Abstract
Background:
Evidence on how decisions regarding escalation to triple therapy and de- or re-escalation are taken and the rationale on which these decisions are based is currently limited in Germany.
Objectives:
The TETRIS study aims to elucidate influences on treatment decisions surrounding triple therapy in a real-world practice setting in Germany.
Design:
TETRIS is an ongoing, multicenter, prospective, observational cohort study recruiting patients with chronic obstructive pulmonary disease (COPD) with or without asthma who have already been treated with triple therapy for 2–48 weeks.
Methods:
For better representation of the treatment reality in Germany, patients are recruited from general practitioners and pulmonologists. Data are collected in two parts. Part 1 involves cross-sectional phenotyping of patients at enrollment. Part 2 involves a 2-year longitudinal follow-up period to monitor/document all visits by the patients during the 24-month observation period per routine clinical practice. Here, we report the demographic and baseline characteristics of 1213 eligible patients recruited to part 1 of the study.
Results:
The mean patient age was 66.4 years overall, and 29.3% (356/1213) of patients had no comorbidities. The mean CAT score was 19.4; the number of exacerbations and hospitalizations due to exacerbations in the past 3 years before starting triple therapy was 0.6 and 0.1, respectively. Dual bronchodilation with a long-acting muscarinic antagonist (LAMA) plus a long-acting β-2 agonist (LABA) was the most common therapy for COPD before initiation of triple therapy in 58.3% of patients.
Conclusion:
In this real-world setting in Germany, patients with COPD have a relatively low reported exacerbation rate but high symptom burden, and over 70% are multimorbid. Triple therapy is initiated in patients who are primarily highly symptomatic despite being on LAMA + LABA. Future prospective studies in patients with multimorbidity are warranted to better understand the treatment landscape across the disease spectrum.
Trial registration:
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a major public health burden globally, given that it is the third leading cause of death and the seventh leading cause of poor health and is also associated with considerable economic and social implications.1–3 Globally, COPD affected 212.3 million individuals and accounted for 3.3 million deaths and 74.4 million disability-adjusted life years in 2019. 4 In Germany, the prevalence of Global Initiative for Chronic Obstructive Lung Disease (GOLD) COPD stage ⩾I was 13.2% (GOLD stage I: 7.4%; II: 5.0%; III: 0.8%; and IV: 0.8%). 5 According to a retrospective claims data analysis in Germany, 14.4% of patients with any COPD diagnosis had at least one COPD-related hospitalization during the first year after a diagnosis of incident COPD. The total direct COPD-related healthcare costs per observed patient-year was €3693, with inpatient costs being the main driver at €2922 per patient-year, accounting for 79.1% of the total costs. Additionally, from a statutory health insurance perspective, COPD-related healthcare costs accounted for 10% of the overall total healthcare cost (€37,061). 6
Pharmacotherapy for COPD is mainly aimed at reducing symptoms and the frequency and severity of exacerbations and improving exercise tolerance and health status. 1 Inhaled bronchodilators are central to the management of COPD symptoms. Bronchodilation with a long-acting muscarinic antagonist (LAMA), a long-acting β-2 agonist (LABA), or a combination of both forms the mainstay of pharmacotherapy in patients with COPD, depending on the severity of breathlessness and the risk of exacerbations.1,7 The GOLD guidelines recommend a step-up to triple therapy with a LAMA plus a LABA plus an inhaled corticosteroid (ICS) for (i) patients with persistent breathlessness or exercise limitation or further exacerbations despite LABA + ICS therapy and (ii) patients who continue to experience further exacerbations despite LAMA + LABA therapy and have an eosinophil count ⩾100 cells/µL.8,9 The German National Care Guideline for COPD (2021) recommends escalation to triple therapy with LAMA + LABA + ICS for patients who experience further exacerbations while on treatment with dual bronchodilation (i.e., LAMA + LABA). The guideline group considers a higher eosinophil count (i.e., >300 cells/µL) in the differential blood count as a possible indication for the additional administration of an ICS. Furthermore, a confirmed diagnosis of asthma or atopy and significant variations in forced expiratory volume in 1 second (FEV1) over a longer period of time (at least 400 mL) are indications for a possibly increased response to the inhalation of steroids. 10 Triple therapy can be administered either through a single inhaler device that combines all three drugs (i.e., LAMA + LABA + ICS) and delivers them in a single dose (referred to as single-inhaler triple therapy (SITT)) or through multiple inhaler devices that deliver separate doses of each of the three drugs (referred to as multiple-inhaler triple therapy (MITT)).11,12 MITT usually involves using three separate inhalation devices to deliver the three single drugs or using two separate inhalation devices to combine ICS+LABA with a LAMA, or LABA + LAMA with an ICS. 12 The use of SITT when compared to MITT has been reported to be simple, potentially cost-effective, and associated with significantly improved treatment adherence.12,13
Available evidence indicates that treatment decisions in routine clinical practice in Germany and elsewhere may be different and can deviate from international and national guideline recommendations.6,14 To determine adherence to guidelines, effect of national disease management programs (DMPs) on treatment outcomes, and understand how patients progress to triple therapy, it is important to assess the characteristics and treatment patterns of patients before the initiation of triple therapy. However, evidence on how decisions regarding escalation to triple therapy and de- or re-escalation are taken and the rationale on which these decisions are based in real-world clinical practice is currently limited in Germany.
The Triple thErapy in paTients with COPD under Real lIve Setting (TETRIS) study aims to provide information on the characteristics and number of patients with COPD (including those with comorbidities like asthma) remaining continuously on triple therapy in Germany. The reasons for treatment changes are hardly known, as are the possible clinical consequences. Therefore, this observational study will also capture the reasons for initiating SITT or MITT considering existing treatment recommendations in Germany. Here, we report the key demographic and baseline characteristics of patients enrolled in the TETRIS study.
