Abstract
Background:
Current guidelines strongly recommend the use of validated classifications to support optical diagnosis of lesions with advanced endoscopic imaging in the lower gastrointestinal tract. However, the optimal strategy in inflammatory bowel disease (IBD) is still a matter of debate.
Objectives:
To analyze the accuracy of endoscopic classifications or single predictors for in vivo lesion characterization during endoscopic surveillance of IBD with advanced endoscopic imaging.
Design:
Systematic review.
Data sources and methods:
Medline and PubMed were used to extract all studies which focused on lesion characterization of neoplastic and non-neoplastic lesions in IBD. The diagnostic accuracy of endoscopic classifications and single endoscopic predictors for lesion characterization were analyzed according to type of patients, lesions, and technology used. When available, the rates of true and false positives or negatives for neoplasia were pooled and the sensitivity (SE), specificity (SP), positive predictive value, and negative predictive value (NPV) were calculated.
Results:
We included 35 studies (2789 patients; 5925 lesions – 1149 neoplastic). Advanced endoscopic imaging included dye-based chromoendoscopy, virtual chromoendoscopy (VCE), magnification and high-definition endoscopy, confocal laser endomicroscopy (CLE), endocytoscopy, and autofluorescence imaging. The Kudo classification of pit patterns was most frequently used, with pooled SE 83%, SP 83%, and NPV 95%. The endoscopic criteria with the highest accuracy, with minimum SE ⩾ 90%, SP ⩾ 80%, and NPV ⩾ 90% were: the Kudo-IBD classification used with VCE (Fuji Intelligent Color Enhancement and i-SCAN); combined irregular surface and vascular patterns used with narrow band imaging; the Mainz classification used with CLE. Multiple clinical and technical factors were found to influence the accuracy of optical diagnosis in IBD.
Conclusion:
No single endoscopic factor has yet shown sufficient accuracy for lesion characterization in IBD surveillance. Conventional classifications developed in the non-IBD setting have lower accuracy in IBD. The use of new classifications adapted for IBD (Kudo-IBD), and new technologies based on in vivo microscopic analysis show promise.
Introduction
Patients with long-standing ulcerative colitis (UC) and colonic Crohn’s disease are at increased risk of colorectal cancer (CRC). Consequently, surveillance colonoscopy is recommended by international inflammatory bowel disease (IBD) guidelines for the early detection of neoplasia,1–3 with the aim of reducing CRC mortality. 4 Surveillance colonoscopy should ensure high detection rates of all visible polypoid and non-polypoid neoplastic lesions, with accurate differentiation of neoplastic from non-neoplastic lesions. Lesion characterization, also called ‘optical diagnosis’, is of critical importance because non-neoplastic (particularly inflammatory) lesions are more frequent than neoplasia in IBD and can share similar macroscopic characteristics.5,6 Accurate characterization is essential as only neoplastic lesions should be removed, thereby optimizing resource utilization including endoscopic treatment and follow-up, and pathology workload. Endoscopic characterization should have both a high sensitivity and negative predictive value (NPV) for neoplasia, allowing identification of all neoplastic lesions, with those confidently characterized as non-neoplastic left in situ without the need for biopsy or resection. 7
However, the optimal endoscopic surveillance approach in IBD remains a matter of debate with multiple endoscopic imaging technologies now available. While first studied in CRC screening in the general population, a variety of advanced endoscopic equipment (also termed ‘powered endoscopy’) has now been tested in the endoscopic surveillance of UC. These include standard-definition (SD) or high-definition (HD) white-light endoscopy (WLE), dye-enhanced chromoendoscopy (DCE), or virtual chromoendoscopy (VCE), also supported by SD/HD or magnification, confocal laser endomicroscopy (CLE), endocytoscopy (EC), and autofluorescence imaging (AFI).
For lesion detection, the current standard of care is HD colonoscopy with or without DCE or VCE; the latter includes narrow band imaging (NBI; Olympus), Fuji Intelligent Color Enhancement (FICE; Fuji), i-SCAN (Pentax), and linked color imaging (LCI)/blue light imaging (BLI) (Fuji).8,9
For optical lesion diagnosis, the 2015 SCENIC guidelines were largely silent on endoscopic classification criteria for suspected neoplasia due to the inconsistent application of endoscopic criteria in the original chromoendoscopy literature. 1 More recently, European Society of Gastrointestinal Endoscopy (ESGE) strongly recommended the use of ‘validated’ classification systems to support optical diagnosis with advanced endoscopic imaging in the lower gastrointestinal tract.10,11 However, even the most recent guidelines do not recommend use of a specific classification as the standard of care in IBD.8,11 Furthermore, use of endoscopic criteria to rule out non-neoplastic lesions remains controversial, with considerable uncertainty around a ‘diagnose and leave’ strategy for low-risk lesions in IBD.
In this systematic review, we analyze the diagnostic accuracy of endoscopic classifications and single predictors of lesion characterization in the surveillance of IBD, stratifying their performance according to type of patients, lesions, and technology used.
Methods
A systematic review following the rules of the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement was performed. Medline and PubMed were used to extract eligible studies, from database inception to February 2023. The terms ‘IBD’ AND [‘chromoendoscopy’ OR ‘advanced endoscopic imaging’ OR ‘powered endoscopy’ OR ‘NBI’ OR ‘FICE’ OR ‘i-SCAN’ OR ‘BLI/LCI’ OR ‘confocal laser endomicroscopy’ OR ‘endocytoscopy’ OR ‘polyps’ OR ‘Kudo’ OR ‘NICE’] were matched. Three independent gastroenterologists (LC, MB, and VZ) performed the initial search. The initial review was performed by two independent authors (AC and MP) through analysis of titles, abstracts and references, identification of additional studies and exclusion of duplicates, reviews, overlapping and inappropriate records, non-English language, and pediatric literature. All authors then analyzed the specific indications for which advanced endoscopic imaging was used, including only studies which specifically analyzed lesion characterization of neoplastic and non-neoplastic lesions in IBD. When available, the rates of true and false positives or negatives for neoplasia were pooled for quantitative analyses, by calculating the sensitivity (SE), specificity (SP), positive predictive value (PPV), NPV, and accuracy (ACC) of the specific classification or single predictor. When comparisons were performed, a p value ⩽0.05 was considered statistically significant. A formal meta-analysis was not performed due to the expected high heterogeneity of clinical, endoscopic, and technical aspects among studies and populations.
Results
The initial search yielded 864 studies. A further search of their references identified an additional 95 studies. After the exclusion of studies as outlined above (Figure 1), a total of 35 studies (16 prospective, 6 multicenter) were included for qualitative analyses, comprising 2789 patients and 5925 lesions (1149 neoplastic; 1.6% cancers) (Table 1). Data from 32 studies were used for quantitative analyses when 2 × 2 contingency tables were available.

Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) flow diagram.
Studies included in the qualitative analysis of optical diagnosis in IBD surveillance.

AFI, autofluorescence imaging; ALM, adenoma-like mass; BLI, blue light imaging; CD, Crohn’s disease; CV, crystal-violet; DALM, dysplasia-associated lesion or mass; DCE, dye-based chromoendoscopy; DM, digital magnification; EC, endocytoscopy; e-CLE, endoscope-based confocal laser endomicroscopy; p-CLE, probe-based confocal laser endomicroscopy; ESD, endoscopic submucosal dissection; FACILE, Frankfurt Advanced Chromoendoscopic IBD LEsions; FICE, Fuji Intelligent Color Enhancement; HD, high definition; HGD, high grade dysplasia; IBD, inflammatory bowel disease; IC, indigo-carmine; JNET, Japan NBI Expert Team; MB, methylene-blue; NBI, narrow band imaging; n.r., not reported; OM, optical magnification; SD, standard definition; SSL, sessile serrated lesions; UC, ulcerative colitis; UCEIS, ulcerative colitis endoscopic index of severity; VPI, vascular pattern intensity; WLE, white-light endoscopy.
The endoscopic criteria for lesion characterization included the Kudo classification (n = 24 studies), modified versions of the Kudo classification (n = 2), the Frankfurt Advanced Chromoendoscopic IBD LEsions (FACILE) classification (n = 1), the Mainz classification with its variants (n = 7), and other criteria including the accuracy of single endoscopic morphological factors such as the Paris classification of macroscopic aspects, vascular patterns, and color of lesions (Table 1).
