Abstract
Background:
The extrafine single inhaler triple therapy (efSITT) containing beclomethasone dipropionate/formoterol fumarate/glycopyrronium 87/5/9 μg has proved to be efficacious in patients with chronic obstructive pulmonary disease (COPD) in randomized control trials.
Objective:
TRIWIN study evaluated the effectiveness of efSITT delivering beclomethasone dipropionate/formoterol fumarate/glycopyrronium 87/5/9 μg in COPD patients previously treated with multiple-inhaler triple therapy (MITT) in a real-world study in Greece.
Design:
Prospective, multicenter, observational, non-interventional study was conducted over 24 weeks.
Methods:
A total of 475 eligible patients had moderate-to-severe COPD, an indication for treatment with efSITT, and were symptomatic despite receiving MITT. COPD Assessment Test (CAT) score, pulmonary function parameters, use of rescue medication, and adherence to inhaler use were recorded at baseline (Visit 1), 3 (Visit 2), and 6 months (Visit 3) after treatment.
Results:
Mean CAT score decreased from 21.4 points at Visit 1, to 16.6 at Visit 2 and 15.1 at Visit 3 (p < 0.001 for all pair comparisons). At Visit 3, 79.8% of patients reached a CAT improvement exceeding minimal clinically important difference (⩾2), compared to baseline. Mean forced expiratory volume in 1 s (%pred.) increased from 55.4% at Visit 1 to 63.5% at the end of study period (p < 0.001), while mean forced vital capacity (%pred.) increased from 71.1% at Visit 1, to 76.7% at Visit 3 (p < 0.001). The mean Test of Adherence to Inhalers score increased from 42.5 to 45.3 and 46.3 points, for the three visits, respectively (p < 0.001 comparing Visits 1/2 and Visits 1/3; p = 0.006 comparing Visits 2/3). The percentage of patients showing good adherence rose from 33.7% at baseline to 58.3% at Visit 3. The percentage of patients using rescue medication during the last month dropped from 16.2% to 7.4% at the end of study period (p < 0.001). Pulmonary function parameters also improved.
Conclusion:
The TRIWIN results suggest that extrafine beclomethasone dipropionate/formoterol fumarate/glycopyrronium is effective in improving health status, pulmonary function, and adherence and in reducing rescue medication use in COPD patients previously treated with MITT, in a real-world setting in Greece.
Keywords
Background
Chronic obstructive pulmonary disease (COPD) can have a major effect on patients’ health status and quality of life, imposing a significant burden on public health, due to its increasing morbidity and mortality, especially when associated with episodic exacerbations. 1 Indeed, prevalence of COPD at a global level outreaches 10% 1 and COPD is the third leading cause of death worldwide, with more than 3.2 million deaths being attributed to this respiratory disease. 2 The aim of current treatment is to improve symptoms and quality of life and prevent exacerbations and disease progression.1,3–6 Current guidelines suggest that triple therapy with a long-acting beta-2 agonist (LABA), an inhaled corticosteroid (ICS), and a long-acting muscarinic antagonist (LAMA) should be used in COPD patients with exacerbations despite LABA/LAMA combination or long-acting bronchodilator monotherapy (when blood eosinophils are ⩾100 and ⩾300, respectively). 1 Furthermore, it is now recommended to initiate treatment with triple therapy in patients with exacerbations and high blood eosinophil count, irrespective of symptom level, although evidence is still missing to support this practical guidance. 1 Previous recommendations have suggested that triple therapy should also be considered for those who remain symptomatic despite dual therapy with LABA/LAMA or LABA/ICS.1,7 In the same context, NICE suggests a 3-month trial with triple therapy in patients remaining symptomatic despite LABA/LAMA use. 8 Besides recommendations, it seems that in everyday clinical practice, COPD populations that are treated with triple therapy are much larger, with approximately 30% of patients in Greece using triple therapy 9 and almost 100% of UK COPD patients progressing to triple therapy after 8 years. 10
Extrafine triple therapy in a single inhalation device containing beclomethasone dipropionate, formoterol fumarate, and glycopyrronium (BDP/FF/G) has been proven to be efficacious in improving COPD outcomes in large, phase III, randomized controlled trials (RCTs), including more than 6000 patients.11–14 Stepping up from dual combinations or monotherapy (ICS/LABA, LABA/LAMA, or LAMA) was shown to improve lung function and exacerbation rate.11,13,14