Materials and methods
Study design and population
TETRIS is an ongoing, prospective, noninterventional, multicenter cohort study. The reporting of this study follows the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) guidelines for observational studies. 15 The study is recruiting patients with COPD with or without comorbid asthma who were already treated with triple therapy for at least 2 weeks but not longer than 48 weeks. To obtain a better representation of the treatment reality in Germany, patients are recruited from two different physician groups covering all German regions: (i) general practitioners (GPs; primary care) and (ii) pulmonologists (specialists; primary or secondary care).
Data are collected in two parts. Part 1 involves cross-sectional phenotyping of patients at study enrollment (visit 1). Part 2 involves a 2-year longitudinal follow-up period to monitor/document all visits by the study patients during the 24-month observation period (i.e., visits 2, 3, and 4 after approximately 6, 12, and 24 months, respectively) per normal clinical practice. Patients will be followed up for 2 years, and, in total, the data from all four visits will be documented (Figure 1 and Supplemental Table 1). Patient characteristics, including sociodemographic profiles, comorbidities, and treatment history, are recorded at baseline. Here, we report the demographic and baseline characteristics of patients in part 1 of the study.
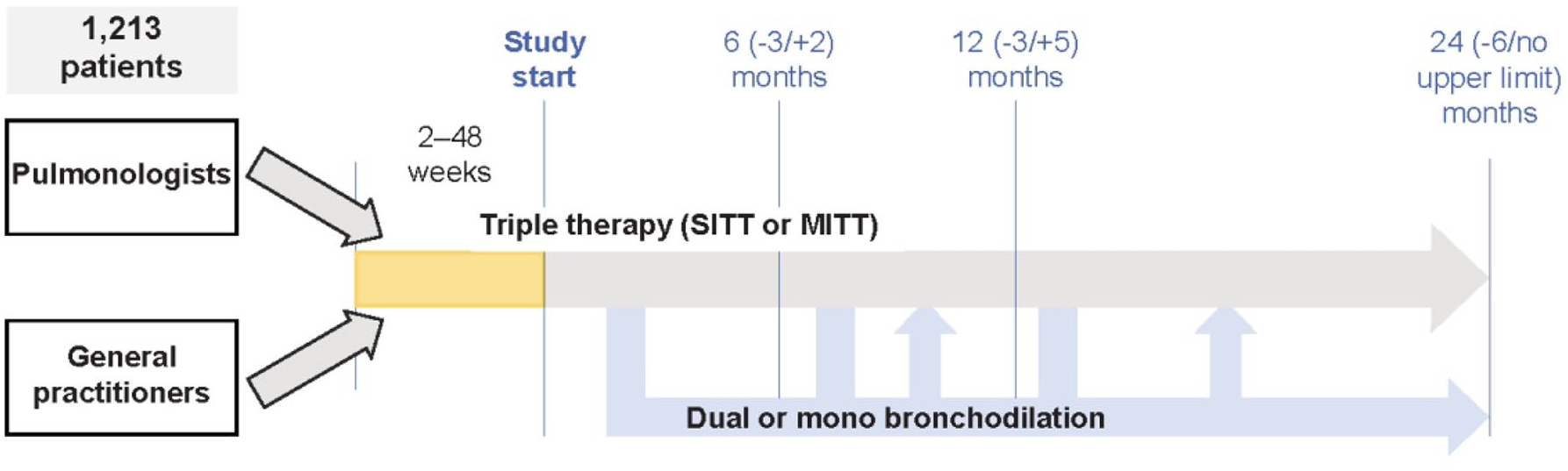
TETRIS—Study design.
Eligibility criteria
Patients were recruited if they were aged ⩾18 years, had a diagnosis of COPD with or without comorbid asthma, and were on triple therapy with LAMA + LABA + ICS for a period of 2–48 weeks before enrollment. Patients were excluded if they had a diagnosis of lung cancer or lung metastasis or only asthma without comorbid COPD. A detailed list of the inclusion and exclusion criteria is provided in Table 1.
Eligibility criteria.
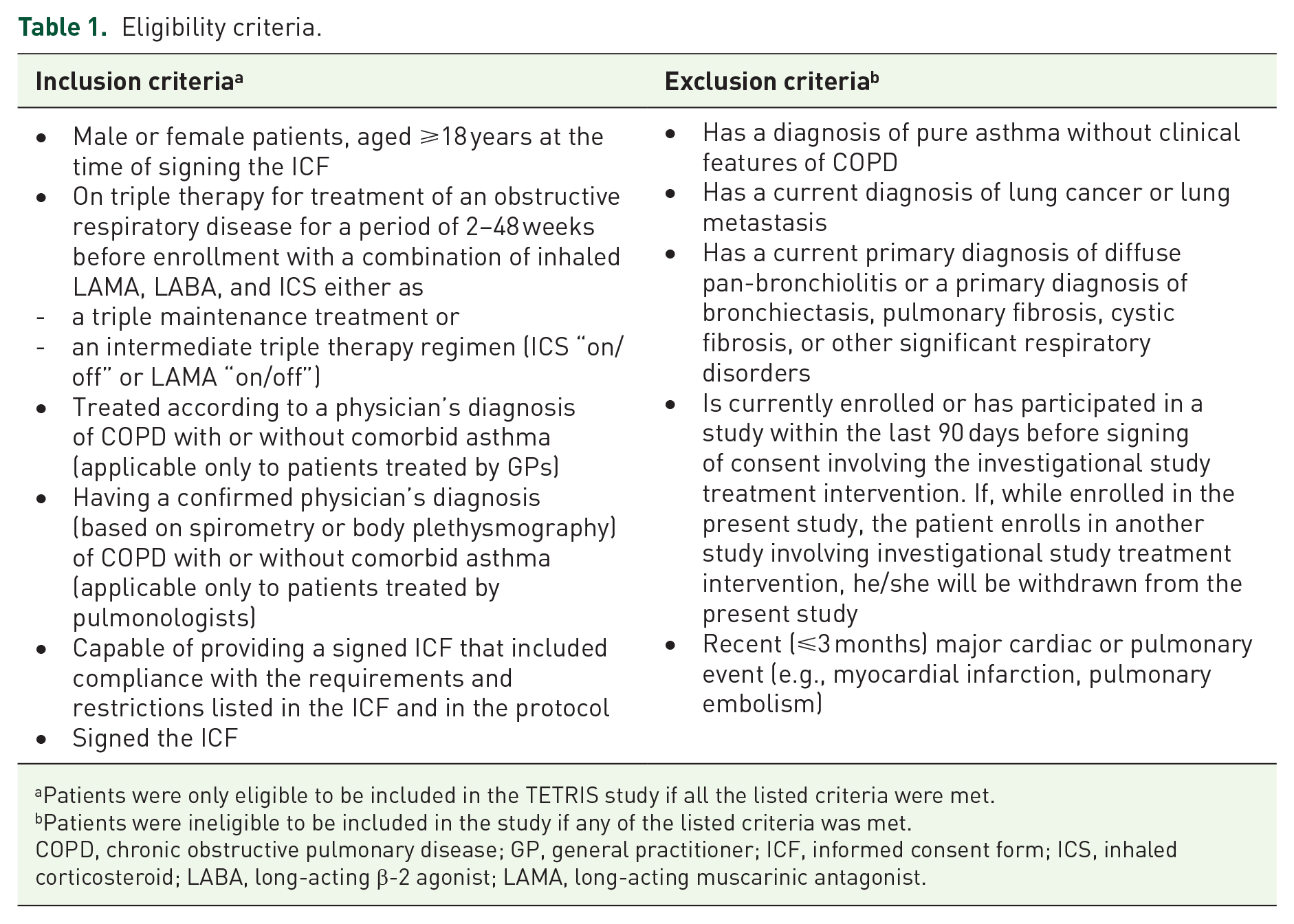
Patients were only eligible to be included in the TETRIS study if all the listed criteria were met.
Patients were ineligible to be included in the study if any of the listed criteria was met.
COPD, chronic obstructive pulmonary disease; GP, general practitioner; ICF, informed consent form; ICS, inhaled corticosteroid; LABA, long-acting β-2 agonist; LAMA, long-acting muscarinic antagonist.
Objectives and endpoints
The primary and secondary objectives and the corresponding endpoints of the TETRIS study are listed in Table 2.
Objectives and endpoints of the TETRIS study.
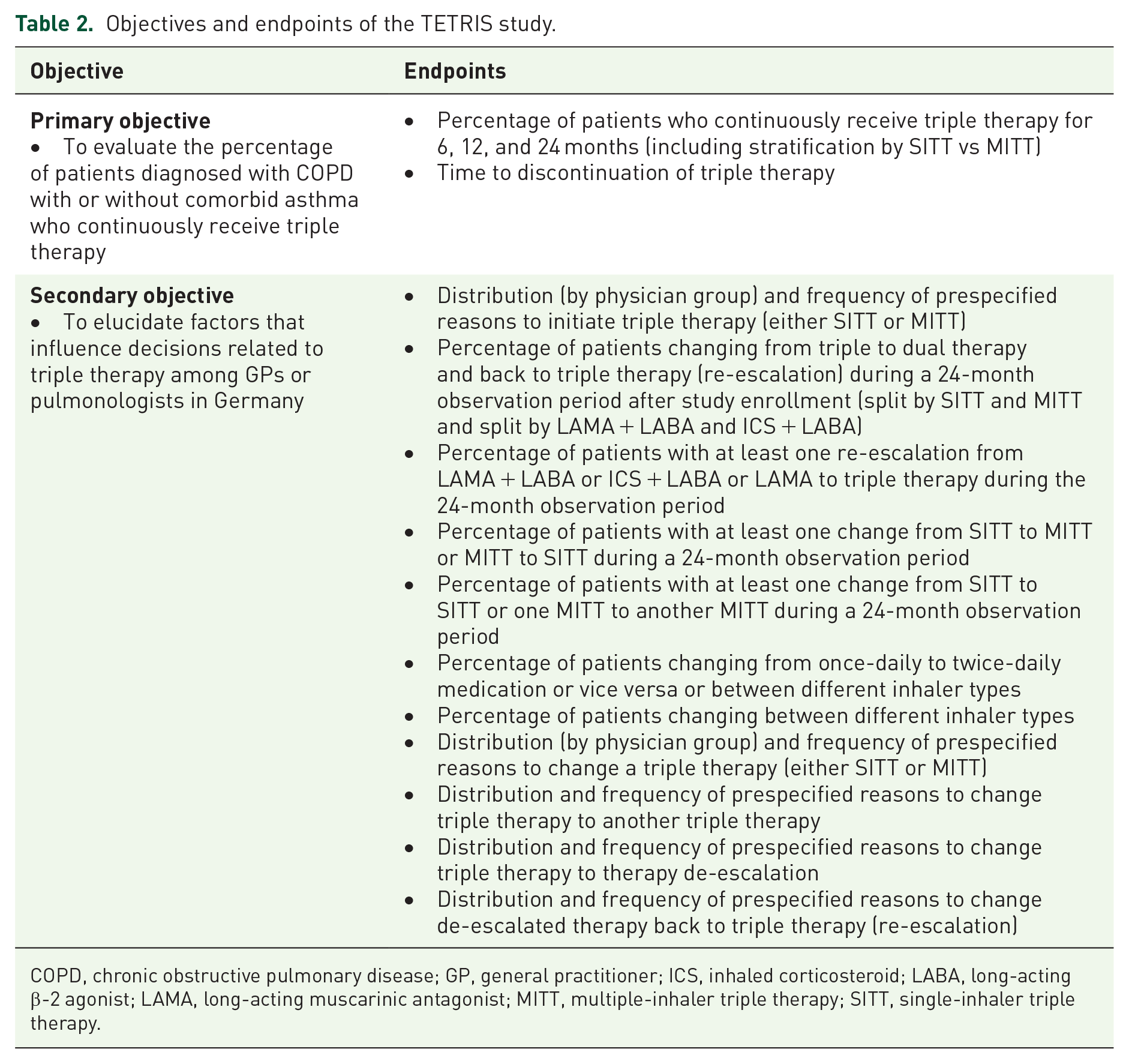
COPD, chronic obstructive pulmonary disease; GP, general practitioner; ICS, inhaled corticosteroid; LABA, long-acting β-2 agonist; LAMA, long-acting muscarinic antagonist; MITT, multiple-inhaler triple therapy; SITT, single-inhaler triple therapy.
