Abstract
Acetylcholine (neuronal and non-neuronal origin) regulates bronchoconstriction, and mucus secretion. It has an inflammatory effect by inducing attraction, survival and cytokine release from inflammatory cells. Muscarinic receptors throughout the bronchial tree are mainly restricted to muscarinic M1, M2 and M3 receptors. Three long-acting muscarinic receptor antagonists (LAMAs) were approved for the treatment of chronic obstructive pulmonary disease (COPD) in Europe: once-daily tiotropium bromide; once-daily glycopyrronium bromide; and twice-daily aclidinium bromide. All have higher selectivity for M3 receptors than for M2 receptors, and dissociate more slowly from the M3 receptors than they do from the M2 receptors. Some LAMAs showed anti-inflammatory effects [inhibition of neutrophil chemotactic activity and migration of alveolar neutrophils, decrease of several cytokines in the bronchoalveolar lavage (BAL) including interleukin (IL)-6, tumor necrosis factor (TNF)-α and leukotriene (LT)B4] and antiremodeling effects (inhibition of mucus gland hypertrophy and decrease in MUC5AC-positive goblet cell number, decrease in MUC5AC overexpression). In the clinic, LAMAs showed a significant improvement of forced expiratory volume in 1 second (FEV1), quality of life, dyspnea and reduced the number of exacerbations in COPD and more recently in asthma. This review will focus on the three LAMAs approved in Europe in the treatment of chronic airway diseases.
Keywords
Introduction
Asthma and chronic obstructive pulmonary disease (COPD) are both characterized by airflow limitation, chronic inflammation and structural changes to the airways. The patterns of inflammation and remodeling have different effects on the bronchial tree and the alveoli in both diseases. Clinical heterogeneity is associated with various mechanisms recently recognized as endotypes. Innate immunity and remodeling of the bronchial epithelium and smooth muscle cells may have different effects on asthma and COPD. Overall, in the future we should better discriminate those entities according to their pathophysiology rather than their response to bronchodilators. Despite these descriptive definitions and still poor understanding of the mechanisms, most of the treatments used today have dramatically improved overall management. However, none of the current therapies interfere with the natural history of the diseases. Inhaled corticosteroids (ICSs) are the cornerstone in the management of persistent asthma according to the current guidelines [GINA, 2012]; however, bronchodilators are the main recommended treatment in pharmacological therapy for COPD [GOLD, 2013], as they improve symptoms, lung function, quality of life, and reduce the frequency of exacerbations in COPD patients with a forced expiratory volume in 1 second (FEV1) <60% predicted [GOLD, 2013].
Two classes of long-acting bronchodilators are now available: long-acting beta2 agonists (LABAs) and long-acting muscarinic antagonists (LAMAs). At present, three LAMAs have been approved for the treatment of COPD in different countries within Europe: once-daily tiotropium bromide (Spiriva®, Boehringer Ingelheim Pharmaceuticals Inc); once-daily glycopyrronium bromide (Seebri® Breezhaler, Novartis Pharma AG); and twice-daily aclidinium bromide (Eklira® Genuair, Almirall). Recently, several studies described the potential use of these treatments in the management of asthma where they are mainly used in the most severe form of the disease without formal health-authority approval. This review will focus on the profile of LAMAs in the treatment of chronic airway diseases.
Acetylcholine and muscarinic receptors
Acetylcholine is the primary parasympathetic neurotransmitter in the airways and a paracrine/autocrine hormone released from non-neuronal origins, Acetylcholine is synthesized from choline and acetyl-CoA mainly by the enzyme choline acetyltransferase (ChAT) [Wessler and Kirkpatrick, 2008]. Airway epithelial cells express ChAT and release acetylcholine [Proskocil et al. 2004]. Other cells (macrophages, mast cells, lymphocytes, granulocytes, fibroblasts, and smooth muscle) have been suggested to express ChAT [Wessler and Kirkpatrick, 2008] but release of acetylcholine from these cells has not yet been demonstrated directly. The parasympathetic network penetrates deep into the airway wall, and regulates bronchoconstriction, the release of mucus from submucosal glands, and from goblet cells in the airway epithelium [Gosens et al. 2006]. The distribution of muscarinic receptors throughout the bronchial tree is mainly restricted to muscarinic M1, M2 and M3 receptors [Gosens et al. 2006; Belmonte, 2005] (Figure 1). Muscarinic M1 receptors are expressed by epithelial cells, where they play a modulatory role in electrolyte and water secretion, and in the ganglia, where they facilitate parasympathetic neurotransmission. Muscarinic M2 receptors are expressed by neurons, where they function as autoreceptors, inhibiting the release of acetylcholine from both preganglionic nerves and from parasympathetic nerve terminals. Muscarinic M2 autoreceptors are dysfunctional in allergic asthma due to eosinophil-derived release of major basic protein, which acts as an allosteric antagonist of the M2 receptor [Jacoby et al. 1993], augmenting acetylcholine release. Furthermore, M2 receptors are widely expressed by airway mesenchymal cells such as fibroblasts and smooth muscle cells [Gosens et al. 2006]. Muscarinic M3 receptors are probably the best characterized subtype and are the dominant receptor subtype in the regulation of mucus secretion from submucosal glands and airway smooth muscle contraction [Gosens et al. 2006]. As a result, muscarinic M3 receptors are the primary target for LAMAs.
