Abstract
Background:
We hypothesized a class effect of currently available long-acting muscarinic antagonists (LAMAs; i.e. tiotropium as a dry powder inhaler or a soft mist inhaler, aclidinium bromide, and glycopyrronium) in preventing chronic obstructive pulmonary disease (COPD) exacerbations. The hypothesis was tested with a network meta-analysis.
Methods:
Several databases and manufacturer’s websites were searched for relevant clinical trials. Randomized, controlled trials, of at least 12 weeks duration, comparing a LAMA with placebo or another LAMA were included. Moderate-to-severe and severe exacerbations were chosen as the outcome assessment criteria. The data were pooled using network meta-analysis.
Results:
A total of 27 studies with 48,140 subjects were included. All LAMAs reduced moderate-to-severe exacerbations compared with placebo. However, there were no statistically significant differences in preventing moderate-to-severe or severe exacerbations among LABAs. In a subgroup analysis restricting studies to those that had a minimum of 6 months of treatment, glycopyrronium was associated with the least-effective strategy and aclidinium was associated with the greatest probability of being the best therapy in preventing severe exacerbations. Our meta-regression analysis suggested that the prevention of COPD exacerbations were less effective in studies which allowed concomitant use of a long-acting beta agonist (LABA).
Conclusion:
All LAMAs were equally effective in preventing moderate-to-severe exacerbations. Aclidinium was associated with the lowest risk for severe exacerbations when treatment duration was 6 months or longer. The concomitant use of LABA may not enhance the efficacy of LAMAs in preventing COPD exacerbations. More studies are needed to further examine above findings.
Introduction
Chronic obstructive pulmonary disease (COPD) will likely become the third leading cause of death by 2030 according to World Health Organization and continues to be a major cause of disability and rising health care costs worldwide [WHO, 2004]. The total cost of COPD in 2010 was US$49.9 billion, including health care expenditures of US$29.5 billion in direct health care costs, US$8.0 billion in indirect morbidity costs, and US$12.4 billion in indirect mortality costs in the USA [Dalal et al. 2010]. These costs were the highest among common lung diseases. Most of the annual direct costs of COPD in high-income countries are attributable to hospitalizations. Since most hospitalizations result from exacerbations, preventing them is the key to controlling the cost of managing COPD [Oba, 2007]. COPD exacerbations are associated with an accelerated deterioration in lung function, reduced health related qualify of life, mortality and significant healthcare expenditures, thus the prevention of COPD exacerbations is recognized as a key goal in COPD disease management. Current guidelines developed by Global Initiative for COPD recommend a maintenance therapy either with a long-acting muscarinic antagonist (LAMA) or a long-acting beta agonist (LABA) in symptomatic patients with moderate or more severe COPD [Vestbo et al. 2013].
Currently, there are four LAMA formulations available for clinical use in COPD: tiotropium (TIO) as a dry powder inhaler (DPI) or a soft mist inhaler (SMI), aclidinium bromide (AB), and glycopyrronium (GPM). TIO-DPI (Handihaler) 18 μg once daily (qd) is approved for clinical use in the United States while TIO-SMI (Respimat) 5 μg qd (2.5 μg two puffs qd) has been used in Europe. The recommended dose of AB and GPM is 400 μg twice a day (bid) and 50 μg qd.
TIO-DPI was associated with reduced COPD exacerbations and related hospitalizations, improved quality of life and symptoms and GPM was shown to reduce COPD exacerbations and improved lung function, exercise tolerance and symptoms as compared with placebo [Barr et al. 2006; Karner et al. 2012; Ulrik, 2012]. Karabis and colleagues conducted a network meta-analysis comparing AB, TIO-SMI, TIO-DPI and GPM and concluded that AB was as effective as other LAMAs in improving lung function, health-related qualify of life and symptoms [Karabis et al. 2013]. However, COPD-related exacerbations were not included in their analysis.