Endoscopic classifications in IBD surveillance
Kudo classification
The Kudo et al. 47 classification focuses on the morphology of ‘pit patterns’, which are the mucosal openings of the colorectal tubular glands. The Kudo classification has been shown to have high PPV, NPV, and ACC for neoplastic lesions and their invasion depth in the non-colitis setting.48,49
As normal glands are simple cylindric structures, at endoscopy their superficial openings appear as small round orifices which should be homogeneously distributed on the lesion surface; this is the type I Kudo pit pattern, similar to the surrounding normal mucosa. 50 Conversely, when there is neoplastic or hyperplastic cellular growth at the glandular level, or infiltration of the mucosa by inflammatory or neoplastic cells, the normal tubular glands can become distorted or stretched thereby losing their homogenous roundish mucosal openings. The morphological result is the following pit patterns (Figure 2):
– Type II = Stellar or papillary pits, characteristic of hyperplastic lesions
– Type IIIL = Tubular or round pits that are larger than normal pits, characteristic of adenomas
– Type IIIS = Tubular or round pits that are smaller than normal pits and can be associated with invasive lesions
– Type IV = Branch-like or gyrus-like pit pattern, characteristic of adenomas
– Type V = Irregular, non-structural pits, characteristic of invasive cancer

Conventional Kudo classification and modified Kudo classification.
The original Kudo et al. 50 classification was developed using stereo-endomicroscopy with magnification and DCE such as indigo-carmine (IC), methylene-blue (MB), and crystal-violet (CV). Further studies and routine clinical practice have, however, shown that pit pattern can be visualized even without magnification, particularly when supported by modern HD equipment.51,52 Therefore, VCEs such as NBI, FICE, and i-SCAN have also assessed these mucosal pit patterns for optical diagnosis.35,53,54
Twenty-four studies reported the use of Kudo classification for lesion characterization in IBD (predominantly patients with UC).6,7,12–16,20,22,23,28,31,33–38,40–42,44–46 Eighteen studies used DCE (IC: 13, MB: 5, CV: 6), whereas 11 studies used VCE (NBI: 7, FICE: 2, i-SCAN: 2); magnification was used in 17 studies, and HD in 19 studies (Table 1).
In all studies, Kudo pit patterns III–V were considered suspicious for neoplasia, whereas Kudo pit patterns I–II were combined and considered non-neoplastic as previously described in the non-IBD literature. 55 When the frequency of these groups of pit patterns was clearly reported (2296 lesions – 19% neoplastic, from 13 studies; Table 2),6,7,12,13,15,16,20,22,23,28,34,40,42 most lesions (65%) had non-neoplastic type I–II pits, whereas 26% were suspicious for neoplasia (Kudo III–V patterns). However, when the Kudo patterns were compared to histology (1645 lesions from 12 studies clearly reporting the rates of true and false positives/negatives),6,7,12,13,15,16,20,22,23,28,40,42 4% of Kudo I–II patterns were neoplastic, whereas only 44% of Kudo III–V were neoplastic. False negative and true positive rates for neoplasia using the Kudo classification were therefore 3% (range 0–20%) and 16% (range 6–42%), respectively.
Diagnostic accuracy of the Kudo classification in studies with 2 × 2 contingency tables.

ACC, accuracy; DCE, dye-based chromoendoscopy; FICE, Fuji Intelligent Color Enhancement; FN, false negatives; FP, false positives; HD, high definition; M+, with magnification; NBI, narrow band imaging; NPV, negative predictive value; n.r., not reported; PPV, positive predictive value; SD, standard definition; SE, sensitivity; SP, specificity; TN, true negatives; TP, true positives.
Overall, SE, SP, PPV, NPV, and ACC of the Kudo classification were highly variable across the studies, with SE 36–100% (mean 80%), SP 0–98% (mean 74%), PPV 26–96% (mean 57%), NPV 0–100% (mean 87%), and ACC 40–98% (mean 79%) (Table 2). After pooling the 12 series with complete 2 × 2 contingency data, the corresponding rates were 83%, 83%, 54%, 95%, and 83% (Table 2).
Five studies (one case report, three prospective, and one large retrospective series; Table 3)6,16,28,34,38 reported the frequency of each type of Kudo pit pattern (total 2365 lesions), with type I most common (mean 46%), followed by type II (mean 33%), IIIL (mean 8%), IIIs (mean 4%), IV (mean 2%), and V patterns (mean 0.25%). Only three studies described their association with histology (total 354 lesions, 13% neoplastic).6,16,28 The mean false negative rate for Kudo type I was 3.5% (range 0–11%), whereas the false positive rate for Kudo type II was 7% (range 5–100%). For type III–V lesions, the mean true positive rate was markedly lower for type IIIL (38%) than IV (69%). Kudo type V lesions were always neoplastic.
Prevalence of each class of Kudo pit patterns and their association with neoplasia.
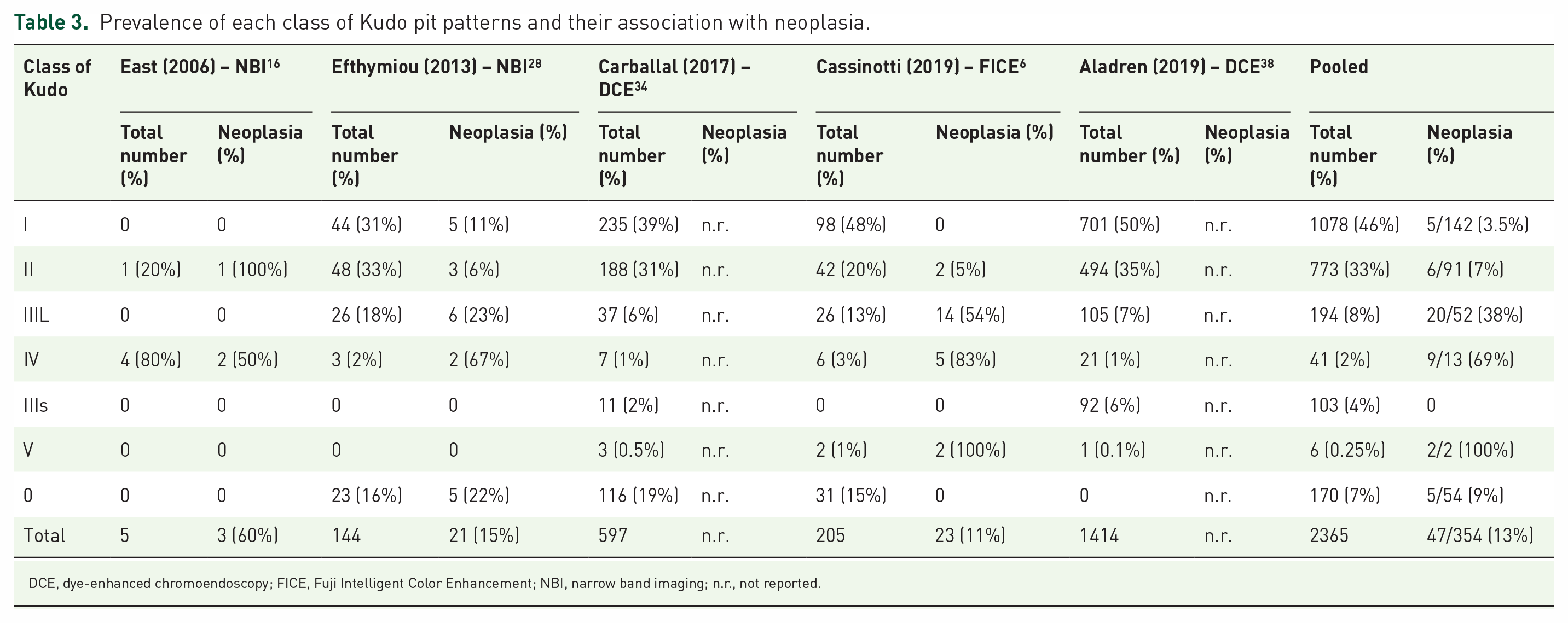
DCE, dye-enhanced chromoendoscopy; FICE, Fuji Intelligent Color Enhancement; NBI, narrow band imaging; n.r., not reported.
Notably, a number of lesions were unclassified by conventional Kudo pit pattern type, with type ‘0’ reported in seven studies,6,13,23,28,31,34,37 with mean prevalence of 13% (range 2–21%). The only two studies reporting their association with histology described opposite results, with no neoplastic lesions in one study that used magnification and HD-FICE, 6 compared with 22% (5/23) neoplastic lesions in the second study, 28 which used non-magnified NBI (Table 3).