The TRINITY study assessed the efficacy of extrafine single inhaler triple therapy (efSITT) versus a triple combination in two separate devices with a non-inferiority analysis and effects were shown to be similar. 12 Nevertheless, it must be highlighted that in this study the compliance to treatment outreached 94%. 12 Outside the setting of a clinical trial, in everyday clinical practice, the use of multiple inhalers has been linked with reduced adherence to treatment while it is quite common that COPD patients using multiple inhalers make critical errors associated with the use of two different devices.15,16 This may possibly interfere with treatment effectiveness regarding symptoms and/or exacerbations. 17 In this context, the Global Initiative for Chronic Obstructive Lung Disease (GOLD) suggests the use of a single inhaler therapy in managing COPD to improve convenience and disease outcomes. 1
The efSITT has been also evaluated in terms of effectiveness in real-world studies that included broad populations in different European countries.18–20 TRICOP 18 and TriOptimize 19 studies included mixed patients previously treated with dual or open triple combinations, whereas the first study evaluating a single inhaler triple therapy in Greece, TRIBUNE, included data from patients with moderate-to-severe COPD with history of at least one exacerbation despite dual therapy. 20 Results confirmed improvements in health status, lung function, and rescue medication use.18–20
We conducted a prospective, multicenter, non-interventional, observational 24-week study aiming to evaluate the effectiveness of efSITT in changing health-related quality of life in symptomatic moderate or severe COPD patients, despite treatment with triple therapy via multiple devices, in Greece. Furthermore, after 24 weeks of efSITT treatment, adherence to therapy, pulmonary function changes, and rescue medication utilization were evaluated.
Methods
Study design
TRIWIN was a multicenter, non-interventional study with an observational and prospective design, which included 475 patients all over Greece. In total, 55 pulmonologists/investigational sites and 2 coordinating hospitals participated in recruiting patients and collecting data. Collection of data was performed from December 2020 (first patient first Visit) to January 2022 (last patient last Visit) and covered a 24-week span of the patient’s treatment period. Three study points included Visit 1 (enrollment to the study/baseline), Visit 2 (12 weeks after enrollment), and Visit 3 (24 weeks following patients’ enrollment). The medicinal product under observation was Trimbow® 87/5/9 μg, delivering at each dose 87 μg of beclomethasone dipropionate, 5 μg of formoterol fumarate, and 9 μg of glycopyrronium via a pressurized metered dose inhaler (pMDI). This was administered in consonance with the product’s summary of characteristics (SmPC) in the customary manner within the routine of medical practice. The decision for enrollment in the study was made independently of the physician’s decision to prescribe treatment with study medication to the patients. This was administered, as per SmPC, meaning two inhalations twice daily. The patients participating in this study had not undergone any other procedures for the diagnosis and monitoring, beyond those used in standard practice.
Patients
Eligible patients were at least 40 years old and no more than 75 years old, with a diagnosis of moderate-to-severe COPD [30% ⩽ forced expiratory volume in 1 s (FEV1) < 80% pred.] who remained symptomatic, having a CAT score ⩾10, in spite of receiving treatment with multiple-inhaler triple therapy (MITT, meaning ICS/LABA plus LAMA or LABA/LAMA plus ICS). Therefore, a single inhaler delivering BDP/FF/G was needed as a maintenance treatment for those patients, according to the judgment of the treating physician. Diagnosis of asthma, and other respiratory or unstable concurrent conditions, that could interfere with study results, were among the exclusion criteria. Lower respiratory tract infection or disease exacerbation treated with antibiotic and/or oral steroids within 8 weeks before screening, participation in interventional studies within 30 days in advance of enrollment, and long-term oxygen therapy also prevented patients from being enrolled in this study. The study was conducted according to the guidelines of the Declaration of Helsinki, Good Clinical Practice guidelines (ICH/GCP), and was approved by the Institutional Review Boards (or Ethics Committees) of Euromedica General Clinic, Pulmonary Department, Thessaloniki (protocol code 00351/23-12-2020) and Athens Medical Center – Palaio Faliro Clinic (record 70, 03-12-2020). All participants provided their informed consent to participate.
