Abstract
Background:
Potassium-competitive acid blocker-based regimens, such as tegoprazan, have demonstrated promising efficacy for Helicobacter pylori eradication. In regions with high antibiotic resistance, such as Gansu Province, bismuth-containing quadruple therapy (BQT) remains the recommended first-line treatment. However, evidence on the efficacy and safety of high-dose tegoprazan–amoxicillin dual therapy (TA) remains limited.
Objectives:
To compare the eradication rates and safety profiles of TA and BQT.
Design:
Prospective, multicentre, randomised controlled trial.
Methods:
Patients were enrolled from 10 centres across Gansu Province, China, between September and December 2024. Participants were randomised 1:1 to receive either BQT (esomeprazole 20 mg twice daily + bismuth potassium citrate 600 mg twice daily + amoxicillin 1000 mg twice daily + metronidazole 400 mg four times daily) or TA (tegoprazan 50 mg twice daily + amoxicillin 750 mg four times daily) for 14 days. The primary outcome was eradication rate. Secondary outcomes included adverse event (AE) rates, treatment compliance and cost-effectiveness.
Results:
A total of 288 patients were randomised, with baseline characteristics well balanced between the TA and BQT groups (p > 0.05). Eradication rates for TA versus BQT were 68.06% (98/144) versus 81.25% (117/144) (difference, −13.19%; 95% confidence interval (CI): −22.95 to −3.43; p = 0.009; non-inferiority p = 0.736) in the intention-to-treat analysis; 69.01% (98/142) versus 81.82% (117/143; difference, −12.81%; 95% CI: −22.70 to −2.90; p = 0.011; non-inferiority p = 0.711) in the modified intention-to-treat analysis; and 72.59% (98/135) versus 89.31% (117/131; difference, −16.72%; 95% CI: −26.01 to −7.43; p < 0.001; non-inferiority p = 0.924) in the per-protocol analysis. AEs were less frequent with TA (8.30% vs 25.70%, p < 0.0001), with similar compliance rates (95.07% vs 91.61%, p = 0.241). Multivariate analysis indicated that long-term residence in the southern region of Gansu Province (including Longnan City and Tianshui) was significantly associated with higher eradication rates compared with the Hexi region (adjusted odds ratio = 5.46, p = 0.04), suggesting that geographical factors independently influenced treatment outcomes.
Conclusion:
TA did not achieve non-inferiority to BQT and yielded significantly lower eradication rates. The inferior eradication efficacy of TA precludes its recommendation as a first-line therapy in high-resistance settings, such as Gansu.
Trial registration:
This trial was registered on the Chinese Clinical Trial Registry with the registration number ChiCTR2400081873.
Keywords
Background
Helicobacter pylori (H. pylori) is a globally prevalent pathogen, infecting approximately 4.4 billion people worldwide. In China, the overall infection rate is estimated to be 50%. 1 The northwestern regions, particularly Gansu Province, have a significantly higher prevalence, with an infection rate of 57.2%, ranking third nationwide. 2 Gansu also has the highest incidence and mortality rates of gastric cancer in China. 3
Strong evidence links H. pylori infection to a wide spectrum of gastrointestinal diseases, including gastric adenocarcinoma, peptic ulcers, chronic active gastritis and gastric mucosa-associated lymphoid tissue lymphoma, as well as various extraintestinal conditions.4,5 Current international and national guidelines recommend eradication therapy for all individuals infected with H. pylori, regardless of the presence or absence of symptoms or complications.6–8 However, the marked increase in antibiotic resistance has led to a decline in global eradication rates. Primary resistance to clarithromycin has increased by more than 10% over the past decade in Europe (reaching 22.4%) and exceeds 90% in certain Asian regions9,10 Levofloxacin resistance also shows wide geographical variation, surpassing 40% in North America and 35% in China, thereby restricting treatment options and thereby limiting eradication effectiveness.11,12 The American College of Gastroenterology (ACG) guidelines, the Toronto Consensus, and the Maastricht VI/Florence Consensus recommend a 14-day bismuth-containing quadruple therapy (BQT) as the first-line treatment in regions where clarithromycin resistance exceeds 15%.6,7,13–15 Similarly, the Sixth Chinese National Consensus Report on H. pylori management supports BQT as an empirical therapy for both initial treatment and retreatment. 16 Although BQT has been shown to achieve high eradication rates,17–19 it has certain limitations: regional unavailability of key drugs such as bismuth, tetracycline or furazolidone; complex dosing regimens; frequent adverse events (AEs); reduced patient compliance; and relatively high costs. 20 Moreover, treatment failure with BQT may promote further antibiotic resistance, thereby limiting future therapeutic options. 21 These challenges highlight the urgent need for novel regimens with high efficacy, reduced antibiotic burden and improved tolerability.