Modified Kudo classification (Kudo-IBD)
In a sub-analysis of a prospective study of consecutive lesions (n = 205; 23 neoplastic, 164 inflammatory, 18 hyperplastic) from 59 patients with UC undergoing surveillance endoscopy with FICE, Cassinotti et al. 6 assessed the performance of Kudo classification combined with other endoscopic markers in the prediction of neoplasia. Four markers were selected for their potential use in a novel modified Kudo classification, with the aim of improving the sensitivity of Kudo types I–II and the specificity of Kudo types III–IV or unclassified lesions:
– Pits heterogeneity (observed in 40% of all lesions), defined as the variable density or size of pits, which was significantly more frequent in neoplastic versus non-neoplastic lesions (91% versus 33%).
– Microvessel positivity, defined as irregular brown/bluish visible capillary vessels, observed in only 12% of lesions but significantly associated with neoplasia (48% versus 7%), with true positive rate of 52% and false positive rate of 7% with conventional Kudo classification.
– The presence of fibrin cap or endoscopic inflammatory activity on the surface of the lesion (16% and 21% of all lesions, respectively), which were negatively associated with neoplasia (0/32 lesions with fibrin cap, 1/43 lesions with endoscopic inflammatory activity) and positively associated with inflammatory lesions (100% and 95%, respectively).
The diagnostic performance of different combinations of these additional markers with conventional Kudo pit patterns was assessed in a post hoc analysis as follows:
– Adding fibrin cap or endoscopic activity as negative predictors of neoplasia among Kudo type III–V and unclassified (type 0) lesions significantly increased SP (96% and 93%, respectively, versus 76% with conventional Kudo), while maintaining similar SE (91%, 87%, and 91%, respectively).
– Adding pits heterogeneity as a positive predictor of neoplasia among Kudo type I–II lesions increased both SE (100%) and SP (84%).
Following this preliminary study, the same group assessed the accuracy of a novel modified Kudo classification for IBD (Kudo-IBD) for the prediction of neoplasia in patients with UC undergoing surveillance colonoscopy with FICE. 42 The diagnostic accuracy of Kudo-IBD with FICE was compared to conventional endoscopic criteria with HD-WLE. One hundred consecutive patients underwent either FICE (46 patients, 136 lesions, 14 neoplastic) or WLE (54 patients, 139 lesions, 14 neoplastic). In this novel Kudo-IBD classification (Figure 2), Kudo pit patterns I–II were considered suspicious for neoplasia if at least one of two further endoscopic factors were present: (1) visible microvessels and (2) pits heterogeneity. Conversely, Kudo pit patterns III–IV or ‘0’ were considered non-suspicious for neoplasia if associated with a fibrin cap. In comparison with the previous study, 6 endoscopic inflammatory activity on the surface of the lesion was not included to avoid false negatives in cases of neoplastic lesions with inflammatory activity such as hyperemia or ulcers. Fibrin cap was preferred due to the known association with inflammatory non-neoplastic lesions in IBD, and the expected better inter- and intra-observer agreement. Finally, Kudo V and IIIs patterns, rarely found in previous IBD series but usually associated with advanced lesions including invasive cancer, were always considered suspicious for neoplasia.
The diagnostic performance of the Kudo-IBD classification versus WLE (per lesion) was: SE 93% versus 64% (p = 0.065), SP 97% versus 86% (p = 0.002), positive likelihood ratio 28.3 versus 4.5 (p = 0.001), negative likelihood ratio 0.07 versus 0.42 (p = 0.092), and NPV 99% versus 96% (p = 0.083). 42
Validation of this classification has been presented by the same group only in abstract form in two prospective series with i-SCAN (287 lesions, 18 neoplastic) and NBI (394 lesions, 21 neoplastic), confirming high SE (94% and 86%, respectively), SP (80% and 79%, respectively), and NPV (100% and 99%, respectively).56,57
FACILE classification
The FACILE classification was developed using four endoscopic criteria based on expert panel opinion and multivariable analyses, and validated in 60 images of polypoid and non-polypoid lesions, including 33 neoplastic (6 cancers) and 12 inflammatory lesions. 39 Images were obtained using different technologies, including HD-WLE, DCE, and VCE, such as i-SCAN and NBI.
The expert panel did not select Kudo pit pattern criteria to avoid diagnostic uncertainty in the absence of magnification, and due to challenges in the interpretation of regenerative changes. Multivariable logistic regression analysis showed that non-polypoid morphology [odds ratio (OR): 3.13, 95% CI: 1.32–7.25], irregular vessel architecture (OR: 3.49, 95% CI: 1.74–7.10), signs of inflammation within the lesion (OR: 2.42, 95% CI: 1.24–4.79), and irregular surface pattern (OR: 8.89, 95% CI: 3.21–25.96) were predictors of neoplasia. These endoscopic criteria were used to create the FACILE classification. SE and SP in multivariate analysis were 94% (95% CI: 90−96) and 51% (95% CI: 43−58), respectively. In the second phase of the study, the diagnostic accuracy of the FACILE classification for neoplasia was assessed through its application by the expert panel to the same 60 lesion images used for initial classification development. SE, SP, and ACC for prediction of neoplastic histology were 72%, 74%, and 72%, respectively. In the third phase of the study, reproducibility of the FACILE classification was assessed among non-experts [consultants (n = 5), trainees (n = 8), and junior doctors (n = 6)] using the same 60 images, with SE ranging from 80% to 90%, SP 56% to 78%, and ACC 77% to 86%.
Sano classification
The Sano classification 58 is the only formal classification of specific vascular patterns analyzed in IBD, using magnified-NBI in the assessment of capillary pattern:
– Type I is characterized by faintly visible microvessels surrounding the pits, consistent with non-neoplastic lesions.
– Type II is characterized by a mesh of capillary vessels, which appear elongated with abnormally large diameters and surround mucosal pits, strongly suggestive of adenomas.
– Type III is characterized by a mesh of capillary vessels with irregular size, complicated and blind ending branching, and disrupted irregular winding, suggestive of cancer. Sano type III is further divided into two subtypes. Sano type IIIA is characterized by high microvessel density with a lack of uniformity, and Sano type IIIB is characterized by a nearly avascular or sparse microvascular area clearly demarcating normal and cancerous mucosa.
The Sano classification has been assessed in two studies.31,36 Yoshioka et al. 31 reported the prevalence of these vascular patterns within a series of 15 exclusively neoplastic lesions (seven cancers): type I = 0.9% (1/15), type II = 27.3% (3/15), type IIIA = 63.6% (7/5); no type IIIB were found. 31 The study by Kinoshita et al., 36 limited to 25 lesions removed by endoscopic submucosal dissection (ESD), did not report the prevalence of these vascular patterns or the association of the Sano classification alone with histology, but instead analyzed the accuracy of combined Sano and Kudo criteria to predict advanced lesions (HGD or cancers) versus LGD, with SE 72%, SP 86%, PPV 93%, NPV 54%, and ACC 76%.
NICE and JNET classifications
The NBI International Colorectal Endoscopic (NICE) classification is a validated NBI-based system for the optical diagnosis of colorectal polyps in the non-colitis setting, based on color, surface, and vascular patterns of the lesions, with a pooled SE of 98% and 95% NPV for lesion characterization.54,59
In this classification, polyps can be divided into three categories: NICE type 1 and 2 are benign; type 3, characterized by disrupted/missing vessel pattern and amorphous or absent surface pattern on NBI, are highly suspicious for deep submucosal invasion.
