Abstract
Background:
Gastrointestinal bleeding (GIB) is a critical clinical emergency associated with high morbidity and mortality. The widespread use of antithrombotic agents, including antiplatelet and anticoagulant medications, has increased the incidence of GIB.
Objectives:
Our study aims to address this gap by evaluating the impact of antithrombotic therapy on both 28-day mortality and rebleeding risk.
Design:
Retrospective cohort study using propensity score-based methods to address confounding.
Methods:
Data were extracted from three independent databases (MIMIC-IV, NWICU, and Xuanwu Hospital) spanning 2008–2022. inverse probability of treatment weighting (IPTW) was applied to balance baseline characteristics. Weighted logistic regression models assessed outcomes across antiplatelet, anticoagulant, and combination therapy subgroups.
Results:
After inverse probability of treatment weighting (IPTW) adjustment, the antithrombotic group maintained a significantly elevated rebleeding rate (19.9% vs 10.5%, p < 0.001) and an increased risk of rebleeding (odds ratio (OR) = 2.118, 95% confidence interval (CI): 1.577–2.845, p < 0.001). Conversely, the 28-day mortality was significantly lower in the antithrombotic group postadjustment (8.2% vs 12.5%, p = 0.022; OR = 0.621, 95% CI: 0.412–0.935, p = 0.023). Notably, early resumption of antithrombotic therapy (within 3 days) significantly increased the risk of mortality.
Conclusion:
Our study suggests that while antithrombotic therapy reduces 28-day mortality, it significantly increases rebleeding risk. Notably, the use of anticoagulants or combination therapy is linked to the highest rebleeding risk, compared to antiplatelets. Additionally, resuming antithrombotic therapy too early (i.e., within 3 days) may further elevate the risk of mortality.
Introduction
Gastrointestinal bleeding (GIB) is a common and potentially life-threatening emergency that poses significant challenges in clinical practice.1,2 The use of antithrombotic agents, including antiplatelets and anticoagulants, is increasing. These medications help prevent cardiovascular events and stroke. However, they also elevate the risk of GIB. 3 While these medications play a critical role in preventing cardiovascular events and stroke, they simultaneously elevate the risk of bleeding within the gastrointestinal tract. 4
When GIB occurs in patients on antithrombotic therapy, clinicians face a complex dilemma: continuing the medication may precipitate rebleeding, whereas discontinuation can increase the risk of thrombotic complications. 5 Especially in elderly patients, the increased incidence of comorbidities and drug interactions related to multiple medications may increase the risk of rebleeding. 6 Moreover, current evidence is largely based on single-center studies with limited sample sizes and often focuses on upper GIB, leaving many aspects of antithrombotic management in GIB unresolved.5,7
Nevertheless, some studies advocate a cautious approach to early resumption of antithrombotic therapy.8,9 Moreover, the effects of antiplatelet agents versus anticoagulants in the context of GIB may differ, and the optimal management strategy for various types of antithrombotic drugs remains to be fully elucidated. 10 Our study aims to address this gap by evaluating the impact of antithrombotic therapy on both 28-day mortality and rebleeding risk in a large, multicenter cohort of GIB patients. We specifically investigate the differential effects of antiplatelet agents, anticoagulants, and their combination, as well as the optimal timing for resuming these therapies.
Methods
Study design
This retrospective, multicenter cohort study used data from MIMIC-IV (2008–2022), NWICU (2020–2022), and Xuanwu Hospital (2017–2022). All three queues are collectively enrolling. The study adhered to the Declaration of Helsinki and obtained ethical approval from institutional review boards.11,12 The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 13
Inclusion and exclusion criteria
The study included patients aged ⩾18 years with a confirmed diagnosis of GIB (based on ICD-9/10 coding, Supplemental Material 1). The exclusion criteria were as follows: (1) Patients with a history of malignancy, including solid tumors of the gastrointestinal, hepatobiliary, respiratory, or neurological systems, as well as hematological malignancies such as lymphoma and leukemia. (2) Patients with other conditions, including HIV infection, congenital immunodeficiency, organ transplant recipients, and individuals with a history of drug abuse. (3) Patients who died within 24 h of admission.
