (OR: 4.622, 95% CI: 1.688–12.658, p = 0.003) were significantly associated with the eradication rate in only the univariate analysis. The other factors, such as alcohol drinking, atrophy, and CPY2C19 genotype, did not affect the eradication rate in this study.
Discussion
To our best knowledge, this trial is the first comparative study to evaluate the efficacies of HDDT and CB-SGT as rescue treatment regimens for H. pylori infection. The main results of this trial are as follows: First, no statistical difference in the H. pylori eradication rate was found between HDDT and CB-SGT according to the ITT, MITT, and PP analyses, indicating that HDDT can be used as a rescue treatment to eradicate H. pylori infection. Second, the overall incidence of adverse effects with HDDT was significantly less than that with CB-SGT (p < 0.001), and third, the cost of HDDT is lower than that of CB-SGT.
In this study, as a second-line or rescue treatment regimen, HDDT achieved an eradication rate of 85% in both the ITT and PP analyses and 91% for patients with CYP2C19 polymorphisms of intermediate/poor metabolizers. Several factors may explain this high eradication rate. First, H. pylori strains have low resistance to amoxicillin in most regions, both in terms of primary and secondary resistance.3,19 In China, the overall rate of resistance to amoxicillin is only 3.23%, and the primary and secondary resistance rates are 2.20% and 6.12%, respectively. 20 Second, amoxicillin is stable in solutions with a pH value greater than 6. Thus, inhibition of gastric acid secretion can improve the efficacy of amoxicillin. Third, esomeprazole metabolism is less affected by CYP2C19 gene polymorphism, and dosing four times daily can maintain a stable high-pH intragastric environment. In addition, amoxicillin is a time-dependent antibiotic, which means its high-dose application can achieve a longer percentage of time with plasma concentrations exceeding the MIC. Similarly, high eradication rates have been reported in other studies. For example, Yang et al. gave patients who had previously received anti-H. pylori therapies a 14-day high-dose and -frequency regimen of rabeprazole (20 mg, four times daily) and amoxicillin (750 mg, four times daily) and reported a high eradication rate both in ITT and PP analyses [89.3% (50/56), 95% CI: 80.9–97.6%] compared with 10 days of sequential therapy and 7 days of levofloxacin-containing triple therapy [51.8% (29/56) and 78.6% (44/56) in ITT, 53.7% (29/54) and 78.6% (44/56) in PP analysis, respectively]. 18
This study demonstrated that triple therapy can still achieve a high H. pylori eradication rate for patients who are sensitive to clarithromycin, metronidazole, or levofloxacin [90.9% (20/22), 95% (19/20), and 100% (1/1) in PP analysis, respectively]. These results show that SGT can reduce unnecessary antibiotic prescriptions while still achieving a high eradication rate. However, for patients with triple antibiotic resistance, the eradication rate achieved with bismuth-containing therapy including high-dose metronidazole was low (69.2% [18/26], 95% CI: 48.2–85.7% in ITT and PP analyses). We chose this regimen for patients with triple resistance according to guideline recommendations 21 and also in consideration of the advantages of low cost and good availability. To our knowledge, increasing the dose of metronidazole can overcome resistance, and the application of bismuth can also indirectly overcome drug resistance. 22 Thus, we expect that increasing the dose of amoxicillin in this regimen may improve eradication rates. For example, as mentioned above, Yu et al. reported a high eradication rate of 95.1% (137/144, 95% CI: 90.2–98.0%) with a bismuth-containing therapy with high-dose amoxicillin (3 g daily) and metronidazole (1.6 g daily) for patients with triple resistance. 7 Therefore, we believe that other combinations of antibiotics, such as amoxicillin plus furazolidone or furazolidone plus tetracycline, may be considered for H. pylori-infected patients with triple resistance. Tetracycline and furazolidone, however, are not available in many medical institutions in China. In addition, CB-SGT may be more effective for treatment-naive patients. The results from Romano et al. show that susceptibility testing-based regimens lead to the significantly higher eradication rates [97.3% (71/73, 95% CI: 91.2–99.5%) in PP analysis and 94.6% (71/75, 95% CI: 87.6–98.3%) in ITT analysis, p < 0.005] and have lower costs when compared with the regimens without susceptibility testing as first-line treatment [79.4% (58/73, 95% CI: 69.1–87.6%) in PP analysis and 77.3% (58/75, 95% CI: 66.9–85.7%) in ITT analysis]. 23
Although the H. pylori eradication rates did not differ significantly between the two treatment groups in our study, the incidence and severity of adverse events were lower in the HDDT group than in the CB-SGT group (p < 0.001). During treatment with HDDT, three (4.1%) cases of dizziness and two (2.7%) cases of abdominal pain were reported, and the other reported adverse events occurred in only one patient each (1.4%). In contrast, in the CB-SGT group, 10 (13.7%) patients reported that they had experienced taste distortion and 9 (12.3%) patients reported that they had experienced darkened tongue or stool during treatment. Almost all patients with altered taste had received the EAC regimen, and all patients with tongue discoloration or darkened stool had received EBAM4, suggesting that these adverse events were caused by clarithromycin and bismuth.