We hypothesized a class effect of currently available LAMAs in preventing COPD exacerbations. When no clinical trials exist that directly compare all relevant treatment options, indirect comparisons can be made by comparing the relative effects of treatments against a common comparator or combining a variety of comparisons (variously referred to as a mixed or multiple treatment comparison (MTC) or network meta-analysis) [Lu and Ades, 2004]. The purpose of this study was to systematically review the efficacy of available LAMA formulations in preventing COPD-related exacerbations with a MTC meta-analysis.
Methods
Identification of trials
We identified all relevant clinical trials which studied clinical efficacies of a LAMA in COPD. Two authors independently searched the Ovid Medline database for studies published from 1946 to 15 May 2014 using the MeSH headings and keywords: randomized controlled trial AND Pulmonary Disease, Chronic Obstructive AND tiotropium, aclidinium, or glycopyrronium. In addition, we searched Scopus, CINAHL, and the internet including the online trial registries of manufacturers of above mentioned LAMA products. Bibliographies of all selected articles and review articles which included information on a LAMA in COPD were also reviewed for other relevant articles. We included any randomized clinical trial, published or unpublished, evaluating COPD patients with a LAMA. Randomized, controlled trials had to be of at least 12 weeks duration. Control interventions included placebo or another LAMA formulation. Two reviewers independently screened studies by title and abstract to evaluate whether a trial met the inclusion criteria. We chose moderate-to-severe and severe exacerbations as the outcome assessment criteria for the purpose of our meta-analysis. We extracted data on COPD exacerbations as moderate and severe. Moderate was generally defined as ‘worsening respiratory status which required treatment with systemic corticosteroids and/or antibiotics’ and severe as ‘rapid deterioration which required hospitalization’. The rate of COPD-related serious adverse events was used when a COPD-related hospitalization rate was not available for severe exacerbations.
Statistical analysis
The primary analyses were conducted with a Bayesian Markov chain Monte Carlo method and fitted with the Bayesian software in WinBUGS version 1.4.3 (Medical Research Council Biostatistics Unit, Cambridge, UK). Bayesian meta-analyses involve data, a likelihood distribution, a model with parameters, and prior distributions. The technique estimates the relative efficacy between treatments that have not been compared directly, and provide the most flexible approach to indirect comparison modeling. For the analyses in WinBUGS, every sample consisted of 100,000 iterations with an initial burn-in period of 10,000 iterations [Toft et al. 2007]. We used a Poisson likelihood and a log link. Each pair of treatments was compared by estimating a hazard ratio (HR) of the outcome. The Poisson model is useful for repeated event data such as number of exacerbations, where each individual may have more than one event [Guevara et al. 2004].
To assess the impact of LAMAs on moderate-to-severe and severe exacerbations, data were extracted in the form of rates and given as the number of events per person-years observed. When the number of events was not available in a given study, we substituted the number of subjects who experienced an exacerbation. We assumed that each of the log hazard ratios had been sampled from a normal distribution and that the hazard was constant in each arm over the follow-up period. We gave vague priors for all trial baselines, treatment effects, and between-trial variances. The probability that each LAMA formulation was associated with being the most efficacious treatment was calculated by counting the proportion of iterations of the Markov chain in which each LAMA formulation had the highest HR. The surface under the cumulative ranking (SUCRA), which is a simple numerical summary of these probabilities, was also calculated. The SUCRA would be 100% when a treatment is certain to be the best and 0% when a treatment is certain to be the worst [Salanti et al. 2011].
The presence of heterogeneity was explored by fitting covariates [i.e. smoking status, forced expiratory volume in 1 second (FEV1) at baseline, treatment duration (a minimum of 6 months), and concomitant use of inhaled corticosteroid (ICS) or LABA] in a meta-regression analysis [Dias et al. 2013a]. The covariates were chosen a priori based on previous studies [Cope et al. 2012; Karner and Cates 2012 ]. A subgroup interaction model was used for the concomitant use of LABA and a continuous covariate model was used for the rest of the covariates. Inconsistency was assessed by comparing the deviance and deviance information criterion (DIC) statistics in fitted consistency and inconsistency models. The posterior mean deviance of the individual data points in the inconsistency model were plotted against their posterior mean deviance in the consistency model [Dias et al. 2013b].