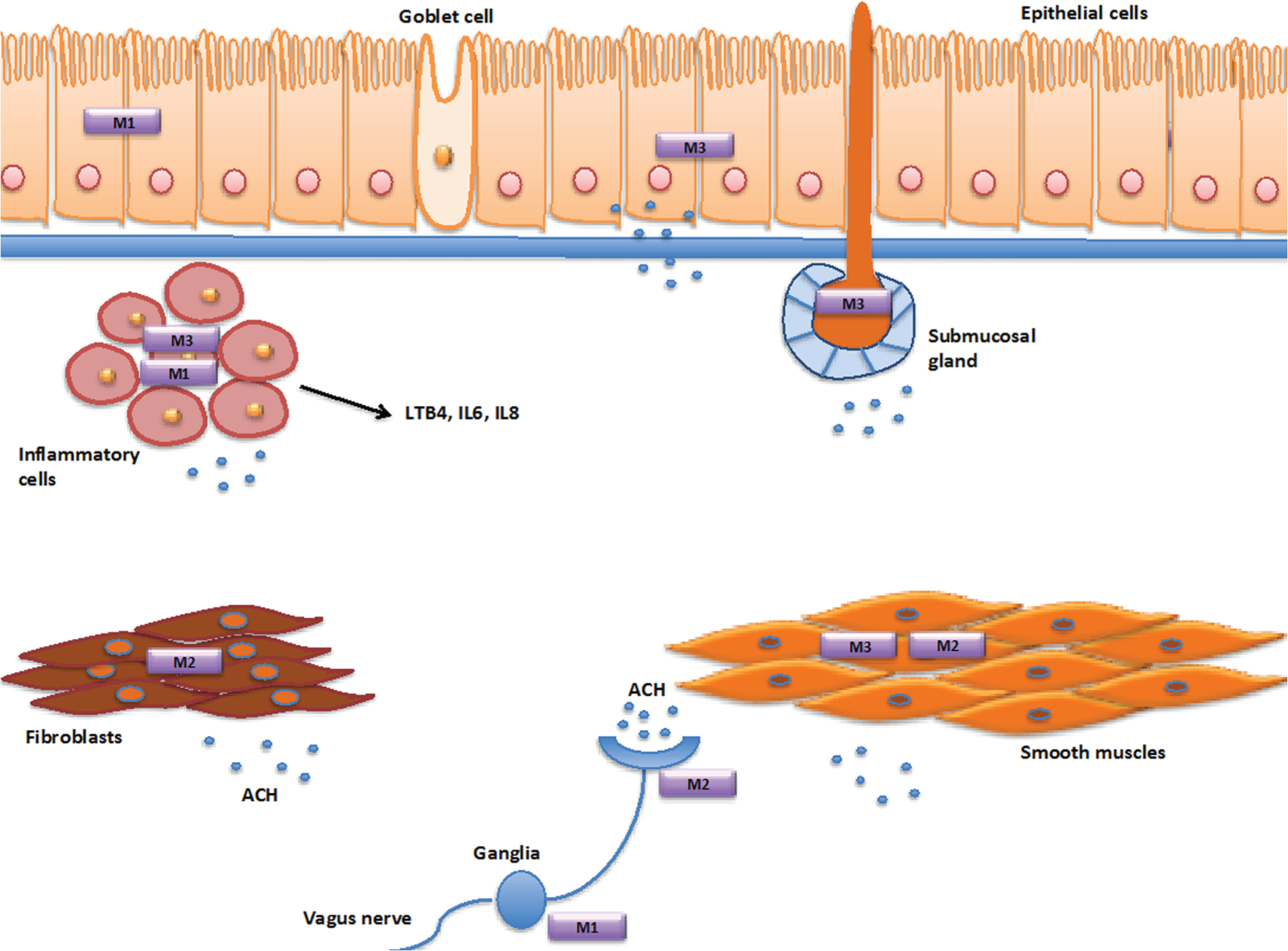
Muscarinic receptors.
LAMAs interactions with muscarinic receptor subtypes
Aclidinium, glycopyrronium, and tiotropium bind to human M1–M5 receptors in a concentration-dependent manner, all of them have higher selectivity for M3 receptors than for M2 receptors, and dissociate more slowly from the M3 receptors than they do from the M2 receptors [Vogelmeier and Banerji, 2011; Gavaldà et al. 2009].
The association rate of aclidinium with the human M3 receptor subtype is 2.6 times faster than that of tiotropium, but both show slow dissociation from M3 receptors, with residence half-lives of approximately 29 and 62 h, respectively. The aclidinium half-life for the M2 receptor is 3.22 times shorter than that obtained for tiotropium [Gavaldà et al. 2009], whereas glycopyrronium bromide associates four to five times faster with the M3 receptor than tiotropium bromide and aclidinium bromide [Sykes et al. 2009; Vestbo et al. 2010; Ulrik, 2012]. The more rapid association of LAMA with the M3 receptor is assumed to underlie its faster onset of action.