No full-text papers have been published on the use of the NICE classification in IBD. A single abstract reported SE 76%, SP 55%, PPV 9%, and NPV 98% in 394 lesions analyzed with non-magnified HD-NBI. 56
The Japan NBI Expert Team (JNET) system is another classification developed for NBI with magnification in the non-colitis setting. 60 It is more frequently used in Asian countries than the Western hemisphere. By focusing on vessel and surface patterns, the JNET system classifies colorectal polyps into four types (1, 2A, 2B, and 3) with types 2–3 considered neoplastic. Similar to NICE, irregular/amorphous vessel and surface patterns suggest a higher likelihood of submucosal invasive cancer. Just two studies on the use of JNET in IBD have been published, with only neoplastic lesions included (17 and 44 lesions, respectively).41,46 JNET types 2–3 were associated with neoplasia, including cancers. In the study by Kida et al. 46 , JNET type 2A was considered an indicator of low-grade dysplasia, JNET type 2B an indicator of high-grade dysplasia, and JNET type 3 an indicator of deep submucosal invasive carcinoma. In UC-associated neoplastic lesions, JNET type 2A had low PPV (50%) but high NPV (95%). 46 In contrast, JNET type 3 had high PPV (75–100%) and NPV (81–88%) for invasive cancer in both studies.41,46
Mainz classification
CLE is a technology which is either integrated into the distal tip of a dedicated endoscope (termed ‘eCLE’; Optivista-Pentax EC3870K, Fort Wayne, NJ, USA) or integrated into a probe which can be passed through the biopsy channel of a conventional endoscope (termed ‘pCLE’; Cellvizio, Mauna Kea Technologies, Paris, France). CLE has typically been applied to circumscribed lesions first detected with DCE and then stained with intravenous injection of the contrast agent fluorescein sodium. 61
As in the non-colitis setting, both eCLE and pCLE equipment have been assessed for the optical diagnosis of lesions in IBD,18,19,22,24–27,30 providing real-time high-resolution imaging (0.8 and 1 μm, respectively) of the gastrointestinal mucosa at the cellular and subcellular levels. However, despite favorable proof-of-concept studies, eCLE is no longer commercially available. 62
The Mainz classification was used for lesion characterization in five studies of eCLE in IBD.18,19,22,24,30 This classifies neoplastic and non-neoplastic images according to cellular and vascular changes. Neoplastic lesions display crypt architecture with a ridged-lined irregular epithelial layer with loss of crypts and goblet cells, or loss of any crypt architecture. Neoplastic vessel architecture has distorted and dilated vessels with increased leakage and little to no orientation to adjunct tissue. 18
Three eCLE series in IBD which used Mainz classification reported sufficient data on the characterization of 306 lesions (28 neoplastic),18,22,30 with pooled SE 93%, SP 98%, PPV 84%, NPV 99%, and ACC 98%.
Three further studies analyzed the accuracy of pCLE in IBD using variants of the Mainz classification25–27 Sufficient data were available for two of these studies, characterizing 63 lesions (22 neoplastic) 25,27: SE, SP, PPV, NPV, and ACC ranged from 65% to 100%, 82% to 90%, 83%, 100%, and 81% to 93% respectively.
Paris classification
In 2015, the SCENIC guidelines recommended that visible dysplasia in patients with long-standing IBD should be endoscopically characterized using a modified Paris classification. 1
The Paris classification, the consensus of an international consortium, has been widely adopted to describe the macroscopic appearance of focal endoscopic lesions. 63 Although one study has shown only moderate agreement among experts using the Paris classification, 64 it serves as a validated and standardized nomenclature that helps categorize colorectal lesions and stratify their treatment according to the risk of CRC. Broadly speaking, superficial lesions are categorized as polypoid (type 0–I, defined as elevated >2.5 mm above the mucosal layer) or non-polypoid (type 0–II). The polypoid type can be either pedunculated (type 0–Ip), sessile (type 0–Is), or semi-pedunculated (0–Isp). Non-polypoid lesions can be further subdivided into superficially elevated (0–IIa), flat (0–IIb), or depressed (0–IIc). Excavated lesions are designated type 0–III.
In IBD, 21 studies used the Paris classification to describe the macroscopic appearance of lesions.6,7,15,19,20,22,23,25,28,30,33–42,44 Pooling data from six studies which clearly described the prevalence of each Paris class among 2619 lesions,6,28,34,38,44,45 the most frequent morphology was Is (40.5%), followed by IIa (29.6%), IIb (21%), Ip (8%), IIc (0.6%), and Isp (0.3%). Slightly elevated (IIa) lesions were however the most frequent morphology (59%) in a series of 74 exclusively neoplastic lesions from two studies, followed by Is (23%), IIb (12%), and Ip and IIc (3%).7,45
Only two studies described the diagnostic accuracy of the Paris classification alone for lesion characterization in IBD, with opposite results.7,34 Vleugels et al. 7 reported a significant association between non-polypoid morphology and neoplasia, with SE 97%, SP 12%, PPV 65%, NPV 60%, ACC 69%. Conversely, Carballal et al. 34 reported a significant association between protruding lesions (Is and Ip) and neoplasia, with SE 56%, SP 68%, PPV 25%, NPV 89%, and ACC 62%. Multivariable analyses from other two studies showed a significant association between polypoid lesions (Paris Is/Ip) and neoplasia, with OR 3.30 (95% CI: 1.26–8.96) 35 and OR 2.751 (95% CI: 1881–4.013 for Paris Is) and OR 7.26 (95% CI: 1.597–33.004 for Paris Ip) in the study by Aladrén et al., 38 while López-Serrano et al. 44 did not report any significant association between Paris morphology and neoplasia.
Notably, an image-based study by Guerrero Vinsard et al. 65 has shown very low inter-observer agreement for the Paris classification and low accuracy for lesion histopathology prediction in IBD.
Single endoscopic predictors of neoplasia in IBD
Surface patterns other than Kudo pits
Matsumoto et al. 17 prospectively classified the surface pattern of 296 lesions (5 neoplastic). Three categories were selected using magnified-NBI: honeycomb-like, villous, and tortuous patterns. The honeycomb-like pattern, defined as crowding of round capillary vessels in honeycomb appearances, was seen in 54% of lesions and was never associated with neoplasia. The villous pattern (n = 85; 29%) was characterized by a cerebriform structure resembling Kudo’s IIIL pit pattern. The tortuous pattern (n = 30; 17%) was defined by round or ovoid structures of various sizes; it was the most frequent pattern among the five neoplastic lesions, with SE 80% and SP 84% for the prediction of neoplasia.
The surface patterns (roundish, villous-regular, villous-irregular, irregular/nonstructural) included in the FACILE classification, 39 were also reported as single predictors of neoplastic or non-neoplastic lesions. When assessed by expert endoscopists, irregular architecture showed SE 85%, SP 70%, PPV 83%, NPV 74%, and ACC 80% for neoplasia.
In the study by Nishiyama et al., 29 61 images of neoplastic (n = 19, 3 cancers) and non-neoplastic (n = 42, all inflammatory) lesions were analyzed by five experts using DCE-IC and magnification. The pit density of neoplastic lesions was found to be significantly greater than that of non-neoplastic lesions (89% versus 60%, respectively); high residual pits had SE 89%, SP 40.5%, PPV 40.5%, NPV 89.5%, and ACC 55.7%. Pit margins were more frequently irregular in neoplastic lesions than in non-neoplastic lesions (63% versus 33%, respectively); irregular pit margins had SE 63%, SP 67%, PPV 46%, NPV 80%, and ACC 65%.
In a second study by the same group using magnified-NBI, irregular/amorphous surface pattern was significantly more common in neoplastic than non-neoplastic lesions (81% versus 18%), with SE 82%, SP 82%, PPV 81%, NPV 82%, and ACC 82%. 32 When combined with irregular vascular patterns, accuracy increased with SE 94%, SP 88%, PPV 88%, NPV 94%, and ACC 91%. 32
In the study by Shinagawa et al., 40 three specific morphological patterns (Pine-cone, Villi, and Open) were validated against 113 endoscopic images (64 from neoplastic lesions). 40 Pine-cone and Villi were considered morphologic patterns representative of Kudo’s type IV pit pattern. Pine-cone was defined as a superficial form like a cone of a pine tree over 1 mm in size, whereas Villi was defined as a collection of fine villous structures each less than 1 mm in size. Open was defined as surface structures with enlarged glandular openings, which were further divided into open ‘homogeneous’ pattern (Open Homo) with regular open pit morphology and arrangement, and open ‘heterogeneous’ pattern (Open Hetero) with irregular open pit morphology and arrangement. For prediction of neoplasia, the Pine/Villi patterns showed SE 44%, SP 92%, PPV 86%, NPV 59%, and ACC 66%, whereas the corresponding values for the open pattern were 3.4%, 91%, 40%, 35%, and 35%. 40
Vascular patterns other than Sano’s
Nine studies reported vascular features in lesion assessment.6,23,31,32,34,36,39,41,46 Overall, pooled ACC of any vascular architecture suspicious for neoplasia was 57–100%, with SE and SP also showing wide ranges of 50–100% and 36–100%, respectively.
