Abstract
Background and objective:
Recently, a large number of trials on proton pump inhibitor-amoxicillin-containing high-dose dual therapy (HDDT) versus bismuth-containing quadruple therapy (BQT) for Helicobacter pylori (H. pylori) eradication have been published with controversial and inconsistent conclusions. The aim of this meta-analysis was to determine the effects of HDDT for H. pylori eradication compared to BQT.
Design:
A systematic review and meta-analysis was conducted.
Methods:
PubMed, Embase, and the Cochrane library database were searched to collect all randomized controlled trials (RCTs) assessing the effects of HDDT versus BQT to H. pylori eradication from inception to September 2022. Meta-analysis was conducted to estimate the pooled relative risk (RR) with 95% confidence intervals (CIs) using a random-effects model. Quality of evidence was appraised using Grading of Recommendations, Assessment, Development and Evaluation system. Trial sequential analysis (TSA) was performed to determine the reliability and conclusiveness.
Results:
A total of 14 RCTs with 5121 patients were included. The results of meta-analysis showed that there was no statistical significance in the eradication rate between HDDT and BQT (intention-to-treat analysis: 86.7% versus 85.1%, RR = 1.01, 95% CI: 0.98–1.04; per-protocol analysis: 89.9% versus 89.4%, RR = 1.01, 95% CI: 0.98–1.03; moderate-quality evidence). The incidence of total adverse effects in HDDT group was significantly lower than in BQT group (5.9% versus 34.1%, RR = 0.42, 95% CI: 0.34–0.50; low-quality evidence). No statistical significance was observed in compliance between HDDT and BQT (RR = 1.01, 95% CI, 1.00–1.03, p = 0.07; low-quality evidence). The TSA result for H. pylori eradication rate indicated that the effect was conclusive.
Conclusions:
Evidence from our updated meta-analysis suggests that HDDT is as effective as BQT in eradicating H. pylori, with fewer adverse effects and similar compliance.
Registration:
Open Science Framework registries (No: osf.io/th4vd)
Introduction
Helicobacter pylori (H. pylori) has become a common global public health problem with reported prevalence rates of 50.8% in developing countries and 34.7% in developed countries. 1 There were approximately 4.4 billion individuals with H. pylori infection worldwide. 2 H. pylori is not only the main cause of upper gastrointestinal diseases, including chronic gastritis, peptic ulcer, and gastric cancer, but also may be associated with many extra-gastrointestinal diseases.3,4
Eradicating H. pylori can prevent these diseases and hinder their progress.
With the continuous increase in antibiotic resistance, the eradication rate of standard triple therapy has been far below 80%. 5 Currently, bismuth-containing quadruple therapy (BQT), consisting of proton-pump inhibitor (PPI), bismuth, and two antibiotics (amoxicillin, clarithromycin, tetracycline, or metronidazole), which has been highly recommended as first-line therapy for H. pylori eradication according to the latest Maastricht VI/Florence consensus report, 3 the Fifth Chinese National Consensus Report, 4 the Toronto Consensus, 6 and ACG clinical guideline. 7 Although the eradication rate has improved, BQT still has some limitations: not available in some regions, complicated dosing regimen, more adverse events, poor drug compliance, and high cost.8,9 Therefore, it is urgent to discover and identify novel regimens with high efficacy, less use of antibiotics, and fewer adverse events to eradicate H. pylori.
Recently, high-dose dual therapy (HDDT), which gives amoxicillin and PPI more than two times daily, has become one of the most discussed hot topics in the field of H. pylori eradication. Of late, Yin et al. 9 conducted a meta-analysis and demonstrated that HDDT is equally effective with BQT in eradicating H. pylori, with fewer side effects and better compliance. However, the search time was from inception to March 2021, and their meta-analysis missed some multicenter trials. Furthermore, the previous study did not provide detailed subgroup analyses and comprehensive sensitivity analyses due to the small number of studies included. In addition, numerous new high-quality randomized controlled trials (RCTs) have been published with controversial and inconsistent conclusions in the latest 2 years. These newly published trials have prompted our interest in updating current evidence. Based on the above considerations, we conducted an updated meta-analysis to assess the efficacy, adverse events, and patient compliance of HDDT compared with BQT for H. pylori eradication.
Methods
Registration of review protocol and implementing guidance
The protocol for this study was registered in advance with Open Science Framework registries (No: osf.io/th4vd). This systematic review was conducted in accordance with the latest Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement. 10
Data sources and literature search strategy
We conducted electronic searches in PubMed, Embase, and the Cochrane library database using pre-established search terms from inception to September 2022. The search terms were as follows: ‘helicobacter pylori’, ‘campylobacter pylori’, ‘H. pylori’, ‘Hp’, amoxicillin, amoxycillin, dual therapy, high-dose dual therapy, high dose dual therapy, bismuth. We used MeSH headings and text word terms searching, without language restriction. The detail of search strategy for each database is shown in Supplemental Table S1. Additional potential eligible studies were manually searched through the reference lists of relevant systematic reviews and included studies.