Inflammation and the cholinergic system
Acetylcholine has been shown to affect inflammatory cells involved in asthma and COPD, by inducing proliferation or cytokine release from these cells [Kistemaker et al. 2012]. The muscarinic agonist, carbachol, can induce proliferation of rodent macrophages in vitro [De la Torre et al. 2008]. Cigarette smoke extract upregulates the non-neuronal cholinergic system in bronchial epithelial cells, and increases the expression of muscarinic M2 and M3 receptors. Consequently, acetylcholine levels in cell extracts were significantly higher after this stimulation. This increase could be reduced by tiotropium [Profita et al. 2011]. On the other hand atropine suppresses the proliferation of T cells [Razani-Boroujerdi et al. 2008]. Acetylcholine is known to induce eosinophil, monocyte and neutrophil chemotactic activity in bronchial epithelial cells [Koyama et al. 1992, 1998]. Neutrophil chemotactic activity induced by acetylcholine could be inhibited by tiotropium [Profita et al. 2008]. The acetylcholine-induced neutrophil chemotactic activity from epithelial cells is partially dependent on interleukin (IL)-8 release, since it is inhibited by an anti-IL-8 monoclonal antibody, and the increase in IL-8 release in response to acetylcholine could be partially inhibited by tiotropium [Profita et al. 2008].
Alveolar macrophages have granulocyte and monocyte chemotactic activity in response to acetylcholine, which is likely explained by cholinergic induction of leukotriene B4 (LTB4) release [Sato et al. 1998; Buhling et al. 2007]. Chemotactic activity induced by acetylcholine could be inhibited by tiotropium [Buhling et al. 2007]. Acetylcholine can also induce the release of LTB4 from sputum cells of COPD patients [Profita et al. 2005]. Tiotropium inhibited alveolar macrophage mediated migration of neutrophils from COPD patients: it was demonstrated that this effect is mediated via the muscarinic M3 receptor [Vacca et al. 2011].
Stimulation of bovine airway smooth muscle strips with the muscarinic agonist carbachol induces pro-inflammatory gene expression, including IL-6, IL-8 and cyclo-oxygenase-2 [Kanefsky et al. 2006], and induces the protein release of IL-6 and IL-8 via muscarinic M3 receptors [Gosens et al. 2009].
Anti-inflammatory effects
Tiotropium treatment in an animal model of cigarette smoke-induced COPD showed a concentration-dependently decreased in cell numbers and neutrophils in the bronchoalveolar lavage (BAL). Furthermore, tiotropium decreased cytokine levels in the BAL supernatant, including IL-6, tumor necrosis factor (TNF)-α, and some proinflammatory mediators such as LTB4 [Wollin and Pieper, 2010].
Tiotropium partially inhibits eosinophilia recruitment in a guinea-pig model of asthma [Bos et al. 2007; Buels et al. 2012]. IL-4, IL-5, and IL-13 levels measured in BAL supernatant were decreased [Ohta et al. 2010].
In a mouse model of Aspergillus fumigatus-induced asthma, aclidinium treatment completely abrogated the methacholine-induced lung resistance, and reduced the numbers of eosinophils in BAL fluid with no significant changes in other cell types or in IL-4 or IL-6 levels. The treatment markedly decreased total protein levels in BAL fluid with a correlation with lung injury and capillary leakage indices [Damera et al. 2010].
Airway remodeling
Airway remodeling in asthma and COPD is characterized by abnormalities of the structure of the airways from the nose to the alveoli including the distal conducting airways. It affects the bronchial and alveolar epithelium with goblet cell hyperplasia and glands hypertrophia, causes profound changes to smooth muscle and extracellular matrix and, in COPD, pulmonary vascular remodeling occurs [Jeffery, 2001; Sköld, 2010].
In asthma, the epithelium and subepithelial layers are abnormal: a considerable thickening of the airway smooth muscle bundle is observed [An et al. 2007]. Airway remodeling correlates weakly with disease severity [Hogg et al. 2004; James et al. 2009]. It is considered as a marker of chronicity and as a potential major component of irreversible airflow limitation in these diseases [An et al. 2007], and may be involved in the faster decline of lung function that is observed [Pare et al. 1997].
Mucus secretion
Mucus hypersecretion is an important feature of most of chronic airway diseases such as COPD and asthma, and contributes to altered quality of life, morbidity, and mortality in affected patients [Voynow and Rubin, 2009]. MUC5AC is the predominant mucin gene expressed in healthy human airway epithelial cells, and its expression is markedly increased in smokers, COPD, and asthmatic patients [Morcillo and Cortijo, 2006].
Mucus secretion can be increased predominantly via muscarinic M3 receptors expressed on the submucosal glands [Rogers, 2001]. In addition, electrolytes and water secretion are regulated by muscarinic M1 and M3 receptors [Ishihara et al. 1992; Ramnarine et al. 1996]. In response to acetylcholine, goblet cells also produce mucus [Rogers, 2001].
Tiotropium reduces allergen-induced mucus gland hypertrophy and the numbers of MUC5AC-positive goblet cell in a guinea-pig model [Bos et al. 2007]. Furthermore, tiotropium inhibits neutrophil elastase-induced goblet cell metaplasia in mice [Arai et al. 2010] and was shown to inhibit the increased MUC5AC expression and mucus gland hypertrophy in a guinea-pig model of COPD [Pera et al. 2011]. In vitro studies have demonstrated that aclidinium suppressed both carbachol-induced MUC5AC overexpression and cigarette smoke-induced MUC5AC expression in human bronchial tissue [Cortijo et al. 2011].