Other vascular features were variably defined as ‘irregular’ or ‘darker’ vessels, using different technologies including NBI and FICE.
Vascular irregularity did not appear a significant predictor of neoplasia, at least when used alone. Nishiyama et al. 32 classified vessel pattern as the same as background mucosa, regular, irregular, or avascular according to magnified-NBI in 33 consecutive lesions. Irregular and avascular vessels were numerically but not significantly more frequent in neoplastic than non-neoplastic lesions (75% versus 41%), with SE 75%, SP 59%, PPV 63%, NPV 71%, and ACC 67%. As also described above, combining surface patterns (irregular and amorphous) with vascular patterns increased accuracy with SE 91%, SP 94%, PPV 88%, NPV 88%, and ACC 94%. 32 Carballal et al. 34 also analyzed the accuracy of abnormal vascularization, found in 4.4% of 597 lesions (94 neoplastic), without further defining their vascular patterns. This marker was not an independent predictor of neoplasia after multivariable analysis. The only study supporting irregular vessel architecture was by Iacucci et al. 39 which reported a significant association with neoplasia (OR: 3.49, 95% CI: 1.74–7.10), with SE 87%, SP 36%, PPV 70%, NPV 60%, and ACC 69%.
Darker colored vessels may be a more accurate marker, based on two studies.6,23 Using definitions analogous to Sano types II–III and with different exclusion criteria for inflammatory lesions, their prevalence ranged from 12% to 34%. In the only study of consecutive lesions during FICE surveillance, microvessel positivity (defined as type III–V Teixeira patterns) 66 was seen in 12% of lesions, and was significantly associated with neoplasia (48% versus 7%); it was associated with true positive rate of 52% and false positive rate of 91% according to the Kudo classification.
There are no data on accuracy when vascular pattern is considered alone or in combination with other classes of conventional Kudo patterns. However, on the basis of preliminary data and previous non-IBD literature, 53 vascular pattern was included in the modified Kudo-IBD classification, described above, as a modifier of neoplastic risk within type I–II Kudo lesions. 42 Furthermore, in the study by van den Broek et al., 23 darker vascular pattern was seen more frequently in neoplastic than non-neoplastic lesions (30% versus 4%). The authors used vascular pattern intensity (VPI) as a predictor of neoplasia,16,53 with SE 80%, SP 72%, and ACC 73%.
Inflammatory activity
Inflammation of the lesion surface as a marker of neoplasia was assessed in two studies, with opposite results.6,39
In the image-based study by Iacucci et al., 39 signs of inflammation were significantly associated with neoplasia (OR: 2.42, 95% CI: 1.24–4.79), although they were the least accurate predictor among four criteria included in the FACILE classification (SE 88%, SP 13%, PPV 63%, NPV 40%, ACC 61%).
In their prospective study of 205 consecutive lesions including all histological types and any clinical and endoscopic activity, Cassinotti et al. 6 defined endoscopic inflammatory activity as the presence of any of friability, erosions, or ulcers at the surface of the lesion, similar to the Mayo endoscopic subscore of disease activity in UC. An additional marker of inflammation was fibrin cap, described as a not removable, circumscribed white exudate at the surface of the lesion, as previously described by Rubin et al. 67 Interestingly, fibrin cap or endoscopic inflammatory activity (16% and 21% of all lesions, respectively) were present in 18% and 23%, respectively of 182 non-neoplastic lesions but were absent in all (for fibrin cap) or almost all (for endoscopic inflammatory activity, 1/23) neoplastic lesions. Their presence was therefore negatively associated with neoplasia and positively associated with inflammatory lesions (100% of lesions with fibrin cap, 95% of lesions with endoscopic inflammatory activity). Following logistic regression, there was a strong negative association between endoscopic inflammatory activity and neoplasia, whereas the presence of fibrin cap was significantly associated with endoscopic inflammatory activity. 6 This data led the authors to include fibrin cap in the modified Kudo-IBD classification described above. 42
Fibrin cap, surface friability, and ulcerations were also associated with non-neoplastic lesions in the study by Sussman et al. 68 that focused on inflammatory polyps, using endoscopic images obtained with HD-WLE, NBI, and DCE. Other endoscopic factors associated with inflammatory polyps were appendage-like appearance, the halo sign with DCE, and a clustering of a multiplicity of inflammatory polys. The overall diagnostic ACC for inflammatory polyp identification was 63% for WLE, 42% for NBI, and 64% for DCE. 68
Inflammation of the background mucosa was not an independent predictor of neoplasia in the study by Nishiyama et al., 32 in which neoplasia rates were similar (35% versus 25%) among 37 lesions with surrounding inflammatory activity and 24 lesions in quiescent disease, analyzed with DCE. Furthermore, ulceration was not an independent predictor of neoplasia in two studies.34,37 Conversely, Aladrén et al. 38 reported that endoscopic activity was an independent risk factor for dysplasia detection (OR: 2.69; 95% CI: 1.47–4.92), whereas Kida et al. 46 reported more severe inflammation in the surrounding mucosa of UC-associated neoplasia than with sporadic adenomas.
Notably, mucosal activity did affect the ACC of Kudo classification, which was lower for lesions occurring in areas of inflammation (44%; 95% CI: 35–51) than for lesions in uninflamed areas (75%; 95% CI: 0.71–0.82). 28
Color of lesion
Seven studies have included lesion color (darker or red versus lighter or pale versus same as surrounding mucosa) in the criteria for morphological description, which varied with different endoscopic imaging equipment.6,29,31,32,36,43,46 Darker or red color was frequently associated with neoplasia, but it was not an independent predictor of histology; indeed, a high rate of false positives among inflammatory lesions was described.
In the study by Cassinotti et al., 6 brown lesion color with FICE was the most frequent endoscopic marker (63% of lesions) of the seven exploratory additional endoscopic factors described above in the assessment of 205 consecutive lesions, most of which inflammatory. However, it was present in both 78% of neoplastic lesions and 61% of non-neoplastic lesions, and was associated with true positive rate of 81% and false positive rate of 91% according to the Kudo classification, and therefore was not a helpful predictor of neoplasia.
Using HD-WLE, three studies have classified lesion color as pale, red, or same as surrounding mucosa.29,32,46 Pooling this data (n = 109 lesions, 50 neoplastic), the prevalence of red, pale, and same colored lesions were 59% (n = 64), 17% (n = 19), and 24% (n = 26), respectively, within all lesions, and 54% (n = 27), 24% (n = 12), and 22% (n = 11) within neoplastic lesions. However, lesion color was not an independent predictor of histology.29,32 Finally, Ikebata et al. 43 found that all 10 cases of flat-type-predominant dysplasia in their series of 38 neoplastic lesions had the endoscopic appearance of demarcated red-colored areas.
The color of neoplastic versus non-neoplastic lesions has also been studied with AFI, which is a technique that differentiates tissues based on their ability to capture and reflect fluorescent light. 69
Four studies used AFI criteria to characterize visible lesions in UC.7,20,21,46 According to the conventional criteria developed for the general population, normal mucosa appears green while neoplasia appears as a purple lesion on the green background fluorescence of normal colonic tissue. 70 In the study by Yoshioka et al., 31 a purple lesion was observed in only 37.5% of neoplastic lesions, indicating that the qualitative analysis of AFI color as green or purple is insufficient for the detection of neoplasia associated with UC. In the study by Matsumoto et al., 21 126 lesions (14 neoplastic) were studied with AFI, with SE 100%, SP 18%, PPV 10%, and NPV 100%. In the study by Vleugels et al., 7 AFI had the highest SE (91.7%) and NPV (98.7%) in comparison to NBI and WLE, but SP was 74.5%, PPV 29.7%, and ACC 76.3%. In the study by van den Broek et al., 20 the SE, SP, and ACC of AFI in 98 lesions (16 neoplastic) were 100%, 42%, and 51%, respectively. Despite these studies, the poor performance of AFI for dysplasia detection in the FIND-UC trial has stopped further development in IBD. 71
Other predictors
The description of lesions margins as distinct versus poorly demarcated has been reported by three studies, with no significant association found with histology.34,37,46 In the study by Yang et al., 37 distinct border had SE 74%, SP 38%, PPV 60%, NPV 54%, and ACC 58%. Kida et al. 46 described lesion borders were more unclear in UC-associated neoplasia than sporadic adenoma (16/21 versus 0/23; p < 0.001).
