Abstract
Background:
Patients with primary sclerosing cholangitis (PSC) and inflammatory bowel disease (IBD) have an increased risk of developing colorectal neoplasia (CRN) in the proximal colon.
Objectives:
To evaluate whether duration and severity of inflammation are linked to the development of CRN in this population.
Design:
Retrospective, case–control chart review of patients with PSC and IBD at a tertiary care center.
Methods:
Disease activity was scored per colonic segment at each colonoscopy prior to the first instance of observed CRN using a modified Mayo endoscopic sub-score and histologic assessment. Patients in the CRN-positive group were compared to controls that did not.
Results:
In all, 72 PSC-IBD patients with no history of CRN were identified, 13 of whom developed CRN after at least one colonoscopy at our institution. Patients in the CRN-positive group had significantly more endoscopic (p < 0.01) and histologic (p < 0.01) inflammation in the right compared to the control group prior to the development of dysplasia. There was significantly greater endoscopic inflammation in the segment of the colon with a dysplastic lesion than other segments of the colon (p = 0.018). Patients with moderate/severe lifetime endoscopic (p = 0.02) or histologic inflammation (p = 0.04) score had a lower probability of remaining free of dysplasia during follow-up. Nearly half of the patients with dysplasia had invisible lesions found on random biopsy.
Conclusions:
Endoscopic and histologic inflammation in the proximal colon are risk factors for CRN in patients with PSC-IBD. PSC-IBD patients frequently have subclinical inflammation, and these findings support the practice of regular assessment of disease activity and random biopsy of inflamed and uninflamed areas in patients with PSC with the goal of reducing inflammation to prevent the development of CRN.
Plain language summary
Patients with PSC and IBD have not been examined as a cohort to assess for risk factors for CRN. We found that severe inflammation in the proximal colon is the main risk factor for CRN in these patients.
Keywords
Introduction
Patients with inflammatory bowel disease (IBD) are at an increased risk of developing colorectal cancer (CRC) and precursory colonic dysplasia, together described as colorectal neoplasia (CRN).1,2 Many risk factors for the development of CRN in patients with IBD have been identified. The duration, severity, and extent of inflammation, as well as a younger age of diagnosis are all associated with an increased risk of developing CRN3–7 in cohorts of IBD patients.
Primary sclerosing cholangitis (PSC), a disease process of the biliary system characterized by biliary duct inflammation and fibrosis, is associated with IBD in 70–90% of cases.8–10 Patients with PSC-IBD have a distinct phenotype characterized by pancolitis, backwash ileitis, and rectal sparing.8,11,12 Notably, patients with IBD and PSC often have subclinical inflammation, primarily in the proximal colon, and mild symptoms, which may lead to a delay in diagnosis or lack of treatment escalation.13,14
In a meta-analysis of 16 observational studies, PSC was associated with an increased risk for the development of dysplasia and CRC in patients with IBD, with an odds ratio of 3.24. 15 As a result, it is recommended that patients with PSC-IBD undergo annual colonoscopy for surveillance. 16 The exact mechanism by which PSC heightens the risk of CRN in the context of IBD is unknown. Moreover, it is unknown whether the predictors of CRN in IBD cohorts also apply to patients with PSC. As such, we examined a population of patients with PSC and IBD and assessed whether factors predictive of CRN in IBD, extent of disease, duration of disease, age of diagnosis, and severity of disease, would also apply to patients with PSC.
