Abstract
Thalidomide is effective in inducing and maintaining clinical remission, as well as mucosal healing, in patients with refractory Crohn’s disease (CD). However, long-term use of thalidomide has raised concern because of the high incidence of adverse events. Cardiovascular events induced by thalidomide have been reported in patients with multiple myeloma, amyotrophic lateral sclerosis, and transfusion-dependent refractory anemia. We report here an extremely rare case of sinus bradycardia induced by thalidomide in an adult patient with CD. This patient’s heart rate converted back to a normal sinus rhythm after withdrawal of thalidomide, but recurred after restarting of thalidomide. Cardiac toxicity should be closely monitored when using thalidomide in patients with CD.
Keywords
Introduction
Crohn’s disease (CD) is a chronic inflammatory bowel disease (IBD) characterized by clinical relapse and progression. The incidence and prevalence of CD are increasing globally.1–3 Biologics and thiopurines are widely used to treat CD with convincing efficacy.4,5 However, refractory disease, including non-response or intolerance to thiopurines, and loss of response to biologics remain a clinical challenge.6,7 With potential effects of immunomodulation and suppression of tumor necrosis factor-α (TNF-α), thalidomide has been shown to be effective in treating refractory CD in retrospective studies and in a landmark randomized, controlled trial.8–14 Recently, the role of thalidomide in inducing and maintaining mucosal healing in patients with CD has been reported in case series and a prospective open-label study that was performed at our center.11–13,15,16 Moreover, endoscopic and histological healing induced by thalidomide was recently reported in multicenter trials. 17 However, long-term use of thalidomide is limited owing to the high incidence of adverse events, including peripheral neuropathy, somnolence, and constipation.8,9,11,14 Thrombosis is the most common cardiovascular adverse event induced by thalidomide, with an incidence ranging from 2.3% to 22.5%.18–20 Some rare cardiovascular events, such as arrhythmia, induced by thalidomide have been reported in patients with multiple myeloma, amyotrophic lateral sclerosis, and transfusion-dependent refractory anemia.21–23 However, arrhythmia induced by thalidomide in patients with IBD has been seldom reported. 24 Asymptomatic bradycardia was previously reported as an adverse event in a child with CD. 14 We report here an extremely rare case of sinus bradycardia induced by thalidomide in an adult patient with CD. The patient’s heart rate converted back into normal sinus rhythm after thalidomide withdrawal, but recurred after reuse of thalidomide.
Case report
In May 2015, a 32-year-old man with established CD for 4 years presented to our IBD outpatient clinic with a clinical flare-up during maintenance therapy with azathioprine (2.0 mg/kg/day). The Crohn’s disease activity index was 232 with ulceration in the terminal ileum and colon as shown by colonoscopy (Figure 1a, 1b). A surface electrocardiogram (ECG) in the patient’s regular clinic visits showed sinus rhythm during maintenance use of azathioprine in the previous 4 years. Biologics were not reimbursed by the patient’s medical insurance. Therefore, thalidomide was administered at an initial dose of 50 mg/day in May 2016 and increased to 100 mg/day 4 weeks later, while the dose of azathioprine was unchanged. After 6 months of treatment with thalidomide, clinical and endoscopic remission was achieved on a repeated ileocolonoscopy (Figure 1c, 1d). One year after initiation of thalidomide, the patient underwent an ECG in a clinic visit and this showed sinus bradycardia with a heart rate (HR) of 44 beats/minute compared with 66 beats/minute before thalidomide use. Holter ECG showed sinus bradycardia, with an average HR of 49 beats/minutes, and the lowest HR was 34 beats/minute. Evidence of structural cardiac disease was excluded by cardiac ultrasonography. Thalidomide was withdrawn because of the low HR and a potential risk of cardiovascular events. One month after discontinuation of thalidomide, the patient showed conversion back to a normal sinus rhythm with a HR of 76 beats/minutes. However, the patient experienced disease relapse 4 months after withdrawal of thalidomide, and use of thalidomide was reinitiated (50 mg/day). One month after restarting thalidomide, the patient developed sinus bradycardia again with a HR of 45 beats/minute, as measured by surface ECG. There were no symptoms, such as palpitation, dizziness, and syncope. The patient had refractory CD and refused to use methotrexate. Therefore, the dose of thalidomide was reduced to 25 mg/day and HR increased to approximately 55 beats/minute 1 month later (Figure 2). The patient has been in clinical remission since treatment with thalidomide (25 mg/day) combined with azathioprine (2.0 mg/kg/day).