Fibroblasts and airway smooth muscle cells
Fibroblasts and airway smooth muscle cells contribute to airway remodeling by means of an increase of their proliferation and the release of various inflammatory mediators, including the matrix metalloproteinases (MMPs). MMPs play a key role in the dynamic of the extracellular matrix by affecting elastic and collagen fiber turnover. They have been shown to be associated with some single-nucleotide polymorphism (SNIP) in COPD genomic analysis and are key players in neutrophilic inflammation and emphysema [Lagente and Boichot, 2010]. In COPD patients, increased expression levels of MMP-1, MMP-2 and MMP-9 have been reported in the sputum [Cataldo et al. 2000; Imai et al. 2001]. In asthma, MMP-9 is the predominant proteinase and its expression may increase when patients have spontaneous acute exacerbations or in response to a local allergenic challenge [Kelly and Jarjour, 2009]. The biological activity of the MMPs can be inhibited by tissue inhibitor of matrix metalloproteinases (TIMPs) [Lagente and Boichot, 2010]. TIMPs bind MMPs in a 1:1 fashion; thus, an increase in the molar ratio of MMP/TIMP may favor tissue destruction, while the reverse could be associated with marked fibrosis [Kelly and Jarjour, 2009]. It was found that tiotropium inhibited transforming growth factor (TGF)-β-induced protein expression of both MMP-1 and MMP-2 in human lung fibroblasts, but had no effect on the TGF-β-induced TIMP-1 and TIMP-2 expression [Asano et al. 2008, 2010]. Therefore, these data suggest that treatment with tiotropium may improve the balance between MMPs and TIMPs, inhibiting some profibrotic responses [Kistemaker et al. 2012].
In vitro studies have provided evidence that the stimulation of muscarinic receptors from fibroblasts induces cell proliferation and the synthesis of collagen through muscarinic M2 receptor activation [Haag et al. 2008; Matthiesen et al. 2006]. Recently, it was demonstrated that aclidinium can inhibit the transition from human fibroblast to myofibroblast. The cells were obtained from smokers and COPD patients, and the transition was induced in vitro by exposure to carbachol, TGF-ß1, and cigarette smoke extracts. On the other hand, this drug dose-dependently reduced fibroblast proliferation and migration [Milara et al. 2012, 2013].
In a guinea-pig model of asthma using repeated challenges with ovalbumin, treatment with tiotropium significantly inhibited airway smooth muscle remodeling and inhibited the overexpression of the contractile protein ‘myosin’ [Gosens et al. 2005]. Increased peribronchial collagen deposition was prevented in a guinea-pig model of COPD [Pera et al. 2011]. Tiotropium also significantly inhibited smooth muscle thickening and the release of TGF-β1 in BAL fluid [Ohta et al. 2010] in a murine model of asthma
Clinical efficacy of LAMAs in chronic airway diseases
Effects on pulmonary function tests
Tiotropium
In COPD, The Understanding Potential Long-term Impacts on Function with Tiotropium (UPLIFT) study [Tashkin et al. 2008] demonstrated that in the tiotropium group, the mean values for FEV1 and forced vital capacity (FVC) before and after bronchodilation showed significant improvements that were maintained at all timepoints after randomization. Mean improvements in FEV1 in the tiotropium group, as compared with the placebo group, ranged from 87 to 103 ml before bronchodilation and from 47 to 65 ml after bronchodilation (p < 0.001). The rate of decline in the mean values for FEV1 and FVC either before or after bronchodilation from day 30 to the end of study was not affected by the treatment.