Study selection
Two reviewers (ZBG and MYZ) independently screened the studies by reading titles and abstracts, and obtained full texts of potentially relevant studies using the following selection criteria. Any disagreement was resolved by consensus. If any overlapping studies were identified, only the study with the most comprehensive data was included. For relevant studies with multiple arms, the data were combined to create a new study group and a new control group according to the following inclusion criteria. Inclusion criteria were as follows: (a) participants: H. pylori-infected patients; (b) intervention: HDDT group: PPI plus amoxicillin, PPI and amoxicillin were both administered 3 or 4 times a day and amoxicillin >2.0 g/day; (c) comparator: BQT group: PPI, bismuth, and two antibiotics; (d) outcomes: (i) primary outcome: H. pylori eradication rate; (ii) secondary outcomes: adverse effects and compliance; (e) study design: RCTs. Exclusion criteria were set as below: (a) non-RCTs, animal studies, comments, editorials, letters, reviews, and meta-analyses; (b) amoxicillin was not administered at a dosage >2.0 g/day and either PPI or amoxicillin was given no more than twice daily; (c) regimens contained probiotics or herbs; (d) duplicate publications; and (e) studies without sufficient data.
Data extraction
Two reviewers (ZBG and MYZ) independently extracted the data from eligible studies using predesigned data extraction form. The following information was extracted from each eligible study: surname of the first author, year of publication, country of study, study design, participant characteristics, number of participants, specific details of eradication regimens (dose, frequencies, and duration), diagnostic tests to confirm H. pylori infection before and after eradication, confirmed testing time after eradication, H. pylori susceptibility to antibiotics, relevant data of the eradication rate, adverse effects, and compliance. Any discrepancies were resolved by a third author (ZM).
Risk of bias and grading the strength of evidence
The risk of bias was independently appraised using the Cochrane Collaboration’s tool for RCTs. 11 We developed summary of findings tables with the use of the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) profiler sofware (GradePro Version 3.6.1). The quality of evidence was categorized into four levels (i.e. very low, low, moderate, and high) in the GRADE system. 12 Two reviewers (ZBG and MYZ) independently appraised the risk of bias of each study and quality of evidence. Consensus between two reviewers was used to resolve any disagreement.
Statistical analysis
We used the Review Manager software (Version 5.3, The Cochrane Collaboration, Nordic Cochrane Centre, Copenhagen, Denmark) to calculate pooled relative risk (RR) with 95% confidence intervals (CIs) for each study.
The data were analyzed with intention-to-treat (ITT) analysis and per-protocol (PP) analysis for H. pylori eradication rate. Data were pooled using the random-effects model to give a more conservative estimate. 13 The standard chi-square and I2 statistic were used to evaluate the statistic heterogeneity among studies. p < 0.10 for the Q test was considered statistically significant. I2 values of 0–25%, 26–50%, 51–75%, and more than 75% indicated insignificant, low, moderate, and high heterogeneity, respectively. 14 Subgroup analyses based on ITT analysis were further utilized to explore potential sources of heterogeneity and possible factors affecting the overall results. Sensitivity analyses were conducted for H. pylori eradication rate (ITT data) as follows: (a) using fixed-effect model; (b) excluding conference abstracts; (c) excluding studies published in non-English; and (d) omitting one study at a time. Publication bias was assessed by Begg’s test and Egger regression asymmetry test using STATA/SE 12.0 (STATA Corporation, Texas, USA).
Meta-analyses may result in type I and type II errors due to the increased risk of random error when analyzing sparse data and due to repeated significance testing when a cumulative meta-analysis is updated with new trials.15,16 To assess the risk of type I and type II errors, a trial sequential analysis (TSA) was performed for the primary outcome of H. pylori eradication rate. This analysis was conducted using TSA software (version 0.9.5.10, Copenhagen Trial Unit, Copenhagen, Denmark).
Results
Literature search results and study selection process
The literature search identified 360 potentially relevant records. Among these records, 137 duplicate records were excluded, 181 records were further excluded by reading titles or abstracts, leaving 42 potential eligible articles. Of these articles, 28 articles were further excluded based on the selection criteria (characteristics of the excluded studies are shown in Supplemental Table S2). Finally, 14 RCTs17–30 were included in the meta-analysis. The article selection process is presented in the PRISMA flowchart in Figure 1.