Sample size
To obtain information regarding the long-term maintenance of triple therapy, a total observation period of 24 months was chosen for this noninterventional study. Within the primary endpoint “description of the percentage of patients with a physician’s diagnosis of COPD with or without comorbid asthma who continuously receive triple therapy for 6, 12, and 24 months after study enrollment,” we therefore focused on the percentage of patients on a continuous triple therapy regimen for 24 months. The sample size calculation was based on the precision of the estimate for this endpoint.
Based on the research data, it is assumed that approximately 35% of all patients will continue to remain on triple therapy 2 years after study enrollment. For a sample size of 740 patients with a target width of 7%, the corresponding 95% confidence interval (CI) ranges from 31.6% to 38.7%. Since data are needed for 2 years for this planned analysis, a sample size of approximately 1100 patients was chosen, considering approximately 30% study dropouts. 16
Statistical analysis
TETRIS is a descriptive study designed to estimate the primary endpoint with specific precision. The study is not powered to detect differences, and no formal statistical hypothesis testing is performed for any of the endpoints. All endpoints are analyzed descriptively. Continuous measures are analyzed by mean, median, minimum, maximum, and standard deviation (SD); discrete and categorical measures by frequencies and percentages; and event/survival measures by event rates per participant-year and Kaplan-Meier estimates.
Two interim analyses are planned. The first interim analysis is intended to collect and analyze cross-sectional data/baseline characteristics and is planned when at least 825 patients (~75%) of the total study population have been recruited. The second interim analysis is intended to include 6- and 12-month data for a subset of the study population and is planned after full recruitment. The final analysis will be performed after a 24-month follow-up from study enrollment for all patients.
Results
Demographic and baseline characteristics
Between January 2021 and June 2022, 1273 patients with COPD were screened, among whom 1213 were considered eligible and recruited for the study. Patients were counted as MITT users if more than one inhaler was used 2–48 weeks before the start of the study. Demographics and baseline characteristics are summarized overall, by treating physician (GPs vs pulmonologists), and by type of triple therapy (SITT vs MITT) in Table 3.
Demographic and baseline characteristics.

N = 1136; bN = 451; cN = 685; dN = 171; eThe nationwide DMP was introduced in Germany in 2006 primarily to avert exacerbations and thereby enhance the quality of life. Evidence indicates that implementation of DMP has been associated with improved adherence to COPD treatment guidelines and self-management education thereby translating to enhanced quality of care;17fN = 1005; gN = 379; hN = 626; iN = 853; jN = 152; kN = 842; lN = 297; mN = 545; nN = 723; °N = 119; pN = 888; qN = 329; rN = 559; sN = 759; tN = 129.
CAT, COPD Assessment Test; COPD, chronic obstructive pulmonary disease; DMP, disease management program; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; GOLD, Global Initiative for Chronic Obstructive Lung Disease; GP, general practitioner; ICS, inhaled corticosteroid; LABA, long-acting β-2 agonist; LAMA, long-acting muscarinic antagonist; MITT, multiple-inhaler triple therapy; SD, standard deviation; SITT, single-inhaler triple therapy.
Among the 1213 patients recruited, more than half were treated by pulmonologists (n = 741/1213), and majority of them were on SITT versus MITT (n = 965 vs n = 248). In addition, we investigated the subgroups of COPD patients with and without asthma (Supplemental Table 2). Focusing on the overall population, the mean age was 66.4 years, and the sex distribution was well balanced, with 49.2% male and 44.4% female patients; for 6.3% of the patients, data were missing. Nearly half of the patients (48.5%) had a former smoking history, while almost one-third (35.7%) were current smokers. A patient was categorized as former smoker if he/she smoked in the past but did not smoke at the time of enrollment into the study. More than half of the patients (55.0%) were participating in a DMP. Dual bronchodilation with LAMA + LABA was the most common therapy for COPD before the initiation of triple therapy in 58.3% of patients, followed by ICS+LABA in 23.9% of patients overall (Figure 2). Similar findings were noted among patients treated by GPs versus pulmonologists. The mean number of exacerbations and hospitalizations due to exacerbations in the past 3 years before starting triple therapy was 0.6 (SD = 1.3) and 0.1 (SD = 0.4), respectively. The mean-predicted FEV1 was 57.4%, the mean forced vital capacity (FVC) was 71.6%, and the mean COPD Assessment Test (CAT) score was 19.4, indicating a moderate impact of COPD. More than half of the patients had moderate-to-severe COPD (GOLD stages II and III; 59.5%).

Last pretreatment before start of triple therapy among patients on SITT versus non-SITT.