Loss of innominate lines was reported in 45% of 272 lesions in the study by Carballal et al. 34 ; it was significantly associated with neoplasia (OR: 1.95; 95% CI: 1.06–3.58), with SE 56%, SP 56%, PPV 19%, NPV 87%, and ACC 57%. Finally, Lopez-Serrano et al. 44 did not report any significant association between dysplasia and lesion size.
Comparison of classifications and single endoscopic predictors
Only two studies included comparative analyses of different classifications. In one series using HD-NBI, the accuracy of the Kudo classification was similar to VPI. 23 In a second study with FICE, the Kudo-IBD classification performed significantly better than the conventional Kudo classification and conventional WLE with higher SP and PPV. 42
Table 4 summarizes the pooled accuracy of each classification system or single endoscopic criteria, stratified by technology and number of studies. The endoscopic criteria with highest accuracy, defined as SE ⩾ 90%, SP ⩾ 80%, and NPV ⩾ 90% were:
– The Kudo-IBD classification when used with FICE and i-SCAN, but with borderline values with NBI, according to three series from the same group.42,56,57
– The irregular surface and vascular patterns, when used with NBI, according to the single study by Nishiyama et al. 32
– The Mainz classification, when used with the now abandoned eCLE.18,22,30
Diagnostic accuracy of endoscopic classifications and single endoscopic predictors of neoplasia, stratified according to technology.

Dark gray cells = highest accuracy (SE ⩾ 90%, SP ⩾ 80%, NPV ⩾ 90%). Light gray cells = high accuracy with SE ⩾ 80%, SP ⩾ 80%, NPV ⩾ 90%.
Studies with full data from 2 × 2 contingency tables. The corresponding mean values from all studies (n = 17) were SE 80%, SP 74%, PPV 57%, NPV 87%, ACC 79%.6,7,12,13,15,16,20,22,23,28,33–35,38,40,42,45
Studies with full data from 2 × 2 contingency tables. The corresponding mean values from all studies (n = 11) were SE 85%, SP 80%, PPV 69%, NPV 92%, ACC 84%.6,7,12,13,15,33–35,40,42,45
Studies with full data from 2 × 2 contingency tables. The corresponding mean values from all studies (n = 6) were SE 62%, SP 63%, PPV 34%, NPV 76%, ACC 68%.7,16,20,23,28,33
ACC, accuracy; AFI, autofluorescence imaging; CV, crystal-violet; DCE, dye-based chromoendoscopy; FACILE, Frankfurt Advanced Chromoendoscopic IBD LEsions; FICE, Fuji Intelligent Color Enhancement; IC, indigo-carmine; JNET, Japan NBI Expert Team; NBI, narrow band imaging; NPV, negative predictive value; n.r., not reported; PPV, positive predictive value; SE, sensitivity; SP, specificity; WLE, white-light endoscopy.
The conventional Kudo classification only achieved high accuracy with DCE, as did variants of the Mainz classification applied to pCLE.
Factors affecting the accuracy of advanced endoscopic imaging in the surveillance of IBD
Exclusion of clinical and endoscopic activity
Among the 35 studies included in this review, the majority (n = 21) excluded patients with clinical activity. However, some studies which excluded patients with clinical activity described endoscopic activity in up to 70% of patients.22,24,32,38,40
Only two studies stratified the accuracy of their diagnostic criteria according to clinical and/or endoscopic activity.28,45
In the study by Efthymiou et al., 28 which used HD-NBI, the Kudo classification had significantly higher ACC for lesions occurring in quiescent mucosa than those found within mucosal inflammatory activity (44% versus 75%). Series with a high prevalence of inflammatory lesions and any clinical and endoscopic activity have also reported that the conventional Kudo classification had a low SP for neoplasia.6,42,45
Kudo et al.,72,73 in a recent retrospective study of 103 lesions (23 neoplastic), suggested a novel strategy to overcome the diagnostic limitations of Kudo et al.’s pit patterns for IBD-associated neoplasia, by combining usual pit patterns with EC, an ultra-magnifying contact microscope, which provides visualization of cell nuclei or microvasculature, and detection of neoplasia in non-IBD patients. Their data found that SP was higher in the combined assessment of pit patterns with EC compared to pit patterns alone (84% versus 57%), while maintaining high SE with both analyses (98% versus 100%). 45 However, they also found that the SP may still be affected by active inflammation and that false positive rates were high (16% in type II–V pits with positive irregularly-formed nuclei and 10% in type V). Indeed both SP and ACC were significantly higher with Mayo endoscopic score (MES) 0–1 than MES 2–3 (SP: 93% versus 68%; ACC: 95% versus 74%). The false positive rates were 5% in lesions occurring in segments with MES 0–1 and 26% in those with MES 2–3. 45
Prevalence of non-neoplastic lesions
Only 10 studies which included consecutive patients provided data on the prevalence of different non-neoplastic lesions.6,12,23,25,28,30,34,38,42,44 Three studies excluded an uncertain quote of lesions.23,34,38 In the remaining seven studies which included all lesions (n = 1141), inflammatory lesions were the most frequent histology (40%), followed by hyperplastic (30%) and neoplastic lesions (14%). None of these studies analyzed the accuracy of specific predictors of inflammatory or hyperplastic lesions versus neoplasia with the aim of improving the SP and PPV of lesion characterization. A high false positive rate was reported by Kiesslich et al. 12 in a small group of inflammatory lesions (6/8) analyzed according to the Kudo III–V criteria. 12 The validation study of the Kudo-IBD classification found it was associated with significantly lower false positive rates than both Kudo and conventional WLE for inflammatory lesions, but not hyperplastic or neoplastic lesions. 42
As expected, studies which excluded all patients with any clinical activity12,25,28 had a significantly lower prevalence of inflammatory lesions than studies which included patients with clinical and endoscopic activity (21% versus 75%).6,30,42
In the few studies available on their characterization, hyperplastic lesions were classified prior to the current 2019 WHO classification. Therefore, scarce data are available on the prevalence and endoscopic predictors of colorectal serrated lesions, which now include hyperplastic polyps, sessile serrated lesions (SSL) with or without dysplasia (replacing the previous concept of sessile serrated adenomas/polyps – SSA/P), traditional serrated adenomas, and serrated adenomas unclassified. 74
Only five studies have reported serrated lesions, with a pooled total of 62 lesions (range 1–33).7,33–35,44 Vleugels et al. 7 described 18 SSL according to the 2010 WHO classification. Paris IIa or IIb morphology was seen in 83%, while all lesions had Kudo type II pit pattern on DCE. Iacucci et al. 35 described 33 SSA in 21/270 patients: 16 had Is/Ip and 17 IIb morphology. The most frequent pit pattern was type IIO, as proposed by Kimura et al. 75 in 28 lesions, followed by III–V in 4 and I–II in 1 lesion. In a prior study, Iacucci et al. 76 assessed the prevalence and endoscopic features of SSA in 14 lesions from 87 IBD patients undergoing surveillance colonoscopy using HD alone or with i-SCAN or MB-DCE. The endoscopic characteristics of SSA were: non-polypoid appearance (86%), predominant localization in the proximal colon (79%), >6 mm in size (79%), cloudy cover (64%), Kudo pit pattern type IIO (86%), and irregular spiral vascular pattern (79%). These characteristics had SE 93% and SP 93% for prediction of a histological diagnosis of SSA, with PPV 87% and NPV 97%.
Inclusion of high-risk patients
When considering clinical features associated with a high risk of colorectal neoplasia in IBD,2,34 primary sclerosing cholangitis (PSC) was reported by 14 studies,7,12,15,18,20,22,23,25,27,28,34,35,37,44 first-degree family history of CRC by 12,6,12,17,21–23,28,30,34,35,42,44 and previous neoplasia by 9.6,7,20,23,28,34,35,37,44 These high-risk patients comprised a minority of the total cases included, with patient numbers ranging from 1 to 29 for PSC, 1 to 38 for first-degree family history of CRC, and 7–60 for previous neoplasia. No studies stratified the accuracy of diagnostic criteria for lesion characterization according to these high-risk factors.