The autocorrelation plots showed that throughout the iterative process the autocorrelation was satisfactorily reduced to a nominal amount and the Brooks–Gelman–Rubin plots showed that the model had converged satisfactorily [Toft et al. 2007]. Assessment of model fit was based on comparison of residual deviance to the number of unconstrained data points, assessment of the DIC, and between-study standard deviation. We assessed the fit of our model using the DIC; a measure of model fit that penalizes model complexity. This criterion advocates selecting the model with the lowest DIC value among a series of competing models for the same data, as this model is believed to provide the best fit to the data [ Spiegelhalter et al. 2002 ]. All results for the network meta-analysis were reported as posterior means with corresponding 95% credibility intervals (CrIs). CrIs are the Bayesian equivalent of classical confidence intervals.
Results
Study selection
The electronic database searches identified 217 citations. A total of 168 studies were excluded on abstract review. The remaining 49 studies were reviewed for further details. Additional 27 studies were excluded for various reasons as shown in Figure 1. Further search identified five additional studies including one unpublished study. We included 27 studies comparing four different LAMA formulations for moderate-to-severe exacerbations and 23 studies for severe exacerbations including a total of 48,140 and 44,250 patients, respectively [Ambrosino et al. 2008; Bateman et al. 2010a, 2010b, 2013; Beeh et al. 2006; Brusasco et al. 2003; Casaburi et al. 2002; Chan et al. 2007; Chapman et al. 2014; Cooper et al. 2013; Donohue et al. 2010; D’Urzo et al. 2011; Dusser et al. 2006; Freeman et al. 2007; Jones et al. 2011, 2012; Kerwin et al. 2012a, 2012b; Niewoehner et al. 2005; Novartis, 2012; Powrie et al. 2007; Rennard et al. 2013; Tashkin et al. 2008; Tonnel et al. 2008; Vogelmeier et al. 2008; Wedzicha et al. 2013; Wise et al. 2013]. The study and patient characteristics are presented in Table 1 and S1 (in the Online Supplement). The mean ages (range: 63.5–65 years), proportion of male patients (range: 65–73.8%), the mean baseline FEV1 (range: 1.02–1.5 liters) and FEV1 per cent predicted (range: 38.7–53.6%) were comparable across the studies. The definitions of moderate and severe exacerbations used in the included studies were presented in Table S2 (in the Online Supplement). The definitions of COPD exacerbations were similar across the included studies. The network of treatments is displayed in Figure 2. The treatments formed a closed network, which was amenable to a MTC analysis.

Flow of study selection.
Study characteristics of included trials that provide data on the total number of exacerbations and/or the mean annual rate of exacerbations.

AB, aclidinium bromide; bid, twice a day; DPI, dry powder inhaler; FEV1, forced expiratory volume in 1 second; GPM, glycopyrronium; ITT, intention to treat; NR, not reported; PLB, placebo; qd, once a day; SMI, soft mist inhaler; TIO, tiotropium; % pred, percentage of the predicted value.
≠ Unclear for exacerbation outcomes. ¶ Modified Full Analysis Set was used which included all patients in the full analysis set except patients from a site, which had major issues with Good Clinical Practice compliance. † Number of patients included in this analysis. * Tiotropium was administered open-label. § Post-bronchodilator.