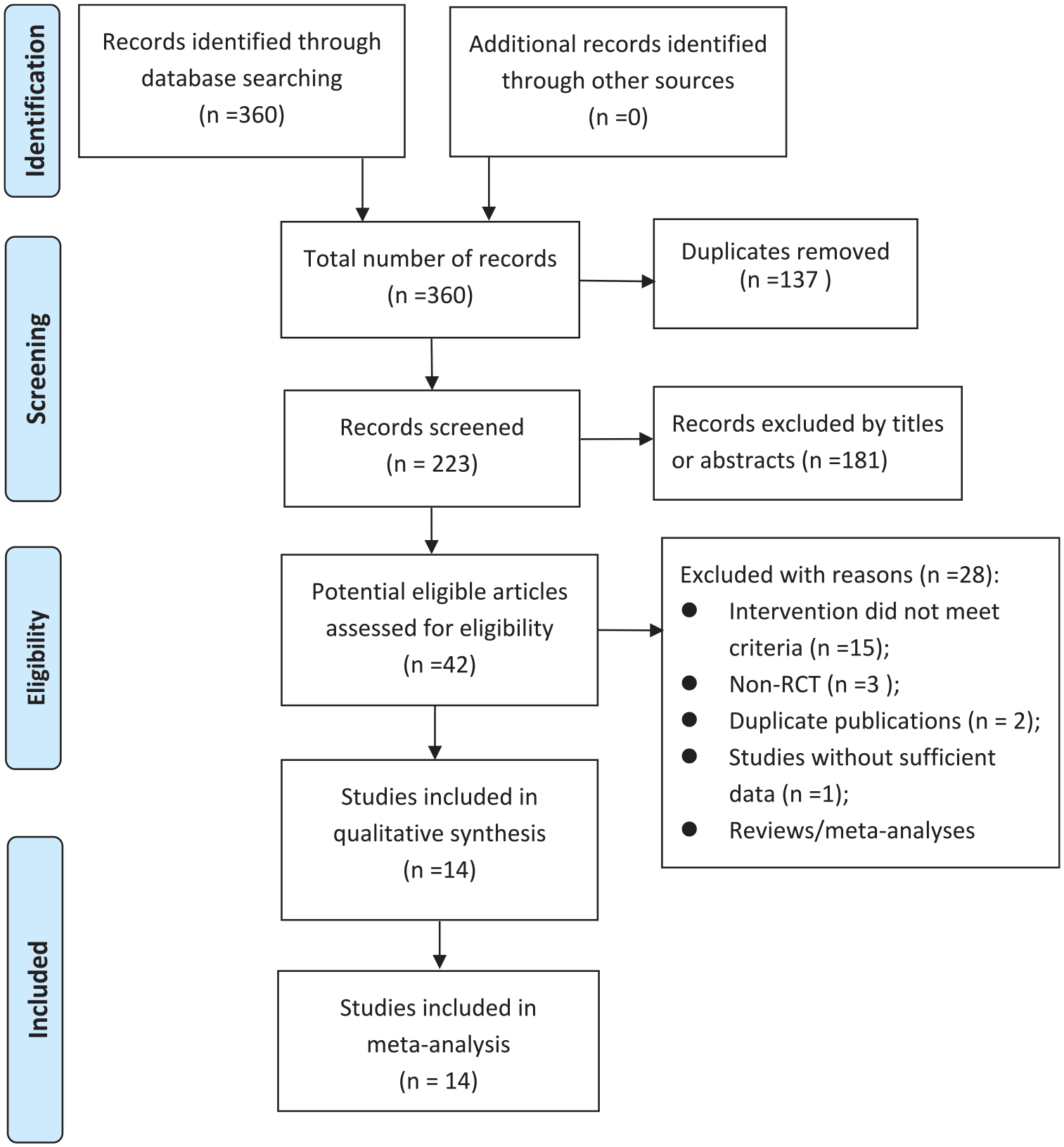
PRISMA flowchart of study selection process.
Study characteristics of included studies
A total of 14 studies17–30 involving 5121 patients were eligible for our meta-analysis. All included studies were published between 2003 and 2022, of which six were multicenter RCTs17,18,22,25,26,29 and eight were single-center RCTs.19–21,23,24,27,28,30 Among these included studies, 11 studies were performed in China,18,19,21–26,28–30 one in Germany and Austria, 17 one in Turkey, 20 and one in Colombia. 27 Concerning publication language, 12 studies were published in English,17–20, 22–26, 28–30 one in Chinese, 21 and one in Spanish. 27 Among these studies, three of which were published as conference abstracts.18,22,25 The sample size of included studies ranged from 84 to 971 patients. As to H. pylori eradication treatment experience, two studies were rescue therapy17,22 and these remaining studies were initial therapy. Two studies17,30 performed antibiotic susceptibility tests, two studies23,24 conducted antibiotic susceptibility tests and CYP2C19 gene polymorphism analysis simultaneously. With regard to HDDT regimen, the types of PPI are mainly esomeprazole or rabeprazole, and only one early study 17 used omeprazole to eradicate H. pylori; the total dose of amoxicillin was 3000 mg, administered three or four times daily. The duration of HDDT regimen for all included studies were 14 days. Regarding the duration of BQT regimen, three studies were 10 days,18,22,25 and the remaining studies were 14 days. The main characteristics of included studies are summarized in Table 1.
Characteristics of included studies in the meta-analysis.