Procedures
The documentation period lasted 24 weeks and comprised three visits to the pulmonologist, as per usual practice. At Visit 1, anthropometric and socio-demographic data such as age, gender, Body Mass Index (BMI), smoking history, and comorbidities were collected. Disease-specific information included duration of COPD, number of disease exacerbations during the prior year, and previous COPD treatment. Each patient was given thorough information about the collection, use, and disclosure of their personal data before being enrolled, and they were required to provide signed informed permission. The COPD Assessment Test (CAT) was used to measure each patient’s disease burden at baseline, after 12 weeks, and after 24 weeks. Additionally, measurements of pulmonary function, including FEV1 and forced vital capacity (FVC), were taken at baseline and at subsequent visits. Furthermore, the ‘Test of Adherence to Inhalers’ (TAI) questionnaire was administered in all visits to detect level of adherence to inhalation therapy in everyday situations. Finally, the use of rescue medication was recorded at all visits, corresponding to the last 4 weeks before the relevant visit (the patient was advised to keep a note whenever use of rescue medication was performed).
Outcomes
The main objective of TRIWIN study was to evaluate the change in health status from baseline, as measured by the CAT score, 24 weeks following the start of treatment with BDP/FF/G, with measurements being included in all three study visits. CAT is a validated, short (8-item), and simple questionnaire completed by the patient, with good discriminative properties, developed for use in everyday clinical practice to evaluate COPD patients’ health status. 21 Each of the eight questionnaire items refers to a specific COPD symptom and is scored by a six-point grading scale (0 = absence of symptom, 5 = most severe symptom). The sum of the eight answers comprises the CAT total score (minimum = 0, maximum = 40), with lower values indicating lower disease burden. The minimal clinically important difference (MCID) for the total CAT score is 2 points according to the literature. 22
Changes in pulmonary function measures (FEV1 and FVC% pred.) and treatment adherence as measured by the TAI were the secondary objectives. Latest American Thoracic Society (ATS) and European Respiratory Society (ERS) guidelines were followed in performing spirometry. 23 TAI questionnaire is a validated questionnaire designed to detect level of adherence to inhalation therapies in everyday practice. 24 The questionnaire which was used in the present study comprises 10 items and the scoring for each question extends from 1 showing worst adherence to 5 denoting best adherence. Good adherence is indicated by a score of 50 points, intermediate adherence by a score of 46–49, and poor adherence by a score of 45 or less. TAI was assessed in all three visits.
Finally, rescue medication was also assessed, as another parameter, looking at the number of days within each month before each one of the three study visits that the participant used rescue medication.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 25 The checklist from STROBE was provided in the Supplemental Material.
Statistical analysis
Sample size calculation
The calculation of the sample size was based on the primary parameter of the study, that is, CAT and more specifically the change in total CAT score between baseline and after 24 weeks of treatment. In the article ‘Minimum clinically important difference for the COPD Assessment Test: a prospective analysis’, 22 three previous studies examining the change of CAT in COPD patients were presented. In these studies, the following 95% CI. Is of the CAT change were calculated: Study 1 (n = 565): −2.5 ± 1.1, Study 2 (n = 147): −3.0 ± 2.8, Study 3 (n = 164): 0.6 ± 1.9.
For the sample size calculation, the information obtained from the second study was used, because it presented the widest confidence interval (CI) and the largest estimation (conservative choice) of standard deviation = 8.66. Finally, the sample size was estimated using a 95% CI for the above-mentioned change in CAT score, and a precision of 0.76 (approximately 40% of the minimum clinically important difference of the CAT score = 2 points).
Thus, when the sample size is 500, a two-sided 95.0% CI for the difference in paired CAT mean scores will extend 0.76 from the observed mean, assuming that the standard deviation (SD) is known to be 8.66 and the CI is based on the large sample z statistic.
To calculate the sample size, the statistical software used was nQuery Advisor 7.0 (Statistical Solutions Ltd. 4500, Rathmacullig West, Cork, T12 NX7D, Ireland).
Statistical methods
The continuous study parameters (e.g. age) were presented using mean ± standard deviation, while the nominal ones (e.g. gender) were presented using tables of frequencies.
All CIs were of normal approximation, 95%, two-sided.
For all statistical tests, α = 0.05.
A 95% CI was calculated for the mean difference between the values of the total CAT score at baseline (Visit 1), and its values after the 6-month treatment (Visit 3).
A 95% CI was computed for the percentage of patients who achieved an improvement in CAT score of 2 points (minimum clinically important difference) or more, on the third visit.