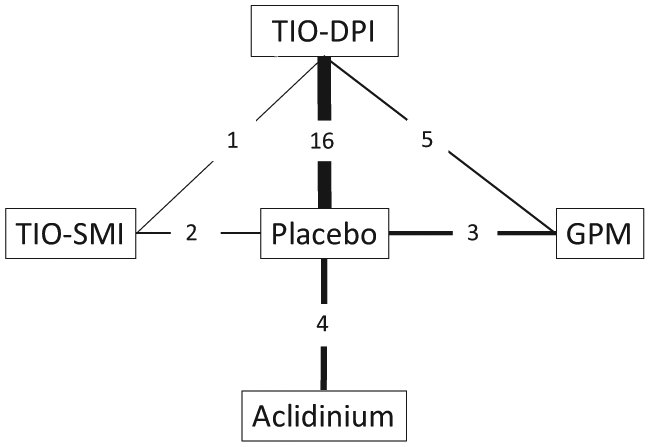
Diagram displaying the network of five arms involved in the Bayesian analysis. The links between nodes are used to indicate a direct comparison between pairs of treatments. The numbers shown along the link lines indicate the number of trials comparing pairs of treatments head-to-head. DPI, dry powder inhaler; GPM, glycopyrronium; SMI, soft mist inhaler; TIO, tiotropium.
Methodological quality of included studies
In general, the risk of bias in the included studies appeared moderate to low. Allocation concealment was appropriate in 19 studies, and unclear in eight studies. A total of 23 out of 27 studies presented intention-to-treat analyses and 24 studies were double blinded (Table 1). In the opinion of the authors, there were no studies that clearly should have been excluded from the analysis because of differences in baseline characteristics or poor quality.
Comparison of LAMAs on moderate-to-severe exacerbations
All studies were included in the evaluation for moderate-to-severe exacerbations. A random model was used for above analyses because of lower DIC scores as compared with a fixed model. Results of Bayesian network meta-analyses are presented in Table 2 and Figure 3. All LAMAs significantly reduced moderate-to-severe exacerbations compared with placebo (Figure 3). Stepwise comparison of all active treatment strategies did not reveal significant differences in rates of moderate-to-severe exacerbations (Table 2). TIO-SMI was associated with the greatest probability of being the best therapy and SUCRA value among all treatment strategies evaluated, 61.1% and 84.1%, respectively. However, the 95% credible intervals of the posterior distribution for the ranking suggested that any of the LAMA formulations could be the best therapy among all comparators (Table 3).
Treatment comparisons within the network meta-analysis for (a) COPD exacerbations and (b) restricting studies to those that had a minimum of 6 months of treatment. Bold type font indicates significant values.
(a).

(b).

TIO-DPI, tiotropium-dry powder inhaler; TIO-SMI, tiotropium-soft mist inhaler.

Pooled effect estimate on COPD exacerbations for long-acting muscarinic antagonists (LAMAs) versus placebo. DPI, dry powder inhaler; SMI, soft mist inhaler; TIO, tiotropium.
Probability of best therapy, SUCRA values and ranking of therapy in preventing chronic obstructive pulmonary disease exacerbations.

CrI, credible interval; TIO-DPI, tiotropium-dry powder inhaler; TIO-SMI, tiotropium soft mist inhaler.
Comparison of LAMAs on severe exacerbations
A total of 23 studies were included in the evaluation of severe exacerbations. A random model was used for above analyses because of lower DIC scores as compared with a fixed model. TIO-DPI is the only LAMA formulation which reduced severe exacerbations (HR 0.73; 95% CrI 0.60–0.86; Figure 3). Stepwise comparison of all remaining treatment strategies did not reveal significant differences in rates of severe exacerbations (Table 2). AB was associated with the greatest probability of being the best therapy and SUCRA value among all treatment strategies evaluated, 68.4% and 81.3%, respectively. However, the 95% credible intervals of the posterior distribution for the ranking suggested that any of the LAMA formulations could be the best therapy among all comparators. (For details, see Table S3 in the Online Supplement.)