Comorbidities and COPD phenotypes in the overall population
Among the 1213 patients, 356 (29.3%) had no comorbidities, 225 (18.5%) had chronic bronchitis, and 164 (13.5%) had comorbid asthma. Heart diseases, followed by respiratory, thoracic, and mediastinal disorders; metabolism and nutrition disorders; and musculoskeletal, connective tissue, and bone diseases were the most common comorbidities noted overall (Figure 3).

Comorbidities recorded in the overall population.
Diagnostic tests
The most common tests to diagnose COPD in the overall population, among patients treated by pulmonologists, and among patients on SITT versus MITT were spirometry, followed by body plethysmography, diffusing capacity of the lungs for carbon monoxide (DLCO), and blood eosinophil evaluation. However, among patients treated by GPs, there was a slight variation in the diagnostic approach, with spirometry followed by blood eosinophil evaluation, body plethysmography, and high-sensitivity C-reactive protein (hs-CRP) being the most common tests (Table 4).
Diagnostic tests.

Multiple responses possible.
CT, computed tomography; DLCO, diffusing capacity of the lungs for carbon monoxide; FeNO, fractionated exhaled nitric oxide; GP, general practitioner; hs-CRP, high-sensitivity C-reactive protein; MITT, multiple-inhaler triple therapy; SITT, single-inhaler triple therapy.
Concomitant respiratory medications
Overall, approximately one-third of the patients (n = 420; 34.6%) were on concomitant respiratory medications. Among them, 123 (29.3%) were on oral betamimetics, whereas only 10 (2.4%) were on oral glucocorticosteroids (Table 5).
Concomitant respiratory medications in the overall population.

Multiple responses possible.
LTRA, leukotriene receptor antagonist.
Reasons for triple therapy initiation
Being symptomatic despite previous dual therapy with LAMA + LABA was the most common reason for initiating triple therapy in the overall population (49.9%), among patients treated by GPs versus pulmonologists (51.1% vs 49.1%, respectively), and among patients on SITT versus MITT (55.4% vs 28.2%, respectively). The other most common reasons were worsening quality of life (QoL) and/or lung function despite long-term therapy, followed by being symptomatic despite previous dual therapy with ICS+LABA in the overall population (29.8% and 20.4%, respectively) and among patients treated by pulmonologists (34.3% and 20.1%, respectively). However, among patients treated by GPs, the other most common reasons for initiating triple therapy were patients’ wish to change medication/device (35.2%), followed by exacerbation prophylaxis (27.3%; Table 6). Notably, there were differences in the reasons for triple therapy initiation among patients with comorbid asthma versus those without comorbid asthma. Being symptomatic despite previous dual therapy with ICS+LABA (41.4%) and worsening QoL and/or lung function despite long-term therapy (40.4%) were the most common reasons for triple therapy initiation among patients with comorbid asthma. On the contrary, being symptomatic despite previous dual therapy with LAMA + LABA (58.8%) was the most common reason for triple therapy initiation among patients without comorbid asthma (Supplemental Table 3).
Reasons for initiating triple therapy.
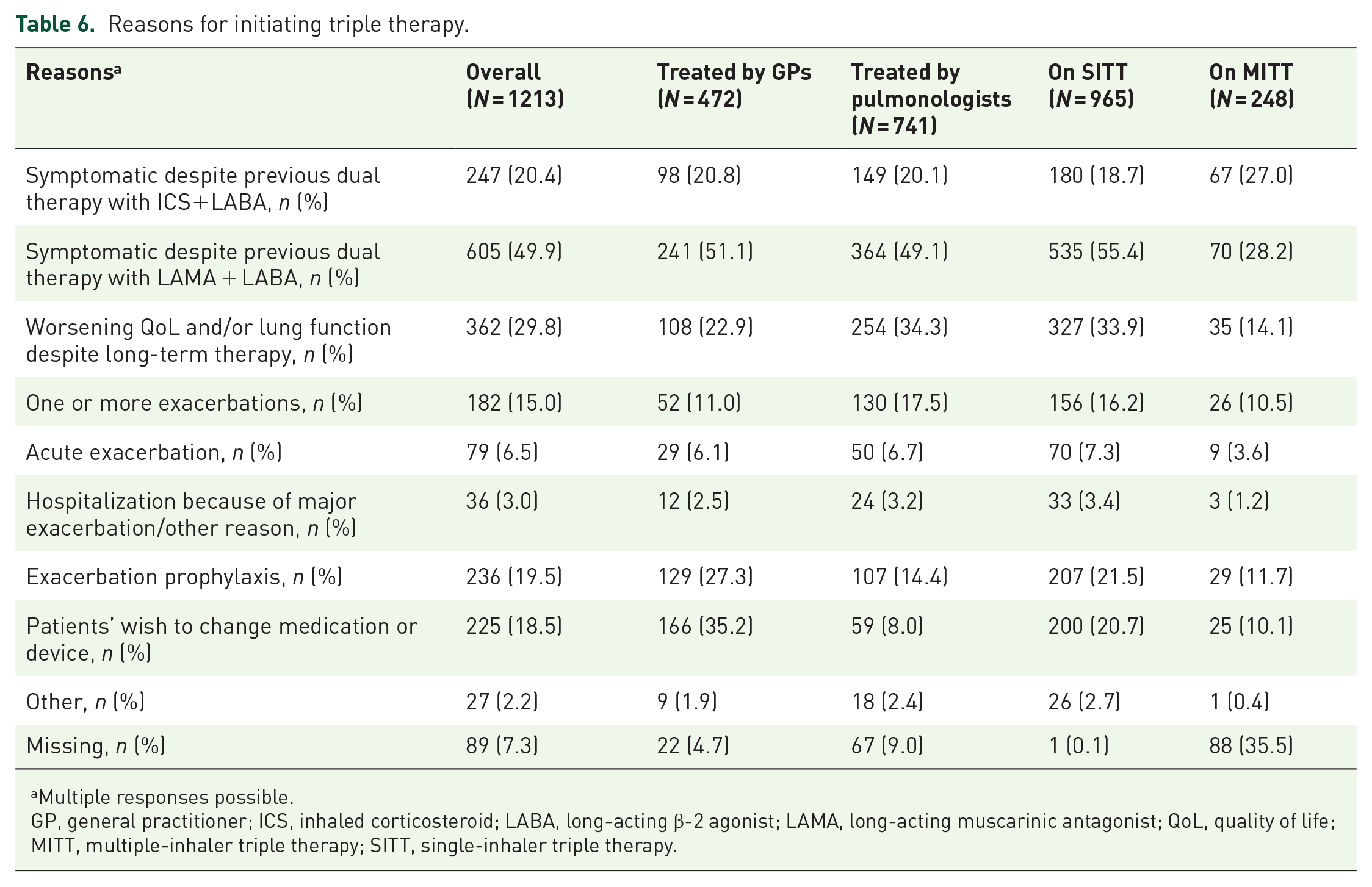
Multiple responses possible.
