Abstract
Objective
The vascular anatomic variations of the right colon present a challenge for colorectal surgeons. However, there have been few detailed studies of the variations in the anterosuperior pancreaticoduodenal vein (ASPDV).
Methods
We studied consecutive patients with right colon cancer who underwent laparoscopic right hemicolectomy at Peking University First Hospital (N = 117) between January 2018 and June 2021.
Results
The variations in the ASPDV were classified as type I (n = 101, (86.3%)), defined as ASPDVs draining into the gastrocolic trunk of Henle (GCT); type II (n = 10, (8.5%)), defined as ASPDVs draining into the superior mesenteric vein (SMV); or type III, defined as ASPDVs draining into both the GCT and SMV. For type I, subtypes were defined according to the branching of the ASPDVs: subtype a, with one branch (n = 87, (86.1%)); subtype b, with two branches (n = 12, (11.9%)); and subtype c, with more than two branches (n = 2, (2.0%)). Type I was also subtyped according to the confluence of the ASPDV and GCT, with subtype 1 being defined by a proximal site (n = 96, 95%) and subtype 2 by a distal site (n = 5, 5.0%).
Conclusions
We have characterized the variations in ASPDVs encountered during laparoscopic right hemicolectomy, which should provide a reference for colorectal surgeons.
Keywords
Introduction
Colorectal cancer (CRC) has become the third most common type of cancer and the second leading cause of cancer-associated mortality. 1 Laparoscopic mesocolic excision and central vascular ligation remains the standard treatment. 2 However, the numerous variations in the vessels encountered during laparoscopic right hemicolectomy may predispose toward significant hemorrhage, and the risks may be greater for inexperienced surgeons. Accidental hemorrhage associated with aberrant variations in such vessels is a common reason for the open conversion of laparoscopic colectomies. 3 There have been previous studies of variations in these arteries and the topographical relationships between the common arteries and veins that are encountered during right hemicolectomy.4,5 The gastrocolic trunk of Henle (GCT) is present in 70% and 100% of patients, and is an important anatomical landmark for colorectal surgeons. 6 However, few previous studies have characterized the anatomical variations in the GCT, and existing knowledge is principally derived from findings obtained from studies of cadavers and computed tomography and radiologic imaging.7 –9 In addition, the anterosuperior pancreaticoduodenal vein (ASPDV) is a common contributor to the GCT, and variants of these veins may increase the challenge associated with right hemicolectomy and predispose toward significant hemorrhage.10,11 However, the variations in the ASPDV have not been characterized in detail during laparoscopic studies.
In the present study, we aimed to retrospectively review high-resolution videos of laparoscopic right hemicolectomy procedures and document the observed variations in ASPDVs. We consider that a detailed characterization and classification of ASPDVs would help reduce the risk of unexpected hemorrhage during laparoscopic right hemicolectomy.
Methods
Study design
The reporting of this study conforms to the STROBE (V4) guidelines. 12 Patients who underwent radical laparoscopic right hemicolectomy at the Division of General Surgery of Peking University First Hospital were enrolled between May 2018 and July 2021. This study was approved by the institutional review board (approval number 2018022; 6 February 2018) and all the procedures were performed according to the ethical principles of the relevant clinical studies committee and the Declaration of Helsinki.
The procedures included were consecutively performed by a single team of surgeons at Peking University First Hospital. The following were used as inclusion criteria: (1) pathologically confirmed malignant colonic lesion; (2) tumor located at the ileocecal junction, ascending colon, or hepatic flexure; (3) standard D3 lymph node dissection and central vascular ligation were undertaken according to the principles of complete mesocolic excision; and (4) laparoscopic hemicolectomy was performed between May 2018 and July 2021. The following were used as exclusion criteria: (1) video records were unclear or incomplete; (2) blurred video records, because of hemorrhage or excessive fat; and (3) palliative, rather than radical hemicolectomy was performed.
