Abstract
Background:
Elective switching from intravenous (IV) to subcutaneous (SC) infliximab (IFX) has shown efficacy in patients with inflammatory bowel disease (IBD). However, long-term outcomes for patients not in remission remain unclear.
Objectives:
We evaluated the effectiveness of SC IFX switching in both remission and non-remission patients.
Design:
This study was a retrospective multicentre study conducted across five tertiary hospitals in Korea.
Methods:
Patients with IBD who switched to SC IFX between January 2021 and January 2023 were included. Clinical remission was defined as a Crohn’s Disease Activity Index of <150 or a partial Mayo score of <2. Biochemical remission was defined as faecal calprotectin of <250 µg/g and C-reactive protein of <0.5 mg/dL. We investigated the treatment persistence rate of SC IFX and trends in pharmacokinetics, clinical indices and biomarkers over 1 year of follow-up, analysing the data based on the baseline remission state.
Results:
Among 127 patients included, 90 (70.9%) were in clinical remission, and 37 (29.1%) were not at the time of switching. The one-year treatment persistence rate was 92.1%, with no significant difference between the clinical remission and non-remission groups (p = 0.139). Persistence was also unaffected by baseline biochemical remission status. IFX pharmacokinetics and biomarkers improved significantly in both clinical groups over 12 months (p < 0.005). Disease activity indices remained stable in the remission group and decreased in the non-remission group after switching. Previous biologics exposure was the only significant predictor of treatment persistence (hazard ratio, 5.634; 95% confidence interval, 1.357–23.384; p = 0.017). Adverse events related to SC IFX occurred in 15.7% of patients. The optimal SC IFX cutoff levels associated with clinical and biochemical remission were 11 and 17 μg/mL, respectively.
Conclusion:
Switching from IV to SC IFX during maintenance therapy demonstrated high treatment persistence and safety, irrespective of clinical and biochemical remission status.
Introduction
Inflammatory bowel diseases (IBDs), which include Crohn’s disease (CD) and ulcerative colitis (UC), are chronic disorders that can lead to gut and systemic inflammation and exhibit a relapsing-remitting course.1,2 Newer drugs, such as biologics and small molecules, have been recently introduced, and this expansion of therapeutic options has reshaped the therapeutic strategy for IBD.3–7 Among the current therapeutic options, infliximab (IFX), a monoclonal antibody targeting tumour necrosis factor-alpha, is the most effective biologic in inducing and maintaining remission in patients with IBD.8,9 However, a significant portion of patients receiving intravenous (IV) IFX experiences loss of response during maintenance treatment, with many cases attributed to immunogenicity caused by anti-drug antibodies.10–12
Recently, a new subcutaneous (SC) formulation of IFX has been introduced and widely used in clinical practice.13–16 A randomized controlled trial regarding switching from IV to SC IFX in patients with IBD demonstrated pharmacokinetic non-inferiority of SC IFX compared to IV IFX, with comparable efficacy and safety profiles. 14 Furthermore, real-world studies have highlighted the benefits of SC IFX, showing superior outcomes in terms of IFX pharmacokinetics, reduced immunogenicity and patient acceptability.17–19 Based on these findings, SC IFX has been approved in both Europe and the United States. However, most studies have focused on elective switching in patients who are in remission.17–19 Studies on switching from IV maintenance to SC IFX in patients who are not in remission are very limited to date. Given the stable serum levels provided by SC IFX, switching to SC IFX may offer additional clinical and biochemical improvements, even in non-remission patients. However, the potential benefit over a long-term period has not been reported in the literature. 20
Our study evaluated the effectiveness of switching from IV maintenance therapy to SC IFX in patients with IBD, both in remission and non-remission states at the time of SC switching. We investigated the treatment persistence rate of SC IFX and trends in pharmacokinetics, as well as clinical and biochemical indices, over 1 year of follow-up. In addition, we analysed optimal SC IFX concentration cut-offs for clinical/biochemical remission.
