Abstract
Background:
Children with Crohn’s disease (CD) experience gastrointestinal symptoms that impair nutrition, growth and quality of life. Exclusive enteral nutrition is recommended as a first-line remission induction treatment. However, compliance is challenging due to monotony and the social impact of excluding whole foods, increasing interest in whole-food diet therapies.
Objectives:
This systematic review aimed to summarise current evidence regarding the impact of whole-food therapies on clinical remission (as defined by each study using validated measures) and related health outcomes in children with CD.
Design:
We performed a systematic review of studies assessing whole-food interventions in children with CD.
Data sources and methods:
A systematic search was conducted in electronic databases for research published in English from 1 January 2012 to 16 August 2024. Randomised controlled trials (RCTs), quasi-experimental studies, cohort studies, case-control studies and case series were included.
Results:
Twenty-eight studies (n = 4 RCTs, n = 3 non-RCTs, n = 16 observational studies, n = 5 post hoc analyses) met inclusion criteria and examined six types of diets. Most of the children being treated had uncomplicated, mild–moderate disease activity and were on concomitant medications. Those on whole-food therapies demonstrated a median clinical remission rate of 75% (interquartile range 62%–85%; n = 18 studies), mucosal improvement and healing (n = 5/6 studies), improved inflammatory biomarkers (n = 18/19 studies) and enhanced growth parameters (n = 11/13 studies). Outcomes related to microbial changes were inconsistent. Overall, studies were low–medium quality due to small, non-randomised, uncontrolled studies using a variety of concomitant medications and different definitions for clinical remission, preventing definitive conclusions.
Conclusion:
The findings suggest whole-food diet therapies can potentially be used to treat children with mild to moderate CD and that a flexible, nutrient-balanced dietary approach tailored to the individual child may be possible. However, large-scale, RCTs with standardised outcome measures are needed to further support the routine use of whole-food therapies in paediatric CD.
Trial registration:
PROSPERO registration number CRD42024580134.
Keywords
Introduction
Crohn’s Disease (CD) is an inflammatory bowel disease (IBD) characterised by transmural inflammation and lesions presenting in various parts of the gastrointestinal tract. 1 The pathogenesis of CD is a complex interplay of genetic predispositions, gut microbiome, the immune system and environmental factors. 2 Dysbiosis, characterised by an imbalance in the gut microbial composition and function, is a central feature in the progression of CD. 2 Metagenomic studies have demonstrated that individuals with CD exhibit reduced overall microbial diversity, an increase in potentially pro-inflammatory microbiota and a decrease in beneficial microorganisms. 3 The dysbiosis can trigger an excessive immune response against the body’s own cells, leading to inflammation and damage to the mucosal barrier.2,3
While the pathogenesis of CD in adults and children may be comparable, paediatric-onset CD is often more aggressive, characterised by rapid progression and increased disease activity, resulting in symptoms, including abdominal pain, rectal bleeding and diarrhoea. 4 Children with CD face additional challenges such as chronic malnutrition, sarcopenia, poor bone density and growth failure during critical developmental periods.4,5 Consequently, treatment must address multiple objectives: symptom relief, inflammation reduction, clinical remission, mucosal healing, improvement of growth and quality of life (QoL). 6 Treatment efficacy is evaluated through clinical assessment of symptoms, growth parameters and validated indices like the Paediatric Crohn’s Disease Activity Index (PCDAI), alongside laboratory inflammatory biomarkers C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), faecal calprotectin (FC). Endoscopic evaluation and imaging studies are also employed to assess objective signs of disease activity and mucosal healing. 4
Extensive evidence has shown that exclusive enteral nutrition (EEN) is as effective as corticosteroids in inducing remission in children with CD. 7 EEN is recommended as a first-line induction treatment for children with active, mild to moderate luminal CD due to fewer side effects, better support for growth and superior mucosal healing.7–9 However, the monotony, poor palatability and social challenges associated with EEN result in poor compliance and mental health impacts making it unsuitable for long-term use. 10 Studies also consistently report inappropriate dietary habits among children and adolescents with IBD, marked by imbalanced intake of energy, macro and micronutrients.11,12 Common findings include insufficient intake of carbohydrates, fibre, calcium and vitamin A compared to recommended levels and healthy controls. 12 Additionally, Italian children with IBD showed poor adherence to a Mediterranean diet (MD) pattern, highlighting potential suboptimal dietary quality in this population. 11 Given diet is a modifiable risk factor for managing intestinal dysbiosis and supporting nutrition and growth, 13 interest is rising in the use of whole-food diet therapies for treating paediatric CD.
While individual studies have investigated these whole-food diet therapies, to date, there has been no attempt to synthesise and evaluate their impact on disease management and health, specifically in children and adolescents with CD. This systematic literature review aims to (1) summarise the current evidence on the effectiveness of whole-food diets in achieving or maintaining clinical remission and (2) explore related outcomes, including any impacts on mucosal healing, inflammatory biomarkers, growth and nutritional parameters and gut microbiome and metabolites in children with CD.
Materials and methods
This systematic review adhered to the Preferred Reporting Item for Systematic Reviews and Meta-Analyses (PRISMA) statement 14 (Table S1) and was registered on the International Prospective Systems Review Registry (PROSPERO; #CRD42024580134).
Search strategy
A comprehensive and systematic search of Medline, Embase and CINAHL online databases was undertaken for articles published in English between 1 January 2012 and 16 August 2024. A preliminary scoping review found the earliest eligible study on whole-food dietary interventions for paediatric CD was published in 2014, with increasing scientific attention from the mid-2010s. Consequently, we set the search period to begin in 2012. The search used a combination of subject headings, keywords and synonyms related to ‘paediatric CD’ and ‘dietary therapy’, as shown in Table S2. Full search description is provided in Table S3. The reference lists of potential eligible papers were manually screened (backward citation search) and citation lists of potential eligible papers in Web of Science were also scanned (forward citation search). Relevant reviews were manually screened for individual studies. Abstracts and study protocols led to searches for eligible full-text articles of the same studies.
Eligibility criteria
Type of participant
Children (⩽18 years) diagnosed with CD. For studies with mixed populations (e.g. children and adults or paediatric IBD subtypes), stratified results for children with CD were required.
Types of interventions and comparisons
Whole-food diets designed for CD, either as monotherapy or in combination with partial enteral nutrition (PEN), were included. Comparators could include any alternative or variation, including regular diets. Studies without a comparator group were also considered.