GP, general practitioner; ICS, inhaled corticosteroid; LABA, long-acting β-2 agonist; LAMA, long-acting muscarinic antagonist; QoL, quality of life; MITT, multiple-inhaler triple therapy; SITT, single-inhaler triple therapy.
Impact of COPD exacerbations on health-related QoL
The impact of COPD on health-related quality of life (HRQoL) was evaluated using the EuroQoL-5 Dimension 5 Level (EQ-5D-5L) tool. EQ-5D-5L/Visual Analog Scale (VAS) data were available for 842 (69.4%) patients. The EQ-5D-5L mean (SD) and the minimum to maximum score (based on the German value set, model 3b) were 0.8 (0.3) and −0.4 to 1.0, respectively. When evaluated specifically for the anxiety/depression subitem, 49.9% of the patients reported “I am not scared or depressed,” while the remaining 50.1% were observed to have either mild/moderate/severe levels of anxiety/depression (Table 7). The EQ-5D-VAS data were available for 842 patients, and the mean (SD) score was 59.7 (18.0).
EQ-5D-5L anxiety/depression subitem scores in the overall population and per physician stratification.

EQ-5D-5L, EuroQoL-5 Dimension 5 Level; GP, general practitioner.
Discussion
To date, TETRIS is the first study to provide prospective, real-world data on the medical landscape of COPD patients on triple therapies available in the German market. Overall, the baseline characteristics of patients in the TETRIS study indicate that in this real-world setting in Germany, nearly 60% of patients had moderate-to-severe COPD and more than 70% of patients had comorbidities. The exacerbation rate was reportedly low, whereas the symptom burden was high. The primary reason for stepping up patients from dual therapy to triple therapy was the high symptom burden despite being on LAMA + LABA. More than half of the patients with COPD were treated by pulmonologists (n = 741/1213), majority of whom were on SITT versus MITT (n = 965 versus n = 248).
The baseline demographics, COPD status, and burden of comorbidities among patients in the TETRIS study are generally consistent with those of patients recruited in two other real-world COPD studies in Germany – DACCORD and TriOptimize (Table 8).18,19
Comparison of baseline characteristics of patients with COPD in real-world observational studies in Germany.

N = 5918; bN = 4992.
CAT, COPD Assessment Test; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 s; GOLD, Global Initiative for Chronic Obstructive Lung Disease; NR, not reported; SD, standard deviation.
COPD is associated with a wide range of pulmonary (e.g., asthma and bronchiectasis) and extrapulmonary (e.g., cardiovascular and musculoskeletal diseases; metabolic, psychiatric, and neurological disorders; and cancer) comorbidities.4,20,21 Similar to our study, the DACCORD and TriOptimize studies also reported comorbidities in more than 70% of patients with COPD, with cardiac diseases being the most common.18,19 In the DACCORD study, cardiovascular diseases were reported in 51.9% of patients, whereas in the TriOptimize study, arterial hypertension followed by coronary heart disease were reported in 49.4% and 16.4% of patients, respectively.18,19 The findings of our study are also in line with the German nationwide claims data study by Akmatov et al., which reported that 13% of patients with COPD had comorbid asthma. This study also reported that diseases of the circulatory system (82%), metabolic disorders (77%), and musculoskeletal diseases (77%) were the most prevalent comorbidities among patients with COPD and comorbid asthma. 22 The presence of comorbidities in patients with COPD increases the utilization of healthcare resources and the risk of hospitalizations and is also associated with impaired QoL and higher mortality.20,21 A real-world study using health administrative data in a North American population quantified the burden of comorbidities in terms of healthcare service utilization among patients with COPD. According to this study, healthcare service utilization was disproportionately higher among patients with COPD with comorbidities than among those without comorbidities, translating to a significant burden on the healthcare system. Hospitalization for cardiovascular diseases was over 40%, while that for lung cancer and lower respiratory tract infections was over 60% among patients with COPD than among those without. Additionally, comorbid cardiovascular diseases contributed to the highest crude claim rate for hospitalizations per 1000 person-years (91.9; 95% CI: 91.6–92.3), while diabetes mellitus and psychiatric diseases contributed to the highest crude claim rate for emergency department visits (108.3; 95% CI: 108.0–108.7) and ambulatory care visits (1505.9; 95% CI: 1504.6–1507.2) per 1000 person-years, respectively. 21
Although mental illness was reported in only about 5.4% of patients in TETRIS, an evaluation of the EQ-5D-5L anxiety/depression subitem revealed that nearly 50% of patients with COPD suffer from at least some anxiety and/or depression. The EQ-5D-5L is a generic tool for measuring HRQoL and uses a score ranging from 0 to 1, where a score of 0 indicates death and a score of 1 indicates perfect health. 23 While the mean score of 0.8 noted in our study may not indicate a significant impact of COPD on HRQoL, the response of more than 50% of patients on the EQ-5D-5L anxiety/depression subitem highlights the burden of anxiety/depression in these patients. Anxiety is associated with a very high risk for mortality in female COPD patients. 24 Similar to our study, the German claims study by Akmatov et al. also reported that more than 50% of patients with COPD had comorbid mental and behavioral disorders. 22 In the DACCORD study, psychiatric disorders were recorded in 11% of the overall population. However, when evaluated specifically with regard to CAT score, psychiatric disorders were approximately five-fold higher among patients with a CAT score > 30 (23.2%) compared with those with a score < 10 (5.3%). 18 The high proportion of comorbidities observed in our study and other real-world studies can serve as one of the key contributors to impaired QoL, which is typical in patients with COPD.