In asthma, several studies have investigated the efficacy of tiotropium in asthma (versus placebo, salmeterol or doubling ICSs); see Table 1. We will present the results of two studies.
Efficacy of tiotropium in asthma.
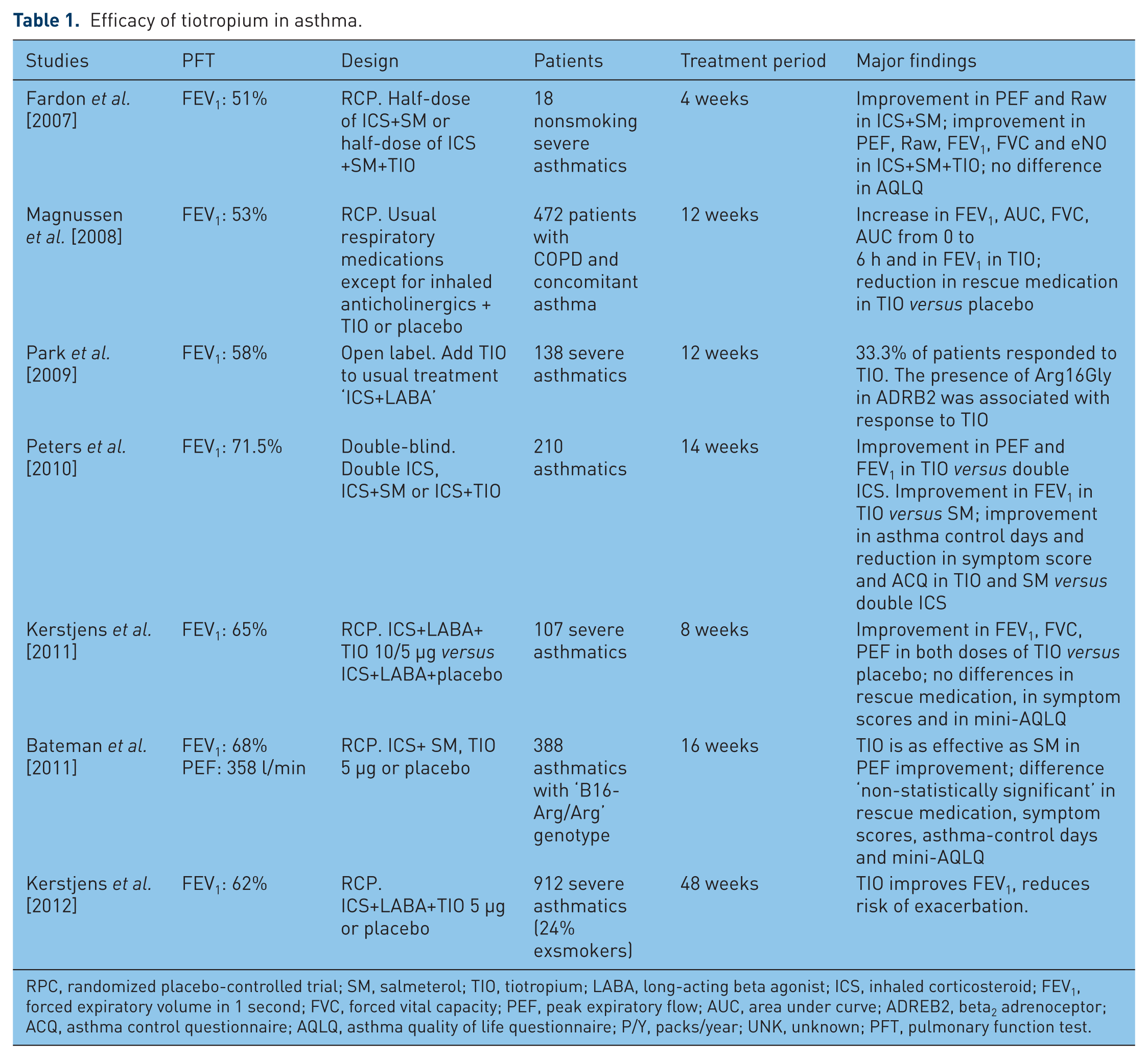
RPC, randomized placebo-controlled trial; SM, salmeterol; TIO, tiotropium; LABA, long-acting beta agonist; ICS, inhaled corticosteroid; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; PEF, peak expiratory flow; AUC, area under curve; ADREB2, beta2 adrenoceptor; ACQ, asthma control questionnaire; AQLQ, asthma quality of life questionnaire; P/Y, packs/year; UNK, unknown; PFT, pulmonary function test.
In the first study [Peters et al. 2010], the researchers examined the effects of adding salmeterol 100 μg daily or tiotropium 18 μg daily (via a Handihaler®) versus doubling the dose of extra-fine hydrofluoroalkane (HFA)-beclometasone (160 μg) over 14 weeks. Tiotropium showed significant superiority for the primary outcome (morning peak expiratory flow [PEF]) over doubling the dose of HFA-beclometasone, amounting to a mean difference of 26 l/min, as well as noninferiority versus salmeterol with a mean difference of 6 l/min. For the secondary endpoint of FEV1, mean differences with the addition of tiotropium were significantly improved by 100 ml versus doubling the dose of HFA-belcometasone and by 110 ml versus adding salmeterol. There were no significant differences between tiotropium treatment and salmeterol treatment with respect to the evening PEF (p = 0.05), the proportion of asthma control days (p = 0.78), the score for daily symptoms (p = 0.10), and the score on the Asthma Control Questionnaire (p = 0.18).
In the second study [Kerstjens et al. 2012], the researchers randomized 912 patients (in two trials) with severe uncontrolled asthma despite treatment with inhaled glucocorticoids and LABAs, of whom 24% were exsmokers (COPD with asthmatic component), to receive tiotropium 5 μg daily (via Respimat®) or placebo for 48 weeks as add on to pre-existing ICS/LABA. At 24 weeks, the mean [± standard error (SE)] change in the peak FEV1 from baseline was greater with tiotropium than with placebo in the two trials: a difference of 86 ± 34 ml in trial 1 (p = 0.01) and 154 ± 32 ml in trial 2 (p < 0.001). The predose (trough) FEV1 also improved in trials 1 and 2 with tiotropium, as compared with placebo: a difference of 88 ± 31 ml (p = 0.01) and 111 ± 30 ml (p < 0.001), respectively.
Aclidinium
Two phase III studies (ACCLAIM I and II) [Jones et al. 2011] evaluated the efficacy and safety of aclidinium 200 μg, showed a statistically significant improvement of FEV1 compared with placebo at weeks 12 and 28, in ACCLAIM I (by 61 and 67 ml, respectively; both p < 0.001) and ACCLAIM II (by 63 and 59 ml, respectively; both p < 0.001). In both studies, this effect was maintained over the 52-week study period. A recent phase III study [Kerwin et al. 2012a] evaluated the efficacy and safety of aclidinium 200 and 400 μg twice daily versus placebo (ACCORD COPD I). At week 12, aclidinium 200 and 400 μg showed a significant improvement in FEV1 by 86 (45–127) ml and 124 (83–164) ml, respectively, compared with placebo.