Study variables
The primary exposure variable is the use of antithrombotic medications after admission, which includes antiplatelet agents (such as aspirin and clopidogrel), anticoagulants (such as warfarin and dabigatran), or their combination (Supplemental Material 2). The outcome variables are 28-day mortality and the risk of rebleeding. Rebleeding is defined as a decrease of ⩾2 g/L compared to admission. Fully adjusted model, including all relevant covariates (gender, age, myocardial infarct, CHD, cerebrovascular disease (CVD), hypertension, diabetes, renal disease, chronic pulmonary diseases (CPD), liver disease, white blood cell (WBC), hemoglobin, platelets, blood urea nitrogen (BUN), aspartate transaminase (AST), total bilirubin, alkaline phosphatase (ALP), creatinine, albumin, calcium, temperature, heart rate, systolic blood pressure (SBP), respiratory rate, location, group (different centers)).
Statistical analysis
Statistical analyses were performed using R software. Categorical variables were compared using the Chi-square test or Fisher’s exact test, while continuous variables were analyzed using the t test or Mann–Whitney U test. Based on its flexibility in handling continuous exposure variables and its ability to handle nonoverlapping areas of the sample, 14 this study chose inverse probability of treatment weighting (IPTW; link = logit) instead of stratification or matching methods, ensuring a standardized mean difference (SMD) <0.1. Validate the inverse probability weighted results using Love Plot and Propensity Score Distribution Plot.
A multivariable weighted logistic regression model assessed the impact of antithrombotic therapy on mortality and rebleeding risk. Robust standard errors were calculated using 500 bootstrap samples, and results were reported as odds ratios (ORs) with 95% confidence intervals (CIs). Model fitting was conducted using a quasi-binomial family to account for the weighted study design. Missing data have not been interpolated and have been processed for deletion.
Sensitivity analysis and robustness assessment
Sensitivity analyses were performed by extreme weights trimming (EWT) at the 1st–99th and 2nd–98th percentiles to reduce the potential instability associated with the IPTW method. Additional sensitivity analyses were conducted by fitting models adjusted for various covariates. Furthermore, the distribution of propensity scores was examined to assess the degree of common support between the exposed and control groups, identify any extreme values or nonoverlapping regions, and confirm that weighting improved the balance of covariates.
Results
Baseline characteristics
A total of 5532 patients with GIB were initially screened (Supplemental Figure 1), of whom 951 were excluded based on the predefined criteria. Ultimately, 4581 patients were included in the final analysis, with 1409 receiving antithrombotic therapy and 3172 not receiving such treatment. To reduce baseline imbalances between groups, IPTW was applied. Before weighting, patients in the antithrombotic group were significantly older, and they had higher incidences of myocardial infarction, congestive heart failure, CVD, and diabetes compared to those not on antithrombotic therapy. Moreover, renal and CPD were more prevalent in the antithrombotic group, whereas liver disease was more common in the nonantithrombotic group. Laboratory findings revealed that the antithrombotic group had higher white blood cell (WBC) counts, while hemoglobin levels were similar between groups; platelet counts, and BUN levels were also elevated in the antithrombotic group. Additionally, significant differences were observed in the bleeding sites between the two groups. After IPTW adjustment, the baseline characteristics were well balanced, with SMDs <0.1 for all variables (Table 1 and Figure 1). The distribution of propensity scores (Figure 2) confirmed that IPTW effectively improved covariate balance.
Baseline characteristics of patients with gastrointestinal bleeding.

ALP, alkaline phosphatase; AST, aspartate transaminase; BUN, blood urea nitrogen; CHF, congestive heart failure; CPD, chronic pulmonary disease; CVD, cerebrovascular disease; GCS, Glasgow coma scale; INR, international normalized ratio; PVD, peripheral vascular disease; SBP, systolic blood pressure; WBC, White blood cell count.