Repeated measures analysis of variance tested the change of each study parameter over time. Paired samples t-test was used for the (three) pairwise comparisons between visits (using Bonferroni correction).
The relation of the differences of each study parameter between the first and the third visit (dependent variable) was tested using multiple linear regression, with the following parameters: (1) Gender, (2) Age, (3) baseline BMI, (4) smoking habit, (5) number of exacerbations/hospitalizations due to COPD the last 12 months, (6) previous COPD treatment, (7) average TAI score during the study, and (8) baseline values of the corresponding parameter (independent variables). It must be noted that the parameter of concomitant COPD medications was not used as an independent variable, since only one patient received theophylline during the study and only one patient received roflumilast.
Cochran’s Q test tested the change in the proportion of patients receiving rescue medication over time (Visits 1, 2, 3), while McNemar test was used for pairwise comparisons between visits (using Bonferroni correction).
Role of the funding source
The study’s sponsor was in charge of its design and analysis, oversaw the execution, and was responsible for preparing the study report. All authors could access all study data.
Results
Patient characteristics
A total of 475 patients meeting the inclusion criteria participated in the TRIWIN study, in an observation period that lasted 24 weeks. Only five patients (i.e. 1.1% of the sample), proceeded to premature treatment discontinuation. Table 1 displays the main demographic data and the characteristics of the patients included. Most of the patients were males (64%) with a mean age of 64.9 ± 8 years. Of them, 51.6% were active smokers, while 48.4% were ex-smokers, and the mean duration of COPD from diagnosis to study enrollment was 6.8 ± 5.7 years. Most of the study participants (69.9%) experienced at least one exacerbation/hospitalization due to COPD the year before, while 30.1% reported no exacerbations in the previous year. Moreover, the most common treatment schemes previously received by the patients were ICS/LABA (fixed combination) plus LAMA (66.7%) and LABA/LAMA (fixed combination) plus ICS (29.7%). Additionally, 149 patients (31.0%) had at least one comorbidity and the most frequent disease reported was Arterial Hypertension (17.7%).
Population demographics and traits (N = 475).

COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroid; LABA, long-acting beta-2 agonist; LAMA, long-acting muscarinic antagonist.
Eight patients had “Underweight” BMI classification.
Smokers were approximately four years younger than ex-smokes (t-test p-value<0.001)
COPD Assessment Test
The use of efSITT was associated with significant improvement (i.e. reduction) in CAT score of participating patients in all study visits. The mean CAT score was reduced from 21.4 points at Visit 1, to 16.6 at Visit 2 and 15.1 at Visit 3 (p < 0.001 for all pair comparisons) [Figure 1(a)]. CAT score decreased on average by 6.3 ± 0.8 points (95% CI: 5.5–7.1) at Visit 3 compared to Visit 1, which is almost 3 times the CAT MCID of 2 points. It must also be mentioned that the precision of the above-mentioned CI using two decimal places, is 0.79, which is very close to the original planned precision of 0.76, used for sample size calculation. 79.8 ± 3.6% of patients (N = 379, 95% CI: 76.2–83.4) experienced a CAT drop of MCID or more at Visit 3 compared to baseline. A statistically significant higher CAT improvement at Visit 3 compared to baseline, was achieved by younger patients (p < 0.018), patients with higher baseline CAT score (p = 0.018), and smokers (p = 0.014), as per multiple linear regression analysis. Importantly, all CAT score single items showed a decrease over time (for the difference between Visits 1 and 3, p < 0.001 for all items). ‘Phlegm’ (second item), ‘I walk up a hill’ (fourth item), and ‘Cough’ (first item) showed the largest reductions, while the item with the lowest decline was ‘I leave my home’ (sixth item) [Figure 1(b)].