High-dose dual therapy (HDDT) is a promising regimen. Randomised controlled trials (RCTs) conducted in Turkey and China have demonstrated that HDDT, using high-dose amoxicillin (AMX) and a proton pump inhibitor (PPI), achieves comparable eradication rates and safety profiles comparable with those of BQT.22,23 Effective acid suppression is essential for successful HDDT. Although PPIs are widely used, their efficacy is limited by several pharmacokinetic drawbacks.24,25 Amoxicillin’s bactericidal activity is time-dependent, relying on prolonged serum levels above the minimum inhibitory concentration (MIC).26,27 Increasing the dosing frequency from twice daily to three or four times daily extends the time above the MIC and enhances effectiveness. Additionally, as a pH-dependent antibiotic, amoxicillin requires the gastric pH to be maintained at approximately 6 for optimal activity against H. pylori.28,29 Therefore, maximising eradication requires a combined strategy of increased dosing frequency, optimised dosage and sustained intragastric pH control. However, PPIs have intrinsic limitations; they are prodrugs requiring activation in an acidic environment, have a delayed onset and short plasma half-lives, and often fail to suppress nocturnal acid production. Furthermore, their metabolism through the cytochrome P450 (CYP) 2C19 pathway is influenced by genetic polymorphisms – particularly prevalent among Asian populations – which may result in suboptimal acid suppression in rapid metabolisers.30,31
Potassium-competitive acid blockers (P-CABs), such as tegoprazan, offer potential solutions. These agents directly inhibit both active and inactive H⁺/K⁺-ATPase pumps, providing rapid, potent and sustained acid suppression that is independent of CYP2C19 metabolism.32,33 In a meta-analysis of 15 RCTs, vonoprazan, a first-generation P-CAB, demonstrated superior efficacy in H. pylori eradication with intention-to-treat (ITT) and per-protocol (PP) eradication rates of 85.0% and 90.0%, respectively. 34 Tegoprazan, a second-generation P-CAB, was approved in South Korea in 2018 for the treatment of acid-related disorders, including gastro-oesophageal reflux disease (GERD), erosive oesophagitis and non-erosive reflux disease.35–37 Tegoprazan-based BQT has achieved eradication rates exceeding 95.8% in PP analyses, meeting current guideline recommendations.38,39 Recent multicentre RCTs conducted in several regions of China – including Chongqing, Sichuan and Shandong – have shown that tegoprazaná–amoxicillin HDDT is non-inferior to BQT in terms of efficacy and safety,40,41 suggesting it as a well-tolerated and effective alternative. International guidelines (Maastricht VI/Florence Consensus Report and ACG) also include HDDT with a P-CAB combined with amoxicillin as a first-line treatment recommendation.14,15 The latest Chinese guidelines affirm the efficacy and potential of this regimen. 42
Despite these promising developments, data on tegoprazan–amoxicillin dual therapy (TA) in high-resistance regions, such as Gansu Province, remain limited. Gansu is characterised by a high prevalence of H. pylori, elevated antibiotic resistance, and relatively poor eradication outcomes.2,43,44 Therefore, this multicentre RCT was designed to evaluate the efficacy and safety of tegoprazan–amoxicillin HDDT compared with BQT for H. pylori eradication in Gansu Province. These findings will inform the development of optimised treatment strategies in regions with high resistance rates.
Materials and methods
Study design
This prospective, multicentre, open-label, RCT was conducted across 10 centres in Gansu Province, China, between September and December 2024. The trial was conducted in accordance with the ethical principles of the Declaration of Helsinki. Reporting adhered to the CONSORT 2025 guidelines (see Supplemental Material). 45
Study participants
Inclusion criteria were as follows: (1) Age: 18–70 years, male or female; (2) Patients confirmed positive for H. pylori by urea breath test (UBT); (3) Treatment-naïve patients for H. pylori infection; (4) Patients or their legal representatives have signed the informed consent form, understanding and agreeing to comply with the study requirements. Exclusion criteria: (1) Use of PPIs, P-CABs, antibiotics, H2-receptor antagonists, bismuth, probiotics or herbal medicines within 4 weeks before enrolment; (2) Pregnancy, lactation or plans for pregnancy during the study; (3) Concurrent use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), corticosteroids, anticoagulants or alcohol abuse; (4) Allergy to study medications; (5) Severe comorbidities (e.g. hepatic, cardiac, renal diseases, malignancies, psychiatric disorders); (6) History of gastric or oesophageal surgery; (7) Participation in other clinical trials within the past 3 months; (8) Inability to complete follow-up or poor compliance.