The medication cost for HDDT ($81.71) was lower than those for CB-SGT ($202.02, $164.04, $147.95, and $156.35 for EAC, EAL, EAM, and EBAM4, respectively). Therefore, for individuals with susceptibility to clarithromycin, levofloxacin, or metronidazole, HDDT offered savings of $120.31, $66.24, or $66.24 compared with the different CB-SGT regimens, respectively. Also, for patients with triple resistance, HDDT cost $74.64 less than BC-SGT.
While CB-SGT has many advantages, it also requires additional endoscopy. 24 In addition, culture is time-consuming. Fortunately, culture is not the only useful method for obtaining antibiotic susceptibility information. Molecular methods based on stool or gastric biopsy samples may provide a more convenient and time-saving method and promote the applicability of SGTs. 25 A previous study reported that for refractory H. pylori treatment, susceptibility therapy guided by molecular methods is an acceptable choice compared with the medication history-guided empirical therapy. 26
In this study, the resistance rates in treatment-experienced patients were significantly higher than in those in treatment-naïve patients. By comparison, our previous study found resistance rates of 29.7%, 39.7%, and 96.6% for clarithromycin, levofloxacin, and metronidazole in H. pylori treatment-naïve patients, respectively. Moreover, the double resistance rates for clarithromycin and metronidazole, clarithromycin and levofloxacin, and metronidazole and levofloxacin were 29.3%, 13.8%, and 36.2%, respectively, whereas the triple resistance rate was 13.4%. 27 In this study, the secondary drug resistance rates were high in patients previously treated with clarithromycin, levofloxacin, and metronidazole (80.7%, 100%, and 91.9%, respectively), and multiple resistance rates were also high according to prior treatment with dual or triple therapies (70.5% for clarithromycin–metronidazole, 42.5% for clarithromycin–levofloxacin, 47.3% for metronidazole–levofloxacin, and 41.8% for clarithromycin–metronidazole–levofloxacin). Comparison of the results from our previous study in H. pylori treatment-naïve patients and the current study in patients with a history of H. pylori treatment failure shows that antibiotic resistance is likely to develop after treatment failure. Consistently, a previous systematic review showed that among 1748 patients who received first-line regimens containing clarithromycin, 58.7% developed clarithromycin resistance after treatment, while 89.7% of 429 patients developed metronidazole resistance after receiving first-line regimens containing metronidazole. 28 Taken together, these results indicate that drug resistance data or previous eradication data of patients must be obtaining when choosing rescue treatment regimens to avoid using antibiotics to which patients are resistant. Local, regional, or national data on susceptibility to H. pylori also need to be regularly assessed, especially in areas with high resistance rates.
This study has some limitations. First, the sample size was relatively small. In future studies, it may be better to randomize groups at 1:3 to receive HDDT and SGT, as this would ensure enough samples for analysis of each regimen of SGT. Second, in areas with high amoxicillin resistance, such as Pakistan or Iran,3 HDDT is not suitable as an empirical treatment regimen. Third, HDDT cannot be used for patients who are allergic to penicillin. Third, in other populations with poor adherence, the reproducibility of our results may not be assured because patients are not able to accept this complex regimen, even if it has a low incidence of adverse events.
In conclusion, our research results show that HDDT offers comparable efficacy at a lower cost and with fewer side effects for eradication of H. pylori in patients with a history of treatment failure. Therefore, we propose that HDDT can serve as an alternative rescue regimen for H. pylori infection.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221145566 – Supplemental material for High-dose dual therapy versus culture-based susceptibility-guided therapy as a rescue regimen for Helicobacter pylori infection: a randomized controlled trial
Supplemental material, sj-docx-1-tag-10.1177_17562848221145566 for High-dose dual therapy versus culture-based susceptibility-guided therapy as a rescue regimen for Helicobacter pylori infection: a randomized controlled trial by Zhe Zhao, Pei-Ying Zou, Na-Yun Su, Yan Guo, Xing-Wei Wang, Jing-Tao Zhao, Hao Mei, Qing Shi, Bin Wang, Dong-Feng Chen and Chun-Hui Lan in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors thank the physicians in the Department of Gastroenterology of the Daping Hospital for patient screening. We also thank the endoscopists and pathologists in the Department of Gastroenterology of the Daping Hospital for performing endoscopy, mucosal sample collection, and pathological diagnosis. We are indebted to our patients for their willingness to participate in this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
