Abstract
In the management of difficult-to-treat and severe asthma, the incorporation of a Long-Acting Muscarinic Antagonist (LAMA) into a regimen of Inhaled Corticosteroids plus Long-Acting β2 agonists (ICS/LABA) represents a viable add-on therapeutic strategy. Historically, this approach required the use of separate inhalers; however, the recent advent of “single-inhaler triple therapy” (SITT) provided a valuable alternative. One such formulation is the extrafine combination of beclomethasone dipropionate (BDP), fluticasone furoate (FF), and glycopyrronium bromide (GB), which is delivered via a single pressurized metered-dose inhaler (pMDI). Clinical trials, including the TRIMARAN and TRIGGER studies, alongside subsequent post-hoc analyses, have elucidated the benefits of this SITT at both 87/5/9 μg and 172/5/9 μg dosing regimens administered daily. Findings indicated a significant improvement in respiratory function and a reduction in the frequency of exacerbations among patients with uncontrolled asthma. The BDP/FF/GB SITT confirmed efficacy and safety across various ethnic groups (including Caucasian, Japanese, and Chinese populations) and across different age cohorts (adults and adolescents), although it still remains unapproved for individuals under 18 years of age. The use of a single pMDI facilitates the deposition of extra- fine particles from all three active ingredients in the small airways enhancing therapeutic effectiveness. Moreover, the consolidation of medications into one device may improve patients’ adherence by mitigating the risks associated with device mismanagement and ensuring optimal drug delivery. The cost-effectiveness analysis of the BDP/FF/GB SITT suggests favorable outcomes compared to traditional ICS/LABA and ICS/LABA plus tiotropium combinations. Additional data will be forthcoming from the ongoing real-life TRIMAXIMIZE observational study.
Keywords
Introduction
According to the Global Initiative for Asthma (GINA), asthma is a prevalent chronic respiratory disease affecting 1%–29% of the global population, characterized by airway inflammation and airway hyperresponsiveness, defined by a history of variable respiratory symptoms, including wheezing, shortness of breath, chest tightness, and cough. These symptoms may vary in frequency and intensity and are often accompanied by expiratory airflow limitation, which may become persistent in certain cases. Patients may also experience episodic severe exacerbations that can be life-threatening. 1
There are different types of inflammation observed in the bronchial airways, known as “endotypes,” which can lead to differences in the clinical features of asthma seen in patients (“phenotypes”). Identification of an individual’s specific asthma pheno-endotype can help improve disease management. Rather than using the same treatment regimen for all patients, current strategies for asthma management focus on individualizing treatment based on a patient’s phenotype. There are several well-defined asthma phenotypes including allergic, nonallergic, eosinophilic, neutrophilic, exercise-induced, aspirin-induced, adult-onset asthma (often nonallergic and less responsive to low doses of corticosteroids), asthma with persistent airflow limitation, and obesity-related asthma (characterized by prominent respiratory symptoms and a limited eosinophilic pattern of inflammation). 1
First-line therapy for patients with moderate-to-severe asthma (GINA steps 3, 4, and 5) typically involves the daily use of a combination of ICS and LABA as controller therapy. This regimen is designed to manage symptoms and mitigate the risk of future exacerbations. 1 LABAs exert their bronchodilator effects through binding to β2 adrenergic receptors in airway smooth muscle, while ICS reduce airway inflammation, contribute to alleviate symptoms, and decrease the risk of exacerbations (which is associated with lung function decline, and mortality).2 –7