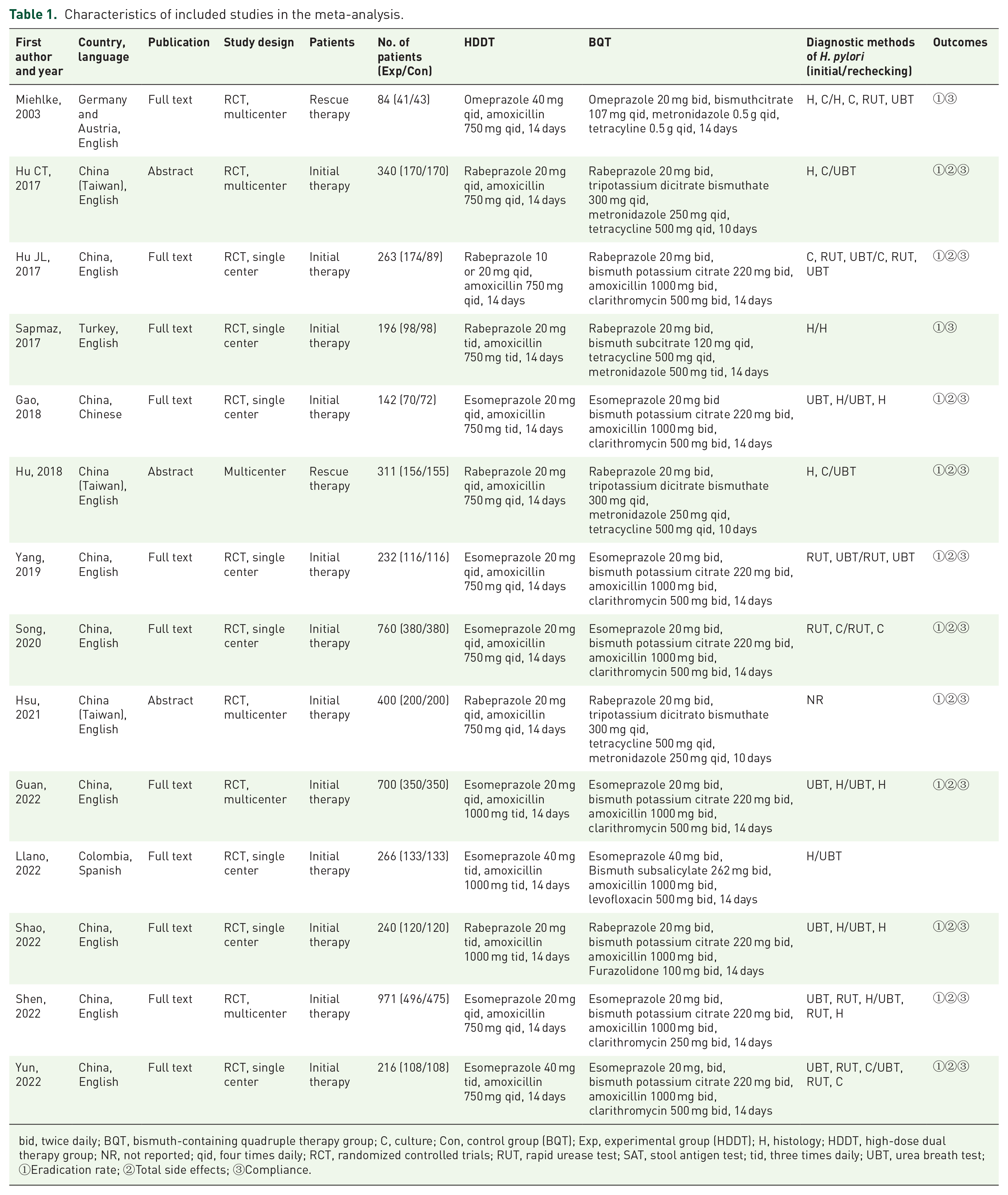
bid, twice daily; BQT, bismuth-containing quadruple therapy group; C, culture; Con, control group (BQT); Exp, experimental group (HDDT); H, histology; HDDT, high-dose dual therapy group; NR, not reported; qid, four times daily; RCT, randomized controlled trials; RUT, rapid urease test; SAT, stool antigen test; tid, three times daily; UBT, urea breath test; ①Eradication rate; ②Total side effects; ③Compliance.
Risk of bias
Supplemental Figures S1 and S2 show the details of risk of bias assessment. Among these 14 studies, nine studies17,19,20,23,24,26,27,29,30 reported sufficient random sequence generation methods (computer-generated randomization and randomized digital table) and were judged as low risk, while the other five studies were judged as unclear risk of randomization. Only two studies24,26 provided sufficient data for allocation concealment and were judged as low risk. With regard to blinding participants and personnel, seven studies were open trial,19,23,24,26,28–30 and the remaining seven studies17,18,20–22,25,27 were judged as unclear risk due to insufficient information. All these studies showed low risk in blinding of outcome assessment, incomplete outcome data, and other bias. As to selective reporting (reporting bias), two studies17,20 were judged as high risk due to the absence of total side effects, all the remaining studies were judged as low risk of bias.
Primary outcome: H. pylori eradication rate
Overall eradication rates
A total of 14 studies17–30 (involving 5121 patients) reported data on H. pylori eradication rate. Based on the data analyzed by ITT analysis, the eradication rate in the HDDT group was higher than BQT group (86.7% versus 85.1%), but there was no statistical significance between them (RR = 1.01, 95% CI: 0.98–1.04, p = 0.36) with low significant heterogeneity (I2 = 40%, p = 0.06) (Figure 2). For the PP analysis, similar result was also observed (89.9% versus 89.4%; RR = 1.01, 95% CI: 0.98–1.03, p = 0.62) with low significant heterogeneity (I2 = 41%, p = 0.05) (Figure 3).