Exploration of heterogeneity and assessment of inconsistency
When examining outcome measures, a fixed-effects model showed largely similar results in terms of treatment effects as the random effects model. Random effects models were preferred in the current analysis since they showed a slightly better model fit as described above. Bayesian random-effects computations were repeated with the aforementioned covariates. The meta-regression models with the covariates fit well as evidence by posterior means of the residual deviances smaller than data points and resulted in minimal changes in shrinkage estimates of the treatment effects. Between-trial heterogeneity was either unchanged or reduced with the introduction of covariates (Online Supplement, Table S3). The meta-regression adjustment for the percentage of concomitant use of ICS, proportion of active smokers, FEV1 at baseline, concomitant use of LABA did not alter the main findings or interpretation. However, an interaction effect was suggested between the concomitant use of LABA and the treatment effects for both moderate-to-severe and severe exacerbations as well as between the treatment duration (a minimum of 6 months versus less) and the effects in severe exacerbations (Online Supplement, Table S3). The effects of preventing COPD exacerbations by LAMAs were greater in studies which prohibited concomitant use of LABA as compared with those allowed it (Figures 4 and 5). AB was associated with the lowest risk for severe COPD exacerbations when treatment duration was 6 months or longer and the only LAMA that ranked best in preventing severe exacerbations as far as 95% CrIs of ranking were concerned (Table 3). There was no suggestion of such interactions with other covariates.

Subgroup analysis examining the effect of concomitant long-acting beta agonist (LABA) use in preventing moderate-to-severe exacerbations by long-acting muscarinic antagonists (LAMAs). AB, aclidinium bromide; CrI, credibility interval; DPI, dry powder inhaler; GPM, glycopyrronium; SMI, soft mist inhaler; TIO, tiotropium.
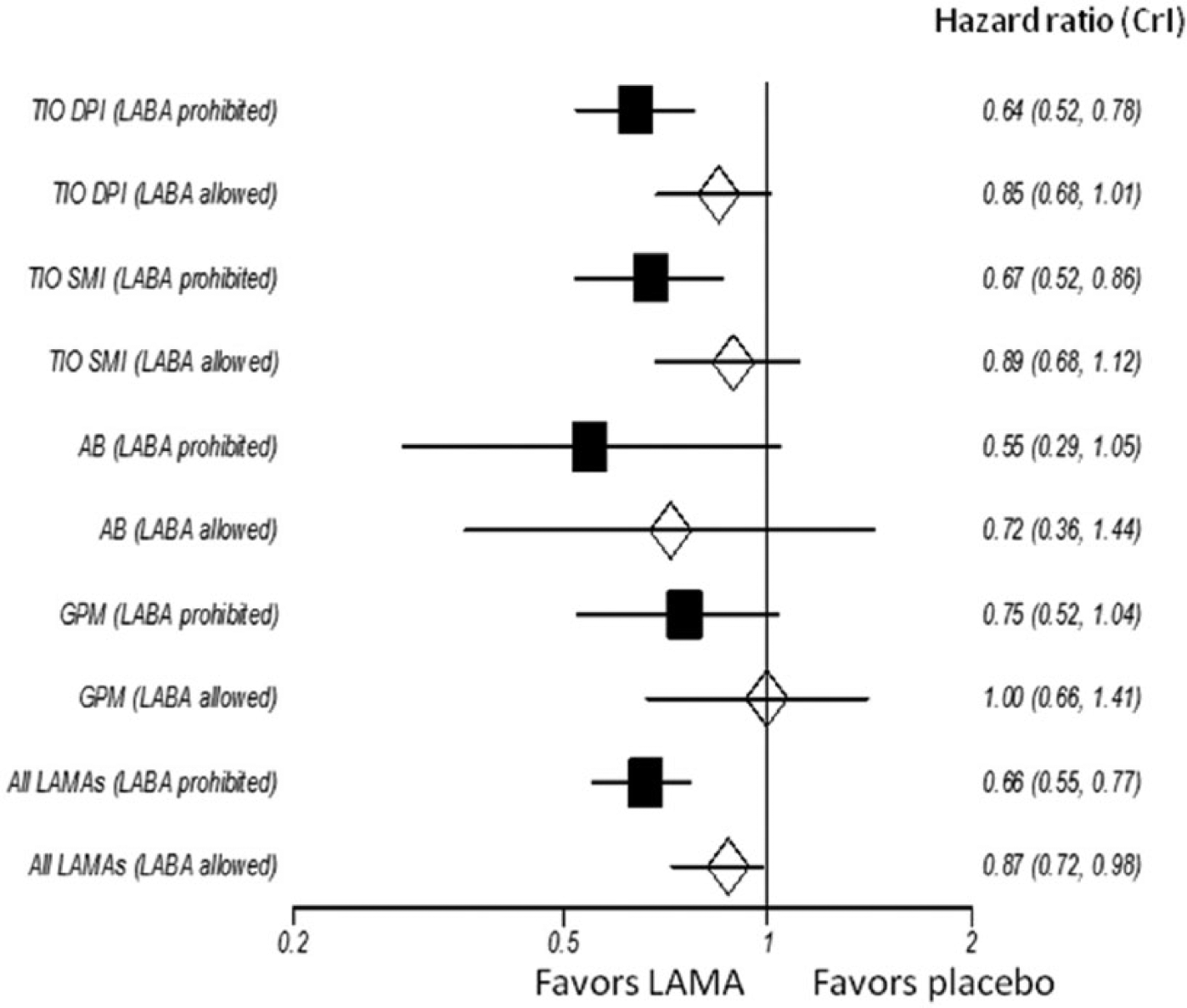
Subgroup analysis examining the effect of concomitant long-acting beta agonist (LABA) use in preventing severe exacerbations by long-acting muscarinic antagonists (LAMAs). AB, aclidinium bromide; CrI, credibility interval; DPI, dry powder inhaler; GPM, glycopyrronium; SMI, soft mist inhaler; TIO, tiotropium.
In moderate-to-severe exacerbations, the consistency model has a similar total residual deviance of the residual deviance, between-study standard deviation, and DIC (Online Supplement, Table S4). There were a couple of data points which showed a slightly lower value of the posterior mean deviance in the inconsistency model, suggesting that a consistency model did not fit these points that well (Online Supplement, Figure S1). These points corresponded to the trial of Ambrosino