Abstract
Background:
The preoperative determination of visceral pleural invasion (VPI) in patients with malignant solitary pulmonary nodules (SPNs) is essential for determining the surgical range and selecting adjuvant chemotherapy.
Objectives:
This study aimed to systematically investigate risk factors of VPI in patients with SPN and construct a preoperative predictive model for such patients.
Design:
This is a retrospective study. The clinical, radiological, and pathological characteristics of study subjects were reviewed, and the groups with and without VPI were compared.
Methods:
Multivariate logistic analysis was utilized to identify independent risk factors for VPI. Moreover, a predictive nomogram was constructed to assess the likelihood of VPI occurrence.
Results:
Of the 364 enrolled cases, SPNs adjacent to the pleura with VPI were found in 110 (30.2%) patients. By incorporating four preoperative variables, including tumor diameter (>2 cm), maximum computed tomography value (>200 Hu), air bronchogram sign, and age, a preoperative predictive nomogram was constructed. The nomogram demonstrated good discriminative ability, with a C-index of 0.736 (95% CI (0.662–0.790)). Furthermore, our data indicated that the air bronchogram sign (odd ratio (OR) 1.81, 95% CI (0.99–3.89), p = 0.048), a maximum diameter >2 cm (OR 24.48, 95% CI (8.43–71.07), p < 0.001), pathological type (OR 5.01, 95% CI (2.61–9.64), p < 0.001), and Ki-67 >30% (OR 2.95, 95% CI (1.40–6.21), p = 0.004) were overall independent risk factors for VPI.
Conclusion:
This study investigated the risk factors for VPI in malignant SPNs touching the pleural surface. Additionally, a nomogram was developed to predict the likelihood of VPI in such patients, facilitating informed decision-making regarding surgical approaches and treatment protocols.
Introduction
Lung cancer accounts for the most cancer-related deaths worldwide, with an estimated 1.76 million deaths each year. 1 Lung adenocarcinoma is the most common type of non-small cell lung cancer (NSCLC). Visceral pleural invasion (VPI) is defined as a tumor invasion of the visceral pleural elastic layer. Numerous studies have demonstrated that VPI represents a significant prognostic factor for poor outcomes among individuals with lung adenocarcinoma.2–4 According to the 8th edition of the tumor-node-metastasis (TNM) guidelines, once lung adenocarcinoma less than 3 cm invades the dirty pleura, it should be up-staged from stage T1 to stage T2. 5 Additionally, one study showed that the 5-year survival rate of stage IA1 lung cancer was 91.6%, whereas once VPI was present, the survival rate decreased to 71.5%. 6 Thus, the determination of VPI presence is crucial for accurately assessing tumor stage and prognosis.
Some studies have demonstrated that malignant solitary pulmonary nodules (SPNs) with VPI require more extensive surgery and increased exploration of the lymph nodes.7,8 Additionally, patients with VPI were found to have a heightened risk of recurrence and mortality following sublobar resection, thus suggesting that total lobectomy may confer a significant survival advantage over segmentectomy for malignant SPNs with VPI. 9 Regarding therapeutic selection, one study found that the utilization of adjuvant chemotherapy in operable lung adenocarcinoma patients with high-risk pathological features, including VPI positivity, might significantly improve their prognosis. 10 However, the pathological assessment of VPI often occurs only after tumor resection, resulting in delays in the determination of the surgical margin and selection of adjuvant treatment modalities for patients.
With the extensive utilization of computed tomography (CT), imaging features can offer evidence of potential pathological characteristics before the operation. 1 However, it is extremely challenging to distinguish VPI status based on clinical experience for SPNs adjacent to the pleura due to their unique location. 11 Although several previous studies have proposed predictive models for VPI among malignant SPNs, very few studies have systematically investigated risk factors for SPNs touching the pleural surface.12,13
In this study, we aimed to investigate the clinical, radiological, and pathological characteristics of malignant SPNs touching the pleural surface with VPI. Furthermore, we proposed to establish a clinical–radiological nomogram model for judging the possibility of VPI among malignant SPNs touching the pleural surface before surgery. This will provide new insights into surgical strategies and treatment procedures.