In addition to controller therapies, reliever medications are critical in asthma management. These medications defined as inhalers taken on an as-needed basis achieve rapid bronchodilation and prompt symptom relief. Currently, two primary reliever options are available: low-dose ICS combined with formoterol and short-acting β2-agonists (SABAs). 1 Compared to Short Action β2 agonists (SABA), ICS-formoterol (termed “anti-inflammatory reliever”) has been shown to reduce the occurrence of severe exacerbations, regardless of concomitant maintenance therapy.4,5,8 In addition, both SABA and ICS-formoterol are recommended prior to exercise to prevent exercise-induced bronchoconstriction.9,10
An important factor in the pathogenesis of asthma is increased cholinergic tone, which contributes to bronchoconstriction, inflammation, airway remodeling, mucus hypersecretion, and cough. 11 Consequently, anticholinergic medications, specifically Long-Acting Muscarinic Antagonists (LAMAs), are rationally integrated into the treatment of asthma as add-on therapy for patients with difficult-to-treat or severe asthma.1,12
GINA guidelines recommend the use of LAMAs as an add-on treatment for patients at step 4 receiving medium-dose ICS/LABA and for patients at step 5 receiving high-dose ICS/LABA. In children aged 6 years and older, separate inhalers are required, whereas a combination inhaler may be used for adults aged 18 and above. 1 A meta-analysis encompassing over 11,000 patients revealed that although there were no significant differences in quality of life, triple therapy with LAMA as an add-on was associated with improved control and a 17% reduction in severe exacerbations compared to dual ICS/LABA therapy. 13
The synergistic interactions among ICSs, LABAs, and LAMAs showed significant clinical benefits in asthma management.13 –16 Specifically, the interplay between inhaled anticholinergic agents and ICSs facilitates airway relaxation. 17 Furthermore, the combined low-dose administration of β2-agonists and anticholinergics inhibits the release of acetylcholine from bronchial tissues, promoting airway smooth muscle relaxation. 18 These interactions may also enhance bronchial anti-inflammatory and anti-remodeling activities, leading to bronchorelaxation effects demonstrated in ex vivo studies.19,20 Consequently, triple therapy showed overall a greater efficacy than ICS/LABA combinations alone in preventing asthma exacerbations, even in the absence of type 2 airway inflammation or in cases of fixed airflow limitation.13 –16
In comparison to open-inhaler triple therapy (ICS/LABA + LAMA), SITT; ICS/LABA/LAMA) is associated with more substantial improvements in lung function, asthma control, and health-related quality of life, potentially leading to enhanced medication adherence. 21 The need for multiple inhalers for LABA/ICS and LAMA, each with distinct designs, dosing regimens, and inhalation techniques, may inconvenience patients, resulting in suboptimal adherence and persistence, as well as increasing the risk of improper drug delivery and adverse clinical outcomes. 22
Currently, there are three SITT options available for asthma management: fluticasone furoate/umeclidinium/vilanterol (FF/U/V), approved in many countries, for example, by the U.S. Food and Drug Administration, by Canada’s Drug Agency, and by Japanese Drug Use System; indacaterol/glycopyrrolate/mometasone furoate (I/GB/MF), approved by the European Medicines Agency (EMA), Canada’s Drug Agency, and Japanese Drug Use System; and beclomethasone dipropionate/formoterol fumarate/glycopyrronium bromide (BDP/FF/GB), approved by the EMA23 –25 and also by Australian Pharmaceutical Benefits Scheme (PBS). The BDP/FF/GB combination is indicated for adults whose asthma is inadequately controlled despite treatment with LABA plus medium-to-high doses of ICS, and who have experienced at least one exacerbation in the previous year. 25
Notably, BDP/FF/GB is particular among SITT options as it is formulated as an extrafine aerosol delivered via a pressurized metered-dose inhaler (pMDI). 25 Depending on symptom severity, clinicians can select from two dosages (87/5/9 μg and 172/5/9 μg). 25
This review aims to synthesize the current evidence regarding this single-device triple therapy, with a specific focus on its efficacy and safety profile, to optimize its integration within the complex landscape of asthma management.
