Abstract
Background:
Helicobacter pylori resistance to amoxicillin remains rare in many regions. Proton pump inhibitor-amoxicillin-containing high dose dual therapy (HDDT) has been proposed to treat H. pylori infection. We aimed to assess the effectiveness and safety of PPI-amoxicillin HDDT for treatment of H. pylori infection in comparison with other regimens.
Methods:
Databases, including PubMed, Embase, and the Cochrane Register of Controlled Trials, were searched to find relevant publications. Randomized controlled trials comparing HDDT with control regimens for H. pylori eradication in adult patients were included. The primary outcome was eradication rate by intention-to-treat analysis. Adverse events were analyzed as second outcome.
Results:
A total of 15 trials with 3818 patients qualified for inclusion. The eradication rate of HDDT was neither significantly inferior nor superior to the recommended regimens such as triple therapy, bismuth quadruple therapy, and non-bismuth quadruple therapy [relative risk (RR): 1.00, 95% confidence interval (CI): 0.96–1.05, p = 0.870]. This finding was robust through subgroup analyses and sensitivity analyses. Trial sequential analysis showed that HDDT was equivalent to control regimens, and further similar trials were unlikely to alter the conclusions of this analysis. The frequency of adverse events was significantly lower in HDDT group (RR: 0.48, 95% CI: 0.37–0.64, p < 0.001).
Conclusion:
HDDT was equivalent to recommended first-line or rescue regimens with fewer adverse effects. The evidence from this meta-analysis supports the use of HDDT as first-line or rescue treatment for H. pylori infection.
Trial registration:
PROSPERO CRD42019133002
Introduction
Helicobacter pylori infection is a considerable public health problem, which affects 24% of people in Oceania, 37% in North America, 47% in Europe, 55% in Asia, 63% in Latin America and Caribbean, and 79% in Africa.1,2 H. pylori is associated with many upper gastrointestinal diseases, such as chronic gastritis, peptic ulcer disease, gastric cancer, and mucosa-associated lymphoid tissue lymphoma; eradication of H. pylori may prevent the development of these diseases.3,4
Standard triple therapy (TT), which consists of proton pump inhibitor (PPI), amoxicillin, clarithromycin, or metronidazole, was recommended as the first-line eradication therapy. 5 Unfortunately, increasing resistance to antibiotics impaired the efficacy of standard TT. 6 The prevalence of clarithromycin and metronidazole resistance was more than 15% in many regions, and standard TT could no longer cure even 80% of infections. 7 Therefore, alternative regimens have been introduced, including bismuth quadruple therapy (BQT), non-bismuth quadruple therapy (non-BQT, also known as concomitant therapy), and sequential therapy (ST). 8 Although these regimens could achieve acceptable eradication rates, the complexity and adverse events of multi-drug regimens may reduce the patient compliance. 9 Moreover, using multi-antibiotics regimens may result in multi-drug resistance and limit the options of antibiotics for subsequent therapy if the eradication treatment is failed. 10
Unlike clarithromycin and metronidazole, H. pylori resistance to amoxicillin has remained rare in many regions. 11 PPI-amoxicillin dual therapy has been used for treating H. pylori infection for decades, but its efficacy remains controversial.12,13 The efficacy of PPI-amoxicillin dual therapy with standard dose and frequency was unacceptable (<80%), whereas higher eradication rates were attained when PPI and amoxicillin were given at high dose and frequency. 14 High dose dual therapy (HDDT), which gives amoxicillin and PPI more than two times daily, was reported equally effective to TT or BQT.15,16 However, some studies showed that the eradication rates of HDDT were less than 80%.17,18 We therefore conducted a systematic review and meta-analysis to compare the efficacy and safety of HDDT with other regimens.
Methods
We followed the recommendations of the Cochrane handbook for systematic reviews of interventions and the preferred reporting items for systematic reviews and meta-analyses (PRISMA) during all stages of the design, implementation, and reporting of this meta-analysis.19,20 The protocol of this study is documented on PROSPERO (CRD42019133002).
Search strategy
Two authors independently searched PubMed, Embase, and the Cochrane Register of Controlled Trials to identify relevant trials without language restrictions (updated to April 2020). The electronic search strategy combined terms related to Helicobacter pylori, proton pump inhibitor, and amoxicillin; the details of search strategy are described in the Supplemental File S1. The major conference proceedings were hand searched to identify further potentially relevant trials for inclusion. We also checked the reference lists of studies and reviews identified by the previous searches to include other potentially eligible trials.