Materials and methods
Patients
Patients who underwent lung adenocarcinoma resection from December 2020 to January 2022 in the First Affiliated Hospital of Soochow University were enrolled in this study. The screening process of the study subjects is shown in Figure 1. To be eligible for inclusion, all the following criteria had to be met: (1) accessible preoperative CT images; (2) the lung nodule adjacent to the pleura according to the preoperative CT images; (3) lung adenocarcinoma being the primary tumor and no previous history of other cancers; and (4) accessible, reliable pathological elastin staining results to evaluate the presence of VPI. The patients were excluded if (1) they presented with pleural effusion; (2) the nodule was touching the interleaf pleura; and (3) the clinical data were incomplete.
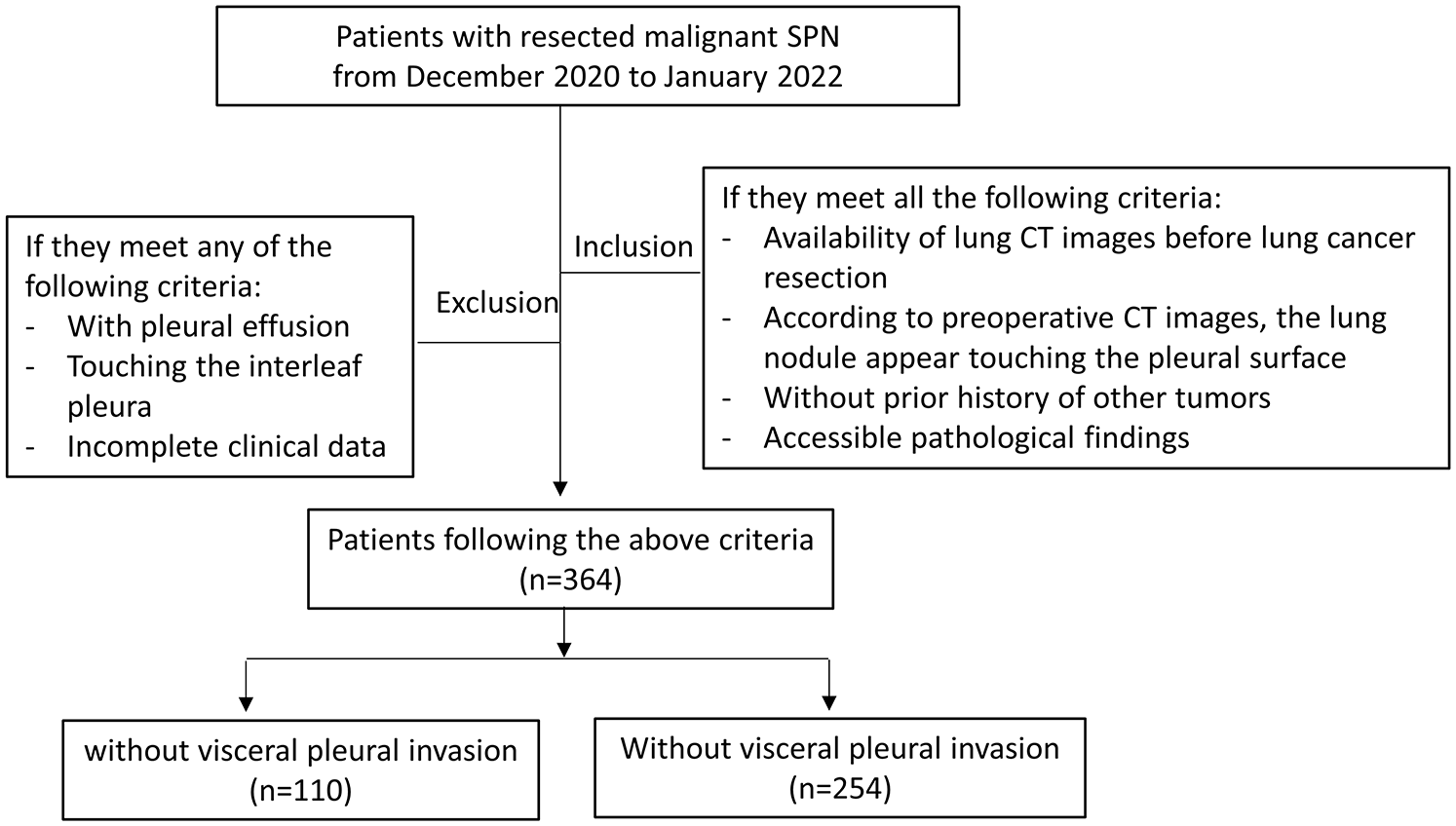
Flowchart of the screening process.