Methods
Study design and patients
We conducted a retrospective multicentre study involving patients who switched from IV to SC IFX at five tertiary hospitals in Korea between January 2021 and January 2023. The decision to switch to SC IFX was made through collaborative discussions between attending physicians and patients, irrespective of their disease activity at the time of the switch. Exclusion criteria were as follows (Supplemental Figure 1): (1) patients who switched to SC IFX immediately after IV induction (n = 52); (2) patients who underwent IV IFX maintenance therapy for <3 months before switching to SC IFX (n = 7); (3) patients who switched back to IV IFX after a single SC injection (for long-term trip and poor acceptance of SC formulation; n = 2) and (4) use of IFX for concurrent ankylosing spondylitis other than IBD (n = 2). All the included patients received IV IFX maintenance therapy for at least 3 months.
We classified subgroups based on disease activity indices and biochemical markers at the time of the switch. Clinical remission was defined as a Crohn’s Disease Activity Index (CDAI) of <150 for CD, and a partial Mayo score of <2 for UC. Biochemical remission was defined as faecal calprotectin (FC) of <250 µg/g and C-reactive protein (CRP) of <0.5 mg/dL.
All patients, except for only one patient lost to follow-up at 9 months, were monitored for 12 months, regardless of whether SC IFX was discontinued. The date of the SC switching was considered as ‘baseline’, and data were collected at 3, 6, 9 and 12 months after the switch. 20 We collected baseline data on patient and disease characteristics, prior exposure to other biologics, concomitant medications, clinical (CDAI and partial Mayo score) or biochemical (FC and CRP) activities and pharmacological data (serum IFX levels and anti-drug antibodies). The ‘immunomodulator (IMM) use’ was defined as the concomitant use of an IMM starting either at baseline or during the follow-up period. The serum levels of IFX and those of antibodies to IFX (ATI) were measured as described in our previous study. 20 These were quantitatively assessed using an enzyme-linked immunoassay platform: RIDASCREEN® IFX Monitoring and RIDASCREEN® Anti-IFX Antibodies (R-Biopharm AG, Darmstadt, Germany). 21 Follow-up data included CDAI, partial Mayo score, biomarkers and serum IFX levels at each specified period. In addition, we documented the details of adverse events (AEs) and IFX discontinuation. The Institutional Review Board of Asan Medical Center approved the study protocol (IRB no. 2023-0404). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 22
IV and SC IFX treatment
The patients were switched from either originator IV IFX (Remicade®; Janssen Biotech, Inc., Horsham, PA, USA) or biosimilar IV CT-P13 (Remsima®; Celltrion Inc., Incheon, Republic of Korea) to SC IFX (CT-P13 SC). Patients with CD maintained a standard dose of 5 mg/kg IV IFX every 8 weeks or received an intensified dose of 10 mg/kg IV IFX every 8 weeks if they experienced a loss of response on the standard dose. In such cases, shortening the dosing interval was not permitted. Patients with UC were not permitted dose escalation with IV IFX in Korea, and they received 5 mg/kg of IV IFX every 8 weeks as maintenance therapy. All the patients who switched to SC IFX received 120 mg injections every other week. At each visit, physicians confirmed adherence to the SC injection schedule and monitored for any AEs.
Outcomes
The primary outcome was 1-year treatment persistence of SC IFX. We assessed differences in treatment persistence among subgroups based on baseline characteristics and identified clinical variables associated with treatment discontinuation. We compared serum IFX levels according to remission state during SC IFX therapy and determined optimal SC IFX concentration thresholds associated with remission.
Statistical analysis
Treatment persistence was estimated using the Kaplan–Meier method, and subgroup comparisons were made using the log-rank test. We performed Cox proportional hazards regression analysis to assess variables associated with treatment persistence. The multivariable Cox analysis included variables that were effective in the univariable Cox analysis (p-value < 0.1) or clinically meaningful. The Wilcoxon signed-rank test was used to assess the changes in pharmacokinetics, disease activity index and biomarkers from baseline to 12 months. The optimal thresholds of SC IFX concentrations to predict the remissions were identified using receiver operating characteristic (ROC) curves based on the Youden index. A two-sided p-value of <0.05 was considered statistically significant. All analyses were performed using IBM SPSS statistics 21.0 for Windows (IBM Corp., Armonk, NY, USA).