While COPD remains the third most common cause of mortality, evidence from national and international surveys of general populations and from primary care settings highlights that the disease is underdiagnosed or misdiagnosed in the general population as well as among those at risk.25–27 COPD is usually diagnosed in the advanced stages, and patients with early COPD are either not diagnosed or not in regular care. 28 Additionally, the diagnosis of COPD at the primary care level by GPs is usually not in line with guideline recommendations. 29 Utilization of correct diagnostic approaches is therefore crucial to prevent underdiagnosis and misdiagnosis of COPD. 30
A retrospective observational study by Greulich et al. evaluated the use of diagnostic tests by private respiratory specialists in the routine clinical care of patients with COPD in Germany. Body plethysmography (100%), followed by chest X-ray (96.3%), spirometry (88.9%), CAT (85.2%), and blood eosinophil evaluation (81.5%) were the most common diagnostic tests used by respiratory specialists. 31 A nationwide prospective cross-sectional COPD questionnaire survey by Glaab et al. also reported that body plethysmography (65.9%) followed by spirometry (32%) were the standard diagnostic tests for COPD used by office-based respiratory specialists in Germany. 32 Although the GOLD and German national guidelines do not mandatorily recommend body plethysmography for COPD diagnosis, analysis of a complete set of lung volumes is important and may be required for further differential diagnosis in some patients. The more frequent use of body plethysmography than spirometry alone by respiratory specialists in Germany in this retrospective study could be attributed to the fact that it is available as an advanced diagnostic aid for respiratory physicians and compulsorily included in the German secondary respiratory care reimbursement system. 31 The GOLD 2019 guidelines recommend the use of blood eosinophil evaluation as a biomarker to identify patients with COPD who would benefit from ICS treatment to prevent exacerbations. 7 Accordingly, the estimated (and not precise cutoff) blood eosinophil counts to select patients with the highest and lowest likelihood to benefit from ICS treatment are ⩾300 cells/µL and < 100 cells/µL, respectively. 33 The increased use of blood eosinophil evaluation as a diagnostic method in the study by Greulich et al. could be attributed to the increasing awareness of this test as a useful biomarker among respiratory specialists. 31 Similar to the study by Greulich et al. and in line with the GOLD and German national guidelines, spirometry (84.6%) was the most common test used to diagnose COPD in the overall TETRIS population. Body plethysmography was the second most common diagnostic test in the overall population (50.9%) as well as in patients recruited by pulmonologists (79.1%), but not in patients recruited by GPs (6.6%). The use of tests such as thoracic computed tomography, DLCO, hs-CRP, blood eosinophil count, and fractionated exhaled nitric oxide in the overall population and among patients diagnosed by pulmonologists or GPs was relatively low. This approach could potentially be attributed to the fact that these tests are not routinely included in the German respiratory care reimbursement system.
The GOLD 2024 guidelines recommend escalation to LABA + LAMA for patients with persistent exacerbations on bronchodilator monotherapy. For patients who continue to experience further exacerbations despite LABA + LAMA therapy, escalation to LABA + LAMA + ICS is suggested. 34 According to a retrospective study of 17,464 patients with an incident diagnosis of COPD in Germany, nearly 30% of patients did not receive a prescription for inhaled maintenance therapy despite severe airflow limitation (i.e., FEV1 < 50%). Only 57.1% of the patients received at least one prescription for inhaled maintenance therapy during a median follow-up of 2 years. Among patients who received a prescription, 25.3% were initiated on LAMA alone, 35.2% on LABA + LAMA, 16.2% on LABA + ICS, and 7.8% on triple therapy with LABA + LAMA + ICS. Among patients initiated on triple therapy, 68.9% had less than two outpatient diagnoses and no inpatient diagnosis of a prior exacerbation. 6 Similarly, evidence from the real-world UK prescription data has shown that the reasons for escalation to triple therapy are not in line with guideline recommendations. 14 However, exacerbations are frequently underreported by patients, 35 which might be even more during the coronavirus disease 2019 (COVID-19) pandemic, suggesting that patients primarily report only about symptom worsening. In TETRIS, nearly half of the overall patients were stepped up to triple therapy mainly because of the continued symptom burden while on LAMA + LABA and not because of exacerbations. Exacerbations (⩾1) were a reason for initiating triple therapy in only 15% of patients overall. Together, these findings indicate that in this real-world population in Germany, patients have a relatively low reported exacerbation rate but a high symptom burden, and triple therapy is initiated in patients who are primarily highly symptomatic. The low exacerbation rate could potentially be attributed to the reduction in transmission of respiratory viruses that trigger exacerbations following the COVID-19 pandemic, as reported in the systematic review and meta-analysis by Alqahtani et al. 36 Other studies have also reported low exacerbation rates in patients with COPD. In the DACCORD study, the annualized exacerbation rate in the overall population during the 1-year follow-up period was 0.384 (95% confidence interval [CI]: 0.356–0.413). On the contrary, the exacerbation rates were 0.259 (0.236–0.284) among patients with no exacerbation at baseline and 0.743 (0.666–0.829) at follow up among patients with at least one exacerbation at baseline. 37 Similar findings were reported in an analysis of the SPIROMICS cohort comprising 1105 COPD patients with a broad range of spirometric severity. During the 3-year follow-up period, 51.3% of patients experienced no acute exacerbations while 48.7% experienced at least one acute exacerbation. Only 2.1% experienced ⩾2 exacerbations every year. 38 Interestingly, patients’ wish to change the medication or device was the second most common reason for GPs to initiate triple therapy in 35.2% of cases, suggesting that treatment decisions in routine clinical practice may be different and can deviate from guideline recommendations. In the ongoing observation, we will assess whether the decision of those patients escalated to triple therapy will have an improved symptom control while maintaining the low exacerbation rate.