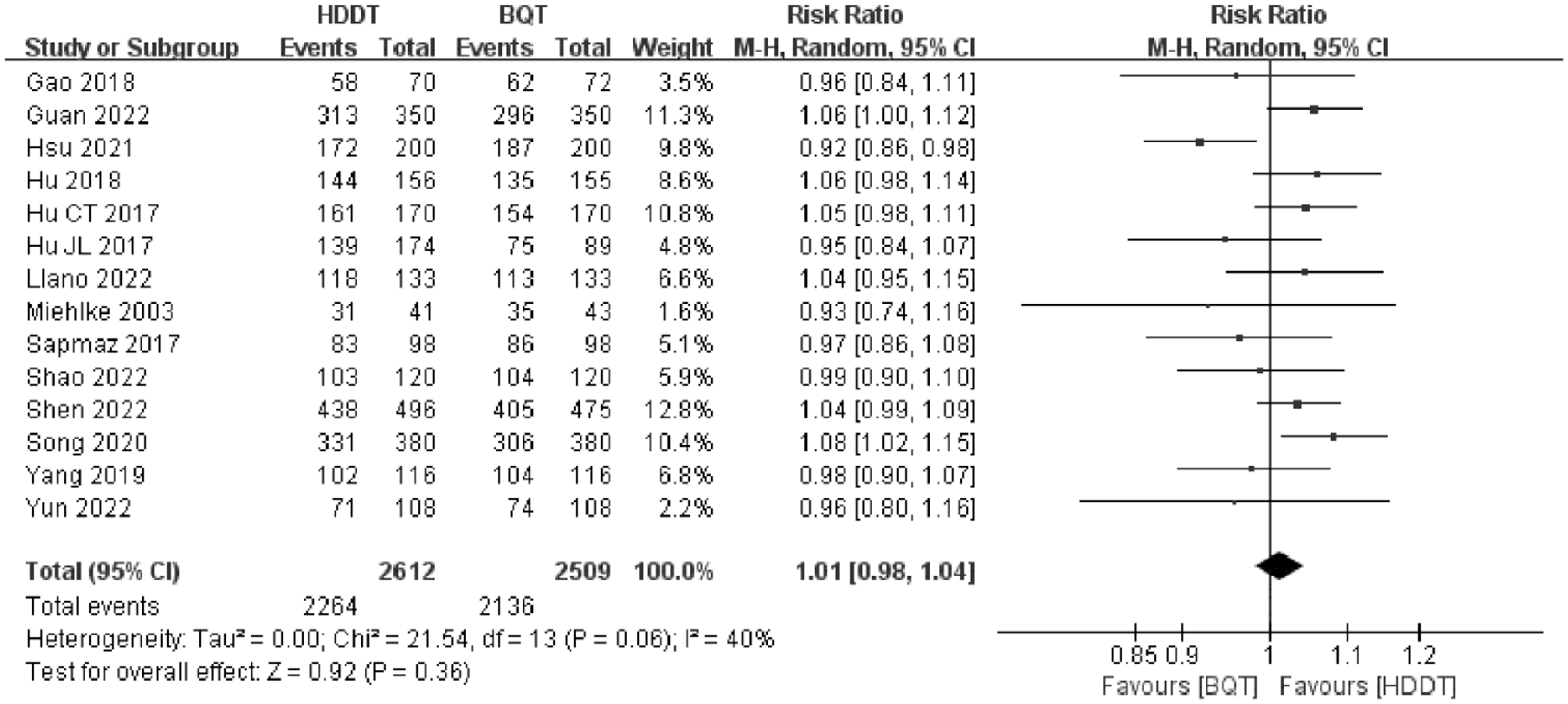
Forest plot of the effect of HDDT versus BQT on H. pylori eradication rate according to ITT analysis.

Forest plot of the effect of HDDT versus BQT on H. pylori eradication rate according to PP analysis.
Trial sequential analysis
Based on ITT data, we conducted a TSA to examine the reliability and conclusiveness. We estimated the required information size (RIS) and adjusted significance thresholds based on the following assumptions: a two-sided adjusted random effects model with 5% risk of type I error, 20% risk of the type II error (power of 80%), an anticipated RR reduction of −5%, and the pooled control group eradication rates of 85.1%. As shown in Supplemental Figure S3, the cumulative z curve did not cross the TSA boundary and the traditional significance boundary, but crossed the futility boundary and RIS line, supporting the findings of the conventional meta-analysis, and also confirming that true negative. The RIS was 3681 patients. The TSA adjusted CI was 0.98–1.05.
Subgroup analyses
We performed subgroup analyses based on study design, study location, treatment experience, frequency of HDDT administration, and BQT duration. The details of the results of subgroup analyses are presented in Table 2. In subgroup analysis based on study design, the eradication rate had no significant difference between multicenter RCTs subgroup (RR = 1.02, 95% CI: 0.97–1.07, p = 0.42) and single-center RCTs subgroup (RR = 1.01, 95% CI: 0.97–1.05, p = 0.61). Similar results were observed when the subgroup analyses were stratified by study location, treatment experience, frequency of HDDT administration, and BQT duration (Supplemental Figures S4–S8).
Results of subgroup analyses for H. pylori eradication rate.