Results
Baseline characteristics
A total of 127 patients with IBD (CD, n = 80; UC, n = 47) were included. The cohort consisted of patients with a mean age of 35.5 years, of which 88 (69.3%) were men. Among the patients with CD, 39 (48.8%) were using the standard dose of IV IFX, and 41 (51.3%) were using an intensifying dose of IV IFX. All the patients with UC were maintained on the standard dose of IV IFX, as mentioned above. At baseline, more than half of the patients (n = 74, 58.3%) were receiving concomitant IMM therapy. Of these, 65 (87.8%) patients continued IMM throughout the 12 months, while 9 discontinued IMM at a median of 9 months of treatment. One patient initiated IMM at week 12 due to elevated FC levels and continued its use until 12 months. Consequently, a total of 75 patients were included in the ‘IMM combination’ group. Ten patients (7.9%) were exposed to other biologics before the IFX therapy. The median IFX trough level at baseline was 3.3 μg/mL (interquartile range (IQR), 1.3–5.1 μg/mL). ATI testing was conducted in only 8 out of 127 patients, with 3 identified as ATI positive (CD: 1/3, UC: 2/5). The baseline characteristics of included patients are listed in Table 1.
Baseline characteristics of patients switched to subcutaneous IFX.

Under the healthcare policy of Korea, patients with UC were not permitted dose escalation with IV IFX.
5-ASA, 5-aminosalicylate; BMI, body mass index; CD, Crohn’s disease; CDAI, Crohn’s Disease Activity Index; CRP, C-reactive protein; IFX, infliximab; IQR, interquartile range; IV, intravenous; MTX, methotrexate; n, number; NSNP, non-stricturing and non-penetrating; SD, standard deviation; UC, ulcerative colitis.
When classified based on disease activity indices, 90 (70.9%) were in clinical remission at the time of the switch, and 37 (29.1%) were in a non-remission state. In the non-remission group, the baseline disease activity was a mean CDAI of 221.1 for CD and a mean partial Mayo score of 4.0 for UC (Table 1). According to biochemical markers at baseline, 61 (48.0%) patients were classified as in biochemical remission, while 66 (52.0%) were categorized as in biochemical non-remission.
Treatment persistence
The 1-year treatment persistence rate was high at 92.1%, with 10 patients discontinuing treatment during the follow-up period. The median treatment duration for those who discontinued SC IFX was 7.3 months (IQR, 6.4–8.6 months). The reasons for discontinuation are summarized in Supplemental Table 1. Notably, the discontinuation group included all three ATI-positive patients, of whom two discontinued due to worsening disease activity and one due to severe localized injection site reaction (ISR). These patients consistently exhibited serum IFX concentrations below 1 µg/mL throughout the follow-up period. In addition, one other patient developed prostate cancer and discontinued SC IFX approximately 6 months after switching despite maintaining stable disease activity.
Figure 1 shows a comparison of treatment persistence for each subgroup based on the baseline characteristics. There was no significant difference between the clinical remission and non-remission group (94.4% vs 86.5%, p = 0.139). Baseline biochemical remission status also had no significant impact on SC IFX persistence (95.1% vs 89.4%, p = 0.239). Patients with UC had significantly lower treatment persistence than those with CD (85.1% vs 96.3%, p = 0.020). Patients with previous biologics exposure also showed significantly lower persistence compared to those without exposure (94.9% vs 60.0%, p < 0.001). Considering that there were relatively more bio-exposed patients in the UC group (n = 7) compared to the CD group (n = 3), we compared persistence rates only among bio-naïve patients, and no significant difference was noted between the UC and CD groups (92.5% vs 96.1%, p = 0.379). Intensified dose of IV IFX and concurrent IMM use showed no significant effect on the persistence of SC IFX.