Study selection
Two reviewers assessed all papers independently using predesigned eligibility forms, according to the eligibility criteria. Disagreements between investigators were resolved by consensus. Studies were included if they met the following criteria: (1) participants: adult patients (18 years or older) with H. pylori infection confirmed by one or more of the standard detection methods (urea breath test, bacterial culture, histological examination, rapid urea test or stool antigen test); (2) intervention: HDDT (PPI and amoxicillin were given more than two times daily; (3) comparison: other eradication regimens; (4) outcome: eradication rate at least 4 weeks after completion of eradication treatment and adverse events; (5) study design: randomized controlled trial (RCT). Studies were excluded if they enrolled pediatric patients or used regimens containing herbs.
Data extraction
Using a predesigned data collection form and working in duplicate, two authors independently extracted the following data from each study: first author, year of publication, geographical location of study, study design, patient characteristics, details of regimens (the doses, frequencies, and duration of medicine dosing), methods for detecting H. pylori infection before and after eradication, time to confirmation of the eradication status, H. pylori susceptibility to antibiotics, and outcome measures. Extracted data were checked by the third author, and any discrepancy was resolved by discussion. Our primary outcome was eradication rate by intention-to-treat (ITT) analysis, and second outcome was adverse events. Authors of the included studies were contacted via E-mail if further study details were needed.
Risk of bias assessment
Risk of bias assessment was independently performed by two authors, and any disagreement was resolved by consensus. The risk of bias was assessed by using the components recommended by the Cochrane Handbook for Systematic Reviews of Interventions: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other sources of bias. 21
Statistical analysis
The statistical analysis was performed by using RevMan software version 5.3 (The Nordic Cochrane Centre, The Cochrane Collaboration 2014, Copenhagen, Denmark), Stata 12.1 (Stata Corporation, College Station, TX, USA), and TSA Viewer 0.9 (The Copenhagen Trial Unit, Copenhagen, Denmark). We estimated the relative risk (RR) with 95% confidence intervals (CI) for outcomes. Data were pooled using the random effects model to give a more conservative estimate, allowing for any heterogeneity between studies. 22 Heterogeneity was expressed as the I2 statistic, and I2 ⩾ 50% indicated significant heterogeneity.
To identify potential sources of heterogeneity, we performed prespecified subgroup analyses according to: dosing frequency of HDDT (three times daily versus four times daily), regimens used in control group (TT versus BQT versus non-BQT versus ST), eradication experience (treatment-naive versus treatment-experienced), and publication type (full-length article versus conference abstract). To assess the independent influence of each study on the overall pooled estimate, we did prespecified sensitivity analyses by omitting one study in each turn.
To reduce the risk of random error we applied trial sequential analysis for the primary outcome. 23 Trial sequential analysis combines an estimation of information size (cumulated sample size of included trials) for meta-analysis with adjusted thresholds for statistical significance (trial sequential monitoring boundaries) as well as non-superiority and non-inferiority (futility boundaries) in the cumulative meta-analysis.24,25 When the cumulative Z-curve crosses the trial sequential monitoring boundary, a sufficient level of evidence for the anticipated intervention effect may have been reached and no further trials are needed. When the Z-curve crosses the futility boundaries, the two interventions do not differ more than the anticipated intervention effect. If the Z-curve does not cross any of the boundaries and the required information size has not been reached, evidence to reach a conclusion is insufficient. The trial sequential analysis was conducted with a desire to maintain a type I error of 5% and a power of 80%, with calculation of information size required to detect 10% relative risk reduction of eradication rate in the HDDT group.
Funnel plots were used to evaluate the publication bias visually. The Egger’s test was used to assess publication bias statistically. Results were considered as statistically significant for p-value < 0.05.
Results
Study selection
A total of 6785 papers were retrieved, and 15 randomized control trials with 3818 patients were eligible for inclusion (Figure 1).26–40 The characteristics of the included trials are shown in Table 1. Of the 15 trials, 11 were conducted in Asia,29–33,35–40 3 in Europe,27,28,34 and 1 in North America. 26 HDDT was given three times daily in four trials,26,28,30,34 and four times daily in the others.27,29,31–33,35–40 Six trials used TT,26,28–31,36 seven used BQT,27,33–35,37,38,40 one used non-BQT, 39 and one used both TT and ST as control regimen. 32 Nine trials recruited treatment-naive patients,30,33–40 four recruited treatment-experienced patients,27–29,31 one recruited both, 32 and one did not report treatment experience of included patients. 26 Of the 15 included trials, 12 were published as full-length articles,26–34,37–39 whereas 3 were conference abstracts.35,36,40

Flow diagram of literature search and selection.