Magnification and high definition
Both image magnification and definition affect the quality of endoscopic visualization. High-definition imaging, which is a function of pixel density, improves the ability to discriminate details, whereas magnification enlarges the image. 77 Optical (or high) magnification endoscopes are defined by the capacity to perform optical zoom, by using a movable lens in the tip of the endoscope which provides a magnified image of the target (usually up to 150×), while maintaining image display quality. This is distinguished from electronic or digital magnification, which simply enlarges the image on the display, with a consequent decreased pixel density and image quality.
Twenty studies used different magnifying equipment to analyze Kudo pit patterns (n = 17)6,7,12,13,15,16,20,23,31,34,36,37,40–42,45,46 or other surface or vascular patterns (n = 9).17,23,29,31,32,36,40–42 No studies performed a direct comparison of magnified versus non-magnified equipment.
An indirect comparison was possible only through pooling studies which clearly reported the true and false positive and negative rates of lesions assessed by Kudo classification with optical magnification + SD or HD,6,7,12,15,16,20,23,40,42,45 versus digital/no-magnification + HD.28,38 After exclusion of two studies that used SD endoscopes no longer available,12,15 HD instruments with optical magnification versus digital or no-magnification showed significantly higher SE (86% versus 36%; p = 0.0000) and NPV (97% versus 92% p = 0.0012), but had lower SP (75% versus 91%; p = 0.0000) (Table 5). The higher SE was shown with both DCE and NBI, whereas lower SP was shown with DCE but not NBI. Notably, the SE of the two non-magnifying endoscopes included was lower than the range (77–92%) shown by two other studies excluded due to missing 2 × 2 tables.33,35 Furthermore, no studies reported the rate of activation of optical magnification during colonoscopy.
Diagnostic accuracy of Kudo classification according to magnifying and HD or SD endoscopes.

p ⩽ 0.05.
ACC, accuracy; DCE, dye-based chromoendoscopy; FN, false negatives; FP, false positives; HD, high definition; NBI, narrow band imaging; NPV, negative predictive value; PPV, positive predictive value; SD, standard definition; SE, sensitivity; SP, specificity; TN, true negatives; TP, true positives.
Technology
The highest SE, SP, and NPV (all ⩾90%) were shown by eCLE with Mainz classification (94%, 98%, 99%, respectively) and by FICE with Kudo-IBD classification (93%, 97%, 99%) (Table 6).
Diagnostic performance of endoscopic imaging technologies (mean values), according to lesion characterization criteria.
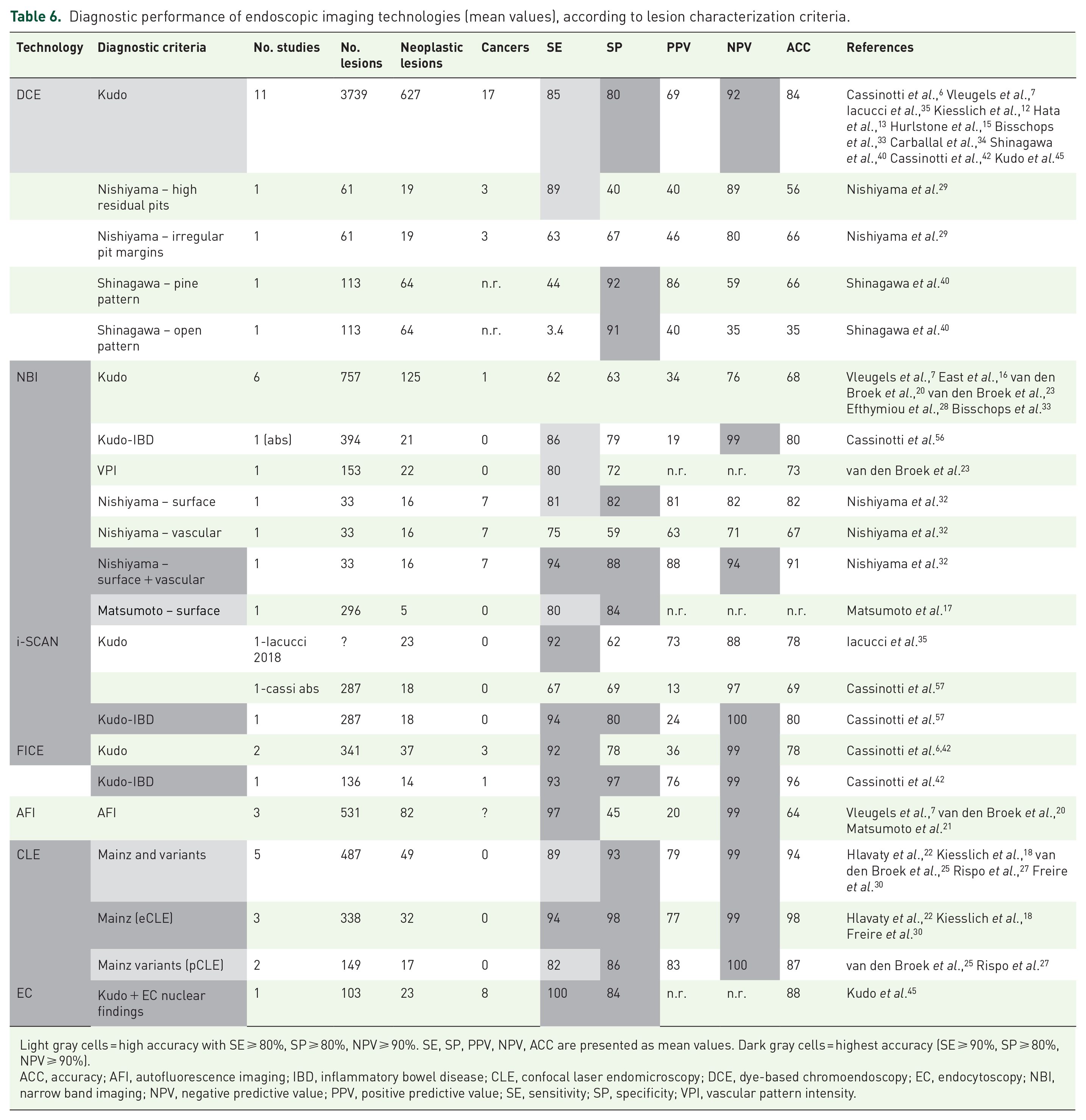
Light gray cells = high accuracy with SE ⩾ 80%, SP ⩾ 80%, NPV ⩾ 90%. SE, SP, PPV, NPV, ACC are presented as mean values. Dark gray cells = highest accuracy (SE ⩾ 90%, SP ⩾ 80%, NPV ⩾ 90%).
ACC, accuracy; AFI, autofluorescence imaging; IBD, inflammatory bowel disease; CLE, confocal laser endomicroscopy; DCE, dye-based chromoendoscopy; EC, endocytoscopy; NBI, narrow band imaging; NPV, negative predictive value; PPV, positive predictive value; SE, sensitivity; SP, specificity; VPI, vascular pattern intensity.
EC had the highest SE (100%) in the only study published to date. 45 NPV was not reported and SP (84%) was significantly lower than FICE with Kudo-IBD classification and CLE.
AFI had higher SE (97%) and NPV (99%) than DCE-Kudo, NBI-Kudo, and pCLE, but was similar to FICE and EC.
DCE with Kudo classification had a non-significantly lower SE (85%) than FICE-Kudo (92%), FICE with Kudo-IBD (93%), and CLE (89%), but a significantly lower NPV (92%) than FICE, CLE, and AFI (all 99%); SP was also significantly lower than FICE with Kudo-IBD and pCLE.
Both i-SCAN and FICE had high SE with Kudo and Kudo-IBD classification (92–94%) but lower SP with Kudo than Kudo-IBD.
Conversely, NBI showed a significantly lower SE compared to all other methods, irrespective of different diagnostic criteria, including Kudo (range 60–81%). NBI only showed improved performance with combined surface and vascular patterns as described by Nishiyama et al. 32 and with Kudo-IBD classification. 42
Experience of the endoscopist
Expert endoscopists performing optical diagnosis were reported in 18 series.6,7,15,19,20,23,24,27–29,32–34,36,37,39,45,46 However, only six studies provided a definition of an expert for the different endoscopic technologies and criteria,7,19,34,37,39,45 ranging from 20 to 500 chromoendoscopies with DCE,7,34,45 80 exams with CLE, or 6–15 years of endoscopic experience,37,39 or at least 11 cases/year of dysplasia. 37
In the study by Carballal et al., 34 experts had performed at least previous 20 DCE for IBD, with no statistically significant difference in accuracy of lesion characterization between expert and non-expert endoscopists. Furthermore, in the study by Efthymiou et al., 28 the low accuracy of the Kudo classification for neoplasia appeared unrelated to a learning curve effect.