Interventions
Patients received one of two treatments: BQT (esomeprazole 20 mg twice daily + bismuth potassium citrate 600 mg twice daily + amoxicillin 1000 mg twice daily + metronidazole 400 mg four times daily) or TA (tegoprazan 50 mg twice daily + amoxicillin 750 mg four times daily) for 14 days. Tegoprazan, esomeprazole and bismuth were administered orally 30 min before meals, whereas amoxicillin and metronidazole were administered after meals. The study drug was esomeprazole (20 mg/tablet, AstraZeneca Pharmaceuticals Ltd., Chinese Yuan (CNY) 9.29 per tablet), tegoprazan (50 mg/tablet, Shandong Luoxin Pharmaceutical Group Co., Ltd, CNY 7.4 per tablet), amoxicillin (250 mg/tablet, Amoxicillin, Federal Pharmaceutical Manufacturing Co., Ltd, CNY 0.81 per tablet), metronidazole (200 mg/tablet, China Resources Double-Crane Limin Pharmaceutical Co., Ltd, CNY 0.067 per tablet) and bismuth potassium citrate (300 mg per capsule, equivalent to 110 mg elemental bismuth per dose, Livzon Pharmaceutical Group Inc, CNY 0.95 per tablet). Drug prices were obtained from the prevailing prices at designated pharmacies listed on the Gansu Medical Insurance Public Service Platform.
Outcomes
The primary outcome was eradication rate, defined as a negative ¹³C/¹⁴C-UBT performed at least 4 weeks after treatment completion. Efficacy was evaluated using ITT, modified intention-to-treat (mITT) and PP analyses. To avoid confounding results, participants were required to refrain from using antibiotics, P-CABs, and PPIs other than the study regimen for at least 6 weeks post-treatment and before the follow-up UBT.
The secondary outcomes were adherence, AEs and cost-effectiveness of each regimen. Adherence was calculated as the percentage of prescribed medication actually consumed by participants, with ⩾80% adherence defined as ‘good’. AEs were reported by participants during follow-up visits or actively assessed during the first and second weeks of treatment. Common AEs (e.g. nausea, vomiting, abdominal distension/pain and fatigue) were categorised as mild (transient, tolerable and no impact on daily life), moderate (physical or psychological discomfort partially affecting daily life) or severe (significant disruption to daily life). AE frequency was recorded as the number of participants experiencing each event, with multiple AEs per participant counted separately. For the cost-effectiveness analysis, only the cost of medications included in each regimen was considered, as laboratory tests and follow-up visits were identical across groups. Total costs were based on the price of each drug in the regimen. The eradication rate from PP analysis in this study was used as effectiveness data. Costs are expressed in CNY, the official currency of China, with all monetary values reported in yuan. The cost-effectiveness ratio (CER) was calculated as the cost per percentage of successful eradication, using the formula: CER = Total cost of medications ∕ effectiveness (%). The incremental cost-effectiveness ratio (ICER) is defined as the ratio of the incremental costs of one eradication regimen compared to another to the relative gain in effectiveness. The formula used for ICER was: ICER = (CostBQT − CostTA) ∕ (EffectivenessBQT − EffectivenessTA).
Sample size calculation
A non-inferiority design was used, with an assumed eradication rate of 91.7% for BQT18,46 and 91.5% for TA (extrapolated from data on esomeprazole–amoxicillin HDDT 47 and tegoprazan trials in non-high-resistance regions 41 ). However, in the absence of RCT data on tegoprazan-based dual therapy efficacy in high-resistance settings such as Gansu, these assumptions may not reflect local effectiveness. With a non-inferiority margin of −10%, power (1 − β) of 80%, and a one-sided α of 0.025, PASS software (NCSS, LLC, Connecticut, USA) estimated that 126 patients per group would be required. Accounting for a 10% dropout rate, the final sample size was 140 patients per group (N = 280).
Randomisation and follow-up
Patients were randomly assigned in a 1:1 ratio to one of the two treatment groups: BQT or TA. The randomisation list was generated by a statistician using the randomisation module in R software (version 4.0, The R Foundation for Statistical Computing), applying a block randomisation method with a block size of six to ensure balance across the two treatment groups. In total, 280 random numbers were generated, each corresponding to a specific treatment regimen. Treatment assignments were placed in sealed opaque envelopes, each labelled with a unique identifier ranging from 1 to 280. A dedicated research assistant, independent of patient screening and treatment, prepared and managed the envelopes. Clinicians assessed eligibility according to the inclusion and exclusion criteria, and once confirmed, informed the research assistant. The assistant then opened the next sequential envelope in numerical order and assigned the treatment regimen accordingly. After allocation, both clinicians and patients were informed of the treatment plan. Technicians performing the 13C/14C-UBT were blinded to the treatment allocation to maintain objectivity in outcome assessment.