Characteristics of included RCTs.
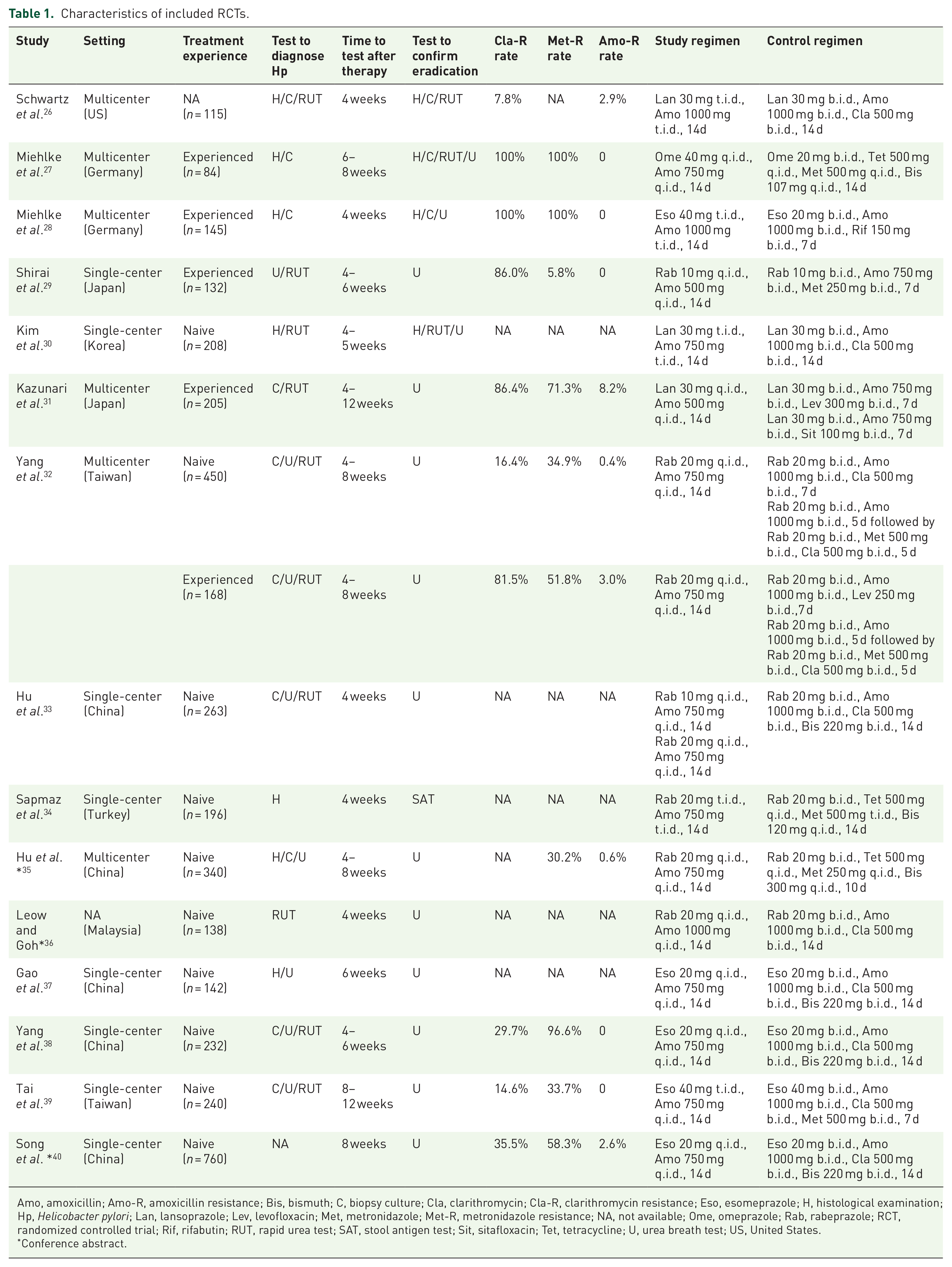
Amo, amoxicillin; Amo-R, amoxicillin resistance; Bis, bismuth; C, biopsy culture; Cla, clarithromycin; Cla-R, clarithromycin resistance; Eso, esomeprazole; H, histological examination; Hp, Helicobacter pylori; Lan, lansoprazole; Lev, levofloxacin; Met, metronidazole; Met-R, metronidazole resistance; NA, not available; Ome, omeprazole; Rab, rabeprazole; RCT, randomized controlled trial; Rif, rifabutin; RUT, rapid urea test; SAT, stool antigen test; Sit, sitafloxacin; Tet, tetracycline; U, urea breath test; US, United States.