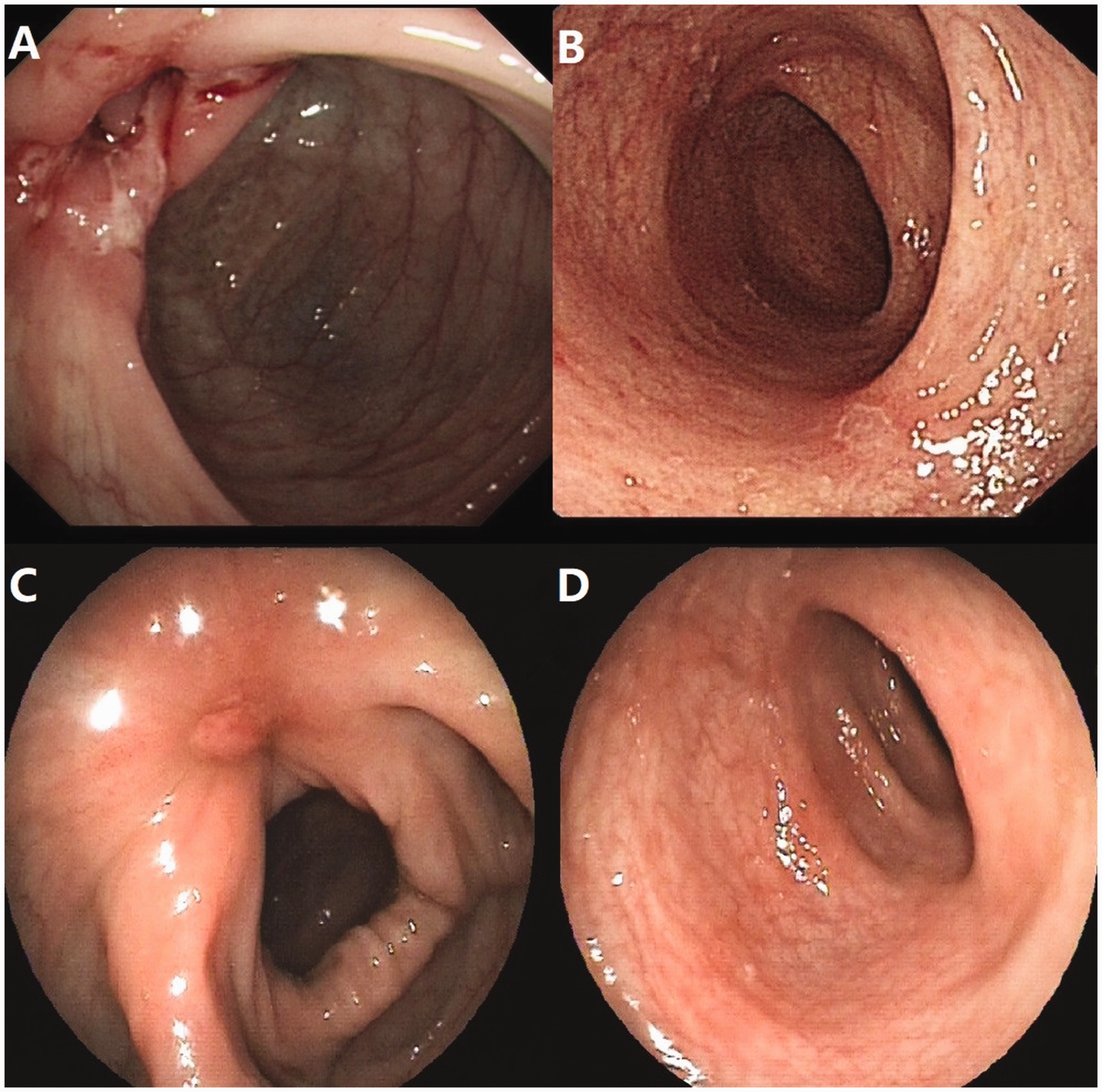
Mucosal healing induced by thalidomide in a patient with Crohn’s disease. (a) and (b) Ileocolonoscopy shows mucosal ulceration before thalidomide treatment. (c) and (d) Mucosal healing after 6 months of treatment with thalidomide.