Types of outcome measures
The primary outcome was clinical remission, defined by study-specific criteria using validated tools (e.g. PCDAI, Harvey Bradshaw Index, etc.). Remission rates were reported using intention-to-treat analysis for comparability. The secondary outcomes, when data were available, included dietary tolerance and adherence, mucosal healing, inflammatory biomarker changes, growth and nutrition and microbiota/metabolite shifts.
Types of studies
Randomised controlled trials (RCT), quasi-experimental studies, cohort studies, case-control and case series were included; however, case-series studies that solely reported individual outcomes were excluded. Both prospective and retrospective designs were considered. Detailed inclusion and exclusion criteria are summarised in Table S2.
Study selection
Retrieved studies were imported into Covidence systematic review software (Veritas Health Innovation, Melbourne, VIC, Australia), and duplicate records were removed. Lead reviewer (C.G.) independently completed title and abstract screening. Two reviewers (C.G. and L.J.R.) independently conducted full-text screening. Results were compared, and any disagreements were resolved by a third reviewer (D.G.) through discussion.
Assessment of evidence quality and risk of bias
The quality of evidence for included studies was evaluated using the National Health and Medical Research Council 6-point scale, with levels I (highest) to IV (lowest) representing levels of evidence. 15 Studies were further assessed for their risk of bias using the ‘Quality Criteria Checklist – Primary Research’ tool created by the Academy of Nutrition and Dietetics Evidence Analysis Library, 16 consisting of 10 validity appraisal criteria. Each study received a quality rating indicating its risk of bias. The three ratings were: ‘Positive’ for low risk of bias with issues clearly addressed, ‘Negative’ for high risk of bias with issues inadequately addressed and ‘Neutral’ for studies neither exceptionally strong nor exceptionally weak and with uncertain risk. The assessment for each study was performed independently by two reviewers (C.G. and L.J.R.). Results were compared and discussed with the research team to achieve an agreement on the final quality rating.
Data extraction
An initial data extraction form was developed and refined by the research team until consensus was reached. Data were extracted independently by C.G. from eligible studies and checked by the research team. Data extracted included study characteristics, intervention characteristics for both intervention and comparator groups (where available) and intervention outcomes of interest.
Data analysis
A meta-analysis of eligible studies was unfeasible due to the heterogeneity in intervention type, duration and outcomes. Instead, a critical synthesis was performed to provide an overview of the impact of whole-food diet therapies on remission and relevant health outcomes in children with CD across multiple studies.
Results
Figure 1 depicts the study selection process (PRISMA flowchart). 14 A total of 1795 records were identified, and 60 studies proceeded to full-text review for eligibility. A total of 28 studies were subsequently included.

PRISMA flowchart of literature search and study selection.
Quality and risk of bias assessment
The 28 studies generally provided low- to medium-quality evidence, with more than half classified as III-3 level of evidence. 15 For risk of bias, six studies received an overall ‘positive’ rating.17–22 One study received a ‘negative’ rating due to higher sources of bias, 23 and the remaining 21 studies were rated as ‘neutral’, mostly due to unclear exclusion criteria, the absence of a comparator, incomparable groups, inadequate intervention descriptions and poor or unclear reporting blinding. Four post hoc analyses24–27 received a neutral rating because not all participants from the original trial were accounted for, and there was uncertainty about the reliability of using the original data to answer post hoc questions. The levels of evidence and quality assessment outcomes for the included studies are summarised in Table S4.
Characteristics of the included studies
Table 1 outlines the characteristics of the 28 included studies. All studies were published within the past decade. These studies examined six unique whole-food therapies, including specific carbohydrate diet (SCD; n = 7, 25%),18,23,28–32 Crohn’s disease exclusion diet (CDED; n = 16, 57%),17,19,20,24–27,33–41 anti-inflammatory diet for Crohn’s disease (AID-CD; n = 2, 7%),21,22 plant-based diet (PBD; n = 1), 42 Crohn’s disease treatment-with-eating diet (CD-TREAT; n = 1) 43 and MD (n = 1). 44 All SCD studies were conducted in the USA. Most CDED studies originated from Europe. Both AID-CD studies were conducted in Slovenia. Seven studies were controlled trials,17,19,21,22,31,39,44 16 were observational studies (n = 2 with comparators)18,23,28–30,32–38,40–43 and 5 were post hoc analyses20,24–27 using subset data from one included RCT. 19
Characteristics of included studies (n = 28).

Baseline demographic is not statistically significant between intervention and control group unless indicated otherwise.
Histologic severity based on the maximal amount of inflammation noted on biopsy.
PCDAI = 15–30 mild, >30 moderate.
PCDAI ⩾10 and ⩽30.
SES-CD = 0 no ulcers, 1–2 inactive disease, 3–6 mild disease, 7–15 moderate disease, ⩾16 severe disease.
Due to a mix reported with adult CD participants or paediatric ulcerative colitis participants.
PCDAI >10 and 45.
PCDAI = 7.5–27.5 mild, 30–37.5 moderate, >40 severe.
PCDAI ⩾10 and ⩽40.
Weighted PCDAI <12.5 remission, 12.5–40 mild, 41–57.5 moderate, >57.5 severe.
PCDAI = 0–7.5 remission, 10–27 mild, 30–40 moderate, >40 severe.
PCDAI <10, faecal calprotectin <250 µg/g, ileocolonoscopy results.
PCDAI <10.
Based on PGA.
CDAI <150 quiescent stage, 150–220 mild–moderate, 220–450 moderate–severe, >450 severe/fulminant.
Weighted PCDAI = 22.5–42.55.
AID-CD, anti-inflammatory diet for Crohn’s disease; CDED, Crohn’s disease exclusive diet; CD-TREAT, Crohn’s disease treatment-with-eating diet; EEN, exclusive enteral nutrition; IQR, interquartile range; KIDMED, Child and Adolescent Mediterranean Diet Quality Index; MD, Mediterranean diet; NG tube, nasogastric tube; NR, not reported; PBD, plant-based diet; PCDAI, paediatric Crohn’s disease activity index; PEN, partial enteral nutrition; PGA, Physician Global Assessment; RCT, randomised controlled trial; SCD, specific carbohydrate diet; SES-CD, Simple Endoscopic Examination for Crohn’s Disease.