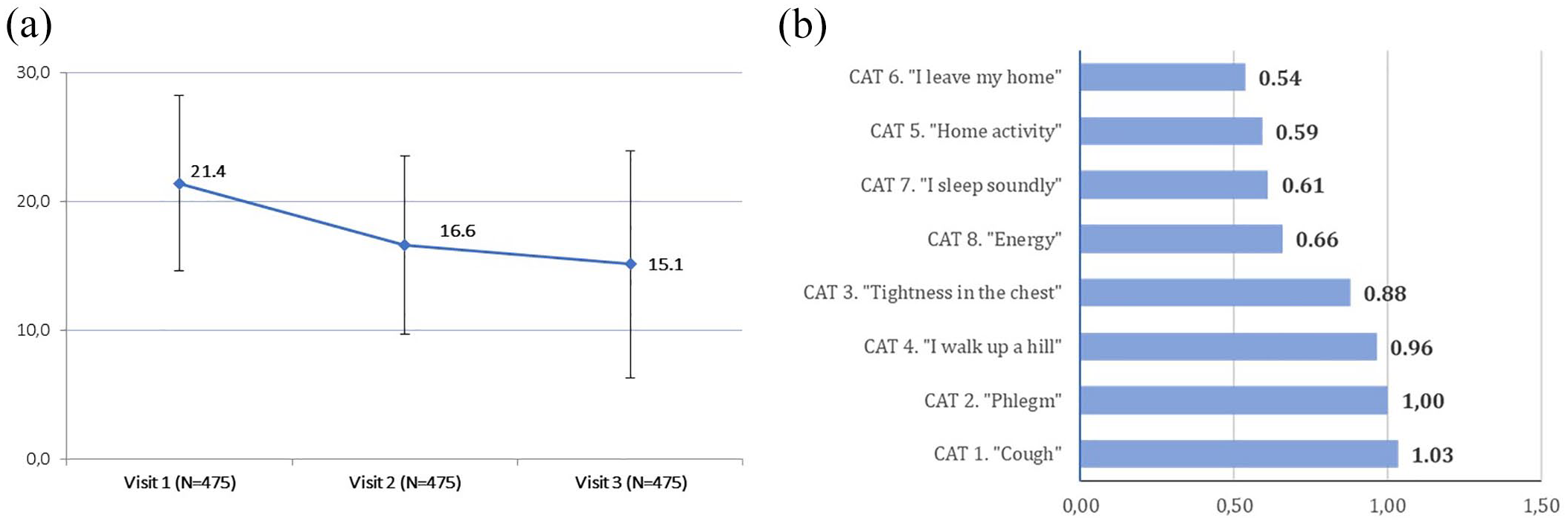
(a) Mean ± SD CAT scores on all study visits. Range of health status: from 0 for best to 40 for worst (p < 0.001 for all pair comparisons). (b) CAT items: mean differences between Visits 1 and 3. Range of health status: from 0 for best to 40 for worst.
Pulmonary function
Spirometry data were available in 264 patients at Visit 1, 65 patients at Visit 2, and 104 patients at Visit 3. Spirometry was performed at all study visits in only 38 patients, while it was performed at both Visits 1 and 3, in 99 patients. Therefore, statistical analysis was applied in this sub-set of 99 participants, excluding Visit 2 measurements. A statistically significant improvement was observed in FEV1 between Visits 1 and 3, with the mean FEV1% predicted rising from 55.4% at Visit 1 to 63.5% at Visit 3 (8.1% increase, p < 0.001). FEV1 increased from 1.7 ± 0.5 to 2.0 ± 0.9 L. Similar changes were observed for the FVC, with mean FVC (% pred.) increasing from 71.1% at Visit 1, to 76.7% at Visit 3 (5.6% increase, p < 0.001) (Figure 2). Multiple linear regression suggested that FEV1% predicted improvement was statistically significantly higher in smokers (p = 0.001) and in patients with previous triple therapy in three separate devices (p = 0.005).

Mean ± SD FEV1% predicted and FVC% predicted, at Visits 1 and 3, p < 0.001 for both comparisons (N = 99 patients with spirometry data available at first and last Visit).
Test of Adherence to Inhalers
The mean TAI score increased from 42.5 points on Visit 1, to 45.3 on Visit 2 and 46.3 on the last one (p < 0.001 for comparison between Visits 1/2 and Visits 1/3 and p = 0.006 for comparison between Visits 2/3) [Figure 3(a)]. ‘Good adherence’ (TAI score of 50/50) was reported by 33.7% of patients at Visit 1, 50.7% at Visit 2, and 58.3% at Visit 3 [Figure 3(b)]. Smokers showed higher TAI score improvement, while patients with higher baseline TAI scores and patients under previous treatment with LABA/LAMA plus ICS, showed lower TAI score improvement at Visit 3 compared to Visit 1.