Baseline data were collected via online questionnaires, and patients were included in a WeChat group for follow-up. Physicians provided detailed health education and electronic medication cards specifying dosing schedules. Compliance reminders and AE monitoring were conducted via phone or WeChat at least three times during the treatment period. Mild-to-moderate AEs were managed with symptomatic treatment, whereas severe AEs warranted treatment discontinuation. Post-treatment H. pylori status was assessed using UBT at least 4 weeks after therapy.
Statistical analyses
Statistical analyses were performed using SPSS (IBM, Armonk, NY, USA) and PASS 2021. Normally distributed continuous variables were expressed as mean ± standard deviation and compared using the t test, whereas non-normally distributed data were expressed as median (interquartile range) and analysed using the Wilcoxon rank-sum test. Categorical variables were reported as numbers (percentages) and compared using Chi-square or Fisher’s exact tests.
Eradication efficacy was assessed using three predefined analysis sets: the ITT included all randomised participants with lost-to-follow-up or untested cases classified as failures; mITT included only individuals who had endpoint measures and received at least one dose of the medication; and PP included only adherent participants (consumed ⩾80% of medications) with completed follow-up. Non-inferiority was evaluated by calculating the risk difference (RD) and its 95% confidence interval (CI) using a one-sided Z-test for proportions, with a prespecified non-inferiority margin of −10%. The null hypothesis of inferiority was rejected if the lower limit of the 95% CI exceeded the −10% margin (equivalent to a one-sided p-value <0.05).
Univariate and multivariate logistic regression analyses were conducted to identify factors influencing eradication rates. Variables with p < 0.1 in univariate analysis were included in the multivariate model. Post hoc power for non-inferiority testing was recalculated using the observed eradication rates, prespecified non-inferiority margin (−10%) and actual sample sizes, with one-sided α = 0.05 in PASS 2021. Except for non-inferiority testing, all analyses were two-sided, with p < 0.05 considered statistically significant.
Results
Baseline characteristics of enrolled patients
As shown in Figure 1, a total of 360 individuals were screened for eligibility between September and December 2024. Of these, 42 were excluded, and 30 declined participation. The remaining 288 eligible participants were randomised in a 1:1 ratio to the TA and BQT groups (n = 144 per group). In the TA group, 142 participants completed post-treatment follow-up, of whom 135 demonstrated good adherence (⩾80%). In the BQT group, 143 participants completed follow-up, with 131 achieving good adherence.

Flow chart of patient enrolment.
Baseline characteristics of the two groups are summarised in Table 1. No statistically significant differences were observed between groups with respect to age, sex, body mass index, residence, dietary habits, smoking or alcohol use, presenting symptoms, endoscopic findings or family history of gastric cancer (all p > 0.05). However, a significant difference was observed in the H. pylori testing status of other household members. Although the proportion of households with positive H. pylori test results was identical between groups (36.11%), the distribution of negative and untested household members differed significantly (p < 0.05).
Baseline demographics and clinical data of patients.

BMI, body mass index; BQT, esomeprazole 20 mg twice daily + bismuth potassium citrate 600 mg twice daily + amoxicillin 1000 mg twice daily + metronidazole 400 mg four times daily; SD, standard deviation; TA, tegoprazan 50 mg twice daily + amoxicillin 750 mg four times daily.
Eradication rates of H. pylori infection
In the ITT analysis, H. pylori eradication was significantly lower with TA than with BQT (68.06% vs 81.25%; RD, −13.19%; 95% CI: −22.95 to −3.43; p = 0.009). The superiority of BQT persisted in the mITT analysis (69.01% vs 81.82%; RD −12.81%, 95% CI: −22.70 to −2.90; p = 0.011) and PP analysis (72.59% vs 89.31%; RD −16.72%, 95% CI: −26.01 to −7.43; p < 0.001). Importantly, TA failed to meet non-inferiority criteria, as all lower CI bounds (ITT: −22.95%; mITT: −22.70%; PP: −26.01%) exceeded the prespecified −10% margin. This is where the data from Table 2 is primarily discussed.
Comparative analysis of eradication rates and non-inferiority assessment.

Non-inferiority margin was set at −10% with one-sided testing at α = 0.025.
BQT, esomeprazole 20 mg twice daily + bismuth potassium citrate 600 mg twice daily + amoxicillin 1000 mg twice daily + metronidazole 400 mg four times daily; CI, confidence interval; ITT, intention-totreat; mITT, modified intention-totreat; PP, per-protocol analysis; TA, tegoprazan 50 mg twice daily + amoxicillin 750 mg four times daily.