The review included a total of 613 enrolled participants with CD (441 in intervention groups and 172 in comparator groups) with sample sizes ranging from 5 to 78 children. In most studies, more males than females participated with ages ranging from 1.5 to 19 years. One study that included a 19-year-old among its 20 participants was included in the review. 29 While most studies included participants with active, mild to moderate CD, 4 studies had 71 participants in remission at baseline, accounting for 12% of the total participants.17,29,38,39 The median proportion of participants on concomitant medication was 60% (interquartile range (IQR) 39%, 85%). The concomitant medications included immunomodulators, biologics, immune suppressors and anti-inflammatory agents.
Dietary intervention protocols, including food selections, use of PEN, intervention durations and delivery methods, varied significantly across the six intervention diets. The median intervention durations of included studies was 12 weeks (IQR 8, 52 weeks), with only eight studies exceeding 24 weeks.18,23,28,29,32,35,36,39 Seventeen studies reported dietitian involvement.17–19,21,22,30–38,40–42 Most dietitians were specially trained in the intervention diets and provided dietary instruction, support and monitoring during fortnightly or monthly follow-ups.
Nineteen studies measured dietary adherence using 24-h dietary recall, food diaries, food frequency questionnaires or patient self-reports.17–19,22,28–36,38,39,41–44 Of the four SCD studies reporting dietary adherence, three indicated over half the participants consumed disallowed food items at various times.18,28,29,32 Studies that assessed dietary adherence of the other five diets found a median adherence rate of 83% (IQR 80%, 100%).
High tolerance rates (median 91%, IQR 80%, 94%) were identified from 10 studies.17–19,21,22,29,31,37,38,43 An RCT demonstrated CDED was significantly better tolerated compared to EEN (98% vs 74%, p = 0.002). 19 The most common reasons for intolerance included having difficulty maintaining the diet, worsening symptoms and experiencing adverse events such as weight loss, nausea and vomiting.
Overview of the included whole-food diet therapies
Figure 2 provides a comparison of the core features of the whole-food diet therapies included in the review. While each diet varies in its structure, level of prescriptiveness and inclusion of PEN, several shared principles emerge: the emphasis on the reduction or exclusion of processed foods, food additives and refined sugars, alongside increased consumption of fruits, vegetables, legumes and healthy fats (e.g. from olive oil and fish). Most diets limit red meat and encourage lean protein sources, with some allowing whole grains, specific dairy products and different amounts of PEN depending on the treatment phase.

Comparison of key features across the included whole-food diet therapies.
The SCD demonstrates a greater degree of restriction by excluding grains, cereals and certain starchy vegetables. 45 The CDED is a phase-wised diet involving three phases: 2 × 6-week induction phases, and a third ‘maintenance’ phase. Phase I consists of 50% of energy requirements from a polymeric formula and 50% whole foods from a list of mandatory or allowed foods, while limiting insoluble fibre to prevent bowel obstructions. Phase II reduces the formula to 25% of requirements and reintroduces small portions of excluded foods such as vegetables. After remission is achieved, phase III introduces an increasing variety of foods to promote dietary flexibility and long-term compliance. 19 The AID-CD is a culturally adapted modification of the CDED, incorporating elements of Slovenian cuisine while excluding fried foods. 21 The CD-TREAT uses whole foods to mimic the nutritional profile of a polymeric formula used in EEN, matching its macronutrient, vitamin and mineral content, except for resistant starch, which is not present in EEN. 43 The PBD is a lacto-ovo-semi-vegetarian diet that includes fish once per week and meat once every 2 weeks. 42
Clinical remission
Table 2 summarises the clinical remission rates in children with CD on whole-food diet therapies. Eighteen studies used various validated measures to assess clinical remission, with a median remission rate of 75% (IQR 62%, 85%).
Clinical remission rates in children with Crohn’s disease on whole-food diet therapies (n = 18).
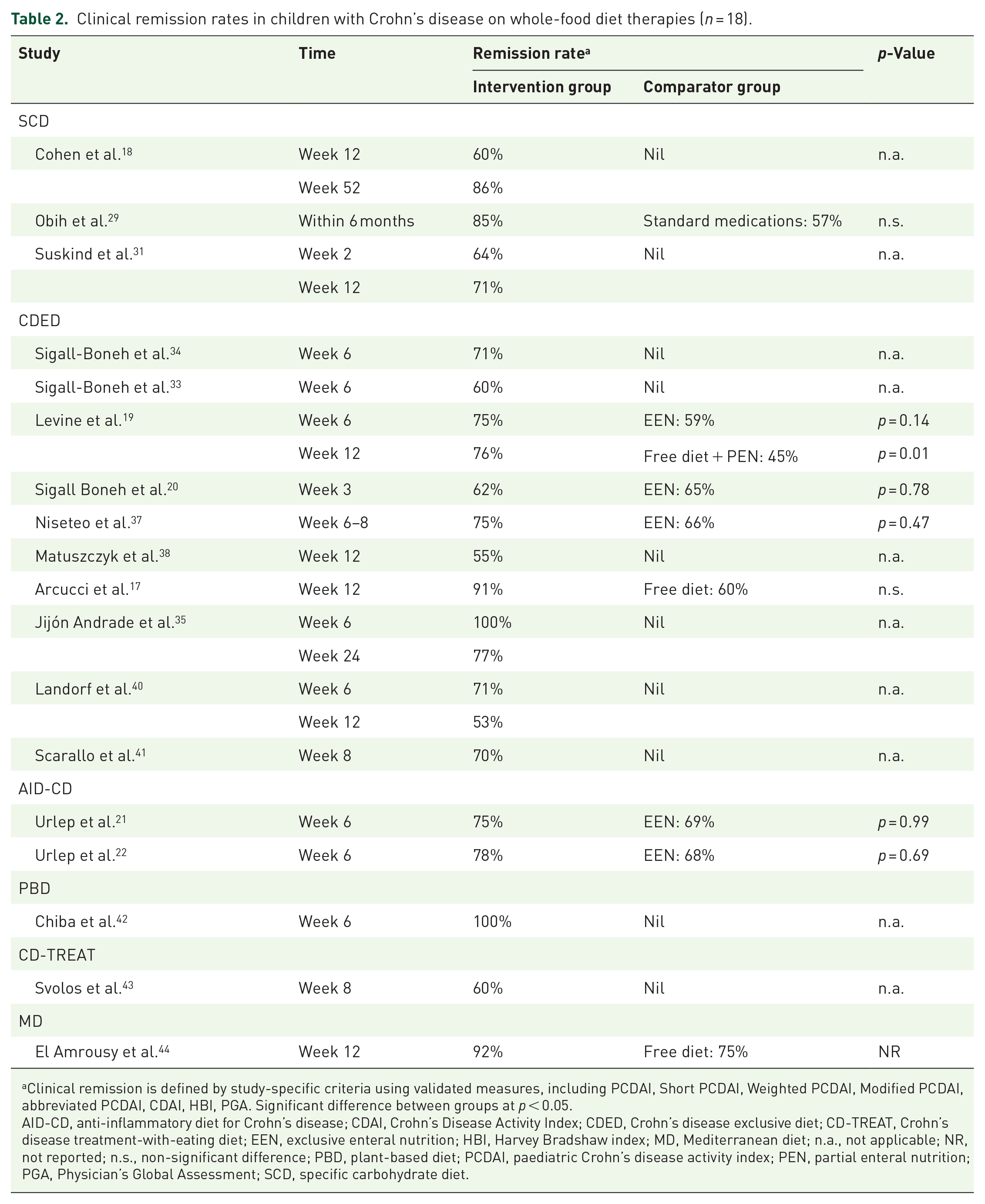
Clinical remission is defined by study-specific criteria using validated measures, including PCDAI, Short PCDAI, Weighted PCDAI, Modified PCDAI, abbreviated PCDAI, CDAI, HBI, PGA. Significant difference between groups at p < 0.05.