Kaplan–Meier curves for treatment persistence with subcutaneous infliximab. (a) Patients with clinical remission and non-remission states at baseline. (b) Patients with biochemical remission and non-remission states at baseline. (c) Patients with CD and UC. (d) Biologics-naïve and biologics-experienced patients. (e) Intravenous standard dose and intensifying dose in patients with CD. (f) IMM combination therapy and monotherapy.
Predictors of treatment persistence of SC IFX
We constructed a Cox proportional hazards regression model to assess the effect of baseline clinical and biochemical variables on treatment persistence. In the univariable analysis, previous surgery, biologics exposure, higher CRP levels, lower albumin levels and lower IFX trough levels at baseline were identified as significant factors of treatment persistence (p < 0.1). Concomitant use of IMM was not a significant factor associated with treatment persistence. In the multivariable analysis, biologics exposure before IFX administration was the only significant risk factor associated with treatment discontinuation (hazard ratio, 5.634; 95% confidence interval, 1.357–23.384; p = 0.017; Table 2).
Risk factors associated with the discontinuation of subcutaneous IFX.
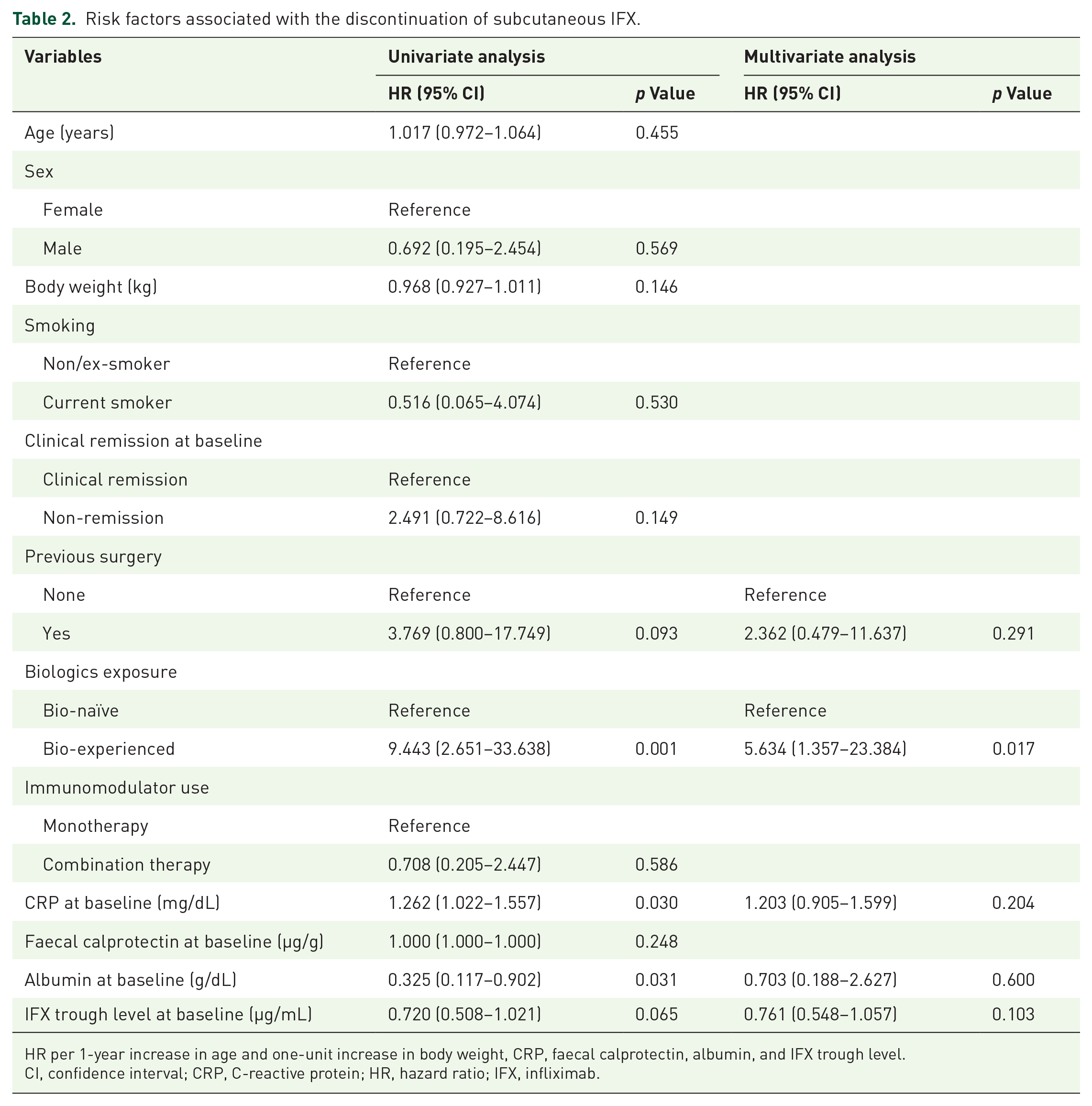
HR per 1-year increase in age and one-unit increase in body weight, CRP, faecal calprotectin, albumin, and IFX trough level.