Pharmacological aspects of the BDP/FF/GB combination
The inhaled triple therapy combination of BDP, FF, and GB in a single device provides concomitant deposition of the three components as extra fine particles. 20 BDP acts as a pro-drug, converting into its active metabolite, beclomethasone-17-monopropionate, through hydrolysis by esterases present in the lungs. The pharmacodynamics of BDP, like other ICS, relies on its affinity for intracellular glucocorticoid receptors responsible for the transcriptional regulation of target genes, notably the attenuation of nuclear factor kappa B activity, resulting in a decreased histone acetylation and chromatin remodeling, ultimately suppressing the expression of activated inflammatory genes. 26
FF is a selective β2-adrenergic agonist that induces relaxation of bronchial smooth muscle in patients with reversible airway obstruction. The onset of the bronchodilator effect occurs rapidly after inhalation (within 2–5 min) with a duration of approximately 12 h.27,28
GB serves as a high-affinity LAMA utilized for bronchodilation, functioning by inhibiting the bronchospastic action of acetylcholine on airway smooth muscle cells, thereby promoting airway dilation. Notably, GB exhibits a high affinity for muscarinic receptors, demonstrating greater than fourfold selectivity for the human M3 receptor over the human M2 receptor, which plays a regulatory role in acetylcholine release. 29
The device employed for administering BDP/FF/GB is a pressurized metered-dose inhaler (pMDI), which facilitates the deposition of extra fine particles with an average mass median aerodynamic diameter of less than 2 μm. 20 This particle size is significantly smaller than that of non-extra particles delivered by other types of SITTs, leading to enhanced intrathoracic deposition relative to extrathoracic deposition (i.e., in the mouth or upper airways), thereby reducing the risk of systemic side effects. 30 Moreover, this characteristic enhances the therapeutic efficacy compared to formulations lacking an extra fine distribution, allowing for improved delivery to peripheral airways and promoting therapeutic activity in small bronchi. 26
The ScinTigraphic assessment Of tRiMbow lung deposition (STORM) study was conducted to evaluate the regional distribution of BDP/FF/GB particles in vivo, utilizing gamma scintigraphy in both healthy volunteers and asthmatic patients (10 and 9 subjects, respectively). A total dose of BDP/FF/GB 400/24/50 μg (administered as four inhalations of BDP/FF/GB 100/6/12.5 μg) was radiolabeled with 99mTc-pertechnetate and delivered via pMDI. Approximately 23% and 26% of the nominal dose were deposited in the lungs of healthy volunteers and asthmatic patients, respectively, with half of the pulmonary dose localized in the peripheral lung region. No significant differences in the standardized central/peripheral ratio values were observed (1.80 for healthy controls and 1.94 for asthmatics), suggesting that the features of asthma do not significantly impact drug deposition. 31 Furthermore, lung function data corroborated the bronchodilator effect of BDP/FF/GB in asthmatic patients, indicating increased forced expiratory volume in one second (FEV1) with a peak at 30 min and sustained elevation over baseline for more than 24 h following a single inhalation. Notably, BDP/FF/GB is typically administered twice daily. 31
Several other clinical studies investigated the pharmacokinetic parameters and safety profile of BDP/FF/GB SITT, assessing potential variations based on ethnicity and age, with all studies involving single administrations.32 –34 A randomized, double-blind, four-way study evaluated the effects of BDP/FF/GB in Japanese healthy volunteers compared to Caucasian counterparts following a single administration of two therapeutic doses (200/12/25 and 400/12/25 µg), a supratherapeutic dose (800/48/100 µg), and a placebo. Overall, plasma exposure of all components following inhalation of BDP/FF/GB was slightly higher in Japanese participants than in Caucasian participants for both therapeutic and supratherapeutic doses, although these differences were not statistically significant. Furthermore, no significant differences were observed in BDP dose-related suppression of plasma cortisol, serum potassium, serum glucose, blood pressure, heart rate, or adverse events, indicating that dose adjustment is unnecessary. 32
Consistent findings regarding adverse events were reported in another study examining the effects of therapeutic and supratherapeutic doses of BDP/FF/GB in Chinese healthy volunteers, with most participants experiencing no adverse events. When adverse events occurred, they were generally mild (e.g., headache).32,33 No adverse events were reported in an adolescent population evaluated by Kuna et al., 34 investigating the pharmacokinetic profile of BDP/FF/GB in adults and adolescents (ages 12–17) trained for a single self-administration of BDP/FF/GB 400/24/50 μg via pMDI.