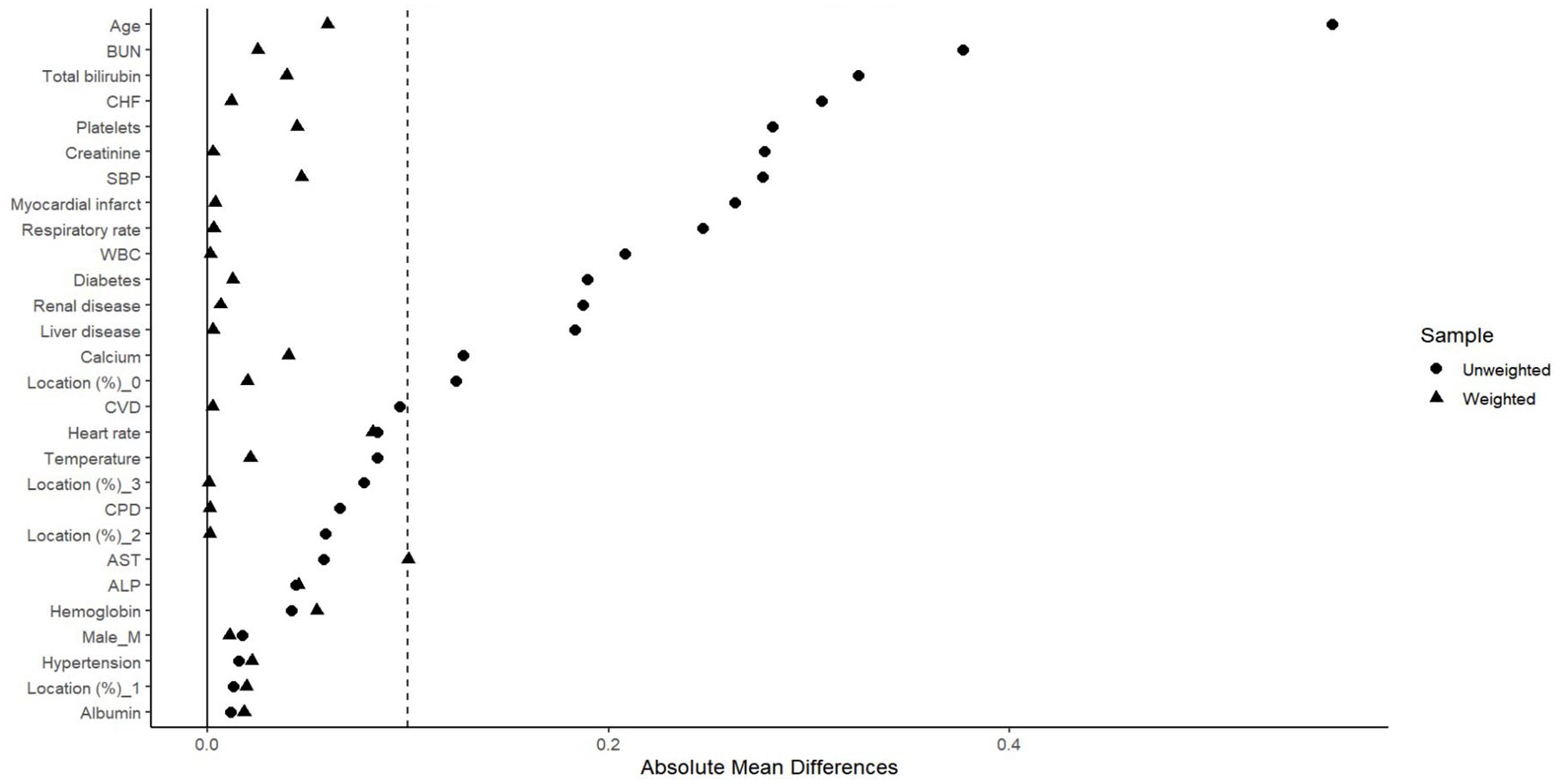
Love plot: covariate balance before and after weighting.

Propensity score distribution: unweighted versus weighted.
Prognosis
Before IPTW adjustment, the rebleeding rate was significantly higher in the antithrombotic group compared to the nonantithrombotic group (22.0% vs 9.7%, p < 0.001; Table 2). Following IPTW adjustment, the rebleeding rate remained significantly elevated in the antithrombotic group (19.9% vs 10.5%, p < 0.001; Table 2). Weighted logistic regression analysis confirmed that antithrombotic therapy was associated with an increased risk of rebleeding (OR = 2.118, 95% CI: 1.577–2.845, p < 0.001; Table 3). This association persisted in sensitivity analyses employing different EWT thresholds (1%–99% and 2%–98%) and adjusting for multiple confounders (OR range: 2.12–2.92, all p < 0.05). In contrast, before the IPTW adjustment, the 28-day mortality rates did not differ significantly between the antithrombotic and nonantithrombotic groups (10.9% vs 11.3%, p = 0.732; Table 2). However, after IPTW adjustment, the 28-day mortality rate in the antithrombotic group was significantly lower than that in the nonantithrombotic group (8.2% vs 12.5%, p = 0.022; Table 2). This reduction was further supported by weighted logistic regression analysis (OR = 0.621, 95% CI: 0.412–0.935, p = 0.023; Table 3). Sensitivity analyses, which included various weight trimming strategies and additional covariate adjustments, consistently demonstrated a reduction in mortality risk (OR range: 0.55–0.64, all p < 0.05).