CI, confidence interval; CRP, C-reactive protein; HR, hazard ratio; IFX, infliximab.
Trends in serum IFX levels, disease activity index and biomarkers
We divided patients into clinical remission and non-remission groups and analysed trends in variables during the follow-up period (Figure 2 and Supplemental Tables 2 and 3). In both the groups, IFX pharmacokinetics significantly improved from baseline to 12 months (clinical remission group: mean 4.6 to 18.4 μg/mL; non-remission group: mean 2.2 to 15.0 μg/mL). Similarly, biochemical remission and non-remission groups also showed significant improvement in IFX pharmacokinetics. Serum IFX levels during the study period were not affected by concomitant IMM use or IV IFX dosing (Supplemental Table 2). The disease activity index (CDAI and partial Mayo score) remained stable in the clinical remission group and significantly decreased in the non-remission group. Supplemental Figure 2 illustrates the individual changes in activity scores during the follow-up period for patients in the non-remission group. Notably, biomarkers showed significant improvement in both groups (Supplemental Table 3).

Trends after switching to subcutaneous IFX. (a) Serum IFX levels according to baseline clinical remission states. (b) Serum IFX levels according to baseline biochemical remission states. (c) CDAI in patients with Crohn’s disease according to baseline clinical remission states. (d) Partial Mayo scores in patients with ulcerative colitis according to baseline clinical remission states. (e) C-reactive protein levels according to baseline clinical remission states. (f) Faecal calprotectin levels according to baseline clinical remission states.
Changes in remission states after switching to SC IFX
We analysed the remission rates during the follow-up period based on the baseline remission state (Figure 3). This analysis included 115 patients with all available measurements at baseline and during the follow-up period. In terms of clinical remission, 90.0% of patients in clinical remission at baseline remained in remission at 12 months, while 54.3% of those not in remission at baseline achieved remission by 12 months. In terms of biochemical remission, 85.7% of patients in biochemical remission at baseline maintained remission at 12 months, and 40.7% of those not in biochemical remission at baseline achieved it by 12 months. Composite clinical and biochemical remission rates after SC switching are also described in Figure 3(c). During the follow-up period, only six patients (4.7%) initiated new corticosteroid use. Among them, two patients discontinued SC IFX due to worsening disease activity despite corticosteroid use, while the remaining four patients continued SC IFX use after showing improvement in disease activity. The initiation of corticosteroids during the follow-up did not significantly affect clinical/biochemical remission rates.

Remission rates during the follow-up period. (a) The proportion of patients with clinical remission according to baseline clinical remission states. (b) The proportion of patients with biochemical remission according to baseline biochemical remission states. (c) The proportion of patients with composite clinical and biochemical remission according to baseline composite remission states.