In the study by Yang et al., 37 endoscopists with at least 6 years’ experience as a staff gastroenterologist showed significantly higher SE in the prediction of neoplasia, but lower SP. When the endoscopists were sub-grouped according to their experience with surveillance endoscopy [>10 patients/year or ⩽10 patients/year or and patients with UC (>50 patients/year or ⩽50 patients/year)], the more experienced endoscopists still showed lower SP (43.7% versus 25.9%; 42.2% versus 27.4%) in the endoscopic prediction of dysplasia. Contrasting results were found by Carballal et al., 34 who reported that expert endoscopists were better at ruling out dysplasia than non-experts (SP 94% versus 88%, respectively; p = 0.01). Finally, in the study by Kida et al., 46 the inter-observer agreement for sporadic neoplasia was higher than for UC-associated neoplasia among both experts and non-experts, irrespective of the classification system used.
Discussion
The accurate differentiation of neoplastic from non-neoplastic lesions found during IBD surveillance endoscopy is of paramount importance, with the presence of mucosal inflammation, scarring, and inflammatory lesions leading to particular challenges unique to IBD. It is therefore of critical importance that validated endoscopic criteria are used in combination with advanced endoscopic techniques to support lesion characterization, with removal of only those lesions strongly suspected to be neoplastic, with the more frequent non-neoplastic lesions left in situ.
The statistical thresholds that represent sufficient accuracy for optical diagnosis remain unclear, with guidelines proposing varying rates of SE (ranging from 80% to 90%), SP (80%), and NPV (90%) to support ‘diagnose and leave’ or ‘resect and discard’ strategies in the non-colitis setting, and considering only diminutive polyps (1–5 mm) in either the recto-sigmoid or entire colon.78,79 These strategies have yet to be validated in IBD, with the failure to demonstrate correlation between lesion size and neoplasia risk in IBD particularly hindering decision making. Notably, it remains unclear which technology to apply for lesion characterization following detection.8,9 Clinical judgment alone, even with experienced endoscopists, is also insufficiently accurate with SE 74% and SP 54%. 80 In the absence of accurate diagnostic criteria, it could therefore be argued that all lesions found during IBD surveillance should be removed and sent for histopathological assessment, which clearly risks overtreatment and resultant complications from procedures such as endoscopic mucosal resection (EMR) or ESD.
Our review provides an important contribution to this debate through the systematic analysis of the many technologies, classifications and single endoscopic predictors for neoplasia in IBD, and the role of the clinical and endoscopic factors that can potentially influence predictive accuracy.
The first take-home message is that in IBD, the criteria used for lesion classification matters much more than the endoscopic imaging technology. This is supported through comparison of the various imaging equipment (Tables 4 and 6), as their accuracy changes significantly when stratified according to the diagnostic criteria used. DCE only meets the minimum accuracy threshold when Kudo classification is used, whereas NBI requires the modified Kudo classification. FICE also shows greater accuracy with Kudo-IBD classification than with conventional Kudo’s.
Conventional Kudo classification appears to have both high SE and NPV that would support a diagnose-and-leave strategy if a lesion can confidently be assessed as non-neoplastic, with pit patterns I–II. However, the low specificity, predominantly due to the high false positive rate of pattern IIIL, significantly hinders useful application and risks overtreatment of inflammatory lesions.
Promisingly, Kudo-IBD classification appears to provide significantly higher SP and therefore better overall accuracy. 42 It should be underlined, however, that the Kudo-IBD classification comes from a single center and still awaits multicenter external validation. Furthermore, it was initially developed with FICE, a technology which has seen reduced investment following the emergence of the novel BLI/LCI. However, two unpublished abstracts suggest that the Kudo-IBD classification may also be applicable with other VCE technologies, such as i-SCAN and NBI.56,57 If the accuracy and reproducibility of the Kudo-IBD classification is validated with multiple endoscopic imaging methods, then it may prove the optimal classification, particularly as the conventional Kudo classification is already widely known with only three additional endoscopic markers required for Kudo-IBD. The FACILE classification also merits particular mention with its use in optical diagnosis of lesions in IBD recommended in recent guidelines. 39 However, in vivo validation is awaited in further studies.
When considering technologies, our review confirms NBI performs poorly when used with conventional classification criteria such as Kudo and NICE, as reported in the very early studies of dysplasia detection in IBD and in a previous systematic review and meta-analysis.81,82 Performance is improved with use of combined surface and vascular pattern criteria (Nishiyama and Kudo-IBD),32,56 and with exclusion of the color component of the NICE classification. These data suggest the need to critically re-evaluate the inclusion of NBI without careful specification of use of endoscopic criteria in current recommendation on optical diagnosis in IBD.
The second take-home message is that no single technology or endoscopic predictor is sufficiently accurate for optical diagnosis of lesions in IBD. It follows that the appropriate diagnostic approach to each lesion encountered during IBD surveillance must be guided by clinical experience and training, and not a rigid application of single technologies or endoscopic factors. The integration of technologies shows particular promise, specifically in vivo ultrastructural lesion analysis with CLE and EC. Indeed, the best performance has been demonstrated with eCLE and Mainz classification, but unfortunately this technology has now been taken off the market. While pCLE technology remains available and is technically easy to apply to any endoscope as it simply involves the use of a probe in the operator channel, the few studies to date in IBD showed lower SE and SP than eCLE. Indeed, pCLE may not be cost effective, with similar accuracy to more popular and established technologies, as previously reported by a meta-analysis conducted in the non-colitis setting. 83 The second and more recent technology that takes advantage of ultrastructural microscopic criteria is EC, which in the only study to date in IBD optimized Kudo’ pit pattern-based classification, significantly increasing SP and maintaining high SE. 45
Regarding clinical and endoscopic factors that may influence diagnostic performance in IBD surveillance, we highlight:
– Endoscopic disease activity, which influences both the prevalence of non-neoplastic lesions, particularly inflammatory, and the interpretation of superficial and vascular patterns, and hence impacts upon the accuracy of optical diagnosis. Consequently, the exclusion of patients with clinical or endoscopic IBD activity from the majority of studies represents a significant bias in the assessment of the true diagnostic accuracy of any technology or endoscopic criteria, with results that may not be applicable to the real-world.
– The need for further studies focusing on endoscopic criteria for optical diagnosis of inflammatory lesions, as these lesions show high false positive rates for neoplasia.
– The scarce data on specific populations of patients at higher risk of malignancy, such as those with PSC, family history of CRC, or previous dysplasia, for whom a different diagnostic approach may be necessary.
– The low number of malignant lesions in the analyzed series. It should however be noted that certain criteria first reported in the non-colitis setting, such as Kudo’s V pattern and Paris depressed IIc morphology, 84 were also frequently associated with malignancy in our IBD series.
– The almost total absence of serrated lesions described according to modern WHO nomenclature in the case series of hyperplastic lesions included in our review.
– The unclear superiority of high-magnification instruments over those with low or no magnification, and the performance of HD alone, as we found no lesion characterization studies with HD-WLE. Although studies suggested high accuracy of non-magnified HD-VCE instruments (e.g. i-SCAN), 35 they were not included in the comparison due to lack of 2 × 2 contingency data.
Finally, operator experience does not appear to play a significant role if the classification criteria for optical diagnosis is clear and correctly applied. It is necessary to optimize training programs with critical appraisal of the technology and classification to be used, 11 and to avoid implementation of strategies that, as shown by our review, do not achieve sufficient accuracy.
Conclusion
In our systematic review, we found that no optical diagnosis classification for colorectal lesions in IBD has been fully validated. The different technologies show variable accuracy depending upon the diagnostic criteria used. Conventional classifications used in the non-IBD setting (such as Kudo, NICE, JNET), or newer classifications (such as FACILE), lack sufficient diagnostic accuracy to support ‘diagnose and leave’ or ‘resect and discard’ strategies required for both resource and outcome optimization. Promising results are suggested by novel classifications adapted to the specific inflammatory scenario of IBD (i.e. Kudo-IBD) and by new technologies based on in vivo microscopic analysis by CLE or EC, whose role deserves further studies in terms of reproducibility, efficiency, and diagnostic efficacy.