Surgical technique
A standard five-port caudal-to-cranial approach was used, with the patients in a dorsal elevated, left-tilted position. 13 The small intestine and mesentery were reflected into the left lower abdomen to expose the surgical field. The mesentery of the ascending colon and ileocecal junction was released and Toldt’s gap was subsequently exposed. After extending Toldt’s gap, the right ureter and reproductive vessels were exposed and secured. Toldt’s gap was then further extended to the level of the descending, horizontal part of the duodenum and pancreatic head. Endoscopic gauzes were used to secure the pancreas. The patients were then positioned with their head elevated. The anterior layer of the ascending colon mesentery was dissected, following the right margin of the superior mesenteric artery (SMA). The superior mesenteric vein (SMV) and GCT were subsequently exposed. The lymph nodes and adipose tissue located at the roots of the ileocolonic vessels were dissected and the vessels were clamped and transected. The path of the SMV was then exposed, followed by the roots of both the SMV and GCT.
After securing the gastric and pancreatic branches of the GCT, the colonic branches of the GCT were clamped and transected. The middle colic vessels were then exposed and the lymphatic tissue in the roots of these vessels was dissected. The right branch of the middle colic artery (MCA) was clamped and transected at its root and the left branch was preserved. The middle colic vein was then clamped and transected at its root. The gastrocolic ligament was lifted up and divided outside the gastro-omental arch. The descending duodenum was then further exposed and the ligamentum hepatocolicum was divided. After closing the peritoneum, a 5-cm median incision was made above the navel to remove the right colon (including the mesocolon, a 15-cm length of the proximal ileocolic mesentery, and sufficient small intestine), and side-to-side anastomosis of the ileum and transverse colon was then performed.
Characterization of the vascular anatomy
The high-resolution videos of the procedures were reviewed by experienced surgeons and screenshots showing the spatial relationships of the vessels were collected and labeled. The data were reviewed and the vascular anatomy was analyzed by three experienced surgeons (Wu YC, Wang X, and Pan YS). In the present study, there were no disagreements regarding the variations in the ASPDVs among the reviewers. The clinical and pathologic characteristics of the patients were collected from their medical records after obtaining their written informed consent.
Statistical analysis
SPSS v.22.0 (IBM Corp., Armonk, NY, USA) and Prism v.8.0 (GraphPad, San Diego, CA, USA) were used for statistical analysis. Continuous data are expressed as mean ± standard deviation (SD) and categorical data are expressed as percentages. P < 0.05 was accepted as indicating statistical significance.
Results
Patient demographics
The clinical and pathological characteristics of the 117 of 126 patients who fulfilled the inclusion and exclusion criteria and were included in the analysis are listed in Table 1. Four patients were excluded because their video records were unclear or incomplete, three because of blurred video records, and two because palliative hemicolectomy was performed. The average age of the patients was 63.8 years. The patients consisted of 67 men (57.3%) and 50 women (42.7%), and 22 had a previous history of abdominal surgery (18.8). Nineteen tumors were located in cecum (16.2%), 83 in the ascending colon (70.9%), 13 in the hepatic flexure (11.1%), and two in the appendix (1.7%).
Characteristics of the 117 participants, who underwent laparoscopic right hemicolectomy.

Complete mesocolic excision (CME) and central vascular ligation were performed in all the patients, using the caudal-to-cranial route, as described in the Methods. The duration of surgery was 168.2 minutes and the mean estimated blood loss was 64.2 mL. The mean duration of hospital stay of the patients was 7.6 days, and severe complications were relatively rare. The mean number of lymph nodes collected was 31.5 and the mean number of pathologically confirmed metastases in the lymph nodes was 2.6.
Classification of the variations in the ASPDVs
The variations in the ASPDV were classified according to its drainage. Type I was defined as an ASPDV draining into the GCT (n = 101, (86.3%)). Subtypes of ASPDV in type I were further defined according to the site of confluence with the GCT and the number of branches. According to the number of branches of the ASPDV in type I, subtype a was defined as only one branch present (n = 87 (86.1% of Type I)) (Figure 1a), subtype b as two branches (n = 12 (11.9% of Type I)) (Figure 1b), and subtype c as more than two branches (n = 2 (2.0% of Type I)). (Figure 1c and d). Type II was defined as an ASPDV draining directly into the SMV (n = 10 (8.5%)) (Figure 1e), and type III was defined as an ASPDV draining into both the GCT and SMV (n = 6, (5.2%)) (Figure 1f). According to the site of confluence of the ASPDV with the GCT in type I, subtype 1 was defined as a proximal site, with the confluence of the ASPDV with the GCT on the left side of the midline between the medial border of the descending duodenum and the right border of the SMV (n = 96, 95% of Type I) (Figure 2a); subtype 2 was relatively rare and was defined as a distal site of confluence on the right side of the midline described above (n = 5, 5.0% of Type I) (Figure 2b). In subtype 1, the ASPDV joined the GCT directly, whereas in subtype 2, the ASPDV joined the superior right colic vein (SRCV).