SC IFX concentration cut-off associated with remission
Serum SC IFX concentrations at the last follow-up according to the presence or absence of clinical remission, biochemical remission and composite clinical and biochemical remission are reported in Supplemental Figure 3. Mean serum SC IFX concentrations were numerically higher in patients with clinical remission than those in patients without clinical remission (17.9 vs 13.4 μg/mL, p = 0.123). SC IFX concentrations were significantly higher in patients with biochemical remission (19.3 vs 13.8 μg/mL, p = 0.011) and composite clinical and biochemical remission (19.7 vs 14.1 μg/mL, p = 0.006) than those in patients who did not achieve these outcomes.
The optimal SC IFX concentration cut-off associated with therapeutic outcomes are summarized in Table 3. Based on the ROC curve analysis, the thresholds for clinical remission were 11 μg/mL, while those for biochemical and composite remissions were 17 μg/mL each.
Serum IFX concentration cut-offs associated with clinical, biochemical and composite remissions at the last follow-up.
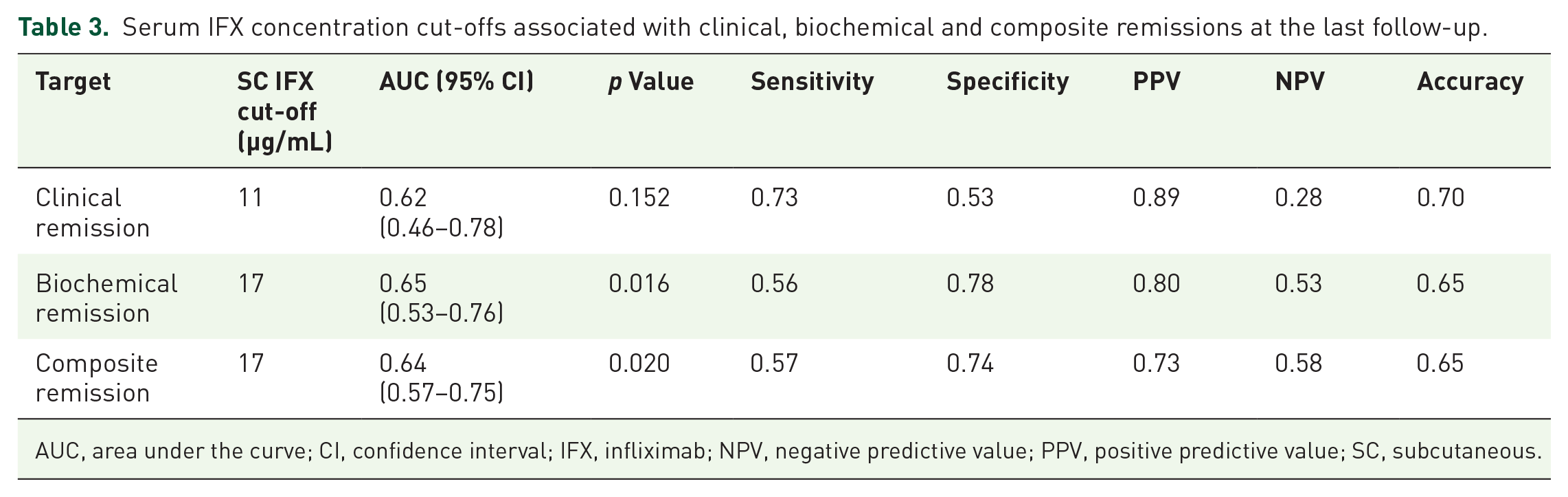
AUC, area under the curve; CI, confidence interval; IFX, infliximab; NPV, negative predictive value; PPV, positive predictive value; SC, subcutaneous.
Safety outcomes
The incidence of SC-IFX-related AE was 15.7% (20 of 127). Localized ISR was the most common AE (n = 15). Among them, one patient experienced severe symptoms leading to treatment discontinuation (ATI positive), while the remaining 14 had mild symptoms and continued SC IFX. Infections occurred in three cases, with one patient discontinuing treatment due to recurrent acute pyelonephritis, one with herpes zoster infection and one with urinary tract infection. Psoriatic skin lesions were observed in two patients (1.6%). During the follow-up period, there were no serious AEs, such as anaphylaxis, severe sepsis episodes or death.