Imaging data collection
The preoperative imaging data of all enrolled subjects with malignant SPNs adjacent to the pleura were comprehensively collected and evaluated. An SPN was considered to touch the pleural surface if no fat space was visible between the tumor and the pleura or if the tumor directly extended into the pleura.14,15 The imaging manifestations of malignant SPNs touching the pleural surface were described in a standardized way, including the location of the lung lobes, tumor diameter, tumor margin (lobulation, spiculation), internal features of the tumor (vacuolar sign, air bronchogram sign, vessel convergence), and maximum CT values. The CT value is a relative value obtained by converting the detector’s X-ray attenuation coefficient into a certain mathematical pattern, measured in Heinz units (Hu). The CT value is determined by identifying the region of interest in CT images. The CT values for all patients in this study were obtained based on thin-layer CT, with a layer thickness of 1.25 mm. Representative imaging findings are shown in Figure 2.
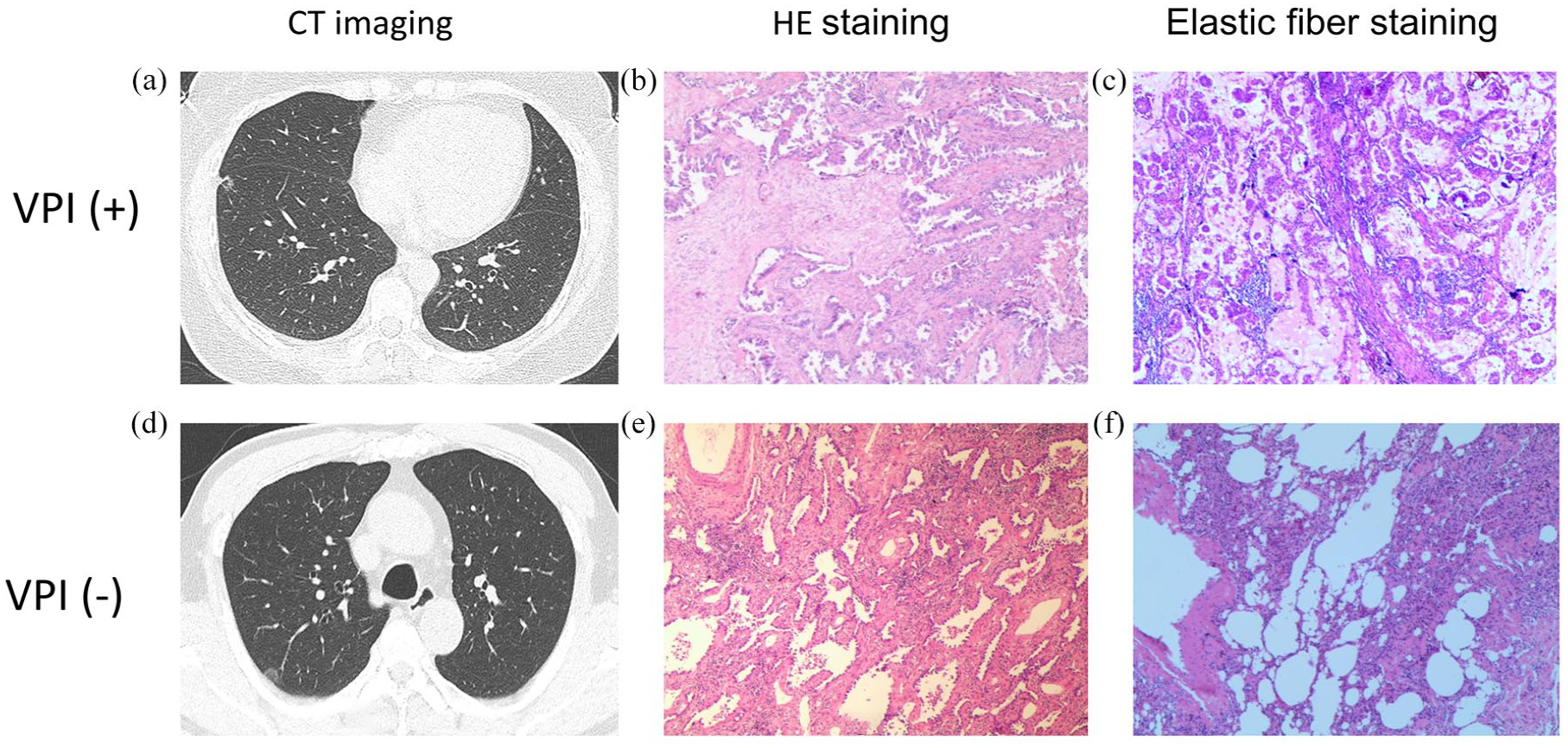
Representative CT and pathological images of malignant SPN that appear touching the pleural surface with and without VPI. (a) A lung nodule in the right middle lobe with microvessels penetrating around. The margin appears blurred and the vacuole is seen inside (transverse CT sections; 2 mm thick). (b) Pathological manifestation of invasive adenocarcinoma. (c) Elastic fiber staining revealed elastic fiber breakage. (d) A pure ground-glass opacity in the right upper lobe with visible vascular penetration. (e) Lung adenocarcinoma with an infiltrative component > 5 mm, which was defined as microinvasive adenocarcinoma. (f) Elastic fiber staining showed no visceral pleural invasion occurred.
Pathological evaluation