Glycopyrronium bromide
In one of the largest published clinical trials of glycopyrronium bromide (GLOW1) [D’Urzu et al. 2011] the researchers randomized patients with stable moderate-to-severe COPD to either glycopyrronium bromide 50 μg once daily or placebo for 26 weeks. Least squares mean trough FEV1 at week 12 was significantly higher in patients receiving glycopyrronium bromide (1.41 l) versus placebo (1.30 l), with a treatment difference of 108 ± 15 ml (p = 0.001). Significant improvements in trough FEV1 were observed at the end of day 1 and sustained through week 26.
In the GLOW2 study [Kerwin et al. 2012b], patients with moderate-to-severe COPD were randomized to either glycopyrronium bromide 50 μg once daily, open-label tiotropium 18 μg once daily, or placebo for 52 weeks. Least squares mean trough FEV1 at week 12 was significantly higher in patients receiving glycopyrronium bromide and tiotropium compared with that in patients receiving placebo, with a treatment difference of 97 ml (p = 0.001) and 83 ml (p = 0.001), respectively. Furthermore, trough FEV1 at weeks 26 and 52 was significantly higher for patients treated with glycopyrronium bromide versus placebo and comparable with tiotropium. At these time points, the treatment difference, compared with placebo, in favor of glycopyrronium bromide, was 134 and 108 ml, respectively (p < 0.001), while the difference in favor of tiotropium was 84 ml and 89 ml, respectively (p < 0.001). On day 1, the least squares mean FEV1 treatment difference for glycopyrronium bromide/placebo and tiotropium bromide/placebo was 87 and 45 ml at 5 minutes, respectively, and 143 ml and 78 ml at 15 minutes, respectively (p < 0.001). The latter finding further supports the notion that glycopyrronium bromide has a faster onset of action than other inhaled antimuscarinic agents, including tiotropium bromide [Ulrik, 2012].
Dyspnea
Tiotropium
Two studies examined the change in the Transition Dyspnea Index (TDI) for tiotropium compared with placebo in COPD patients [Brusasco et al. 2003; Casaburi et al. 2002]. They showed a statistically significant improvement in the dyspnea score in favor of tiotropium compared with placebo. These improvements have not reached clinically significant change (one-unit change).
Aclidinium
In ACCLAIM I, more patients receiving aclidinium versus placebo exceeded the minimal clinically important difference (1 unit) for TDI focal score at 52 weeks (56.4% versus 38.0%; p < 0.0001). In ACCLAIM II, the difference was significant only at week 12 (53.1% versus 42.7%; p = 0.029). The ACCORD study showed a significant improvement of dyspnea for the two doses 200 and 400 mg versus placebo with a TDI focal score (>1 unit) at 12 weeks for aclidinium 400 mg.
Glycopyrronium bromide
In the GLOW1 study [D’Urzu et al. 2011] patients receiving glycopyrronium bromide had a significantly greater TDI focal score at week 26 (1.84) compared with patients receiving placebo (0.80). This difference was statistically and clinically significant; see Table 2. Furthermore, patients treated with glycopyrronium bromide were 1.7-fold more likely to have at least a one-point improvement in transition dyspnea index focal score compared with those on placebo (p = 0.001). Similar findings were reported from the GLOW2 trial [Kerwin et al. 2012b].
Clinical efficacy of long-acting muscarinic receptor antagonists in chronic obstructive pulmonary disease.

FEV1, forced expiratory volume in 1 second; SGRQ, St George’s Respiratory Questionnaire.
Exacerbations
Tiotropium
In COPD, tiotropium treatment significantly reduced the numbers of patients suffering from acute COPD exacerbations. In addition, tiotropium also reduced exacerbations leading to hospitalization but no significant difference was found for hospitalization of any cause or mortality [Karner et al. 2012]. These findings were confirmed by a pooled analysis [Halpin et al. 2009] examining nine studies, which found a significantly reduced risk of COPD exacerbation and the risk of exacerbation-associated hospitalization, in COPD (reduction by 21%). The TIOSPIR study [Wise et al. 2013] showed that tiotropium Respimatat® at a dose of 5 μg is not superior to HaniHaler® with respect to the risk of the first exacerbation [hazard ratio 0.98; 95% confidence interval (CI) 0.93–1.03].
In asthma, tiotropium increased the time to the first severe exacerbation (282 versus 226 days) in patients with severe uncontrolled asthma despite treatment with inhaled glucocorticoids and LABAs, with an overall reduction of 21% in the risk of a severe exacerbation (hazard ratio 0.79; p = 0.03) [Kerstjens et al. 2012].
Aclidinium
The moderate or severe COPD exacerbation rate in ACCLAIM I is nearly the same versus placebo (26.6% versus 25.7%), and so is the duration before the first moderate or severe exacerbation (p = 0.9 versus placebo). In ACCLAIM II, fewer patients in the aclidinium group experienced a moderate or severe exacerbation compared with those in the placebo group (33.2% versus 39.8%, respectively; p = 0.0046). The time to first moderate or severe exacerbation with aclidinium was significantly delayed compared with placebo (p = 0.01). In the ACCORD study, there was no significant difference of the exacerbation numbers but this study was of shorter duration and not powered for this outcome.