Conference abstract.
Risk of bias in included studies
Potential risks of bias within individual trials are shown in Table 2. The included RCTs were at high risk or unclear risk of bias. Blinding of participants and personnel was the main source of potential bias in this meta-analysis. Only one trial was double-blinded, 26 whereas the others were open-labelled.
Risk of bias for the included studies.

Conference abstract.
Meta-analysis of eradication rate
In the meta-analysis of 15 trials with 3818 participants, the eradication rate of HDDT was not significantly different from control regimens (84% versus 81%; RR 1.00, 95% CI: 0.96–1.05; p = 0.870, I2 = 66%). Significant heterogeneity was found among these trials (p < 0.001) (Figure 2).
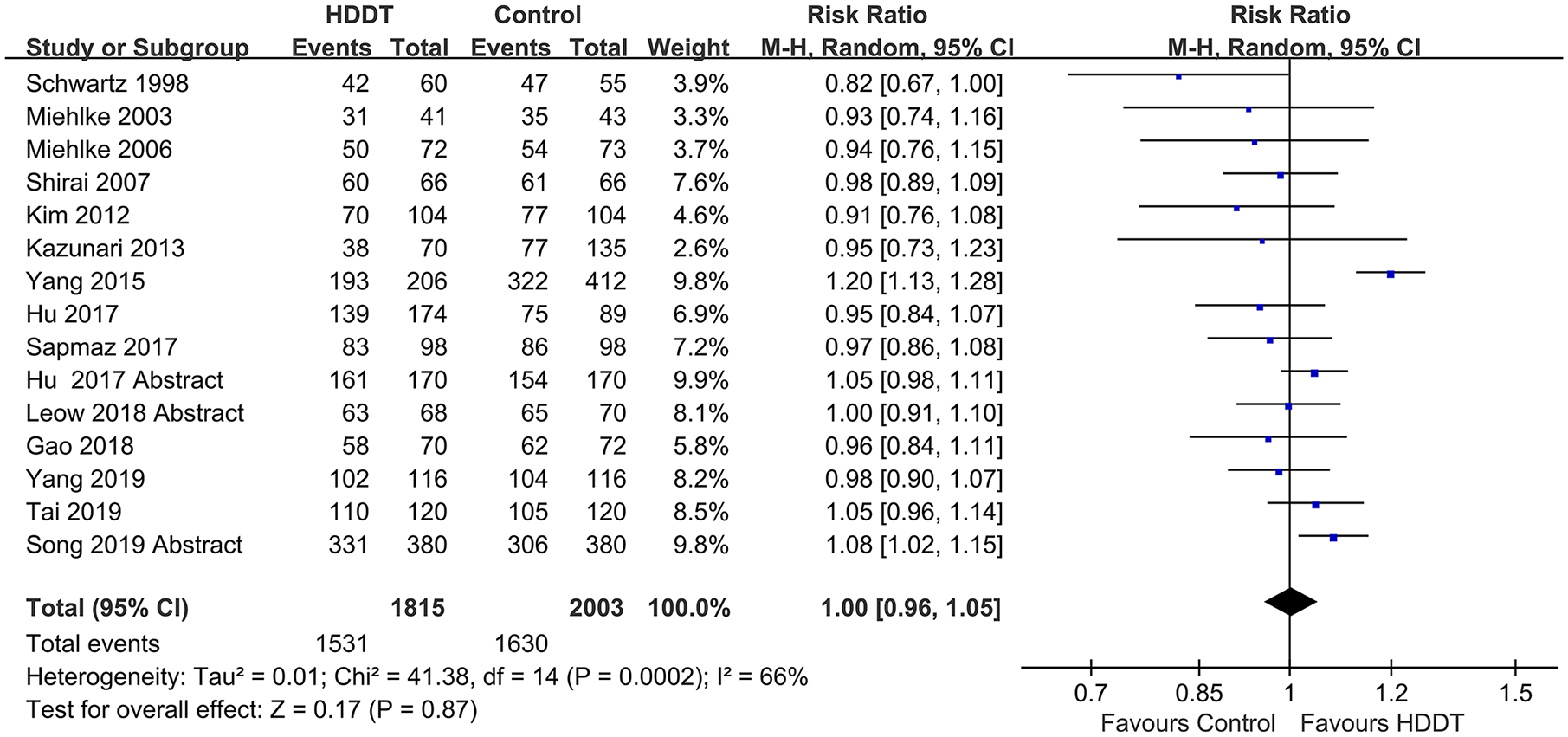
Forest plot of efficacy of HDDT versus control regimens.