AID-CD, anti-inflammatory diet for Crohn’s disease; CDAI, Crohn’s Disease Activity Index; CDED, Crohn’s disease exclusive diet; CD-TREAT, Crohn’s disease treatment-with-eating diet; EEN, exclusive enteral nutrition; HBI, Harvey Bradshaw index; MD, Mediterranean diet; n.a., not applicable; NR, not reported; n.s., non-significant difference; PBD, plant-based diet; PCDAI, paediatric Crohn’s disease activity index; PEN, partial enteral nutrition; PGA, Physician’s Global Assessment; SCD, specific carbohydrate diet.
In the study by Obih et al. 29 a higher proportion of participants achieved remission after an average of 9.6 months on the SCD compared to those who switched to medical therapy (85% vs 57%), no statistical difference was observed between the groups over time. In an RCT, 31 64% of participants achieved remission after 2 weeks on SCD before randomisation to either a modified SCD (adding oats, rice) or a whole-food diet (eliminating wheat, corn, sugar, milk and additives). The remission rate was sustained to week 12, indicating all three diets effectively improved PCDAI. However, statistical tests were not conducted due to small sample sizes.
Eleven CDED studies assessed remission in paediatric patients with diverse baseline disease characteristics, including newly diagnosed cases, relapse from previous treatments, loss of response to biologics and inactive disease. Three studies compared CDED to EEN for inducing CD remission, demonstrating that CDED (phase I) was as effective as EEN in inducing remission at 3 weeks (early remission) and 6 weeks.19,20,37 Of note, in the study by Niseteo et al., 37 80% of participants in the CDED group received 1–2 weeks of EEN before starting CDED. Furthermore, three studies compared CDED to an unrestricted diet for maintaining CD remission. In the RCT by Levine et al., 19 the CDED group transitioned to the second phase of the diet after week 6, while the EEN group started a free diet with 25% PEN. At week 12, the CDED group had a significantly higher corticosteroid-free remission rate than the free diet with PEN group (76% vs 45%, p = 0.01). A subgroup analysis revealed a higher chance of maintaining remission in the CDED group compared to the free diet with PEN group at week 12 (87.5% vs 56%, p = 0.01). Another RCT by Arcucci et al. 17 focused exclusively on children with CD in clinical remission at baseline (PCDAI < 10) and compared CDED to free diets for sustaining remission, showing that only one patient in the CDED group needed intensified biological treatment, compared to eight in the free diet group (p = 0.005) at week 12. Interestingly, one recent retrospective chart review observed participants with severe disease phenotype according to Physician’s Global Assessment were less likely to achieve clinical remission using CDED compared to those with mild to moderate disease (50% vs 80%, p = 0.012). 41 Another retrospective study in Australia found CDED with formula modifications (using cow’s milk-based, rice-based or soy-based) appeared as effective as the original protocol. 40 Even a small subset on CDED without formula saw remission in three of four children with CD. 34
Mucosal healing
Table 3 summarises mucosal healing criteria and rates reported in the included studies. Six studies assessed mucosal healing, five employing endoscopic procedures (reporting healing rates of 0%–60%),18,21,22,32,42 and one using the mucosal inflammation non-invasive (MINI) index (reporting a significant mucosal improvement). 36 Among the five endoscopic studies, two (n = 1 SCD, 32 n = 1 PBD 42 ) used non-validated criteria, defining mucosal healing based on visible inflammation observed during endoscopy. Three studies used validated measures, including the Simple Endoscopic Examination for Crohn’s Disease score of 0 (n = 2 AID-CD) to quantify inflammation in the lower gastrointestinal tract,21,22 and a Lewis Score <135 (n = 1 SCD 18 ) to assess small bowel inflammation via video capsule endoscopy.46,47
Mucosal healing rates in children with Crohn’s disease on whole-food diet therapies (n = 6).