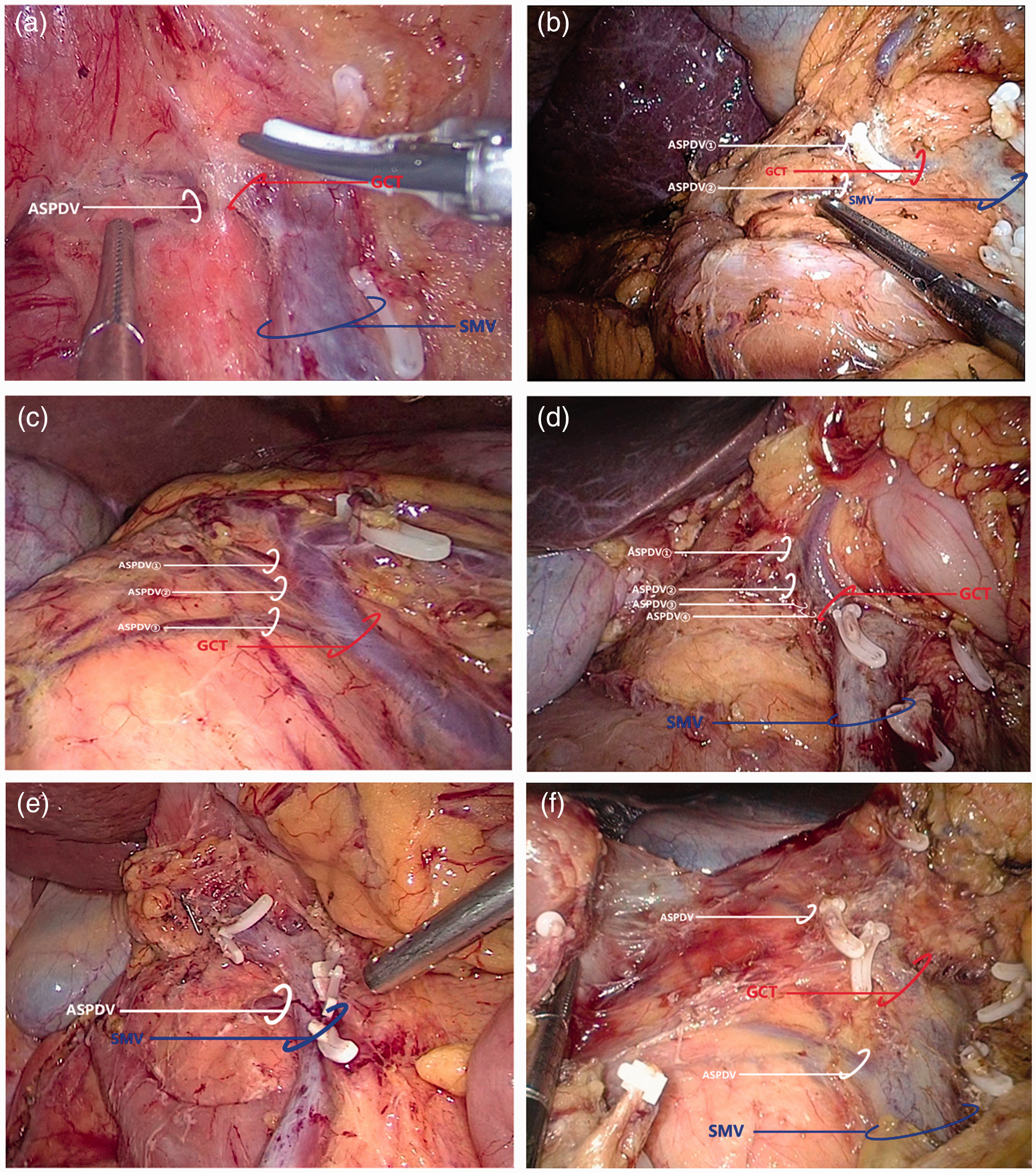
Subtypes of the anterosuperior pancreaticoduodenal vein (ASPDV) type I, according to the number of branches of the ASPDV and their confluence with the superior mesenteric vein (SMV). (a) subtype a, with only one branch; (b) subtype b, with two branches; (c) subtype c, with more than two branches (three branches were present in this case); (d) subtype c, with more than two branches (four branches were present in this case); (e) ASPDV draining directly to the SMV; (f) ASPDV draining to both the gastrocolic trunk of Henle (GCT) and SMV.