Post hoc power reassessment
Post hoc power analysis using the observed ITT eradication rates (TA: 68.06%, BQT: 81.25%) and the prespecified non-inferiority margin (−10%) demonstrated 99.99% power to detect non-inferiority at α = 0.025 (one-sided). This confirmed that the failure to establish non-inferiority was not attributable to an inadequate sample size.
AEs and compliance
As shown in Table 3, the incidence of AEs was significantly lower in the TA group than in the BQT group (8.30% vs 25.70%; p < 0.0001), which was largely attributable to a reduction in the incidence of mild AEs (7.60% vs 21.50%; p = 0.0002). Importantly, no severe AEs were reported in either group. The most frequently reported AEs in the BQT group were nausea, dry mouth and dizziness, all of which occurred at significantly higher rates than those in the TA group. No significant differences were observed between the two groups in the incidence of other adverse symptoms.
AEs and compliance.

Marked empty comparisons with ‘N/A’ where no test was possible.
AEs, adverse events; BQT, esomeprazole 20 mg twice daily + bismuth potassium citrate 600 mg twice daily + amoxicillin 1000 mg twice daily + metronidazole 400 mg four times daily; TA, tegoprazan 50 mg twice daily + amoxicillin 750 mg four times daily.
p < 0.05. **p < 0.01. ***p < 0.001.
Both treatment groups demonstrated high medication adherence, with no significant difference between them (95.07% vs 91.61%; p = 0.241). Although the BQT group exhibited a higher rate of treatment discontinuation due to AEs, the difference was not statistically significant (p = 0.28).
Factors influencing eradication rates
Table 4 presents the associations between baseline factors and treatment outcomes using logistic regression analysis. Long-term residence in the Longnan region of Gansu Province (including Longnan City and Tianshui City) was significantly associated with a higher eradication rate compared to the Hexi region. This association remained statistically significant in both univariate analysis (odds ratio (OR) = 5.33, 95% CI: 1.10–25.99, p = 0.04) and multivariate-adjusted analysis (adjusted odds ratio (aOR) = 5.46, 95% CI: 1.06–28.12, p = 0.04), suggesting that long-term residence serves as an independent predictor of eradication success.
Univariate and multivariate analyses of factors influencing the eradication of Helicobacter pylori infection.

BMI, body mass index; BQT, esomeprazole 20 mg twice daily + bismuth potassium citrate 600 mg twice daily + amoxicillin 1000 mg twice daily + metronidazole 400 mg four times daily; CI, confidence interval; OR, odds ratio; TA, tegoprazan 50 mg twice daily + amoxicillin 750 mg four times daily.
BQT showed a significant advantage over TA in univariate analysis (OR = 2.02, 95% CI: 1.16–3.52, p = 0.01). However, the effect size diminished after multivariate adjustment and did not reach statistical significance (aOR = 1.72, 95% CI: 0.96–3.08, p = 0.07), indicating that its efficacy may be partially mediated by confounding factors, although its potential clinical relevance remains noteworthy.
Additionally, tap water consumption (compared to purified water) and a family history of gastric cancer both exhibited a trend toward lower H. pylori eradication rates in the multivariate model (aOR = 0.56, 95% CI: 0.31–1.02, p = 0.06; aOR = 0.53, 95% CI: 0.24–1.16, p = 0.11, respectively). However, these associations did not reach traditional statistical significance.
Cost-effectiveness analysis of each regimen
The cost-effectiveness analysis indicates that the CERs for BQT and TA were 4.61 and 4.73, respectively. The ICER was 4.08, indicating that compared with TA, BQT required an additional 4.08 CNY for each 1% increase in eradication efficacy (Table 5).
Cost-effectiveness analysis of each regimen.