Synergistic effects of BDP/FF/GB fixed-dose combinations: clinical trials and post hoc analysis
The TRIMARAN and TRIGGER studies investigated the efficacy of a SITT combining BDP, FF, and GB in patients with uncontrolled asthma. Beyond the known beneficial interactions elicited by the combinations of ICS/FF and ICS/GB in promoting bronchodilation,17,35 recent studies have characterized the potential synergistic effect of the triple combination BDP/FF/GB using ex vivo models of human airways passively sensitized by histamine from non-asthmatic and non-COPD donors. 20
In terms of individual components, both BDP and FF demonstrated a clear bronchorelaxant profile, whereas GB exhibited limited potency and only partial efficacy. This supports the rationale for employing a LAMA as an add-on treatment in GINA steps 4 and 5, but not as monotherapy. Notably, Rogliani et al. found that the relaxant effect induced by varying concentrations of BDP/FF/GB at a 100:6:12.5 ratio was significantly greater than the expected additive effects of the individual components. This bronchodilator effect was observed in both medium and small passively sensitized precontracted bronchi. 20
Regarding in vivo studies, Triple in Asthma With Uncontrolled Patients on Medium Strength of ICS/LABA (TRIMARAN) and Triple in Asthma High Strength Versus ICS/LABA HS + Tiotropium (TRIGGER) were the first to assess the efficacy of SITT in asthma. These studies focused on changes from baseline pre-dose forced expiratory volume in one second (FEV1) at week 26 and the number of moderate-to-severe exacerbations at week 52 (Table 1). 36 Moderate exacerbations were defined according to the joint statement by the American Thoracic Society and the European Respiratory Society, 37 while severe exacerbations were characterized as documented worsening requiring systemic corticosteroid treatment for a minimum of 3 days. 36
Summary of efficacy data Phase III SITT RCTs versus LABA/ICS (TRIMARAN e TRIGGER) in patients with poorly controlled asthma.

ICS/LABA, Inhaled Corticosteroids plus Long-Acting β2 agonists.
Both studies spanned 52 weeks and included adult patients with a clinical diagnosis of asthma who were uncontrolled (Asthma Control Questionnaire 7 score ⩾1.5) while receiving dual maintenance treatment with medium doses (in TRIMARAN) or high doses (in TRIGGER) of an ICS/LABA combination. To qualify, patients had to have experienced at least one asthma exacerbation requiring systemic corticosteroid treatment, emergency department visit, or hospitalization within the prior year. Key exclusion criteria included a history of near-fatal asthma, severe exacerbation within 4 weeks prior to study entry or during the run-in period, significant comorbid lung disease, smoking history (current or former with 10 or more pack-years or cessation <1 year prior to screening), and current treatment with biological agents. 36
In TRIMARAN, patients received two twice-daily doses of BDP/FF/GB at 87/5/9 μg (n = 579) compared to two twice-daily doses of a fixed combination of BDP/FF at 100/6 μg (n = 576). The TRIGGER study compared two twice-daily doses of BDP/FF/GB at 172/5/9 μg (n = 573) against two twice-daily doses of BDP/FF at 200/6 μg (n = 576) or with the addition of tiotropium 2.5 μg administered once-daily (n = 288). 36
In TRIMARAN, the BDP/FF/GB 87/5/9 μg group exhibited a significant increase in pre-dose FEV1 at week 26 compared to the BDP/FF 100/6 μg group (185 mL vs 127 mL, respectively). Similarly, the TRIGGER study demonstrated a significant trend in FEV1 improvement for high doses of BDP/FF/GB compared to BDP/FF (229 mL vs 157 mL), although this was not observed in the BDP/FF + tiotropium arm. In TRIMARAN, the use of BDP/FF/GB led to a significant 15% reduction in the rate of moderate-to-severe exacerbations, whereas the TRIGGER study reported a similar but nonsignificant trend (12% reduction), with statistical significance achieved only for severe exacerbations. Notably, no differences were found between the BDP/FF/GB group and the BDP/FF + tiotropium group. Furthermore, adverse events across the five treatment groups were generally mild and not treatment-related. Overall, findings from TRIMARAN and TRIGGER indicate that in adults with uncontrolled asthma treated with medium-to-high doses of ICS/LABA, the addition of a LAMA in a single inhaler (BDP/FF/GB) primarily enhances lung function and reduces severe exacerbations with good tolerability. 