Subtypes of the anterosuperior pancreaticoduodenal vein (ASPDV) type I, according to the site of confluence. (a) subtype 1, with a proximal confluence of the ASPDV with the gastrocolic trunk of Henle (GCT); (b) subtype 2, with a distal confluence of the ASPDV with the superior right colic vein (SRCV).
A schematic representation of the variations in the ASPDV, with the proportions of each of these variations, is shown in Figure 3.
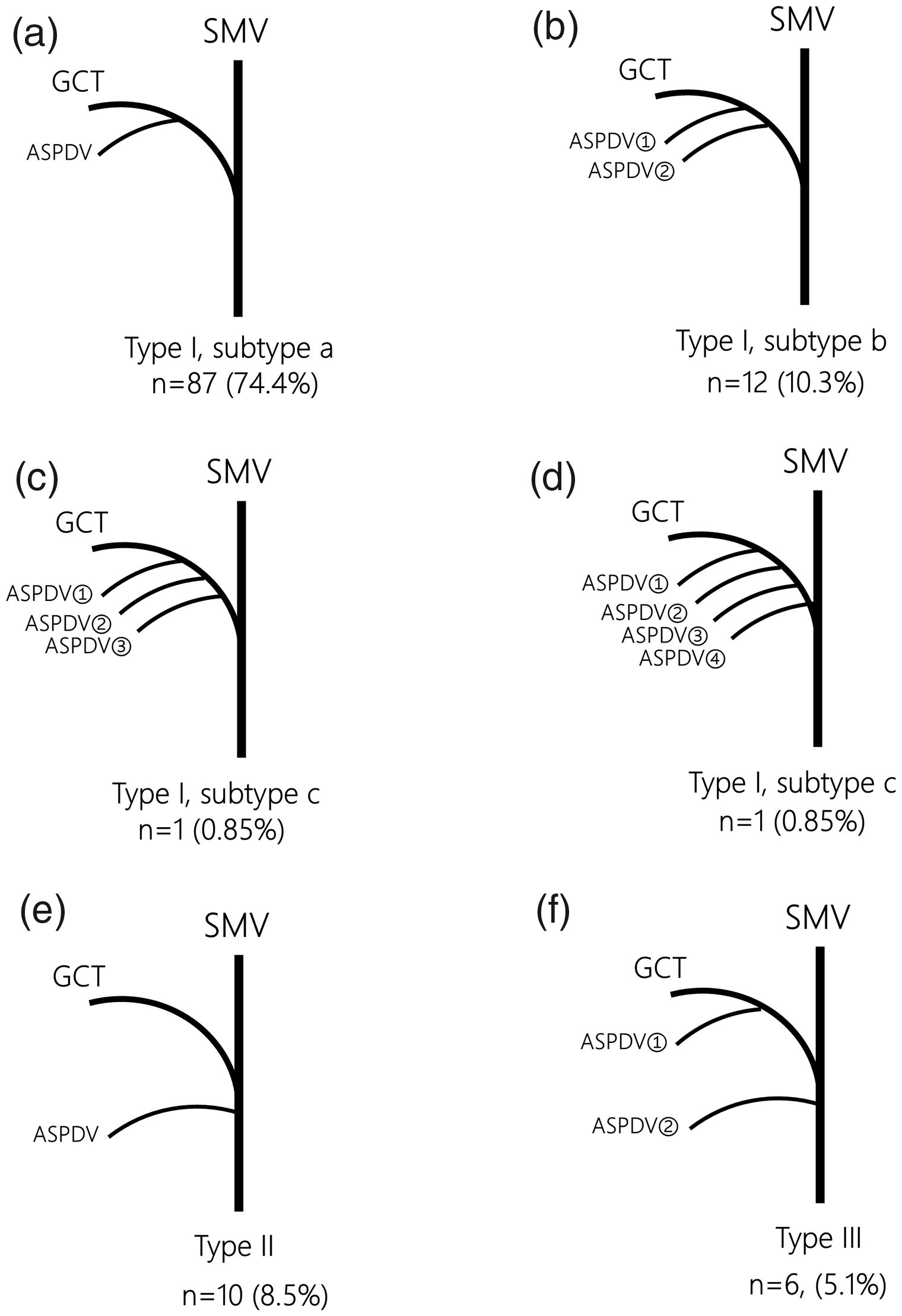
Schematic representation of the subtypes of anterosuperior pancreaticoduodenal vein (ASPDV). (a) Type I subtype a, with only one branch; (b) Type I subtype b, with two branches; (c) and (d) subtype c, with more than two branches (three branches were present in the case described on the left and four in the case described on the right); (e) Type II, with the ASPDV draining directly to the superior mesenteric vein (SMV); (f) Type III, with the ASPDV draining to both the gastrocolic trunk of Henle (GCT) and the SMV.
Discussion
Previous studies performed on cadavers have shown a prevalence of GCT of 70% to 100%. 7 Successful right hemicolectomy requires the clear anatomical identification of the vasculature and variations in this, especially of GCT and its tributaries, because this has a direct effect on the success of the procedure. The GCT is classically described as being formed by the confluence of the right gastro-omental vein and right colic vein, with the ASPDV and ileocolic vein being potential tributaries. 14 The variation in the GCT and its tributaries, sometimes including the ASPDV, is a common obstacle for CRC surgeons performing laparoscopic right hemicolectomy. 15 Previous studies have shown that the ASPDV converges with the GCT in more than half of patients who have noticeable vascular variants. 16
In the present study, we observed that the ASPDV drained directly to the SMV in 8.5% of patients and to both the GCT and SMV in 5.2% of the cohort. In the remaining 86.3% of the present cohort, the ASPDV drained exclusively to the GCT, either directly or via the SRCV. Securing the ASPDV is often essential for clear identification of the colic and gastro-omental tributaries of GCT. Massive hemorrhage can be the result of over-traction or accidental incision of the GCT or its tributaries. 