Meta-analysis of the adverse effects
A total of 14 included trials provided information of adverse events during treatment.26–35,37–40 The occurrence of adverse events was significantly lower in the HDDT group in comparison with the control group (17% versus 37%; RR 0.48, 95% CI: 0.37–0.64; p < 0.001, I2 = 77%) (Figure 3).

Forest plot of adverse events of HDDT versus control regimens.
Subgroup analysis
For the eradication rate, significant interaction was observed between trials dosing HDDT with different frequency, and larger effect was found in trials dosing four times daily (p = 0.030). There was also significant interaction between trials using different regimen in control group, and larger effect was found in trials using sequential therapy as control regimen (p < 0.001) (see Supplemental File S2).
Sensitivity analysis
Sensitivity analyses showed that one study published in 2015 accounted for the majority of the observed heterogeneity. 32 When we included all 15 trials in the analysis, I2 was 66%. I2 decreased to 23% when this study was omitted, and the overall estimate remained consistent. The omission of other studies separately resulted in only tiny variation around the overall estimate for the remaining 14 studies (see Supplemental File S3).
Trial sequential analysis
For eradication rate, the required diversity-adjusted information size (4031 participants) was calculated based on a proportion of 81.4% events in the control group, a RR reduction of 10%, a type I error of 5%, and a power of 90%. The cumulative Z-curve did not cross the trial sequential analysis boundary or the conventional significance boundary, but crossed the futility boundary, supporting the findings of the conventional meta-analysis, and also suggesting that HDDT was non-superior and non-inferior (i.e., equivalent) to the control intervention (Figure 4).

Trial sequential analysis assessing efficacy of HDDT versus control regimens. The cumulative Z-curve did not cross the trial sequential analysis boundary or the conventional significance boundary, but crossed the futility boundary, indicating HDDT was equivalent to the control regimens.
Publication bias assessment
The funnel plot for eradication rate was symmetrically distributed by visual inspection, indicating that publication bias is unlikely to have had a major influence on the analysis of outcome. Moreover, Egger’s test did not indicate significant publication bias (p = 0.090).
Discussion
In this meta-analysis, the efficacy of HDDT was neither significantly inferior nor superior to the recommended eradication regimens, such as TT, BQT, and non-BQT. This result was robust through the subgroup analyses and sensitivity analyses we performed. Trial sequential analysis showed there is reliable evidence that HDDT is equivalent to the recommended regimens.
Differences between the current and previous studies should be noted.15,41 In the previous meta-analyses, only a few trials with a small number of patients were included, which lacked power to draw definite conclusions. In addition, the absence of a prespecified protocol for these meta-analyses could weaken the strength of their findings and increase the risk of bias. In contrast, 15 trials with 3818 patients were included in our meta-analysis, and the protocol of this study was documented online beforehand. Furthermore, trial sequential analysis was performed in this study to ascertain the equivalence between HDDT and the recommended regimens. In previous conventional meta-analyses, which did not consider the accumulated number of events, the effect size, and the information size, non-significant results were often simply inferred as a need for more evidence. 42 By contrast, in the case of non-significant results, trial sequential analysis could reduce the uncertainty by assessing whether this non-significance is due to lack of power or underlying equivalence between the interventions. 43 In the present study, the cumulative Z-curve crossed the futility boundary, suggesting there is definite evidence for equivalence of efficacy between HDDT and control regimens. Further similar trials are unlikely to alter the conclusion of this study.
As both primary and second resistance to amoxicillin remain rare in most countries, HDDT is an accessible and reasonable option for eradicating H. pylori infection. 44 The present study showed that HDDT was comparable with recommended first-line and rescue regimens in treatment-naive and treatment-experienced patients. When comparing with each regimen in subgroup analyses, HDDT was equivalent to the recommended eradication regimens, such as TT, BQT, and non-BQT, but superior to ST irrespective of eradication experience. Thus, HDDT is not inferior to the current widely used regimens, and it is worthwhile to introduce HDDT into clinical practice. Moreover, HDDT, which consisted of PPI and amoxicillin, used less antibiotics than other eradication regimens, reducing the potential for developing multi-drug resistance.