Materials and methods
Patient selection
The Institutional Review Board (IRB) of the University of Chicago (protocol IRB 13-1080) approved the study. All clinical features and values were retrospectively identified and extracted from patients’ electronic medical records. Patient-level data included demographic data, disease activity, disease history, and medication usage. Encounter-level data included endoscopic inflammation scores, histological inflammation scores, and histological neoplasm grades. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist for case–control studies was used to prepare the manuscript. 17
We identified 133 patients with PSC-IBD with a total of 516 encounters for colonoscopy procedures between 1997 and 2019. Patients who underwent only one colonoscopy at our institution as well as those who had CRN detected either prior to their first colonoscopy at our institution or at the time of their first colonoscopy at our institution were excluded from the analysis. It is standard of care at our institution for patients at with PSC-IBD to undergo annual surveillance colonoscopy with random segmental biopsies and targeted biopsies of visible lesions. In 2005, our institution began using high-definition (HD) endoscopes for all exams. A majority of the endoscopic exams in the entire cohort (88%) were completed after the HD endoscopes were introduced. 18 All but two control patients had at least one endoscopic exam completed by HD endoscope. Chromoendoscopy was not used in this cohort. Two patients had lesions indefinite for dysplasia in the setting of histologic inflammation who were found to have no dysplasia on subsequent exams with healing of active histologic inflammation. These two patients were excluded from the analysis. After excluding patients who did not meet the eligibility criteria, 72 patients, including 13 patients who developed neoplasia (cases) and 59 controls, were included in the analysis (Figure 1). All endoscopic examinations were included in the analysis for controls.

Patient inclusion flow diagram.
Endoscopic and histologic scoring
Endoscopic and histologic disease activity was recorded in the cecum, ascending colon, hepatic flexure, transverse colon, splenic flexure, descending colon, sigmoid colon, and rectum. The cecum, ascending colon, hepatic flexure, and transverse colon were grouped as the right or proximal colon. Similarly, the splenic flexure, descending colon, and sigmoid colon were grouped as the left colon. When discussed as a group, the average of each segment was used. The endoscopic and histologic scores of the rectum were analyzed independently of the left colon. All polyps were removed and assessed histologically, and all patients underwent random biopsy throughout the colon during withdrawal regardless of the presence of inflammation.
For endoscopic scoring, an ordinal score (0 = no inflammation, 1 = mild, 2 = moderate, and 3 = severe) was assigned to each segment using a previously described methodology.2,13 Similarly, a histologic ordinal score was assigned to each segment (0 = normal, 1 = quiescent, 2 = mild, 3 = moderate, and 4 = severe) using histologic criteria as previously described.13,19,20 At our institution, the endoscopist routinely grades the severity of inflammation based on the Mayo endoscopy score, 19 and gastrointestinal pathologists use a standardized scoring system as previously described.13,20 If findings were ambiguous for a given segment based on the endoscopic or histologic report, an experienced gastroenterologist with specialization in IBD reviewed the endoscopic report and images, and a pathologist with expertise in GI pathology reviewed all pathology before assigning a final score. When neoplasia was found, a second pathologist reviewed the slides to confirm the findings.
When assessing time to neoplasia, the mean inflammation score for cases prior to developing neoplasia and controls was compared to the lifetime duration of IBD in years. For this analysis, the cohort was broken into clinically useful dichotomous groups, patients with average endoscopic inflammation of normal/mild (⩽1) versus moderate/severe (>1) and average histologic inflammation of quiescent/mild (⩽2) versus moderate/severe (>2) based on the ordinal scoring system described above.
Statistical analysis
Statistical analyses were performed using the R version 4.1.0. 21 Comparison of baseline patient variables between the two groups was performed using the student’s t-test for continuous variables and the chi-squared test for categorical data. The association between the risk of CRN and endoscopic and histologic disease activity was tested using the chi-squared test and t-test. Time to neoplasia was assessed with log-rank survival analysis using the Kaplan–Meier method.
Results
Characteristics of study population
A total of 72 patients with concomitant IBD and PSC were included in the analysis. Among these patients, 13 (18.1%) developed CRN on one or more exams after their baseline examination without neoplasia, and 59 (81.9%) patients with at least two negative endoscopic exams did not develop CRN during the follow-up period. Of the patients who developed neoplasia, 1 was diagnosed with adenocarcinoma, 2 were diagnosed with high-grade dysplasia, and 10 were diagnosed with low-grade dysplasia. Representative endoscopic and histologic images of visible neoplastic lesions are shown in Supplemental Figure 1. The control group had an average of 5.14 colonoscopy exams, while the case group had an average of 5.38 exams (p = 0.785). Two patients developed neoplasia at multiple locations. Of the 13 cases, 6 patients developed neoplasia in the right colon, 2 in the left colon, 3 in the rectum, and 2 patients had lesions both in the left colon and rectum. Of the dysplastic lesions identified, seven were polypoid (54%), five were invisible neoplasia found on random biopsy in an area of inflammation (38%), and one was invisible neoplasia found in an area that otherwise appeared endoscopically normal (8%).