Discussion
This study assessed the long-term outcomes of switching from IV to SC IFX maintenance therapy in patients with IBD, both in remission and non-remission states. The 1-year persistence rate of SC IFX was high, with no significant difference based on the initial remission state. Only exposure to biologics prior to IFX was a factor associated with treatment discontinuation. During the follow-up period, IFX pharmacokinetics significantly improved regardless of the baseline remission state. The disease activity index remained stable in the clinical remission group and showed significant improvement in the non-remission group. To our knowledge, this is the first study to analyse these long-term outcomes based on remission status at the time of switch.
In our study, the 1-year treatment persistence rate of SC IFX was 92.1%, with only 10 of 127 patients discontinuing treatment. Our findings are consistent with a previous multicentre study that reported a 1-year SC IFX persistence rate of 92% in patients with IBD. 17 It is noteworthy that the previous study primarily included patients in remission at the time of the switch, whereas our study included both remission and non-remission patients, yet showed similar persistence rates. Although patients in non-remission exhibited numerically lower persistence rates compared to those who switched while in remission, the log-rank test did not show a statistically significant difference: the persistence rates for clinical and biochemical non-remission groups remain high at 86.5% and 89.4%, respectively.
Most of the previous real-world studies on SC IFX have focused on the outcomes of patients who were maintaining remission with IV IFX.17,18,23,24 These studies have reported superior pharmacokinetics of SC IFX, as well as efficacy and safety.17,18,24 In our study, consistent with existing knowledge,17,18,24 a significant increase in serum IFX concentration was observed in the clinical remission group after switching to SC IFX, along with stable maintenance of clinical activity index. In the non-remission group, IFX pharmacokinetics were also significantly increased after switching. Interestingly, significant reductions in disease activity index and biomarkers were observed in non-remission patients with both UC and CD. Regarding therapeutic outcomes during the follow-up period, approximately half of those in the non-remission group attained new remission at 12 months. These results suggest that SC IFX switching from a non-remission state leads to improvements in pharmacokinetics and allows for the acquisition and maintenance of therapeutic outcomes.
SC IFX demonstrated enhanced pharmacokinetic properties and lower immunogenicity compared to IV IFX, showing potential as ‘Biobetters’ that are equivalent to or better than existing biologics.25,26 From this perspective, there is increasing interest in the role of SC IFX in difficult-to-treat IBD. A case series explored the effectiveness of switching to SC IFX in four patients with CD who experienced IV IFX failure. 27 Similarly, we previously reported the positive efficacy of SC IFX at 6 months in patients with UC who experienced IV IFX failure. 20 Unlike the previous studies, our study has the advantage of analysing long-term outcomes in patients with both UC and CD.