(a) TAI score at Visits 1, 2, and 3: Mean ± SD. Score ranges: 50 points = Good adherence, 46–49 points = Intermediate adherence, and ⩽45 points = Poor adherence. p < 0.001 for comparison between Visits 1/2 and p = 0.006 for comparison between Visits 2/3. (b) Percentage of patients demonstrating various adherence levels, as measured by TAI at Visits 1, 2, and 3. Score ranges: 50 points = Good adherence, 46–49 points = Intermediate adherence, and ⩽45 points = Poor adherence.
Use of rescue medication
The proportion of participants who took rescue medication in the month prior to each study Visit dropped from 16.2% on Visit 1, to 5.5% on Visit 2 and 7.4% on Visit 3 (p < 0.001 for the comparison between Visits 1/2 and 1/3, statistically non-significant for the comparison between Visits 2/3) [Figure 4(a)]. At Visit 1 the mean (±SD) number of times of rescue medication use was 0.8 ± 3.4 while this was only 0.3 ± 1.9 at Visit 2 and 0.4 ± 2.0 at Visit 3 (p < 0.001 for comparison between Visits 1/2 and 1/3, statistically non-significant for the comparison between Visits 2 and 3) [Figure 4(b)]. Statistically, the reductions were significantly greater in patients with higher rescue medication use at baseline (p < 0.001).

(a) Percent (%) of patients who took rescue medication in the month prior to each study Visit. p < 0.001 for comparison between Visits 1/2 and 1/3, non-statistically significant for the comparison between Visits 2/3. (b) Mean ± SD of times of rescue medication use in the month prior to each study Visit. p < 0.001 for comparison between Visits 1/2 and 1/3, non-statistically significant for the comparison between Visits 2/3.
Discussion
This multicenter, non-interventional, prospective, observational study provides valuable insights into the real-world effectiveness of efSITT containing BDP/FF/G, in symptomatic COPD patients previously treated with MITT in Greece. The population studied is representative of the patients with COPD encountered in everyday clinical practice in Greece, with more than 80% of them having moderate-to-severe disease. 9 Therefore, the data are applicable to the COPD population in Greece. We demonstrated that starting treatment with a single inhaler triple therapy improves health status, pulmonary function parameters, and the need for rescue medication in patients who had previously received treatment with MITT. Moreover, the adherence to treatment was significantly improved, as reflected by the increase in mean TAI score.
The efficacy of efSITT containing BDP/FF/G in a pMDI device has been extensively investigated in large RCTs.11–14 However, due to the stringent selection criteria for the participating patients, RCTs are less typical of real-world conditions. 26 Therefore, the use of countrywide data in order to perform real-world observational studies, like TRIWIN, contributes to the evaluation of longer-term outcomes, thus providing valuable evidence to supplement the findings of RCTs. Furthermore, real-life evidence reflecting everyday clinical practice is necessary to demonstrate effectiveness, expand applicability, enrich clinical trial data, and get up-to-date physicians with practice patterns. The final aim is to improve and upgrade the quality of health services received by patients. 26 Observational real-world studies cast a broader analysis network through the evaluation of randomly chosen, representative patients treated in real-world clinical practice. 26
In TRIWIN study, the CAT score at Visits 2 and 3 after the start of efSITT decreased, indicating a significant improvement in health status. In comparison to Visit 1, the average CAT score decreased by 6.3 points at last Visit, exceeding three times the MCID, 22 thus suggesting that patients will notice these beneficial changes. 27 Our results are in agreement with those of the first real-life study assessing efSITT, namely TRICOP study, 18 as well as with the TRIBUNE study, although the last one only included patients previously treated with dual combinations. 20 On the contrary, these findings outreach CAT score improvement in the cohort previously treated with open triple combinations, in TriOptimize study, a fact that could be attributed to differences in the population studied (lower FEV1, very severe COPD included, at least one exacerbation in the previous year). 19 After receiving therapy with efSITT, a significant shift of 70% of patients who were initially categorized in group GOLD D into group GOLD B was shown in the Austrian cohort. 18 Following the same pattern, and further confirming results of TRIBUNE study, 20 we found that younger patients, smokers, and those with a higher CAT score at baseline showed greater improvements. Given correlations of CAT with disease outcomes28,29 and the wide applicability of the questionnaire in clinical practice, TRIWIN results cannot be overlooked.