Glycopyrronium bromide
In the GLOW 1 trial [D’Urzu et al. 2011], during the 26-week study period, (17.5%) in the glycopyrronium bromide group had at least one moderate or severe COPD exacerbation, compared with 24.2% in the placebo group. Glycopyrronium bromide significantly reduced the risk of COPD exacerbations in terms of time to first moderate or severe COPD exacerbation by 31% compared with placebo (p = 0.023). Furthermore, a significant reduction was also observed in the future risk of a COPD exacerbation leading to hospitalization in the glycopyrronium bromide group versus placebo (p = 0.022).
In the GLOW2 trial [Kerwin et al. 2012b], glycopyrronium significantly reduced the risk of exacerbations in terms of time to first moderate or severe exacerbation by 34% compared with placebo (p = 0.001) over 52 weeks, thereby demonstrating similar results to those reported for tiotropium, which provided a 39% risk reduction versus placebo (p = 0.001). Further, a 35% reduction was observed in the rate of moderate-to-severe COPD exacerbations in the glycopyrronium group compared with placebo (0.54 versus 0.80 per year, p = 0.003).
Quality of life
Tiotropium
Two meta-regression analyses examined quality-of-life of studies that compared tiotropium with placebo. The first [Yohannes et al. 2011] examined 20 studies (22,468 COPD patients) and found that the proportion of COPD patients who obtained a clinically significant change (a four-unit change) in the St George’s Respiratory Questionnaire (SGRQ) score was significantly higher in the tiotropium group compared with the placebo group. The second [Karner et al. 2012] examined 22 studies (23,309 COPD patients) and reported that patients treated with tiotropium obtained a statistically significant difference in the SGRQ over placebo, but this difference was not clinically relevant. The UPLIFT study [Tashkin et al. 2008] demonstrated a statistically significant difference of the mean quality of life, the overall mean between-group difference in the SGRQ total score at any time point was 2.7, which was not clinically significant.
Aclidinium
In ACCLAIM I and II [Jones et al. 2011], significantly more patients receiving aclidinium had an improvement in SGRQ total score ≥4 units versus placebo; at week 52, the percentage of patients achieving this improvement was 48.1% versus 39.5% for aclidinium versus placebo, respectively (p = 0.025), in ACCLAIM I and 39.0% versus 32.8% for aclidinium versus placebo, respectively (p = 0.074), in ACCLAIM II. The difference observed between the two studies is probably due to the higher number of aborts in the placebo group of ACCLAIM II.
Glycopyrronium bromide
In GLOW 1 [D’Urzu et al. 2011] patients treated with glycopyrronium bromide had a significantly lower SGRQ total score at week 26 than patients treated with placebo. The treatment difference was −2.81, which was statistically but not clinically significant. The percentage of patients achieving a >4-point reduction in SGRQ score was significantly greater with glycopyrronium bromide than with placebo (56.8% and 46.3%, respectively, p = 0.006). Similar findings were reported from the GLOW2 trial [Kerwin et al. 2012b].
Safety issues
Tiotropium
The most commonly reported adverse event (AE) in a meta-analysis performed by Yohannes and colleagues [Yohannes et al. 2011] was dry mouth: 10 studies reported the incidence of dry mouth. The cumulative incidence was 7.4% with tiotropium and 2.0% with placebo. In this meta-analysis, the summary odds estimate for the number of patients who experienced a serious AE (SAE) was not statistically significant when tiotropium was compared with placebo.
A recent meta-analysis [Singh et al. 2011] investigated the safety profile of five randomized controlled trials (n = 6522) examining tiotropium delivered using the Respimat® inhaler in comparison with placebo. They found that using the tiotropium Respimat® inhaler was associated with a significantly increased risk of mortality. Another recent meta-analysis examined 42 randomized controlled trials (n = 52516) of tiotropium Soft Mist Inhaler, tiotropium HandiHaler®, LABAs, ICSs, and LABA–ICS combination with at least a 6-month treatment duration [Dong et al. 2013]. Tiotropium Soft Mist Inhaler was associated with a universally increased risk of overall death compared with placebo [odds ratio (OR) 1.51; 95% CI 1.06–2.19], tiotropium HandiHaler (OR 1.65; 95% CI 1.13–2.43), LABA (OR 1.63; 95% CI 1.10–2.44), and LABA–ICS (OR 1.90; 95% CI 1.28–2.86). The risk was obvious for cardiovascular death, in patients with severe COPD, and at a higher daily dose. However, the UPLIFT [Tashkin et al. 2008] study showed that tiotropium was superior in reducing all causes of mortality by 11% compared with placebo.
A more recent randomized, double-blind, parallel-group trial ‘TIOSPIR’ [Wise et al. 2013] involving 17,135 patients with COPD (mean FEV1 after bronchodilation was 48%), 10% of them have a previous stable and non-life-threatening arrhythmia and 15% have a previous ischemic heart disease, treated with tiotropium HandiHaler® 18 μg once daily, tiotropium Respimat® 5 μg once daily, or tiotropium Respimat® 2.5 μg once daily, evaluated the risk of death (noninferiority study, Respimat® at a dose of 5 or 2.5 μg versus HandiHaler®). This study showed that Respimat® was noninferior to HandiHaler® with respect to the risk of death (Respimat® at a dose of 5 μg versus HandiHaler®: hazard ratio 0.96; 95% CI 0.84–1.09; Respimat® at a dose of 2.5 μg versus HandiHaler®: hazard ratio 1.00; 95% CI 0.87–1.14), Respimat® was not associated with higher mortality than HandiHaler® among patients with previous cardiac disease, including stable arrhythmias at baseline, and was also not associated with a higher incidence of arrhythmias. There were no significant differences among the three study groups in terms of SAEs and nonfatal and fatal major adverse cardiovascular events. The limitations of this study are the absence of a placebo group and the exclusion of patients with unstable cardiovascular conditions (myocardial infarction within the previous 6 months, hospitalization for class III or IV heart failure, or unstable or life-threatening arrhythmia) or moderate or severe renal impairment, so the study findings cannot be extended to these populations.