and colleagues. Exclusion of this study improved model fit but did not alter main findings. The hazard ratios were also similar for both models and there was considerable overlap in the 95% CrIs, suggesting there was no evidence of inconsistency in the network (Online Supplement, Table S4). In severe exacerbations, the heterogeneity estimates, the posterior means of the residual deviance and the DICs were very similar for both models, although both were slightly lower for the inconsistency model (45.0 versus 46.1 and 78.7 versus 79.5, respectively). A plot of the individual data points’ posterior mean deviance contribution showed data points with lower values in the inconsistency model (Online Supplement, Figure S2). Exclusion of the outliers improved model fit. However, the results did not change substantially. The HRs were also similar for both models and there was considerable overlap in the 95% CrIs, suggesting there was no evidence of inconsistency in the network (Online Supplement, Table S4).
Discussion
To the best of the authors’ knowledge, this is the first study to examine a class effect of LAMAs on COPD exacerbations using meta-analytic pooling. Our network analysis suggested that there were no statistically significant differences in preventing COPD exacerbations among LAMAs. Although TIO-DPI was the only LAMA which reduced severe exacerbations as compared with placebo, the 95% CrIs of the posterior distribution for the ranking suggested that any of the LAMA formulations could be the best therapy among all active comparators for the prevention of severe exacerbations. In a subgroup analysis restricting studies to those that had a minimum of 6 months of treatment, GPM was associated with the least-effective strategy and AB was the only strategy that could the best therapy among all LAMAs in preventing severe exacerbations (Table 3). Our meta-regression analysis suggested that the efficacy of LAMAs in preventing COPD exacerbations was greater in studies which prohibited concomitant use of LABA. The difference was most prominent with TIO-DPI. This would raise a possibility that the concomitant use of LABA may not enhance the efficacy of LAMAs in preventing COPD exacerbations. Above observation could explain why LAMA/LABA combinations failed to reduce COPD exacerbations as compared with a LAMA or a LABA alone in a recent meta-analysis [Karner and Cates 2012]. A similar phenomenon was observed among short-acting bronchodilators. Only ipratropium containing arms reduced COPD exacerbations and adding albuterol to ipratropium did not reduced COPD exacerbations compared with ipratropium alone [Friedman et al. 1999]. It was speculated that alterations in mucus production, rheology by glands, or mucus clearance in small airways were primarily responsible for COPD exacerbations which were favorably affected by anticholinergics rather than by beta-2 agonists.