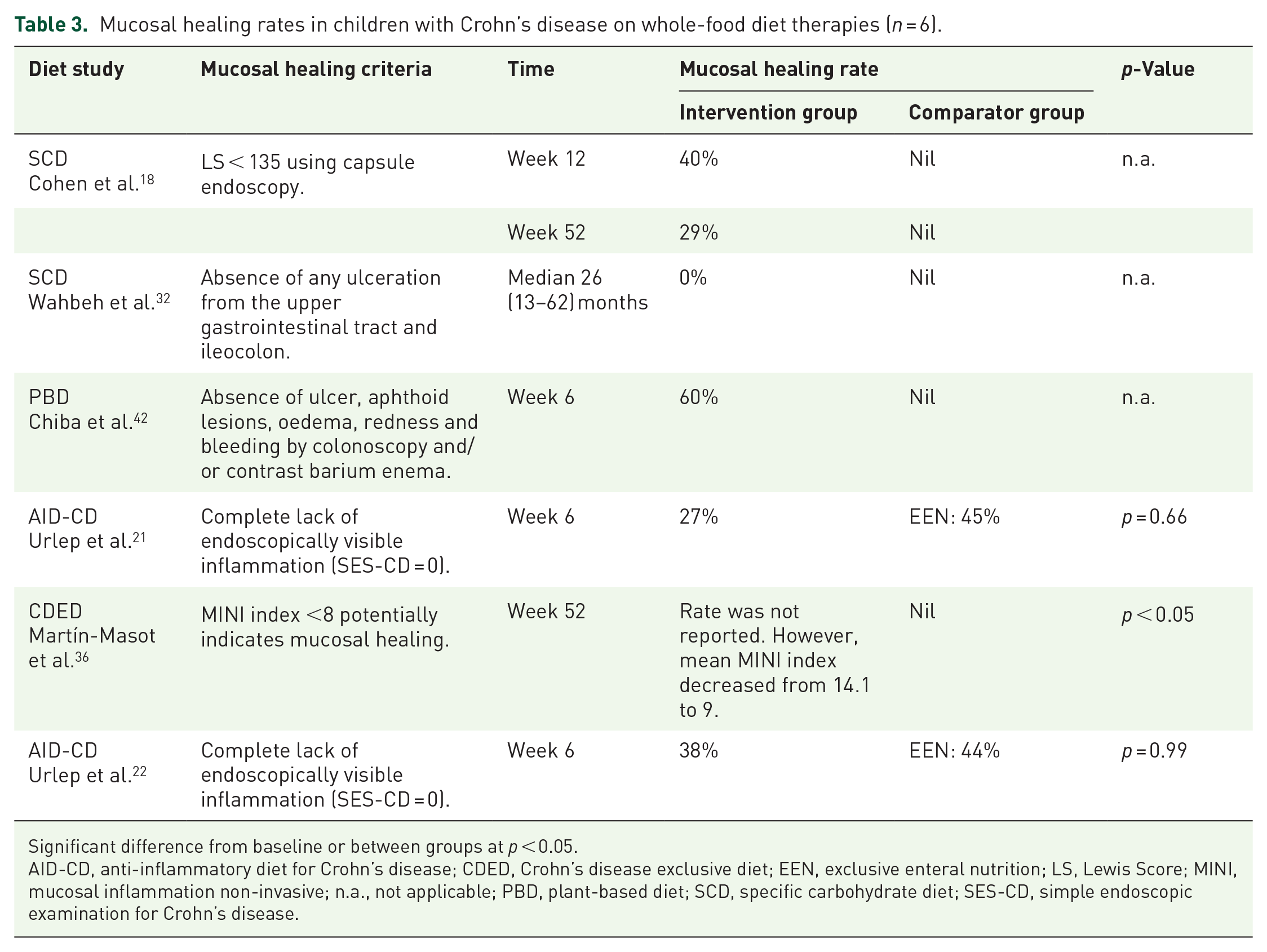
Significant difference from baseline or between groups at p < 0.05.
AID-CD, anti-inflammatory diet for Crohn’s disease; CDED, Crohn’s disease exclusive diet; EEN, exclusive enteral nutrition; LS, Lewis Score; MINI, mucosal inflammation non-invasive; n.a., not applicable; PBD, plant-based diet; SCD, specific carbohydrate diet; SES-CD, simple endoscopic examination for Crohn’s disease.
The timing of endoscopic assessments varied across studies, ranging from 6 weeks to a median of 26 months. The highest mucosal healing rate (60%) was observed in the PBD study, 42 which assessed healing at 6 weeks using non-validated measures to assess the lower gastrointestinal tract. Notably, all participants in this study received concomitant infliximab with the PBD intervention. 42 In contrast, the lowest healing rate (0%) was reported in an SCD study with the longest intervention duration of 13–62 months (median 26). 32 This study applied a stricter, non-validated definition of mucosal healing requiring the absence of ulceration in both the upper and lower gastrointestinal tracts. Despite the resolution of active CD symptoms and normalisation of inflammatory biomarkers, none of the participants achieved complete mucosal healing. 32 Importantly, this SCD study used dietary intervention exclusively, without concomitant medications. 32
Laboratory inflammatory biomarkers
Table 4 provides an overview of the outcomes of laboratory inflammatory biomarkers reported in the included studies. Most participants on whole-food diet therapies achieved improvements in the inflammatory biomarkers, including FC, CRP, ESR and albumin.
Outcomes of laboratory inflammatory biomarkers in children with Crohn’s disease on whole-food diet therapies (n = 19).

Significant difference from baseline in both groups (p < 0.05), unless otherwise specified.
Significant difference between groups (p < 0.05) for changes.
Significance not reported.
Treatment naïve subgroup refers to newly diagnosed patients who have not received any prior treatment for CD, such as biological agents.
AID-CD, anti-inflammatory diet for Crohn’s disease; CDED, Crohn’s disease exclusive diet; CD-TREAT, Crohn’s disease treatment-with-eating diet; mSCD, modified specific carbohydrate diet; n.s., non-significant difference from baseline in both groups; NR, not reported; PBD, plant-based diet; SCD, specific carbohydrate diet.
Burgis et al. 28 found that while participants on a strict SCD showed significant improvements in ESR and albumin, levels remained stable after the diet was liberalised. Two studies demonstrated that CDED was as effective as EEN in decreasing CRP levels.19,37 Chiba et al. 42 reported that participants on a PBD plus Infliximab achieved a significant reduction in the mean CRP values, decreasing from 5.2 mg/dL at baseline to 0.2 mg/dL by week 6.
Twelve studies tracking FC showed reduced levels from baseline.17,19,21–23,29,35,36,38,40,41,43 Levine et al. 19 found that CDED reduced FC levels comparably to EEN by week 6 (CDED: 3126–1744 µg/g; EEN: 2647–1021 µg/g). However, while the CDED group continued to show further reductions between weeks 7 and 12, FC levels in the EEN group rebounded to baseline after resuming a free diet. This finding aligned with those of Arcucci et al., 17 where participants on CDED experienced a significant FC reduction, while FC levels remained unchanged in those on free diets. Three CDED studies defined FC normalisation targets, achieving rates of 35% (<250 µg/g) by week 12, 38 27% (<150 µg/g) by week 8 41 and 29% (<100 µg/g) by week 12. 40
Growth and nutritional parameters
Eleven studies reported increases in mean or median weight and body mass index (BMI) following whole-food dietary interventions.17–19,21,23,28,29,37,38,40,41 Levine et al. 19 reported significant improvements in weight z-scores for both CDED and EEN by week 6 (p < 0.001), while Niseteo et al. 37 found that CDED provided superior support for growth compared to EEN. In addition, Lev-Tzion et al. 26 demonstrated significant improvements in bone formation following either CDED or EEN intervention. Martín-Masot et al. 36 assessed children with CD adhering to CDED for up to 52 weeks, finding significant improvements in dietary habits and patterns.