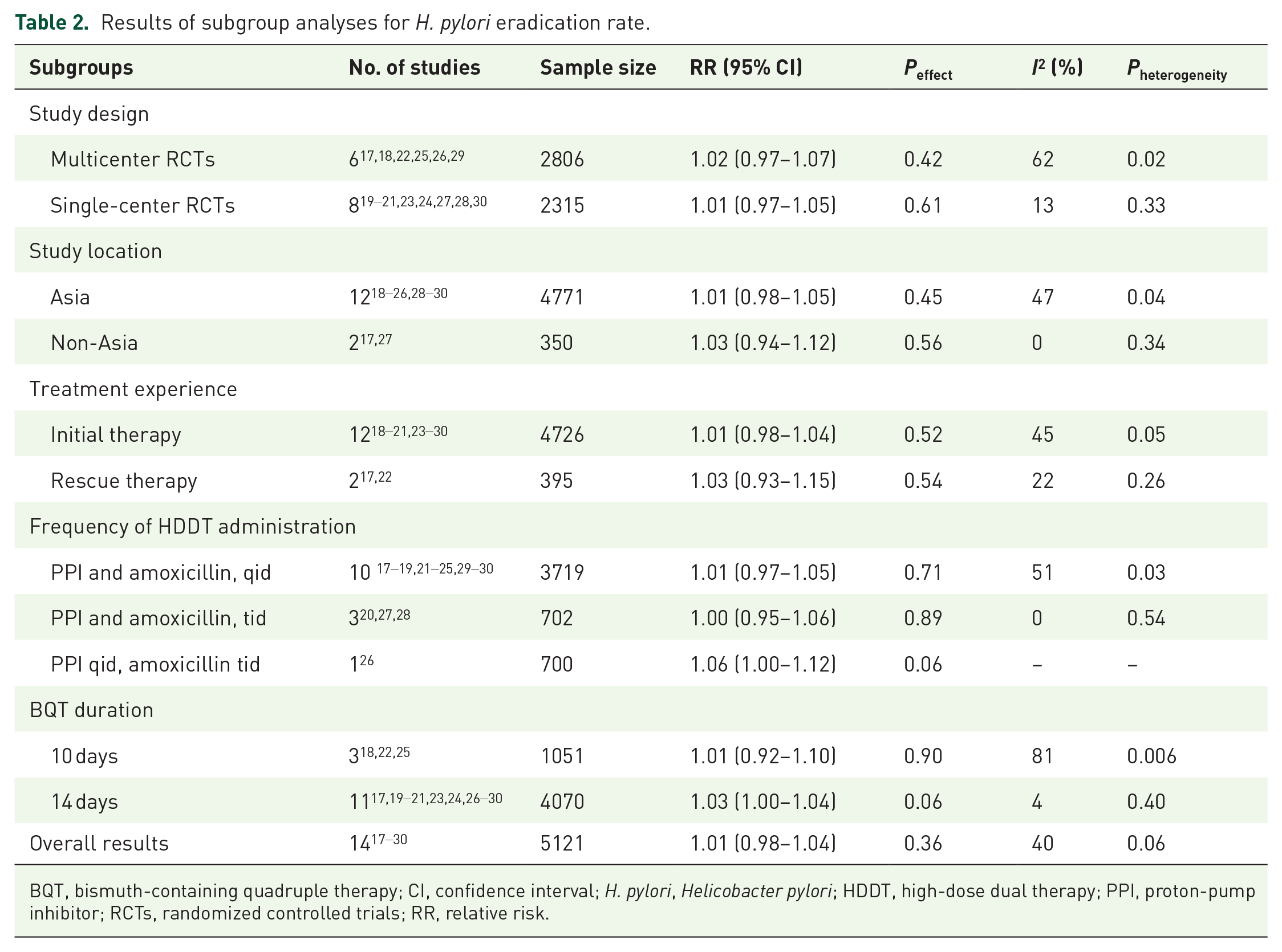
BQT, bismuth-containing quadruple therapy; CI, confidence interval; H. pylori, Helicobacter pylori; HDDT, high-dose dual therapy; PPI, proton-pump inhibitor; RCTs, randomized controlled trials; RR, relative risk.
Sensitivity analyses
Sensitivity analyses were conducted to confirm the robustness of overall results and explore potential sources of heterogeneity. Similar results were observed for H. pylori eradication when using fixed-effect model, excluding conference abstracts, and excluding studies published in non-English. When omitting one study at a time and combining the remaining studies for sensitivity analyses, except for the study conducted by Hsu et al., 25 exclusion of any other study did not significantly alter the overall results. Notably, when excluding conference abstracts and the study conducted by Hsu et al. 25 (published also as conference abstract), these I2 were dramatically reduced to 4%, 0%, respectively, indicating that the study may be a source of statistical heterogeneity. These results of sensitivity analyses are presented in Supplemental Table S3.
Secondary outcomes
Total adverse effects
In all, 12 studies18,19,21–30 including 4767 patients reported the incidence of total adverse effects. The incidence of total adverse effects in HDDT group was significantly lower than in BQT group (5.9% versus 34.1%, RR = 0.42, 95% CI: 0.34–0.50, p < 0.00001). There was moderate statistical heterogeneity between studies (I2 = 59%, p = 0.005) (Figure 4).

Forest plot of the effect of HDDT versus BQT on total adverse effects.
Compliance
In all, 14 studies17–30 involving 5121 patients reported data on patient compliance. The compliance in HDDT group was statistically higher than that of the BQT group (96.7% versus 94.7%), but there was no significant difference (RR = 1.01, 95% CI, 1.00–1.03, p = 0.07) with low statistical heterogeneity (I2 = 45%, p = 0.04) (Figure 5).

Forest plot of the effect of HDDT versus BQT on compliance.
Quality of evidence
We assessed the quality of evidence using the GRADE approach. For H. pylori eradication rate based on ITT data, the GRADE assessment of the quality of the evidence was judged as moderate quality, mainly because of risk of bias in study design (unclear random sequence generation and allocation concealment, unclear or no blinding, and selective reporting). For the incidence of total adverse effects and compliance, the quality of the evidence was rated as low quality, which were mainly due to risk of bias in study design, statistical heterogeneity, or reporting bias. The details of GRADE evidence profiles for all these outcomes are presented in Supplemental Table S4.