As seen in Table 1, there was no difference in disease extent, age at IBD diagnosis, age at PSC diagnosis, IBD duration, follow-up time (first endoscopic exam until the last endoscopic examination in the control group, and the first endoscopic exam until the last endoscopic exam without neoplasia in the case group), number of endoscopic examinations or medication usage between PSC-IBD patients with or without neoplasia.
Patient cohort characteristics.

IBD, inflammatory bowel disease; PSC, primary sclerosing cholangitis.
The lifetime use of ursodiol, which has previously been observed to have a chemoprotective effect in PSC-UC patients, 22 was not significantly different between the two groups (p = 0.171) in this cohort. The use of other therapies (biologics, immunomodulators, and 5-aminosalicylic acid) was also not statistically different between the two groups.
Inflammation prior to the diagnosis of neoplasia
When comparing mean inflammatory scores from endoscopic exams prior to the development of neoplasia in the cases to controls, the cases were found to have significantly more endoscopic inflammation in the right colon (p < 0.001) seen in Figure 2. In contrast, there were no statistically significant differences between controls and cases in endoscopic severity in the left colon or rectum. Mean histologic inflammation was also significantly higher in cases than in controls prior to the diagnosis of neoplasia in both the right (p < 0.001) and left colon (p = 0.03) also seen in Figure 2.

Comparing endoscopic and histologic inflammation in cases and controls prior to the development of neoplasia in all segments of the colon.
Mean endoscopic and histologic inflammation in the segment of the colon that subsequently harbored neoplasia was also compared to inflammatory scores in the same segment in the control group. Prior to the development of neoplasia, the cases with lesions in the right colon had significantly more endoscopic and histologic inflammation than controls in the right colon (p < 0.001) seen in Figure 3. In contrast, the severity of endoscopic and histologic inflammation did not differ in the left colon in patients who subsequently developed neoplasia in this segment compared to controls. Patients with lesions in the rectum were found to have no difference in endoscopic inflammation in the rectum prior to developing neoplasia but had significantly more histologic inflammation (p = 0.003) also seen in Figure 3.
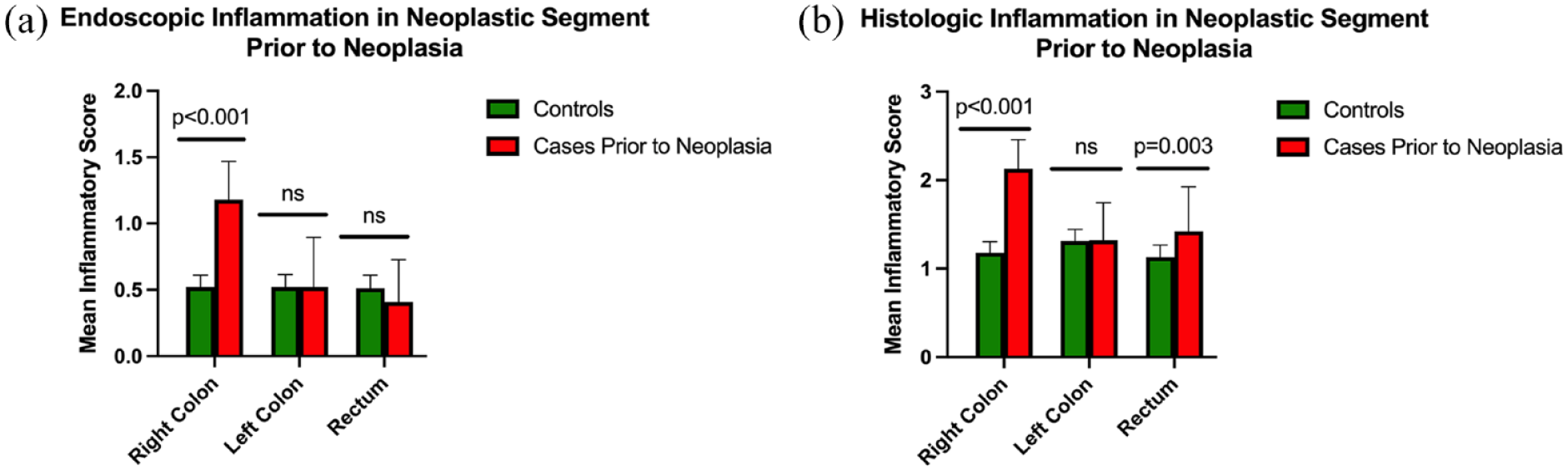
Comparing endoscopic and histologic inflammation in cases and controls prior to the development of neoplasia in only the segment with the dysplastic lesion.