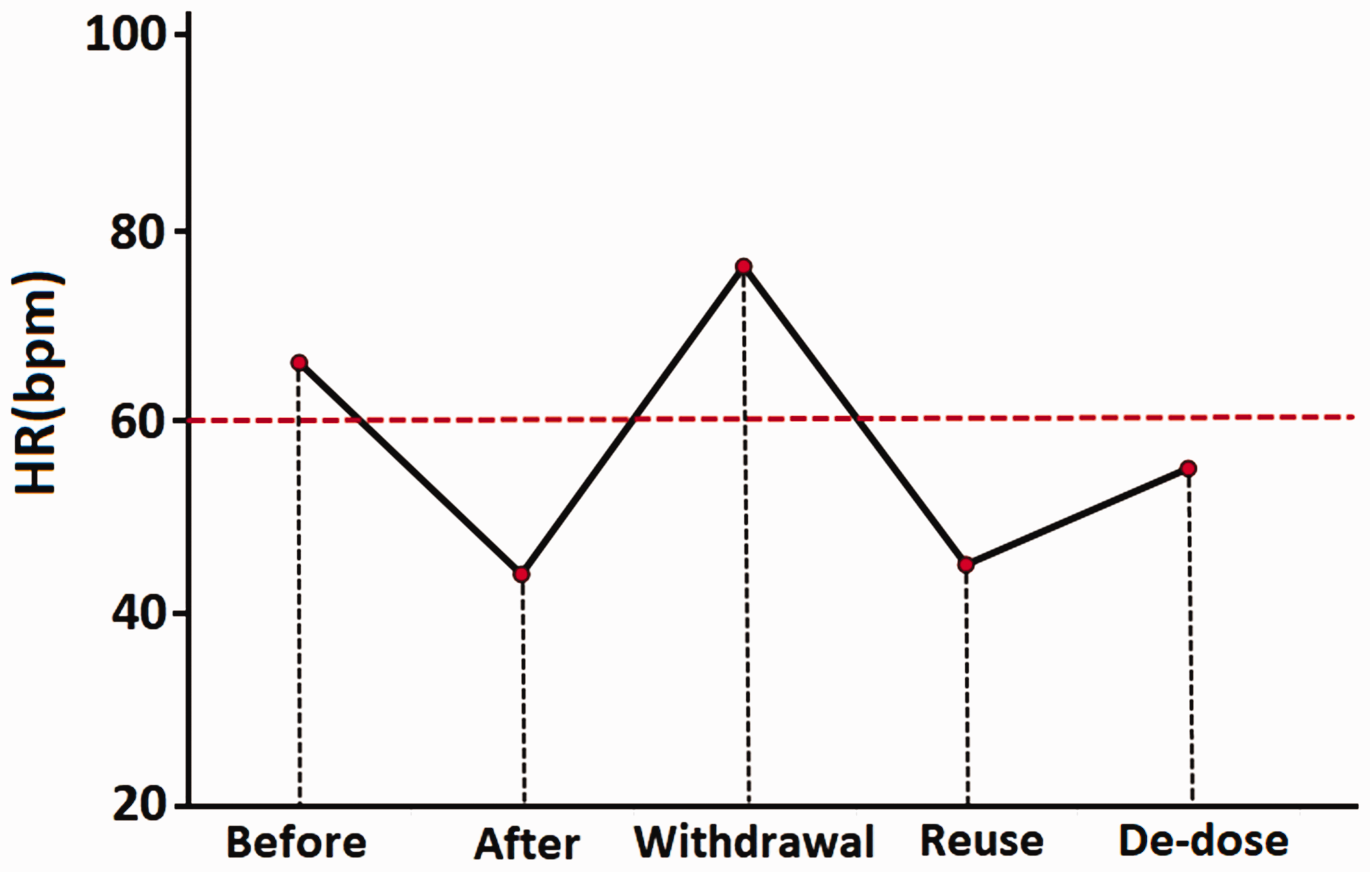
Change in the patient’s HR over the course of thalidomide use. HR: heart rate; bpm: beats per minute; de-dose: the dose of thalidomide was reduced from 50 mg/day to 25 mg/day.
Ethical permission from the local ethics committee was not necessary. Consent from the patient was obtained.
Discussion
In the present case of refractory CD, the patient suffered from sinus bradycardia after using thalidomide for 12 months. His HR converted back to a normal sinus rhythm after withdrawal of thalidomide. However, the patient developed sinus bradycardia again after restarting thalidomide. Furthermore, we found that his HR was able to partially restored while reducing the dose of thalidomide. Therefore, thalidomide-induced sinus bradycardia might have a dose-dependent effect. Additionally, clinical remission and mucosal healing were achieved by thalidomide in this patient, which is consistent with findings from previous studies.11–13,15,16
Although thalidomide-induced sinus bradycardia in a child with refractory CD was previously reported as an adverse event, 14 the potential mechanism remains unclarified. Sinus bradycardia is usually caused by autonomic nervous system dysfunction or failure of impulse generation and conduction of the sinus node. 25 Several studies have shown that some drugs, including cocaine, crizotinib, and corticosteroids, can lead to sinus bradycardia by affecting function of the sinus node or the autonomic nervous system.26–28 Most of the physiologically identified dorsal motor neurons, which are part of the nucleus of the vagus nerve, are rapidly and completely inhibited by exposure to TNF-α. 29 Infliximab and thalidomide can inhibit expression and activity of TNF-α. Infliximab can cause sinus bradycardia. 30 Therefore, sinus bradycardia induced by thalidomide might also result from its inhibitory effect on TNF-α expression and activity. This may lead to overactivity of the parasympathetic system. 21 Another potential mechanism might be associated with inhibition of the nitric oxide (NO) signaling pathway. A previous study reported that 31 the increase in HR was due to stimulation of the inward current I(f) via the NO signaling pathway. Several studies have shown that thalidomide inhibits the NO signaling pathway by reducing the activity of soluble guanylyl cyclase.32,33 Therefore, thalidomide-induced sinus bradycardia might be due to inhibition of the NO signaling pathway. Nonetheless, the precise mechanism of thalidomide-induced bradycardia in CD remains to be clarified in future studies.
In summary, we report a rare case of sinus bradycardia induced by thalidomide in an adult patient with CD and analyzed its potential mechanism. Clinical remission and mucosal healing can be potentially achieved by thalidomide in refractory CD; however, adverse events, including the risk of cardiovascular disease, should be closely monitored.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