Faecal microbiota and metabolite
Two SCD30,31 and five CDED studies19,24,25,27,39 examined changes in faecal microbiota and/or metabolites associated with the dietary interventions. The SCD studies found highly individual changes in microbial composition but observed increased community diversity and improved dysbiosis after 12 weeks.30,31 The treatment was associated with an increase in Blautia, Lachnospiraceae, Faecalibacterium, Roseburia, Anaerobutyricum and Eubacterium and a decrease in Escherichia coli and Faecalibacterium prausnitzii. 31 Suskind et al. 31 also found that a strict SCD decreased the metabolism of starch and galactose while increasing the metabolism of plant polysaccharides, and caused a shift from amino acids biosynthesis to catabolism.
Levine et al. 19 reported a decrease in Proteobacteria in participants from both CDED and EEN groups after 6 weeks. From weeks 7 to 12, the CDED group continued to show a decrease in Proteobacteria and an increase in microbial diversity. Conversely, the EEN group, following the reintroduction of free diets, had the microbial composition returning to baseline levels, notably with a major rebound in Proteobacteria. A subsequent post hoc analysis of metagenome sequences in faecal samples found that CDED with PEN increased Firmicutes, particularly Clostridiales, driven by genera including Roseburia, Oscillibacter, Anaerotruncus and Ruminococcus. It also decreased the relative abundance of Proteobacteria to the levels close to healthy controls, except for E. coli. 27 Interestingly, Stein et al. 39 found that the abundance of certain gut microbial gene pathways related to the metabolism of amino/nucleotide sugar and galactose from the baseline samples could predict a CD relapse at 52 weeks with 80% accuracy.
Ghiboub et al.24,25 observed a reduction in metabolites of the kynurenine pathway and an increase in those of the serotonin pathway in participants on CDED or EEN and in remission by week 12. However, in participants who failed to sustain remission, the metabolomic profile rebounded to baseline. Furthermore, Verburgt et al. 27 found that although the concentration of short-chain fatty acid (SCFAs) was not associated with remission achieved by CDED with PEN by week 12, an increase in the SCFA synthesis pathway was observed.
Discussion
This systematic review aimed to summarise the existing evidence regarding the impact of whole-food therapies on clinical remission and related health outcomes in children with CD. In total, 28 studies examined 6 unique types of diets that focus on modifying diet quality by incorporating nutrient-rich foods while excluding pro-inflammatory foods or components in children living with CD, demonstrating effects on clinical remission (n = 18/18 studies), mucosal improvement and healing (n = 5/6 studies), improved inflammatory biomarkers (n = 18/19 studies) and enhanced growth parameters (n = 11/13 studies). Outcomes related to gut microbiome changes, however, were inconsistent, and results should be interpreted with caution as most participants were on concomitant medication regimes and evidence overall was low to medium quality due to small, non-randomised, uncontrolled studies using varied definitions for clinical remission and mucosal healing outcomes. No conclusions could be made regarding the effects of whole-food diets on children with severe CD and complications, as most studies excluded this disease phenotype. Therefore, to overcome these methodological flaws more high-quality controlled intervention trials are needed to further support the efficacy of whole-food diet therapies in managing paediatric CD.
The included studies on whole-food diet therapies primarily focus on inducing clinical remission in children with uncomplicated, mild to moderate CD. A median remission rate of 75% (IQR 62%, 85%) was reported for these diets, which are comparable to the combined remission rates of 50%–90% observed for EEN in the literature. 48 However, unlike EEN studies, most included studies on whole-food diets involved participants receiving additional medications. To minimise potential confounding effects from medications, most studies prohibited starting new medications or modifying existing doses during the dietary intervention or within a defined stabilisation period before initiating the diets. Participants, however, were typically allowed to continue stable doses of maintenance immunomodulators or biologics during the intervention. While this approach complicates the attribution of clinical remission solely to the diet, it reflects real-world clinical practice, where dietary interventions (except for EEN) are rarely used as standalone therapies for remission induction due to a lack of high-quality evidence. Further research is needed to investigate whole-food diets as adjuvant therapies alongside medications, including for severe CD. This approach may enhance real-world applicability and integration into routine CD management by leveraging the synergistic effects of medications targeting the immune response and dietary interventions addressing environmental triggers. 7
In addition to combining with medications, most included studies, particularly those investigating the CDED and AID-CD protocols incorporated 25%–75% of total caloric intake from PEN. Although CDED with or without PEN demonstrated comparable efficacy in inducing remission in adults with mild to moderate biologic-naïve CD, 49 its effectiveness in paediatric populations remains underexplored. The inclusion of PEN continues to be recommended in dietary protocols for children, as it helps meet daily energy and macro and micronutrient requirements (e.g. calcium) that may be difficult to achieve through CDED alone. 34
Not surprisingly, remission rates for whole-food diet therapies varied significantly across studies, likely due to heterogeneity in study design, subjects enrolled, interventions and clinical remission criteria. Despite these variations, these diets share a focus on modifying diet quality by reducing processed foods high in fat, salt, refined sugar and additives that may exacerbate gut inflammation, 13 while promoting nutrient-rich whole foods that positively influence the gut microbiota, metabolites and barrier function. 50 While improving diet quality naturally minimises exposure to heavily processed foods, food additives and chemicals, the SCD is particularly restrictive by also excluding grains and starchy vegetables. This increased degree of restriction may contribute to its relatively lower adherence compared to other dietary therapies, as three studies reported over half of children consumed disallowed foods such as oats and potatoes during the intervention.18,28,29 However, adding these ‘illegal’ foods did not appear to compromise clinical remission rates or improvements in inflammatory biomarkers.28,29 These findings highlight the need for further research to understand the mechanisms driving gut inflammation and how these mechanisms may vary or align with individual biology, suggesting that some dietary restrictions may be unnecessary, and a more flexible diet tailored to the individual child might effectively achieve remission.