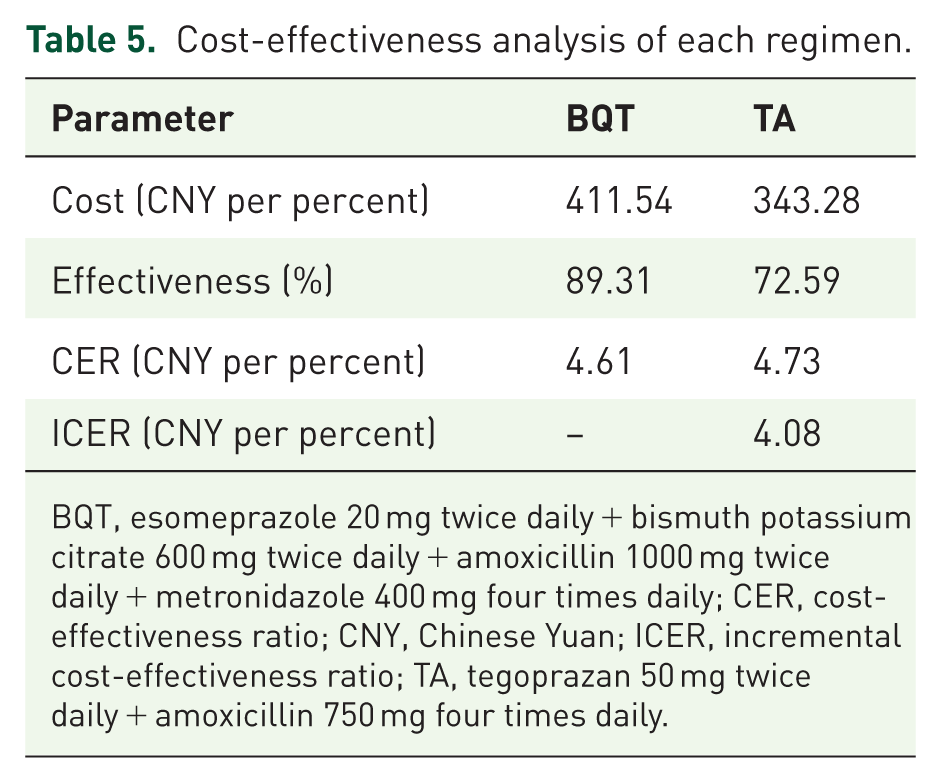
BQT, esomeprazole 20 mg twice daily + bismuth potassium citrate 600 mg twice daily + amoxicillin 1000 mg twice daily + metronidazole 400 mg four times daily; CER, cost-effectiveness ratio; CNY, Chinese Yuan; ICER, incremental cost-effectiveness ratio; TA, tegoprazan 50 mg twice daily + amoxicillin 750 mg four times daily.
Discussion
This prospective multicentre RCT demonstrated that, in Gansu Province – a region characterised by high H. pylori prevalence and elevated antibiotic resistance – BQT achieved significantly higher eradication rates than TA across all analysis sets (ITT: 81.25% vs 68.06%, p = 0.009; mITT: 69.01% vs 81.82%, p = 0.011; PP: 89.31% vs 72.59%, p < 0.001). The trial failed to demonstrate the non-inferiority of TA to BQT, as the lower bounds of the 95% CIs (−22.95% to −26.01%) exceeded the prespecified −10% margin. The eradication rates were substantially lower than those assumed efficacy eradication rates during sample size calculation (91.5% for TA vs 91.7% for BQT), which had been extrapolated from studies conducted in non-high-resistance areas. This discrepancy between anticipated and observed efficacy underscores the critical influence of regional antibiotic resistance patterns and highlights the inability of standard dosing regimens from low-resistance areas to achieve satisfactory outcomes in our high-resistance population. In contrast, multiple studies conducted across different regions of China have reported higher eradication rates with TA therapy. For instance, Liu et al. observed ITT and PP eradication rates of 83.9% and 88.3% in Chongqing, 40 respectively, while a trial by Lin et al. in Fujian Province yielded PP rates exceeding 91%. 41 Similarly, Kong et al. reported a PP rate of 88.5% in Shandong Province. 48 Moreover, consistent with local evidence, previous trials in Gansu echo our findings by reporting suboptimal dual therapy efficacy (vonoprazan-amoxicillin ITT = 58.3–63.5% 49 ; PPI-amoxicillin ITT = 65.7% 44 ). This is further supported by evidence from South Korea, where studies have shown that tegoprazan-based regimens yield low eradication rates (74.77%–78.6%) without demonstrating superiority over PPI-based therapies,50,51 collectively suggesting that regional resistance patterns fundamentally compromise the efficacy of amoxicillin-based monotherapy approaches. The markedly low eradication rates observed in our study may be attributed to several factors. First, the pharmacokinetic and pharmacodynamic properties of tegoprazan may differ from those of vonoprazan, particularly in terms of acid suppression potency and duration, which are critical for optimising the activity of time-dependent antibiotics, such as amoxicillin. Second, regional variations in H. pylori antibiotic resistance, particularly resistance to amoxicillin, could have contributed to reduced efficacy. Although Gansu Province has limited surveillance data on resistance patterns, environmental and socioeconomic factors may result in higher baseline resistance rates compared to coastal regions. Third, host-related and bacterial factors such as H. pylori virulence genotypes, CYP2C19 metaboliser status and gastric mucosal conditions may differ geographically and influence treatment outcomes. Finally, differences in study design, including treatment duration, dosing schedule, patient inclusion criteria and adherence monitoring, may partially explain the discrepancies in the results. Taken together, these findings underscore the need for region-specific validation of dual therapy regimens and highlight the need for further pharmacological and microbiological investigations to optimise acid inhibition-based H. pylori eradication strategies.