Clinical outcomes of patients with gastrointestinal bleeding.

Weighted logistic regression analysis of antithrombotic therapy on 28-day mortality and rebleeding.

Model 1: Antithrombotic therapy only. Model 2: Adjusted for group (different centers). Model 3: Adjusted for bleeding location. Model 4: Adjusted for age, sex, group, and bleeding location. Model 5: Fully adjusted model, including all relevant covariates.
CI, confidence interval; EWT, extreme weights trimming; OR, odds ratio.
Subgroup analyses
Subgroup analysis based on the bleeding site (Table 4 presents part, Supplemental Material 3 presents whole) indicated that while antithrombotic therapy was negatively associated with 28-day mortality, the differences did not reach statistical significance (p > 0.05; due to limited sample sizes in certain subgroups, EWT was not performed). Furthermore, subgroup analyses showed that, in patients with nonvariceal upper GIB and unknown/middle GIB, antithrombotic therapy significantly increased the risk of rebleeding (p < 0.05). In contrast, in patients with variceal upper gastrointestinal bleeding and lower GIB, the association between antithrombotic therapy and rebleeding risk was positive, but not statistically significant (p > 0.05).
Subgroup analysis on 28-day mortality and rebleeding.
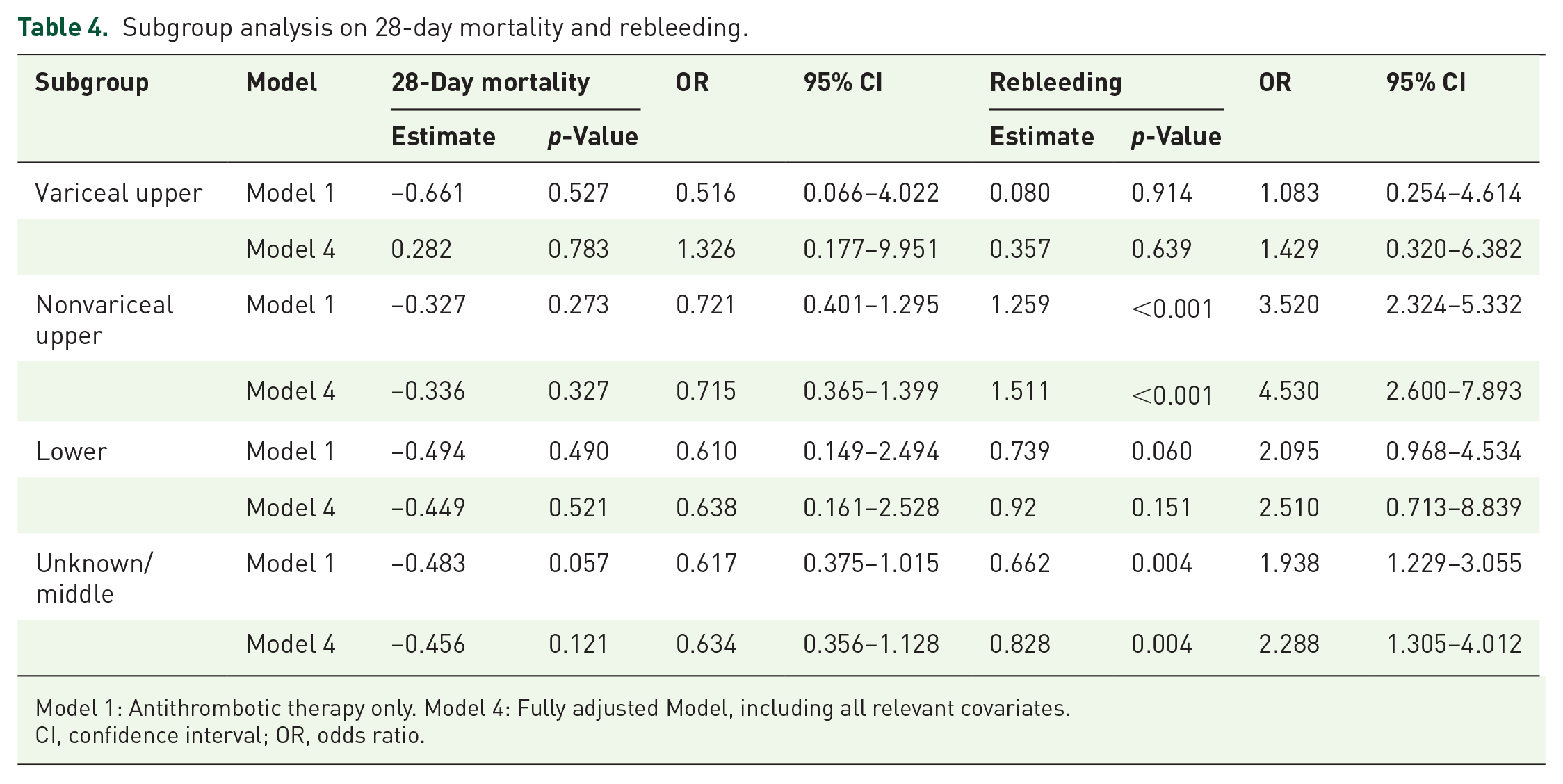
Model 1: Antithrombotic therapy only. Model 4: Fully adjusted Model, including all relevant covariates.
CI, confidence interval; OR, odds ratio.
Impact of resumption timing on outcomes
An additional analysis examined the effect of the timing of antithrombotic therapy resumption on patient outcomes (Table 5). In the subgroup of 1409 patients receiving antithrombotic medications, the mean time to resumption was 3.15 days. Patients were divided into two groups based on whether therapy was resumed in ⩾3 or <3 days. After adjusting for baseline differences by IPTW and ensuring the robustness of the results through EWT, it was found that early resumption (<3 days) did not significantly increase the risk of rebleeding (p > 0.05). However, early resumption was associated with a 2.9- to 4.2-fold increase in mortality risk (p < 0.05).