In conclusion, in light of these results, we can conclude that tiotropium Respimat® may be prescribed to patients with COPD with a good tolerance, even in the case of stable ischemic heart disease or stable cardiac non-life-threatening arrhythmia. However, we do not actually recommend that tiotropium Respimat® be prescribed in cases of unstable ischemic heart disease, unstable or life-threatening cardiac arrhythmia.
Aclidinium
The rapid plasma hydrolysis of aclidinium results in minimal to no systemic exposure 15 minutes following a dose, suggesting a low potential for extrapulmonary side effects regardless of renal impairment [Prat et al. 2009; Sentellas et al. 2010].
In phase I studies in humans, aclidinium had no significant effect on heart rate or QT interval at doses up to 800 μg [Lasseter et al. 2011; Schelfhout et al. 2010]. In phase I–III studies in humans [Schelfhout et al. 2010; Chanez et al. 2010; Jansat et al. 2009; Jones et al. 2011; Joos et al. 2010; Maltais et al. 2011], non-SAEs were generally comparable in incidence to placebo, and included headache, dry mouth, cough, sore throat, upper respiratory tract infection, diarrhea, myalgias, and arthralgias. In particular, the incidence of anticholinergic side effects in the phase III study was very low and not significantly different from placebo [Jones et al. 2011]. None of the studies reported SAEs that were thought to be related to study drug.
Glycopyrronium bromide
In the GLOW1 trial [D’Urzu et al. 2011], the incidence of AEs was lower in patients receiving glycopyrronium bromide (57.5% of patients), compared with placebo (65.2%). Other AEs, including those typically associated with anticholinergics (gastrointestinal disturbances, urinary difficulty, urinary retention, dry mouth), occurred at low frequencies in the glycopyrronium bromide and placebo groups. SAEs and discontinuations due to AEs were less frequent in patients receiving glycopyrronium bromide than in those receiving placebo. The number of deaths was low in the glycopyrronium bromide group (3 patients, 0.5%) and in the placebo group (3 patients, 1.1%). Notable QTcF intervals were reported in 4.0% of patients treated with glycopyrronium bromide and 1.1% of those receiving placebo. Changes in QTcF interval from a baseline of 30–60 ms or >60 ms occurred in 11.8% of patients receiving glycopyrronium bromide and 8.2% of those receiving placebo.
In GLOW2 [Kerwin et al. 2012b], the overall incidence of AEs was similar across the three treatment groups (glycopyrronium bromide 76.6%, placebo 76.5%, tiotropium 74.2%). Antimuscarinic side effects, such as dry mouth, constipation, urinary retention and urinary tract infections, occurred with a low frequency in the glycopyrronium bromide, placebo, and tiotropium treatment groups. SAEs occurred with a lower frequency in the glycopyrronium bromide group, compared with the tiotropium and placebo groups. The percentage of patients with newly occurring or worsening clinically notable QTcF values was low across treatment groups (glycopyrronium bromide 4.4%, tiotropium 5.3%, and placebo 6%). Seven deaths were reported during the treatment and the 30-day follow-up period; three in the glycopyrronium bromide group (0.6%) and two each in the placebo (0.7%) and tiotropium groups (0.7%). None of the deaths was suspected to be related to the study medication.
Conclusion
Three LAMAs have been approved for the treatment of COPD in Europe: tiotropium bromide, glycopyrronium bromide, and aclidinium bromide. All have higher selectivity for M3 receptors than for M2 receptors, and dissociate more slowly from the M3 receptors than they do from the M2 receptors.
Some LAMAs show anti-inflammatory effects and antiremodeling effects but most of these studies are in vitro on cellular models or in animal models.
Clinically, tiotropium and glycopyrronium have longer half-lives than aclidinium, which allows for a once-daily dose compared with twice daily for aclidinium. Can we use the term LAMA for aclidinium and ‘ultra-LAMA’ for tiotropium and glycopyrronium? Glycopyrronium has a rapid onset of action. In COPD, all three products produced a statistically significant improvement of FEV1 but only glycopyrrinium produced a clinically significant improvement. There was also improvement in the quality of life measured by the SGRQ, but this was not clinically significant. Only glycopyrronium produced statistically and clinically significant improvement of dyspnea. In asthma, only tiotropium has been tested: in patients with severe asthma (not well controlled on medications), it improved FEV1 and the risk of exacerbations.
At present, there is no unique way to select the best safety and efficacy profile among the three products for patients with COPD. In the future, and in the real clinical setting, the satisfaction of the patients according to different parameters including the persistence of night symptoms may favor one of these drugs over the other two. Combinations of LAMAs and LABAs may quickly change the use of these molecules in the treatment of COPD. At present, tiotropium has been found to be efficient in severe uncontrolled asthma.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The author declares that there is no conflict of interest.