The suboptimal eradication efficacy of tegoprazan, a P-CAB, observed in this trial may largely be attributable to insufficient acid suppression resulting from subtherapeutic dosing. Pharmacodynamic studies have demonstrated that higher doses of P-CABs are generally required to achieve sufficient gastric acid inhibition for H. pylori eradication, compared with doses used for acid-related disorders. For example, vonoprazan, a first-generation P-CAB now recommended in international guidelines, is administered at 20 mg twice daily for eradication therapy – four times the standard dose used for GERD management (10 mg once daily).48,52,53 In contrast, the tegoprazan dose used in the present study (50 mg twice daily) represented only a two-fold increase over the standard GERD dose (50 mg once daily), raising concerns regarding its acid-suppressive capacity. This concern is supported by dose–response studies, which have shown that increasing tegoprazan from 100 mg once daily to 200 mg once daily results in a significant increase in the median intragastric pH from 5.2 to 6.4, 52 a threshold for maintaining optimal antibiotic activity. These findings suggest that tegoprazan 100 mg twice daily (200 mg/day) may be necessary to sustain an intragastric pH >6, which is considered optimal for H. pylori eradication. Accordingly, the inadequate efficacy observed in our TA protocol is likely a pharmacological consequence of suboptimal acid inhibition. Our findings are consistent with recent data from Korea, where Kim et al. reported insufficient eradication efficacy associated with standard-dose tegoprazan-based dual therapy, emphasising the need for dose optimisation. 54 Taken together, these findings highlight the importance of further investigations into appropriate tegoprazan dosing strategies to ensure adequate acid suppression and maximise therapeutic outcomes in H. pylori eradication.
Significant geographical and temporal variations in antibiotic resistance patterns of H. pylori have been reported in China. 55 In Gansu Province, a combination of factors, including widespread antibiotic overuse,55,56 socioeconomic disparities and inadequate sanitation, have contributed to markedly elevated resistance rates to first-line eradication antibiotics compared to other regions,43,57 Characterised by a high infection burden, elevated resistance rates and suboptimal eradication efficacy,2,43,44 H. pylori strains in Gansu exhibited alarming resistance levels between 2018 and 2020: 85.86% to metronidazole, 55.05% to clarithromycin and 46.66% to levofloxacin. 43 The amoxicillin resistance rate, while comparatively low, remained notable at 16.16%, a five-fold higher value than the national average (3.23%).43,57 This profoundly challenging resistance landscape is the fundamental reason for the suboptimal eradication rate of BQT observed in our study, which fell below the desired 90% threshold. Nevertheless, the rationale for retaining amoxicillin in the BQT regimen remains scientifically sound. Amoxicillin resistance requires simultaneous mutations at multiple sites within penicillin-binding protein-related genes, thus creating a high genetic barrier to resistance. 58 Although the local resistance rate was elevated, it remained substantially lower than that for clarithromycin and metronidazole. Furthermore, the pH-dependent antibacterial activity was maximised by potent acid suppression. Additionally, although esomeprazole, which exhibits a lower dependence on CYP2C19 metabolism than first-generation PPIs, was used in our BQT regimen, the lack of routine CYP2C19 genotyping means that its efficacy might still be adversely affected by CYP2C19 rapid metabolisers, especially among difficult-to-treat patients. 59 Furthermore, variations in adherence to this complex multidrug regimen may compromise treatment success. 60 The significantly higher eradication rate achieved with BQT than with TA can be explained by its multi-target approach. The BQT regimen employs an optimised high dose of metronidazole (1600 mg/day) to overcome resistance.61,62 Furthermore, bismuth salts confer direct antimicrobial effects and demonstrate synergistic activity with metronidazole and additive effects with amoxicillin, enhancing the eradication efficacy even against resistant strains.63,64 In contrast, TA relied solely on AMX, lacking the synergistic support of bismuth and a second antibiotic.
Safety analysis revealed a significantly higher incidence of AEs in the BQT group (25.70% vs 8.30% with TA; p < 0.0001), which was primarily attributable to metronidazole-related side effects, such as nausea and dizziness – a pattern consistent with previous reports on nitroimidazole-based regimens. 65 Although TA demonstrated better tolerability and marginally higher compliance (95.07% vs 91.61%, p = 0.241), its lower eradication rates preclude its use as a first-line therapy in Gansu.