The pathological results of resected tumors were evaluated by two experienced pathologists. Based on the results of elastic fiber staining, the occurrence of VPI in malignant SPNs was determined. Elastic fiber staining revealing elastic fiber breakage was defined as VPI. 16 This study assessed the pathological types of malignant SPNs as standard. 17 The presence of a micropapillary component, lymph node metastasis, programmed cell death ligand 1 (PD-L1) expression, and Ki-67 expression were also assessed.
Study design and statistical analysis
This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement guidelines for reporting observational studies 18 (Supplemental File 1). All study subjects were divided into two groups according to their pathological elastin staining results: the VPI (+) group and the VPI (−) group. A wide spectrum of clinical, radiological, and pathological characteristics was compared between the two groups. Based on selected preoperative risk factors from univariate analysis, multivariate logistic regression analysis was utilized to identify independent risk factors for VPI.
Statistical analyses were performed using Statistical Product and Service Solutions (SPSS) software (version 25.0; IBM, Armonk, NY, USA). Continuous variables were compared between the VPI (+) group and the VPI (−) group using Student’s t-test or the Mann–Whitney U-test, and categorical variables were compared using the chi-square test or Fisher’s exact test. Multivariate logistic regression was conducted to identify clinical–radiological independent risk factors for VPI in malignant SPNs adjacent to the pleura. Moreover, the rates of progression-free survival (PFS) and overall survival (OS) of the groups were compared by the log-rank test, and the hazard ratio (HR) was calculated. Typically, a two-tailed p-value <0.05 was considered significant.