36 Data encompassing 7204 asthmatics were obtained from the main studies with SITT (TRIMARAN, TRIGGER, IRIDIUM, and CAPTAIN). Comparative analyses between triple therapies and ICS/LABA regimens revealed no elevated risk of total severe adverse events (SAEs) (RR-0.99, 95% CI 0.83–1.18) and cardiac SAEs (RR-0.74, 95% CI 0.39–1.40). Furthermore, the dose of ICSs did not affect the risk of pneumonia. Ultimately, it was concluded that triple therapy represents a safe pharmacological treatment modality in patients with severe asthma, characterized by a favorable safety profile. 37
Post-hoc analyses of selected subgroups from the TRIMARAN and TRIGGER studies support the potential for targeted care utilizing BDP/FF/GB SITT. Specifically, SITT was associated with a greater likelihood of normalizing airflow compared to BDP/FF, with enhanced efficacy in reducing exacerbations noted particularly in patients achieving airflow normalization due to treatment. 38 This effect was also observed in patients with persistent baseline airflow limitation 16 and those with greater reversibility. 39 Importantly, the effectiveness of extrafine BDP/FF/GB compared to BDP/FF was not influenced by eosinophil blood count, further supporting the broad utility of this SITT over ICS/LABA alone. 38
In terms of seasonality, it is well-documented that winter correlates with a higher incidence of asthma exacerbations. 40 A post-hoc analysis confirmed that patients treated with BDP/FF experienced a notable seasonal effect, with the highest rates of moderate-to-severe exacerbations occurring in winter. This occurrence was significantly reduced by 20.3% with extra fine BDP/FF/GB SITT, reaffirming the overall utility of LAMA addition to ICS/LABA maintenance therapy for uncontrolled asthmatic patients. Nonsignificant reductions in exacerbation rates during other seasons ranged between 8.6% and 12.0%. 41
Importantly, among studies evaluating the effectiveness of SITT in asthma—specifically BDP/FF/GB (TRIMARAN and TRIGGER), FF/U/V (CAPTAIN study), and I/GB/MF (IRIDIUM and ARGON studies)—only TRIMARAN demonstrated a significant reduction in exacerbations compared to the respective ICS/LABA combination.36,42 –44
Further considerations about the BDP/FF/GB SITT option in the management of uncontrolled asthma
Adherence
Suboptimal adherence to asthma treatment occurs in approximately 75% of patients. 45 Data specifically addressing adherence and compliance in asthmatic patients using SITT are limited. However, SITT has demonstrated potential for improving treatment adherence by consolidating multiple medications into a single inhaler, thereby reducing the number of devices required for maintenance therapy. This simplification can lead to fewer instructions and a unified dosing regimen, which may decrease handling errors and the selective discontinuation of therapy components. To optimize adherence, the selection of an appropriate inhaler device and the frequency of SITT administration should be collaboratively discussed with patients, taking into account their symptoms and preferences. 46
Given that asthma severity can fluctuate over time, there are concerns that stepping down from triple therapy may offer reduced flexibility in dose adjustments, particularly for ICS and LAMA components. 47 It is speculated that this limitation may be more pronounced for SITT regimens administered once-daily compared to those with a twice-daily schedule (such as BDP/FF/GB); however, targeted investigations are required to further explore this issue.
Regarding SITT adherence trends, Busse et al. performed a retrospective cohort study to compare adherence and persistence among adult patients with asthma receiving single-inhaler FF/UMEC/VI versus multiple-inhaler triple therapy (MITT) in the United States. 47 The study included 1396 FF/UMEC/VI and 5115 MITT initiators. FF/UMEC/VI users were 49% more likely to persist at 12 months than MITT users (25.9% vs 15.1%, adjusted hazard ratio; p < 0.001). The conclusions were that patients with asthma initiating triple therapy with FF/UMEC/VI had significantly better adherence and persistence compared with MITT initiators.