The dose frequency is essential for the efficacy of PPI-amoxicillin dual therapy, as the bactericidal effect of amoxicillin is time-dependent. 45 Four included trials in this meta-analysis giving PPI-amoxicillin three times daily had an unacceptable eradication rate of 73%,26,28,30,34 whereas the remaining trials adopting a four times daily dosing strategy achieved an eradication rate of 87%, suggesting that the latter dosing strategy was more effective in eradicating H. pylori infection. Moreover, a significantly larger effect was observed in trials dosing HDDT four times daily in comparison with trials dosing three times daily in the subgroup analysis. These collectively indicated that HDDT could attain a better eradication rate when PPI-amoxicillin was given four times daily.
HDDT were safe and well-tolerated, and adverse events were significantly fewer in the HDDT group comparing with other regimens. The most frequent adverse effects of HDDT included diarrhoea, nausea, and dizziness, which were mild and disappeared shortly after completion of treatment.
The strength of this study included applying Cochrane methodology and performing trial sequential analysis. We adhered to a prespecified protocol with minimal deviations, and conducted a range of subgroup analyses to examine clinical heterogeneity and robustness of our findings. In addition, we also performed a trial sequential analysis to confirm the finding of conventional meta-analysis and reduce the uncertainty of non-significant results. Despite these strengths, our study also had limitations. First, only one included study was double-blind in design, and the others had high risk of bias for blinding of participants and personnel. Second, we observed significant heterogeneity in primary outcome. The included trials varied in treatment experience, eradication regimens, and dosing frequency. However, results were robust through subgroup and sensitivity analyses. Finally, most of the included trials were conducted in Asia, and thus extrapolation of the result to other regions should be with caution.
Conclusion
Our study showed that HDDT was equivalent to recommended first-line or rescue regimens with fewer adverse effects. The evidence from this meta-analysis supports the use of HDDT as first-line or rescue treatment for H. pylori infection.
Supplemental Material
Supplementary_1 – Supplemental material for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis
Supplemental material, Supplementary_1 for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis by Yang-Jie Zhu, Yi Zhang, Ting-Yi Wang, Jing-Tao Zhao, Zhe Zhao, Jian-Ru Zhu and Chun-Hui Lan in Therapeutic Advances in Gastroenterology
Supplemental Material
Supplementary_File_1_1 – Supplemental material for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis
Supplemental material, Supplementary_File_1_1 for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis by Yang-Jie Zhu, Yi Zhang, Ting-Yi Wang, Jing-Tao Zhao, Zhe Zhao, Jian-Ru Zhu and Chun-Hui Lan in Therapeutic Advances in Gastroenterology
Supplemental Material
Supplementary_File_2 – Supplemental material for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis
Supplemental material, Supplementary_File_2 for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis by Yang-Jie Zhu, Yi Zhang, Ting-Yi Wang, Jing-Tao Zhao, Zhe Zhao, Jian-Ru Zhu and Chun-Hui Lan in Therapeutic Advances in Gastroenterology
Supplemental Material
Supplementary_File_3_1 – Supplemental material for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis
Supplemental material, Supplementary_File_3_1 for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis by Yang-Jie Zhu, Yi Zhang, Ting-Yi Wang, Jing-Tao Zhao, Zhe Zhao, Jian-Ru Zhu and Chun-Hui Lan in Therapeutic Advances in Gastroenterology
Supplemental Material
Supplementary_File_4_1 – Supplemental material for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis
Supplemental material, Supplementary_File_4_1 for High dose PPI-amoxicillin dual therapy for the treatment of Helicobacter pylori infection: a systematic review with meta-analysis by Yang-Jie Zhu, Yi Zhang, Ting-Yi Wang, Jing-Tao Zhao, Zhe Zhao, Jian-Ru Zhu and Chun-Hui Lan in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
YJZ, YZ, and CHL were involved in study conception and design; YJZ, YZ, and TYW performed literature search and data collection; YZ, YJZ, and JTZ performed data analysis and interpretation; YZ, YJZ, and ZZ were involved in drafting of the manuscript; YJZ, YZ, TYW, JTZ, ZZ, JRZ, and CHL were involved in critical revision of the manuscript for important intellectual content; All authors have approved the final version of the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (No. 81472006) and the Chongqing Science-Health Joint Medical Research Project (No. 2018MSXM014). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