Among whole-food diet interventions, studies implementing CDED provided the most detailed dietary information and intervention protocols and have therefore been more widely replicated and investigated, contributing to a growing evidence base supporting its efficacy. Three studies comparing CDED to EEN showed similar remission rates at week 6,19,20,37 but CDED was better tolerated and more effective in sustaining remission by week 12. 19 These outcomes align with recent guidelines, which suggest CDED be considered as an alternative to EEN to induce remission in paediatric patients with mild to moderate CD. 51
The long-term efficacy of whole-food diet therapies in maintaining remission in children with CD remains unclear. Only four of the included studies explored clinical remission beyond 24 weeks, reporting a remission rate of 56%–86%.18,29,35,39 While these results appear promising, the clinical trials were statistically underpowered due to small sample sizes, highlighting a significant gap in understanding the effectiveness of whole-food diets in sustaining remission over time. Nevertheless, given the incomplete understanding of gut inflammation mechanisms, it is important to prioritise diet quality modifications over excessive dietary restrictions in long-term CD management. Diets rich in fruits, vegetables and fish have been linked to increased baseline microbial diversity, fewer disease flares and a delayed need for anti-TNF therapy in paediatric CD. 52 These findings reinforce the value of a flexible, nutrient-balanced diet tailored to individual needs for long-term remission in paediatric CD.
Although dietary interventions show promise in achieving clinical remission in mild to moderate paediatric CD, their impact on mucosal healing remains underexplored. Mucosal healing, an objective treatment target in paediatric CD defined by the absence of ulcers on ileocolonoscopy, strongly predicts sustained clinical remission and better long-term outcomes compared to subjective clinical assessments.6,46 Among the studies reviewed, mucosal healing was only evaluated in six studies, consistently showing lower rates of healing compared to clinical remission.18,21,22,32,36,42 These findings align with the guidelines that highlight clinical scores alone do not adequately reflect mucosal healing. 8 The six studies also demonstrated variability in the criteria used to define endoscopic healing, further complicating cross-study comparisons and the interpretation of results. Additionally, most studies had short follow-up durations (6 or 12 weeks), whereas guidelines recommend endoscopic outcome assessment at 6–9 months following the start of treatment to allow enough time for a treatment effect to be seen. 53 However, this recommendation was tailored to patients treated with anti-TNF agents. 53 In contrast, no trials to date have examined the optimal timing for evaluating mucosal healing in response to dietary interventions.
Although mucosal healing is an important long-term treatment target in paediatric CD, 6 the invasive nature of endoscopy makes repeated assessments impractical and non-standard in most paediatric settings, posing a major barrier to routine monitoring of mucosal improvement. However, there is growing recognition that proxy measures such as the MINI index, FC and bowel ultrasound can serve as feasible alternatives to repeated ileocolonoscopies in children with CD. 54 These non-invasive tools can complement established clinical indices, such as PCDAI, in evaluating treatment response and mucosal improvement. 54 Future research should prioritise the development, validation and integration of non-invasive methods in dietary intervention studies to support the monitoring of mucosal healing over time, reduce procedural burden and enhance the feasibility of long-term disease monitoring in routine clinical practice.
Inflammatory biomarkers, particularly FC and CRP, are widely used to assess disease activity and response to therapy in CD. 55 Most studies reviewed reported decreases in FC and CRP levels following dietary interventions, suggesting whole-food diet therapies may reduce intestinal and systemic inflammation. Current guidelines recommend reducing FC to <250 μg/g as a marker of treatment success, 8 though a reliability study suggested that a combination of PCDAI < 10, CRP < 5 mg/dL and FC < 500 μg/g better predict inflammation resolution. 56 However, variability in measurement criteria across included studies, including normalisation of values based on various thresholds, percentage reductions (e.g. >50% drop) or absolute reductions, introduces heterogeneity that complicates direct comparisons across studies.
Diet significantly influences gut microbiota composition and function, which are key regulators of metabolic and immune responses. 13 Studies in this review found that, despite individual variation in faecal microbiome changes, remission achieved through SCD, CDED and EEN was associated with increased microbiome diversity and decreased abundance of Proteobacteria.19,27,30,31 At the genus level, successful SCD and CDED interventions promoted an increase in Roseburia,27,31 a key butyrate-producing bacterium in the colon that is typically depleted in patients with CD. 57 Butyrate, an SCFA, is essential for maintaining intestinal barrier integrity and reducing inflammation. 58 These results indicate that whole-food diet therapies may drive clinical improvement by restoring gut symbiosis through modulation of microbial composition and function. Notably, participants who relapsed following the reintroduction of regular diets or failed to respond to dietary therapies did not demonstrate those specific changes.19,27 This comparison raises questions about whether disease remission is driven by the modulation of the gut microbiome through dietary interventions or if remission itself leads to microbiome changes. Therefore, further exploration of the mechanistic links between gut microbiome and inflammation in CD is needed.
Adherence to therapeutic diets is often challenging and requires extensive support from family, friends and healthcare providers. 59 A study assessing the feasibility of applying CDED reported 46% of participants had difficulty following the diet precisely, and 43% found it challenging to continue the diet beyond 12 weeks. 60 However, with the exception of SCD, this review found relatively high adherence rates (median 83%, IQR 80%, 100%) across other dietary therapies, likely attributable to the predominantly short intervention durations (⩽12 weeks). It is also important to note that adherence measures in all included studies relied on subjective self-reports, which are prone to recall bias and under-reporting, 61 highlighting the need for objective measures to assess dietary adherence. McKirdy et al. 62 recently used faecal gluten immunogenic peptides (GIP) as an objective biomarker of compliance with EEN and found that 23% of participants deemed compliant by clinical review had detectable GIP, indicating gluten intake during EEN. These participants showed poorer responses to EEN compared to those with undetectable GIP. Since most whole-food diet therapies limit gluten, faecal GIP could potentially serve as a proxy biomarker for adherence to these diets.
Interestingly, even participants who struggled to maintain adherence to CDED beyond 12 weeks showed improvements in dietary habits, including increased fruit and vegetable intake and reduced consumption of processed foods, 63 indicating the potential long-term benefits of these diets. Given the significant role of dietary adherence, dietary education and ongoing support from gastroenterology dietitians are essential for sustaining engagement with these therapies. 60 To further enhance adherence and feasibility in clinical practice, future research should focus on developing clear dietary guidelines that address adherence barriers and enablers. Co-designing these guidelines with end users, including children with CD and their families, could ensure they are practical and adaptable to real-world clinical settings.
Whole-food diet therapies in paediatric CD requires careful consideration of nutritional and growth outcomes, as children with CD are particularly prone to malnutrition. 5 While most studies included in this review assessed changes in growth parameters, such as weight, height and BMI, these measures alone were insufficient to provide a comprehensive evaluation of nutritional adequacy. 64 Micronutrient deficiencies are significantly associated with CD and may persist despite improvements in growth parameters. 51 For instance, a study evaluating SCD in a small cohort of children with IBD found that a significant percentage of children failed to meet daily requirements for calcium and vitamin D, despite achieving weight gain and adequate energy intake. 65 Therefore, it is imperative that children undergoing therapeutic dietary interventions receive close monitoring and follow-up by dietitians (offered in some but not all studies), including anthropometric measurements and detailed evaluations of nutritional intake.