Furthermore, pulmonary function parameters significantly improved after 6 months of treatment, with changes exceeding RCTs11–14 and real-world study results.18–20 Since it is well recognized that the severity of the disease is a significant determinant of increased mortality in COPD,30,31 the recent findings on COPD severity reduction, as indicated by improvements in pulmonary function parameters and CAT scores, acquire extra significance.
This real-life study also showed reduction of rescue medication use in symptomatic patients with moderate-to-severe COPD switching from MITT to efSITT. Rescue medication use is usually assessed in asthma and COPD studies. 32 Even though asthma recommendations aim at reducing the rescue medication need, 33 COPD recommendations do not highlight this patient-reported outcome. 1 Nevertheless, changes in rescue medication use in COPD have been associated with improvements in FEV1, St. George’s Respiratory Questionnaire score, and even rate reduction of moderate/severe exacerbations 31 while onset and duration of medication action have been identified by physicians as important parameters that increase patients’ compliance. 34
Another important finding in our study is the good adherence and persistence with efSITT. The proportion of patients reporting good adherence showed a sharp increase from 33.7% at Visit 1, to 50.7% at Visit 2, further rising to 58.3% at Visit 3 and therefore, adherence to the fixed triple combination of BDP/FF/G in the present study was satisfactory. When different inhaler devices were compared in a significant multicenter trial, it was shown that inappropriate usage of the devices may be highly common in outpatients and, if left uncorrected, may result in poor disease control. 35 Consequently, using a single inhaler to administer a mix of medications may aid in proper dosing, enhance adherence, and may also boost efficacy of the intervention. In patients with COPD, non-adherence to therapy ranges from 28% and 74% in affluent countries, 1 and inhaler mishandling is still a common occurrence in real-life settings. 1 TRIWIN results accompanied by low discontinuation rates (1.1% in the sample) suggest treatment persistence with fixed triple combination of BDP/FF/G, confirming retrospective data from France showing that patients treated with SITT show higher persistence than those treated with MITT. 36
This is the first study evaluating effectiveness of a single inhaler triple therapy in patients previously treated with MITT, using countrywide data in Greece, reflecting daily clinical practice in our country and perceptions of COPD patients, while complementing data of the TRIBUNE study. TRIWIN is also the first study to include exclusive patients switching from MITT to efSITT. It is noteworthy that, according to previous studies, this population consists of marginally 50% of the pool of patients being prescribed BDP/FF/G.18,19 The fact that TRIWIN achieved all primary and secondary outcomes despite that the overall study population was already receiving MITT, underscores the effectiveness of BDP/FF/G combination.
In terms of study limitations, these are inherent to the nature of the real-life settings, including the absence of comparator arm in a matched cohort during the same study period and the observational design. Another limitation of the present study refers to the spirometry data which were available only in a subgroup of 99 patients at Visits 1 and 3, a fact that could be attributed to restrictions in clinical practice due to COVID-19, 37 since our study was conducted during the recent pandemic. As spirometry data were only available for a subpopulation of the study, we included patients belonging to different GOLD groups, without differentiating the results. Consequently, ameliorations of patients’ health status should be interpreted cautiously. After all, instead of focusing on changes in pulmonary function, which is a well-validated outcome for efSITT, the TRIWIN study examined changes in the health-related quality of life as reported by patients in a real-world setting. Finally, rescue medication was reported by patients recalling the number of days of utilization during the last month and therefore, lack of a diary may have had an impact on accuracy of data, though we expect this to be minor due to the short duration of recall.
Conclusion
TRIWIN study provides evidence that efSITT containing BDP/FF/G improves health status, pulmonary function, and adherence to treatment while reducing use of rescue medication in patients with moderate-to-severe COPD who are still symptomatic despite MITT in a real-world setting in Greece.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241263439 – Supplemental material for Extrafine single inhaler triple therapy effectiveness in COPD patients previously treated with multiple-inhaler triple therapy: the TRIWIN study
Supplemental material, sj-docx-1-tar-10.1177_17534666241263439 for Extrafine single inhaler triple therapy effectiveness in COPD patients previously treated with multiple-inhaler triple therapy: the TRIWIN study by Paschalis Steiropoulos, Niki Georgatou, George Krommidas, Konstantinos Bartziokas, Aliki Korkontzelou, Panos Katerelos, Petros Efstathopoulos, Dimosthenis Papapetrou, Stavros Theodorakis and Konstantinos Porpodis in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