Impact of antithrombotic resumption within 3 days on 28-day mortality and rebleeding.

Model 1: Antithrombotic therapy only. Model 2: Adjusted for group (different centers). Model 3: Adjusted for bleeding location. Model 4: Adjusted for age, sex, group, and bleeding location. Model 5: Fully adjusted model, including all relevant covariates.
CI, confidence interval; OR, odds ratio.
Effects of different antithrombotic regimens
Finally, the study evaluated the impact of different antithrombotic regimens on the prognosis of GIB patients. The analysis categorized patients into three groups: 1082 patients (23.6%) received antiplatelet agents, 191 patients (4.2%) received anticoagulants, and 136 patients (3.0%) received combination therapy (antiplatelet plus anticoagulant). A weighted logistic regression model, adjusted by IPTW, was used to assess the effects on 28-day mortality and rebleeding risk (Table 6 presents part, Supplemental Material 4 presents whole). In terms of 28 day mortality, neither antiplatelet nor anticoagulant monotherapy showed statistically significant differences, but the effect values were consistent with the main analysis direction (OR < 1). In terms of the risk of rebleeding, all treatment types significantly increased the risk: antiplatelet drugs had the lowest risk (Model 6 OR = 2.036, 95% CI: 1.389–3.169, p < 0.001), followed by anticoagulants (OR = 2.983, 95% CI: 1.503–6.027, p = 0.001), and combination therapy had the highest risk (OR = 6.088, 95% CI: 2.076–25.175, p = 0.001). It is worth noting that the death protection effect of anticoagulants disappears after adjustment, and the wide CI of combination therapy suggests that sample size limitations may affect estimation accuracy.
Effect of different types of antithrombotic agents on 28-day mortality and rebleeding.

Model 1: Antithrombotic therapy only. Model 6: Fully adjusted model, including all relevant covariates.
CI, confidence interval; OR, odds ratio.