The prevention of COPD exacerbations has become a critical aspect of decision making in the management of stable COPD patients. COPD exacerbation has a large impact on quality of life and resource utilization and strongly predicts a rapid decline in health status and death [Suissa et al. 2012]. US Hospitals with high risk-adjusted, 30-day all-cause unplanned readmission rates after an index hospitalization for a COPD exacerbation will be penalized with reduced reimbursement in near future as a part of Hospital Readmission Reduction Program by Centers for Medicare and Medicaid Services [Feemster and Au, 2014]. Our findings suggest that AB may be associated with the lowest risk for severe exacerbations when treatment duration is 6 months or longer.
Our study has the following limitations. First, we did not include other outcomes for the following reasons. (1) Karabis and colleagues conducted a network meta-analysis which demonstrated that AB produced similar improvements in lung function, health-related quality of life, and dyspnea compared with TIO-SMI, TIO-DPI, and GPM [Karabis et al. 2013]. (2) Pharmacotherapies in COPD have never been shown to improve mortality in clinical trials or meta-analyses. (3) The complexity of exacerbation outcomes associated with the current analysis was considered substantial. (4) We felt that long-term safety of TIO-DPI and SMI were addressed in the previous studies [Tashkin et al. 2008; Wise et al. 2013].
Second, we used the number of exacerbations per person-year observed to assess the exacerbation rates. The efficacy of AB on severe exacerbations derived from four studies which reported the number of subjects instead of exacerbation events for severe exacerbations [Jones et al. 2011, 2012; Kerwin et al. 2012a; Rennard et al. 2013]. We used the number of subjects in place of exacerbation events for this arm because excluding these studies would have precluded the inclusion of AB arm for severe exacerbations. We assumed the point estimate of efficacy would be quite similar when using the number of subjects instead of events because we picked the HR instead of mean difference as the outcome assessment parameter.
Third, the clinical heterogeneity of the trials included in our analysis might have affected the estimates of treatment effects. We conducted meta-regression analyses to adjust for trial-level covariates which did not affect the main results. Patient and study characteristics of the included studies were relatively homogenous but between-trial comparisons are known to be vulnerable to ecologic bias [Salanti et al. 2011]. The meta-regression analysis to assess biases by systematic differences between studies was also compromised due to limited information. For example, some studies reported only post-bronchodilator FEV1 while others report prebronchodilator values. Individual patient data would be necessary to avoid ecological bias and gain a much greater statistical power to detect a true covariate effect.
Fourth, although the network meta-analysis for COPD exacerbations yielded a reasonable fit to the data, there were a few studies that did not fit particularly well. The HRs were similar for both consistency and inconsistency models and there was considerable overlap in the 95% CrIs suggesting there was no evidence of inconsistency. However, this should be interpreted with caution as there may not have been sufficient power to detect inconsistency.
Fifth, the included person-years were greatest with TIO-DPI and least with AB. The precision of the estimates are likely more robust with TIO-DPI and SMI as compared with GPM and AB. Although the included person-years were least with AB, its sample size of 2506 included in this analysis would be still sufficient to achieve 80% power to detect an 20% relative reduction in the percentage of patients with at least one exacerbation when the control event rate is assumed to be 20%.
Conclusions
Our network analysis suggested there were no statistically significant differences in preventing COPD exacerbations among LAMAs. In a subgroup analysis restricting studies to those that had a minimum of 6 months of treatment, GPM was associated with the least effective strategy and AB was associated with the greatest probability of being the best therapy and SUCRA value in preventing severe exacerbations. LAMAs were less effective in preventing COPD exacerbations in studies which allowed concomitant use of LABA suggesting that the concomitant use of LABA may not enhance the efficacy of LAMAs in preventing COPD exacerbations. More studies are needed to further examine above observations.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