5 Attempts have been made in the past to classify and subtype the variations in the GCT.17,18 However, a detailed characterization of the variations in the ASPDV was still lacking. We reasoned that classification of the variants in the ASPDV, both in terms of the number of branches and site of confluence with the GCT or SMV would greatly advance knowledge of the surgical anatomy of the ASPDV. Rare variations in the ASPVD, including the presence of more than two branches and a confluence with both the GCT and SMV, have been revealed by previous studies and the present data, and may increase hemorrhage caused by vessel injury.18,19 Considering the complex spatial anatomic relationships between the ASPDV and the adjacent vessels and organs, the present findings might aid CRC surgeons to avoid accidental hemorrhage related to variations in the ASPDV.
Additional precision and caution are necessary in the exposure and ligation of GCTs in patients with Type I subtypes b/c and 2, referring to multiple branches of the ASPDV and distal confluence with the SRCV, respectively. Owing to the infrequency and relatively short distance of the ASPDV from its start at the pancreas to its confluence with the SRCV, subtype 2 may be associated with a higher risk of hemorrhage if there is a lack of care and/or a limited field of vision; therefore, special attention should be paid to this rare variant. The risks of massive hemorrhage and conversion to open surgery might be increased by the presence of these rare variations.
In conclusion, we have provided new insight into the anatomical variations in the ASPDV that are encountered during laparoscopic right hemicolectomy. We have subtyped the ASPDV according to the number of branches and site of confluence, including rare cases with four branches and a distal site of confluence with the SRCV. Understanding these variations should assist CRC surgeons to minimize the risks of hemorrhage and the need for conversion to open surgery while performing laparoscopic right hemicolectomy.