Beyond nutritional considerations, none of the included studies assessed the impact of whole-food dietary therapies on QoL in children and their families living with CD. As QoL is a key treatment goal, 6 the evaluation of QoL is particularly relevant in studies comparing whole-food diets to EEN, where EEN is often associated with greater psychosocial burden due to the exclusion of solid foods. Future research should incorporate validated QoL measures to investigate how diet therapies affect overall well-being of the child and their family to inform the development of sustainable dietary strategies in paediatric CD.
To the best of our knowledge, this is the first review to comprehensively evaluate all types of whole-food diet therapies studied specifically in the context of paediatric CD, whereas previous systematic reviews have exclusively focused on CDED in both children and adults with CD.66,67 This review highlights critical gaps in the current evidence base and identifies key priorities for advancing research on whole-food diet interventions in paediatric CD. Figure 3 presents a detailed summary of these recommendations, grouped into actionable categories.
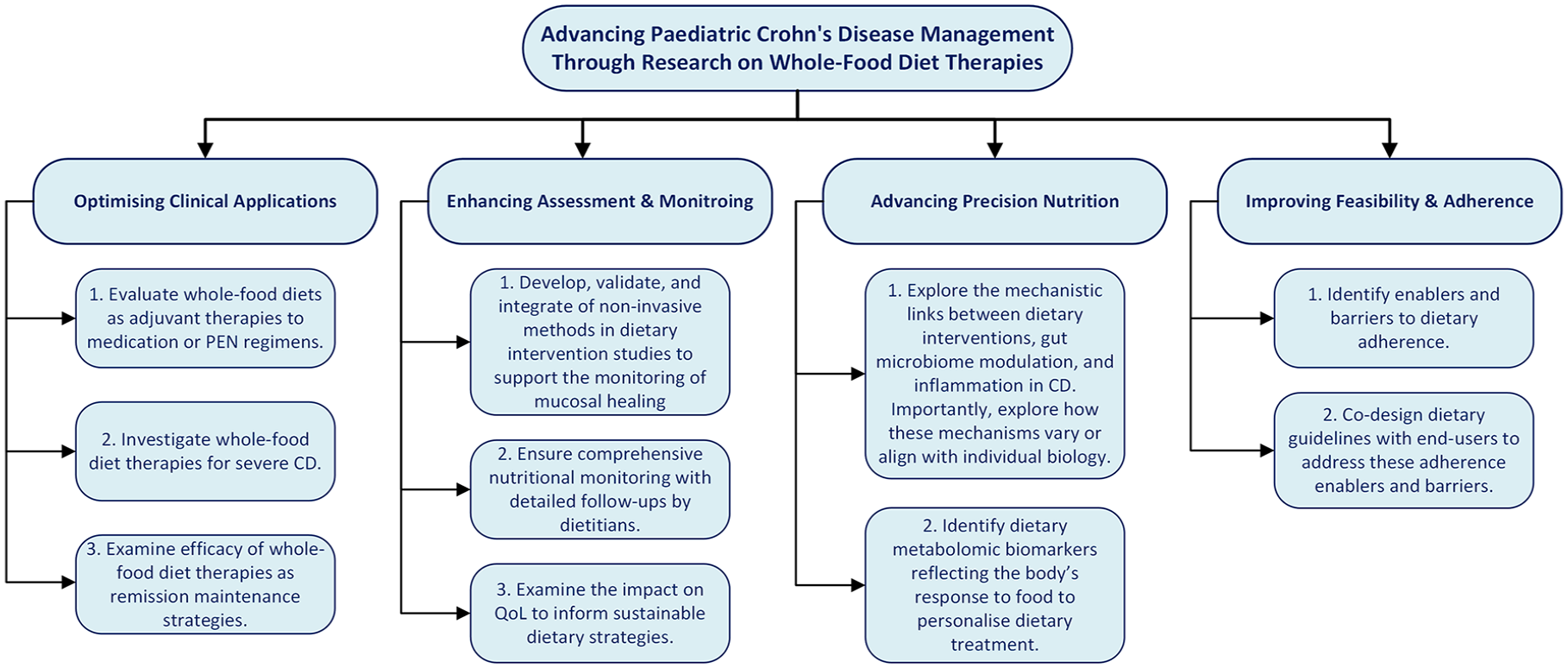
Future research directions for whole-food diet interventions in paediatric CD.
Several limitations remain in our study. The diversity of whole-food diet therapies and heterogeneity in clinical remission criteria, study design and methodology hinder comparability and generalisation of findings; and the ability to perform a meta-analysis of dietary interventions across multiple studies with comparator groups. Additionally, clinical remission rates were often confounded by concomitant treatments, such as medications. Finally, the sample sizes in the included studies were generally small, leading to inadequate statistical power and a high risk of sampling and selection bias. Future research should recruit larger and diverse sample to ensure representativeness and adequate statistical power.
Conclusion
In conclusion, our findings suggest that whole-food diet therapies hold potential for treating children with mild to moderate CD and that a flexible, nutrient-balanced dietary approach tailored to the individual child may be possible. However, the current evidence is limited by small, non-randomised and uncontrolled studies, which have employed inconsistent criteria to define clinical remission and involved varying types of concomitant medication regimes, preventing definitive conclusions. Compared to other whole-food diet therapies for paediatric CD, CDED has the stronger evidence base supporting its efficacy. However, studies examining its effectiveness in maintaining long-term remission remain limited. Therefore, large-scale, RCTs with standardised outcome measures and longer intervention durations are warranted to further support those results and to assess long-term efficacy. Furthermore, future research that explores the impacts of whole-food diets on the QoL and gut microbiome, identifies dietary metabolomic biomarkers for monitoring responses, and develops dietary guidelines that address adherence challenges will advance whole-food diets to precision nutritional therapies in paediatric CD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251355436 – Supplemental material for Whole-food diet therapies for children with Crohn’s disease: a systematic review
Supplemental material, sj-docx-1-tag-10.1177_17562848251355436 for Whole-food diet therapies for children with Crohn’s disease: a systematic review by Cathy Guo, Julia Fox, Kristie Bell, Danielle Gallegos and Lynda J. Ross in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors thank Mr Peter Sondergeld (Queensland University of Technology research liaison librarian) for their assistance with the initial search strategy.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