In this study, biologics exposure prior to IFX was a significant factor associated with treatment discontinuation in multivariable analysis. The treatment persistence rates of patients with biologics exposure were 80% and 60% at 6 and 12 months, respectively. Similarly, Cerna et al. 26 reported the effectiveness of SC IFX in 32 patients with refractory CD previously unsuccessfully treated with at least 2 biologics. They observed a significant increase in IFX trough levels and a decrease in ATI levels following SC IFX administration, although the midterm treatment persistence at 30 weeks was 53%. Based on these results, prior biologics exposure can be considered a major factor contributing to the discontinuation of SC IFX. However, as our study was retrospective and the number of patients in the bio-experienced group was small, these findings should be interpreted with caution; further prospective research with large samples is warranted. On the other hand, the intensified IV IFX regimen did not affect the treatment persistence of SC IFX in patients with CD in this study. There are several studies on SC IFX switching from intensified IV IFX.18,24,28 The REMSWITCH-LT study explored the long-term effectiveness and acceptability of switching from IV to SC IFX in patients with IBD treated with or without an intensified IV regimen. 28 Interestingly, rates of relapse were 13.8% and 18.4% at the last follow-up in those receiving 5 and 10 mg/kg/8 weeks at baseline, respectively. By contrast, among patients with the most intensified regimen (10 mg/kg/4 weeks), 86.7% of them experienced relapse at the last follow-up. In a multicentre Spanish study, patients receiving standard and intensified dosages of IV IFX exhibited high 6-month treatment persistence rates of 95% and 100%, respectively. 24
It is known that good therapeutic outcomes in SC IFX are attributed to higher serum IFX levels.29,30 Robin et al. demonstrated that more stringent outcomes are associated with higher SC IFX concentration thresholds. They proposed thresholds of 12 μg/mL for sustained clinical remission, 16 μg/mL for composite clinical and biomarker remission and 20 μg/mL for deep remission. 29 The REMSWITCH study recommended SC IFX trough levels exceeding 11 μg/mL for sustained response. 18 In our study, we observed similar results for clinical, biochemical and composite clinical and biochemical remissions, with threshold values of 11, 17 and 17 μg/mL, respectively. However, the overall modest AUC with relatively low corresponding sensitivity and specificity values suggests a need for prospective multicentre studies to validate these findings. It is known that IFX concentration increases 3–7 times when switching from IV to SC administration, likely due to different pharmacokinetic mechanism.14,31 SC IFX pharmacokinetics remain high and stable throughout the treatment cycles, suggesting that it may exhibit different effects compared to the IV IFX. 32
The incidence of SC IFX-related AEs in this study was 15.7% (20 of 127). Among these, localized ISR was the most common, occurring in 15 individuals, consistent with previous studies. 17 Only 3 of the 20 patients discontinued treatment due to AEs. According to the pivotal randomized controlled trial, the occurrence of AEs related to IFX, excluding localized ISRs, was comparable between SC and IV IFX groups. 14 Additionally, real-world studies showed a low incidence of AEs that is <10%, and the rate of treatment discontinuation due to AEs was also low.17,18,24
Our study has several limitations. First, this study was a retrospective design, resulting in the lack of data on potentially relevant variables such as ATI and endoscopic findings. In particular, ATI measurements were conducted in a limited number of patients, limiting our ability to evaluate ATI effects on treatment persistence. Second, the non-remission group in this study primarily included patients with moderate CDAI and partial Mayo scores, with mean baseline scores of 221.1 and 4.0, respectively. Therefore, there are limitations in evaluating the effectiveness of SC switching in severely active cases. Third, in the context of our study conducted in Korea, IV IFX dose intensification is permitted only for CD, and not UC. Consequently, all patients with UC received standard IV IFX, potentially introducing differences in characteristics and results between CD and UC.
Conclusion
In conclusion, switching from IV to SC IFX during maintenance therapy in patients with IBD demonstrated high long-term treatment persistence and safety irrespective of baseline remission status. SC IFX could be a reasonable treatment option, as it shows high efficacy not only in clinical and/or biochemical remission but also in non-remission patients.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251333516 – Supplemental material for Comparative efficacy of subcutaneous infliximab switching in remission and non-remission patients with inflammatory bowel disease after intravenous maintenance: 1-year outcome from a multicentre cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848251333516 for Comparative efficacy of subcutaneous infliximab switching in remission and non-remission patients with inflammatory bowel disease after intravenous maintenance: 1-year outcome from a multicentre cohort study by June Hwa Bae, Yoo Jin Lee, Jung-Bin Park, Ji Eun Baek, Seung Wook Hong, Sang Hyoung Park, Dong-Hoon Yang, Byong Duk Ye, Jeong-Sik Byeon, Seung-Jae Myung, Suk-Kyun Yang, Kyeong Ok Kim, Byung Ik Jang, Eun Soo Kim, Hyeong Ho Jo, Eun Young Kim and Sung Wook Hwang in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
None.
Author’s note
Preliminary results of this study were presented as a poster at the European Crohn’s and Colitis Organisation (ECCO) Congress, Stockholm, Sweden, January 2024.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