Inflammation at the time of neoplasia diagnosis
To assess the impact of active inflammation during neoplasia detection, endoscopic and histologic inflammation were also evaluated at the time of diagnosis of neoplasia. There was no significant difference in endoscopic (p = 0.112) or histologic (p = 0.665) inflammation at the time of neoplasia in cases when compared to prior exams. At the time neoplasia was diagnosed, however, there was more endoscopic inflammation in the segment of the colon with the dysplastic lesion compared to the other segments of the colon during the same examination (p = 0.018).
Time to neoplasia
To evaluate the impact of mucosal inflammation on time to neoplasia, subjects with an average lifetime endoscopic or histologic inflammatory score of less than or equal to mild were compared to those with a mean score of moderate/severe using the Kaplan–Meier method. Having moderate or severe endoscopic (p = 0.02) or histologic (p = 0.04) inflammation was associated with a higher probability of developing neoplasia seen in Figure 4.
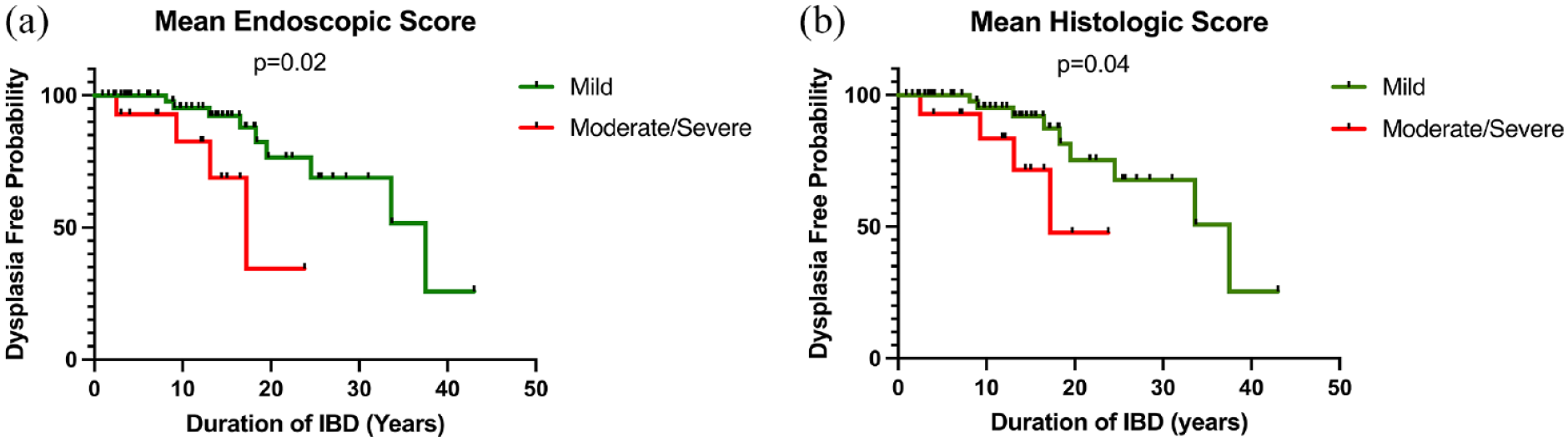
Comparing time to dysplasia between patients with mild versus moderate/severe disease according to duration of disease.