Publication bias
For H. pylori eradication rate based on ITT analysis data, the funnel plot was asymmetrical distribution by visual inspection (Supplemental Figure S9). However, the results of Begg’s test and Egger’s test indicated no evidence of substantial publication bias (Pbegg = 0.08, Pegger = 0.07, respectively). For the incidence of total adverse effects and compliance, the funnel plots were both asymmetrically distributed (Supplemental Figures S10 and S11). The results of Begg’s test and Egger’s test for the incidence of total adverse effects indicate that there may be publication bias (Pbegg = 0.047, Pegger = 0.013, respectively). For compliance, the result of Begg’s test did not confirm the presence of publication bias (Pbegg = 0.189), but the result of Egger’s test showed that there may be publication bias (Pegger = 0.025).
Discussion
Summary of main findings
To the best of our knowledge, this study provides the most comprehensive, systematic, up to date evidence on the effects of HDDT compared with BQT for H. pylori eradication. Our updated meta-analysis demonstrated that, compared with BQT, HDDT has similar efficacy and compliance, with fewer adverse effects in eradicating H. pylori.
For H. pylori eradication rate, similar results were observed in all conducted subgroup analyses and most of sensitivity analyses. The GRADE quality of the evidence was appraised as moderate quality. The result of the TSA was conclusive. For adverse effects and compliance, the GRADE quality of the evidence was appraised as low quality.
Potential explanations and implications
In 1989, Unge et al. 31 initially reported that the dual therapy consisting of amoxicillin (750 mg bid) and omeprazole (40 mg qid) was used to eradicate H. pylori, with low eradication rate of only 62%. Subsequently, a multicenter study conducted by Bayerdörffer et al. 32 in Germany showed that the H. pylori eradication rate was 91% in the omeprazole (40 mg tid) plus amoxicillin (750 mg tid) group. At that time, however, this regimen may not be adopted by physician because of the high dose and frequency of drugs. Thereafter, investigators have further conducted a series of clinical trials on standard dose of dual therapy for H. pylori eradication, but the eradication rate is significantly lower than triple therapy and quadruple therapy.33,34 Therefore, routine dual therapy has not become the standard treatment for H. pylori eradication. In recent years, with the in-depth research on the pharmacokinetics and pharmacodynamics of amoxicillin and PPI, the mechanism of the dual therapy has been gradually clarified, and the HDDT has attracted clinicians’ attention again. The mechanism of the effectiveness of HDDT in eradicating H. pylori was summarized as follows: (i) antibiotic resistance remains the most important challenge to successful H. pylori eradication; however, the resistance rate of H. pylori strain to amoxicillin is very low (<5%).4,35,36 (ii) amoxicillin is a time-dependent antibiotic, and its bactericidal effect depends on the time when the plasma concentration is higher than the minimum inhibitory concentration. 37
After taking amoxicillin for 6–8 h, its concentration decreased significantly, and the effective plasma concentration of amoxicillin could be maintained by increasing the frequency of administration. To increase and maintain blood concentration, amoxicillin should be administered 3–4 times a day to achieve a good eradication effect. (iii) Amoxicillin has been shown to be pH dependent, when the intragastric pH is 6 or greater, H. pylori is in proliferation state and becomes highly susceptible to amoxicillin. Therefore, it is necessary to use high-dose PPI to fully inhibit gastric acid secretion while using amoxicillin.37–39 (iv) The effect of PPI on inhibiting gastric acid secretion is affected by CYP2C19 gene polymorphism. Nevertheless, some studies have shown that when PPI is given at high dose and multiple frequencies, the effect of CYP2C19 gene polymorphism could be basically ignored.40,41 Thus, high-dose and high-frequency administration of HDDT are expected to improve its effectiveness for H. pylori eradication.
In our meta-analysis of 14 RCTs, HDDT achieved similar eradication rate compared to the BQT recommended by current mainstream guidelines. Considering that racial differences may affect the eradication effect of HDDT, 42 we conducted the subgroup analysis based on study location, the result showed that HDDT and BQT have similar eradication rates in both Asia subgroup and non-Asia subgroup. However, in the non-Asia subgroup, there were only two studies17,27 from Europe and Oceania. Thus, the effect still needs to be verified by further large sample RCTs in non-Asian population.
Of the 14 studies included, PPI and amoxicillin were administered at different frequencies in HDDT, four times daily in 10 studies17–19,21–25,29–30 and three times daily in three studies,20,27,28 whereas PPI was administered four times daily and amoxicillin three times daily in one study. 26 We further conducted subgroup analysis based on frequency of HDDT administration, similar eradication rates were observed in these subgroups. Although it is convenient for HDDT to be administered three times a day, which can increase patient compliance and obtain similar eradication rate compared to BQT, the number of clinical trials is small and the sample size is limited, so we cannot draw a definite conclusion. In the future, further clinical trials are needed to verify the efficacy of HDDT administered three times a day compared with BQT in the eradication of H. pylori.
For eradication rate, we further conducted sensitivity analyses to confirm the robustness of overall results. When omitting one study conducted by Hsu et al. 25 and combining the remaining studies for sensitivity analysis, the eradication rate in the HDDT group was higher than BQT group (RR = 1.03, 95% CI: 1.01–1.06). Notably, with the exception of the study conducted by Hsu et al. 25 , I2 were dramatically reduced to 0%, which may be attributed to the study was published as conference abstract, indicating that publication type may be a source of low statistical heterogeneity.