Comprehensive data regarding adherence to therapy, alongside assessments of asthma control, impact on functional respiratory parameters, quality of life, and adverse events in patients utilizing SITT, is anticipated from an ongoing real-world observational study (TRIMAXIMIZE; ClinicalTrials.gov identifier: NCT06100042).
Costs
Asthma exacerbations, particularly those necessitating hospitalization, significantly contribute to direct healthcare costs. 48 Recent research evaluating the use of BDP/FF/GB in England has indicated advantages in terms of cost-effectiveness compared to alternative therapies. 48 Other treatment options for patients with asthma uncontrolled by ICS/LABA combinations include the addition of monoclonal antibodies (which are expensive and indicated only for specific patient subsets) or oral therapies such as leukotriene receptor antagonists or theophylline. These alternatives can increase treatment complexity, potentially impacting adherence and persistence BDP/FF/GB SITT, at both medium and high doses, has been described as cost-effective relative to medium- and high-dose BDP/FF, as well as open triple therapy (BDP/FF + tiotropium) in adults with uncontrolled asthma.
The role of extrafine SITT and small airway dysfunction
A significant feature of BDP/FF/GB SITT is its extrafine formulation. Recent findings have underscored the critical role of small airway dysfunction (SAD) in asthma control, particularly among patients classified within GINA steps 4 and 5.49,50 Consequently, SAD has increasingly recognized as an important “treatable trait” in asthma management (Figure 1). 51 The extra fine formulation facilitates the release of drug particles with a small aerodynamic diameter, optimizing drug deposition in peripheral airways. 31 Significant synergistic interactions among the ICS, long-acting beta-agonist (LABA), and LAMA have been observed in these regions. 20 The acknowledgment of SAD as a treatable trait in asthma therapy further supports the use of extrafine SITT. 52
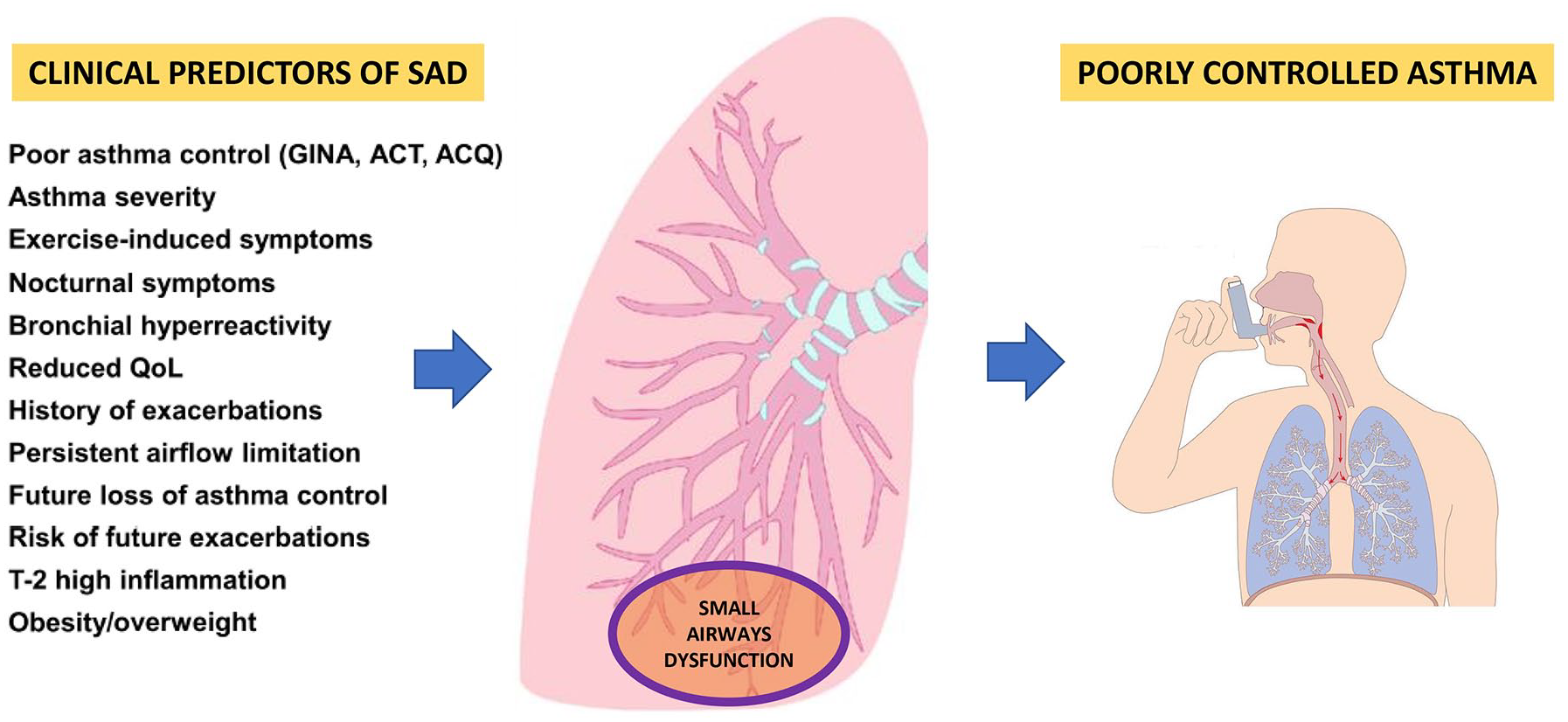
The presence of small airway dysfunction correlates with poor asthma control and several clinical predictors.
Conclusion
The incorporation of a LAMA alongside inhaled corticosteroids (ICS) and LABA represents a significant advancement in the management of uncontrolled asthma. Recent developments in therapeutic options have enabled the formulation of a combination therapy comprising ICS, LABA, and LAMA, notably exemplified by the extra fine BDP/FF/GB combination delivered via a single pressurized metered-dose inhaler (pMDI).