The reasons for escalation to triple therapy differed among patients with or without comorbid asthma. Being symptomatic despite previous dual therapy with LAMA + LABA was the major cause for escalation to triple therapy among patients without comorbid asthma. On the contrary, patients with comorbid asthma were escalated if they remained symptomatic despite previous treatment with ICS + LABA (Supplemental Table 3). Given that the initial treatment regimen for patients with comorbid asthma is ICS + LABA and not LAMA + LABA, escalation to triple therapy if patients remained symptomatic despite treatment with ICS + LABA is in line with the Global Initiative for Asthma and the German National Care guidelines for the management of asthma.39,40
Strengths and limitations
Considering the noninterventional design, results from the TETRIS study will help to gain an understanding of patient journeys depending on healthcare professional (HCP) settings (i.e., GPs or pulmonologists), reasons for prescription of triple therapies by HCPs, daily tools used to assess patient health status (e.g., patient-reported outcomes, medical devices, and blood eosinophil count), and patient health beyond COPD in the context of comorbidities and mental health.
However, the study had some limitations that need to be acknowledged. The recruitment strategy of the study centers used in the current study may lead to a potential bias in generalizing results to the overall patient population in Germany. The availability of data for collected variables may be limited owing to the noninterventional nature of the study, and variables may not be comprehensively recorded for all patients. Therefore, the availability of data for the collected variables may not be complete, which can lead to a bias due to missing data. By participating in a study, the investigators may feel more obliged to follow treatment recommendations. Therefore, there may be deviations from real-world treatment when making therapeutic decisions (Hawthorne effect).
Conclusion
In this real-world setting, patients with COPD have a relatively low reported exacerbation rate but a high symptom burden, and triple therapy is initiated in patients who are primarily highly symptomatic. Baseline data from this prospective, real-world German study reveal that over 70% of patients with COPD are multimorbid. Mental illness was reported as a comorbidity in only 5.4% of the overall population; however, evaluation of the EQ-5D-5L subitem revealed that ~50% of patients suffer from at least some anxiety and/or depression. Data gathered over the next 2 years will provide additional valuable insights into the patient journey and challenges associated with COPD treatment in Germany. Future prospective studies are warranted in patients with early stages of the disease and in those with multimorbidity to better understand the treatment landscape across the disease spectrum. Considering the significant impact of the disease, it is crucial to increase awareness regarding COPD preventive strategies, such as smoking cessation and vaccination, among the general public. In individuals at risk of COPD and those with comorbidities, increased use of basic diagnostic tests among GPs and specialists may aid in early disease diagnosis and timely treatment intervention.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241287621 – Supplemental material for Baseline patient demographics for TETRIS: a prospective, noninterventional study to characterize the use of triple therapy for COPD in Germany
Supplemental material, sj-docx-1-tar-10.1177_17534666241287621 for Baseline patient demographics for TETRIS: a prospective, noninterventional study to characterize the use of triple therapy for COPD in Germany by Claus F. Vogelmeier, Kai-Michael Beeh, Peter Kardos, Thomas Paulsson, Gernot Rohde, Henrik Watz, Chris Compton, Tharishini Mohan and Jing Claussen in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666241287621 – Supplemental material for Baseline patient demographics for TETRIS: a prospective, noninterventional study to characterize the use of triple therapy for COPD in Germany
Supplemental material, sj-docx-2-tar-10.1177_17534666241287621 for Baseline patient demographics for TETRIS: a prospective, noninterventional study to characterize the use of triple therapy for COPD in Germany by Claus F. Vogelmeier, Kai-Michael Beeh, Peter Kardos, Thomas Paulsson, Gernot Rohde, Henrik Watz, Chris Compton, Tharishini Mohan and Jing Claussen in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-3-tar-10.1177_17534666241287621 – Supplemental material for Baseline patient demographics for TETRIS: a prospective, noninterventional study to characterize the use of triple therapy for COPD in Germany
Supplemental material, sj-docx-3-tar-10.1177_17534666241287621 for Baseline patient demographics for TETRIS: a prospective, noninterventional study to characterize the use of triple therapy for COPD in Germany by Claus F. Vogelmeier, Kai-Michael Beeh, Peter Kardos, Thomas Paulsson, Gernot Rohde, Henrik Watz, Chris Compton, Tharishini Mohan and Jing Claussen in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
In memoriam of Professor Tobias Welte, the authors would like to thank him as principal investigator of the TETRIS study for providing significant contributions toward the conceptualization and formal analysis of this study, and review and editing of this manuscript. Furthermore, the authors would like to acknowledge Dr. Michael Henning and Dr. Klaus Hechenbichler for statistical support and Dr. Raj Sharma for reviewing the manuscript. The authors would also want to thank all participating study centers for their contribution to the TETRIS study. Medical writing support (including preparation of the draft manuscript under the direction and guidance of the authors, collating and incorporating authors’ comments for each draft, assembling tables and figures, and referencing) was provided by Dr. Vidya V. Murthy, an employee of GSK, and funded by GSK.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