Discussion
In this retrospective study evaluating patients with concomitant IBD and PSC, more severe endoscopic and histologic inflammation in the proximal colon was identified as a risk factor for the development of future neoplasia at any location in the colon. In addition, endoscopic and histologic inflammation in the proximal colon increased the risk for future CRN in this segment in PSC patients. This is the first study to examine risk factors for colon neoplasia in patients with PSC-IBD. These results have important clinical implications for risk stratification, surveillance, and treatment in this population.
Although recognized as an independent risk factor itself, most studies examining the risk factors for neoplasia in IBD have not focused on patients with PSC.23,24 These previous analyses identified disease extent, duration of disease, age at diagnosis, and severity of inflammation as independent risk factors for CRN in IBD patients without PSC. 25 In our cohort of IBD patients with PSC, the severity of endoscopic and histologic inflammation was the only factor strongly associated with the development of future neoplasia. Other clinical features previously identified as risk factors for neoplasia in IBD patients were not significant in this analysis.
While PSC is considered an independent risk factor for the development of CRN among patients with IBD, the etiology of this pathogenesis remains unknown. One hypothesis regarding the high rate of CRN in patients with PSC is that long-standing subclinical inflammation in PSC-IBD patients is present in the proximal colon before the diagnosis of IBD is made and annual endoscopy initiated.26,27 In addition, clinicians may be less likely to adjust therapy in patients with active inflammation in the absence of clinical symptoms. Although other biologic mediators unique to PSC are likely to be important in the development of neoplasia, these clinical factors may contribute to uncontrolled inflammation, increasing an individuals’ risk of CRN. In this study, nearly half of the patients that developed neoplasia were otherwise invisible dysplastic lesions found on random biopsy. These lesions can easily be missed and may even be brewing prior to the presence of endoscopic inflammation.
Our study was limited by the number of available subjects for analysis and would benefit by additional analyses from other institutions to both increase numbers, but also to account for potential geographic variation. In order to power the study, patients from 1997, prior to the introduction of narrow band imaging and enhanced imaging modalities, were included. 28 As a result, some lesions that may be visible under advanced imaging modalities today may have been described as invisible lesions. We were unable to assess the impact of healing of active inflammation on the risk of neoplasia given the size of the cohort. Lastly, the endoscopic and histologic scoring systems employed in the study used non-validated scoring measures. However, these descriptions of inflammation have been standardized in our institution.
Conclusion
IBD patients with PSC with more severe endoscopic and histologic inflammation in the proximal colon are at an increased risk of developing CRN. The fact that we identified an association between localized mucosal inflammation and neoplasia risk in the proximal colon but no other colonic segments supports the notion that other biologic factors are also likely to be important in the development of CRN in PSC. A second important conclusion from the study is that patients had more significant endoscopic inflammation in the area of neoplasia on examinations when neoplasia was diagnosed, even when the dysplastic lesions were often invisible. Based on these findings, we recommend that clinicians take particular care to take random biopsies throughout the colon on every exam in these high-risk patients regardless of clinical symptoms.
Supplemental Material
sj-tif-1-tag-10.1177_17562848231184985 – Supplemental material for Inflammation in the proximal colon is a risk factor for the development of colorectal neoplasia in inflammatory bowel disease patients with primary sclerosing cholangitis
Supplemental material, sj-tif-1-tag-10.1177_17562848231184985 for Inflammation in the proximal colon is a risk factor for the development of colorectal neoplasia in inflammatory bowel disease patients with primary sclerosing cholangitis by Omar K. Jamil, Dustin Shaw, Zifeng Deng, Nicholas Dinardi, Natalie Fillman, Shivani Khanna, Noa Krugliak Cleveland, Atsushi Sakuraba, Christopher R. Weber, Russell D. Cohen, Sushila Dalal, Bana Jabri, David T. Rubin and Joel Pekow in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The project was conducted in collaboration between the Inflammatory Bowel Disease Center and Digestive Diseases Research Core Center at The University of Chicago Medicine, Chicago, IL, USA.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