Compliance is an important factor affecting the H. pylori eradication rate, adverse effects are the crucial factors that affect patient compliance. 43 Adverse effects and patient compliance were also analyzed in our study. In our meta-analysis, HDDT has a lower incidence of total adverse effects compared to BQT (5.9% versus 34.1%), which may be related to fewer kinds of medications used in the regimen. Nevertheless, no significant difference was observed in the compliance between HDDT and BQT. Although HDDT increased the number of daily doses and frequency of administration, which may decrease patient compliance, HDDT has fewer adverse effects and fewer kinds of medications, so compliance does not decrease compared to BQT.37,42
In view of increasing antibiotic resistance and high incidence of adverse effects of BQT, the results of our meta-analysis have important clinical implications. For one thing, HDDT may be a good alternative treatment for those who cannot obtain bismuth or tolerate the adverse effects of BQT. For the other thing, HDDT is also applicable to patients of rescue treatment, especially when drug susceptibility test cannot be conducted. In addition, vonoprazan, a novel potassium-competitive acid blocker, has been recently attracted considerable attention. A meta-analysis by Lyu et al. 44 showed that vonoprazan-based triple therapy is superior to that of PPI-based triple therapy for first-line H. pylori eradication. Compared to PPI, vonoprazan provides a stronger and longer-lasting effect on gastric acid suppression, and is not affected by diet and CYP2C19 polymorphism. 9 In the future, RCTs on vonoprazan–amoxicillin dual therapy versus BQT for H. pylori eradication should be further conducted.
Comparison with previous studies
In 2019, a smaller meta-analysis conducted by Yang et al., 45 including four RCTs with 829 patients, showed that both HDDT and BQT achieved similar efficacy of eradication rate and compliance, but side effects were more likely in BQT. Recently, Yin et al., 9 who included six RCTs with 1677 patients, demonstrated that HDDT achieved similar eradication rate compared with BQT. However, HDDT showed fewer side effects and better compliance compared to BQT. In these two published meta-analyses, our meta-analysis covers all of their included studies. Compared to previous studies,9,45 our updated meta-analysis further validates these above conclusions and advances the findings of this past work. Our meta-analysis covers the most comprehensive studies (n = 14 RCTs) and greatly increases the total sample size (n = 5121 patients), especially by including the most updated large sample and multicenter studies published between 2021 and 2022, providing the latest, most timely and more sufficient evidence on the topic.
Strengths and limitations
Our study has several strengths. First, our study is the most current and largest meta-analysis to date on the effects of HDDT compared with BQT for H. pylori eradication. The large number of sample size provided high statistical power. Second, we used rigorous and transparent methodology according to the Cochrane Handbook and the PRISMA statement, conducted a limited number of subgroup analyses and comprehensive sensitivity analyses, and assessed the quality of evidence with GRADE approach, which made our results more convincing and reliable. Third, we applied TSA to increase the reliability of the meta-analysis and estimate the RIS, which further draws firm evidence.
Despite these strengths, several limitations should not be neglected. First, many of included studies in our meta-analysis did exist risk of bias, such as unclear randomization, allocation concealment, and no blinding, which might affected the reliability of our results. Further large, well-designed RCTs are needed to validate our findings. Second, there are few trials (only two trials 17,22) regarding HDDT versus BQT for H. pylori rescue therapy. More large sample multicenter trials are needed in the future to verify the effect of HDDT versus BQT as a rescue therapy for H. pylori. Third, for secondary outcomes (total adverse effects and compliance), there may be publication bias, which may have a certain impact on the results. Fourth, this regimen may not be applicable to patients who are allergic to amoxicillin. Finally, study patients in our meta-analysis were mainly from Asia (mostly from China), a paucity of evidence from other regions. Therefore, the evidence of this review is mainly applicable to Asian populations (especially China), whether the evidence are suitable for other populations should be confirmed by conducting further studies. In spite of the limitations aforementioned, we are reasonably confident in our results and provide valuable updated evidence for H. pylori eradication in clinical practice.
Conclusion
In conclusion, evidence from our updated meta-analysis demonstrate that HDDT is as effective as BQT in eradicating H. pylori, with fewer adverse effects and similar compliance. Increasing the total dose and administration frequency of PPI and amoxicillin is more conducive to maintaining a stable blood concentration of PPI, and forming a stable high pH environment in the stomach, which helps H. pylori enter the proliferation state and increase its sensitivity to amoxicillin. In addition, high dose and multiple frequencies of PPI also helps to overcome the impact of CYP2C19 gene polymorphism. The scientific evidence supports that HDDT can be used as an alternative regimen to BQT regimen for H. pylori infection eradication in clinical practice.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221147756 – Supplemental material for High-dose dual therapy versus bismuth-containing quadruple therapy for Helicobacter pylori eradication: a systematic review and meta-analysis with trial sequential analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848221147756 for High-dose dual therapy versus bismuth-containing quadruple therapy for Helicobacter pylori eradication: a systematic review and meta-analysis with trial sequential analysis by Ben-Gang Zhou, Yu-Zhou Mei, Min Zhang, Xin Jiang, Yao-Yao Li and Yan-Bing Ding in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