Multivariate analysis identified the key factors influencing H. pylori eradication efficacy. This study demonstrated that long-term residents in the Southern Gansu Region (including Longnan City and Tianshui City) exhibited a significantly higher eradication rate of H. pylori compared to those in the Hexi region (aOR = 5.46, 95% CI: 1.06–28.12, p = 0.04). These preliminary findings suggest the potential for geographic disparities in efficacy. However, the limited sample size within each region necessitates confirmation in larger regionally stratified cohorts. This disparity likely stems from the synergistic effects of geographical environment, healthcare resource allocation and lifestyle differences. Previous studies have shown that H. pylori can develop genomic polymorphisms through horizontal gene transfer during long-term interactions with the host or cohabiting microbiota, thereby adapting to local environmental pressures that may lead to significant geographical variations in the distribution of virulence genes. 66 Such adaptive selection could potentially explain the regional heterogeneity in eradication rates observed here and elsewhere. Our team’s previous investigation identifies marked substantial regional heterogeneity in H. pylori antibiotic resistance rates across Gansu Province. 43 Differences in antibiotic resistance may directly affect treatment regimen efficacy. In regions with high resistance rates, the continued use of conventional antibiotic regimens may significantly reduce eradication rates. Additionally, treatment regimen selection emerged as an independent predictor, with BQT being significantly associated with higher eradication rates (aOR = 1.72, p = 0.07), aligning with the global consensus that recommends BQT as the preferred regimen in regions with high antibiotic resistance. 15 Exploratory analyses suggested potential associations between tap water consumption (aOR = 0.56, p = 0.06) or family history of gastric cancer (aOR = 0.53, p = 0.11) and reduced eradication of H. pylori. Although both a mechanistically plausible risk of waterborne re-exposure67,68 and inherited mucosal susceptibility 69 have been proposed, these findings require validation in larger cohorts due to limited statistical power in subgroup analyses.
This study has several notable strengths, including its rigorous multicentre randomised design, the use of standardised therapeutic regimens, and the comprehensive evaluation of both clinical and environmental factors influencing H. pylori eradication. However, certain limitations should be acknowledged. First, the absence of in vitro antibiotic susceptibility testing or molecular analysis of resistance genes (such as pbp1A for amoxicillin) precluded confirmation of whether the suboptimal efficacy of the two treatment regimens was directly caused by pre-existing resistance. Future studies should implement dynamic resistance monitoring and adapt treatment strategies based on local resistance profiles to guide individualised therapy. Second, although geographical variations in efficacy were observed, the sample size from specific sub-regions (e.g. only eight patients from the Hexi region) was insufficient to draw reliable conclusions. This limitation affects both the robustness and generalisability of these findings, highlighting the need for validation in larger, regionally stratified cohorts. Third, the suboptimal efficacy of the TA regimen may be partly attributable to the insufficient acid-suppressive effect of tegoprazan dosing (50 mg twice daily) used in this trial. Future studies should investigate optimised P-CAB dosing schedules to achieve more potent and sustained acid suppression, which is critical for successful amoxicillin-based therapies. Finally, the suboptimal efficacy of TA challenges its suitability as a first-line empirical treatment in international settings. Because most evidence on tegoprazan-based regimens originates from Asian countries with variable efficacy, the current regimen is not recommended as a globally competitive alternative. Future applications should be guided by local efficacy and resistance data, and validation through large-scale multi-regional studies is strongly recommended.
In the current epidemiological context in the Gansu Province, BQT remains the preferred first-line regimen for H. pylori eradication. Although TA demonstrates superior tolerability and comparable compliance, the suboptimal eradication rate precludes its recommendation as an empirical first-line regimen in this setting. Future research should focus on developing optimised regimens, which may include tailoring therapy based on local resistance patterns and investigating modified TA dosing protocols to achieve satisfactory eradication rates.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251391276 – Supplemental material for Comparison of tegoprazan-based high-dose dual therapy versus bismuth-containing quadruple therapy for Helicobacter pylori eradication: a prospective, multicentre, randomised controlled trial in Gansu Province, a high-resistance region of China
Supplemental material, sj-docx-1-tag-10.1177_17562848251391276 for Comparison of tegoprazan-based high-dose dual therapy versus bismuth-containing quadruple therapy for Helicobacter pylori eradication: a prospective, multicentre, randomised controlled trial in Gansu Province, a high-resistance region of China by Jinxia Yang, Xiuhong Zhang, Lihong Yang, Ronggui Wu, Hong Fan, Ni Xie, Liping Gao, Yeze Dong, Baoyuan Xie, Yuanyuan Wang, Xinglan Chen, Yi Yu and Dekui Zhang in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We express our sincere gratitude to JueQiong Wang, LiNa Wei, Ming Zhou, XiaoYin Zhang, JianFang Zhao, Fang Wang, JianWei Yun, XiaoChun Wang, ZhaoYuan Fu, XueFeng Ding, YanYan Yang and ZuoHui Yuan for their tireless dedication to patient recruitment.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