The TRIMARAN and TRIGGER studies, alongside subsequent post-hoc analyses, have illuminated the substantial benefits of this SITT in enhancing respiratory function and reducing exacerbation rates among patients with uncontrolled asthma. The BDP/FF/GB SITT demonstrates remarkable efficacy and safety across diverse ethnicities, including Caucasian, Japanese, and Chinese populations, as well as in various age groups, encompassing both adults and adolescents. However, it is noteworthy that current indications do not extend to individuals under the age of 18.
The unique advantage of administering all three therapeutic agents through a single pMDI facilitates the delivery of extra fine particles that can effectively penetrate even the smaller bronchi. This not only optimizes drug deposition but also has the potential to enhance therapeutic adherence by minimizing the complexities associated with multiple devices, thereby reducing the likelihood of suboptimal drug administration due to incorrect inhaler technique.
Furthermore, the cost-effectiveness of the BDP/FF/GB combination therapy adds another layer of appeal in the context of healthcare resource allocation. As the landscape of asthma management continues to evolve, future studies are poised to provide additional insights into clinical outcomes, safety, adherence, and potential new applications of this promising therapeutic strategy.
Another important point to evaluate the impact of SITT in asthmatics is the efficacy of persistent airflow limitation (PAL). Indeed, PAL has emerged as a potential predictor of improved therapeutic response in asthma treatment. In studies comparing extra fine particle BDP/FF/GB to BDP/FF, the efficacy was notably higher in patients exhibiting more reversible airway obstruction compared to those with less reversible obstruction. 53
Furthermore, SITT therapy has the potential to improve asthma symptoms and control postponing the requirement for initiating therapy with biological agents.
Last but not least, the ability to have two different formulations of fixed triple therapy with BDP/FF/GB (100/6/10 mcg vs 200/6/10 mcg) allows the clinician greater flexibility in setting or modifying therapy during follow-up and allows the recommendations of the major asthma guidelines for step-up or step-down to be taken into account.
In summary, the integration of BDP/FF/GB SITT into asthma management heralds a new era of enhanced patient care, offering a multifaceted approach to addressing the challenges of uncontrolled asthma while fostering improved quality of life for patients.