Discussion
Most current studies and guidelines advocate a proactive approach to antithrombotic therapy, recommending the use of antiplatelet agents.15 –17 However, much of the supporting evidence originates from single-center studies with small sample sizes and largely focuses on patients with upper GIB. 18 In contrast, a recent multicenter study from China—including 2299 patients (with 20.1% receiving antiplatelets and 2.9% receiving anticoagulants)—reported an association between antiplatelet or anticoagulant therapy and increased 28-day mortality in GIB patients. 9
In our study, we integrated data from the MIMIC-IV database, the NWICU database, and clinical records from Xuanwu Hospital. This multicenter approach, which encompasses hospitalized patients from various countries and regions and covers a range of bleeding sites, renders our findings more representative. Moreover, we employed the IPTW method to achieve balanced baseline characteristics, thereby reducing the selection bias inherent in observational research. Multiple sensitivity analyses—using different EWT strategies and adjusting for various confounders—confirmed the robustness of our main findings. Overall, the primary analysis revealed that, among patients with GIB, the use of antithrombotic therapy is associated with a reduction in 28-day mortality, albeit at the cost of increased rebleeding risk.
Notably, our study was the first to differentiate between the effects of antiplatelet agents, anticoagulants, and their combination on the prognosis of GIB patients and to quantify the risks associated with each regimen. This approach provides more detailed clinical guidance and enhances causal inference by reducing confounding factors through IPTW. Our results indicate that, compared with antiplatelet agents, the use of anticoagulants or combination therapy leads to a higher risk of rebleeding, while the mortality benefit is not as pronounced. Although antiplatelet or anticoagulant monotherapy did not significantly improve patient mortality within 28 days, their direction of effect is consistent with the main effect, and we believe they have potential protective effects. Similarly, due to the opposite direction of effects of using two drugs simultaneously, this suggests its potential risks. Therefore, larger-scale studies may be needed to investigate the differences in the impact of different types of drugs on patient prognosis.
Lastly, we quantitatively assessed the impact of the timing of antithrombotic therapy resumption on patient outcomes. To our knowledge, few studies have investigated the optimal timing of drug resumption. Current guidelines, due to a lack of robust evidence, only suggest evaluating the resumption of antithrombotic therapy (apart from aspirin) within 7 days based on individual patient benefit.16,19 Our study found that an early resumption (within 3 days) may significantly increase the risk of mortality, underscoring the need for careful assessment of the optimal timing for restarting antithrombotic therapy. This may be related to endothelial repair 48–72 h after acute bleeding. Premature restoration of antithrombotic therapy may interfere with the initial hemostatic mechanism of platelet thrombus formation and vascular constriction, while delaying to restart after 3 days may be more in line with the biological rhythm of tissue repair.
Despite the valuable insights provided by our study on the impact of antithrombotic therapy on the outcomes of GIB patients, several limitations should be noted. First, as a retrospective study, our analysis is susceptible to selection and information biases. Although IPTW was used to adjust for known confounders, the influence of unknown or unmeasured factors cannot be entirely excluded. Second, detailed information regarding drug dosages, duration of therapy, and patient adherence was not available due to data limitations, and these factors may affect patient outcomes. Third, the follow-up period was restricted to 28 days, precluding assessment of the long-term impact of antithrombotic therapy on survival and rebleeding risk in GIB patients. Finally, although some subgroup analyses did not yield statistically significant results, the consistency of their causal direction with the main analysis suggests that the lack of significance may be attributable to limited sample sizes and insufficient statistical power in these subgroups.
Conclusion
The results demonstrate that while antithrombotic treatment is associated with a reduction in 28-day mortality, it significantly increases the risk of rebleeding. Notably, the use of anticoagulants or combination therapy is linked to the highest rebleeding risk, compared to antiplatelets. Additionally, resuming antithrombotic therapy too early (i.e., within 3 days) may further elevate the risk of mortality.
Supplemental Material
sj-jpeg-2-tag-10.1177_17562848251342864 – Supplemental material for Impact of antithrombotic therapy resumption in patients with gastrointestinal bleeding: a multicenter retrospective cohort study
Supplemental material, sj-jpeg-2-tag-10.1177_17562848251342864 for Impact of antithrombotic therapy resumption in patients with gastrointestinal bleeding: a multicenter retrospective cohort study by Ding Peng and Huihong Zhai in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-xls-1-tag-10.1177_17562848251342864 – Supplemental material for Impact of antithrombotic therapy resumption in patients with gastrointestinal bleeding: a multicenter retrospective cohort study
Supplemental material, sj-xls-1-tag-10.1177_17562848251342864 for Impact of antithrombotic therapy resumption in patients with gastrointestinal bleeding: a multicenter retrospective cohort study by Ding Peng and Huihong Zhai in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