Nomogram construction and validation
Regarding nomogram construction, the nomogram is based on multivariable regression analyses. It integrates multiple predictive indicators in a certain proportion and then calculates the total score to predict the probability of occurrence of the outcomes. According to the contribution of the independent risk factors to the outcome variable (regression coefficients) obtained from the logistic multivariable regression model, a score is ascertained for each independent risk factor. Based on the significant preoperative variables identified through multivariate logistic analysis, a nomogram model was proposed and validated for the preoperative prediction of VPI.
The discriminatory ability of the nomogram model is evaluated using the receiver operating characteristic (ROC) curve, while its calibration is generally displayed by the calibration curve.
Moreover, the overall independent risk factors for VPI in malignant SPNs touching the pleural surface were evaluated and weighted. The nomogram was plotted and validated using the R programming language and environment (http://www.r-project.org/). The area under the curve (AUC), equivalent to the C-index, was used to assess the discriminatory performance of the predictive model. The internal validation was implemented using the bootstrapping method by setting the number of iterations to 1000. The calibration curve was used to assess the consistency between the predicted risks and the actual results of the nomogram. A C-index greater than 0.7 indicated a reasonable estimation.
Results
Demographic features of the study subjects
A total of 364 patients subjected to the screening process were enrolled in the study, 30.2% (110/364) of whom had VPI (Table 1). The median age of patients with VPI was 66.1 years, significantly higher than that of patients without VPI (61.7 years, p = 0.0110). The gender proportion was comparable between the two groups (47.3% vs 42.9%, p = 0.442). Regarding smoking history, the proportion of smokers was significantly higher in the VPI (+) group than in the VPI (−) group (23.6% vs 15.7%, p = 0.029).
Clinical and preoperative imaging characteristics of the study subjects.

VPI, Visceral pleural invasion.
P values less than 0.05 are highlighted in bold.
Imaging findings
Imaging features were compared between the two groups. As shown in Table 1, the proportion of lesions with a maximum diameter >2 cm was higher in the VPI (+) group compared to the VPI (−) group (87.3% vs 70.9%, p = 0.001). The distribution of lung lesions in the lobes of the lungs was approximately the same in the two groups (p = 0.854).
Though some marginal and internal imaging features, such as lobulation, spiculation, vessel convergence sign, and vacuolar sign, were comparable between study subjects with and without VPI (p > 0.05 for each), certain imaging characteristics were identified in patients with VPI. Specifically, the VPI (+) group was more likely to manifest an air bronchogram sign, a consolidation ratio >0.5, and a maximum CT value >200 Hu (51.7% vs 26.4%, p < 0.001) than the VPI (-) group. The above data indicate that a higher proportion of solid components may be associated with an increased risk of VPI occurrence.
Pathological evaluation
As depicted in Table 2, the pathological type of invasive adenocarcinoma was more likely to manifest in the VPI (+) group (100.0% vs. 35.0%, p < 0.001). Our data revealed that the micropapillary component, a highly recognized invasive feature, was more likely to be found in the VPI (+) group (80.0% vs 42.9%, p < 0.001). The frequency of lymph node metastasis was also significantly higher in the VPI (+) group (45.6% vs 11.1%, p < 0.001).
Immunohistochemical characteristics of the study subjects.

AIS, adenocarcinoma in situ; IAC, invasive adenocarcinoma; LN: lymph node; MIA: microinvasive adenocarcinoma; PD-L1: programmed cell death-Ligand 1; VPI, Visceral pleural invasion.
The lymph node dissection was performed in 57 patients in the VPI (+) group and 73 patients in the VPI (−) group.
Additionally, a larger proportion of patients in the VPI (+) group tested positive for PD-L1 (>1%) compared with their VPI (−) counterparts (61.8% vs 47.3%, p < 0.001). In terms of Ki-67 expression, the percentage of Ki-67 (+) cases was comparable between patients with and without VPI (99.1% vs 97.6%, p > 0.05). However, the VPI (+) group was more likely to exhibit higher Ki-67 expression (>30%; 35.5% vs 9.1%, p < 0.001). Collectively, the above findings indicated that malignant SPNs with VPI showed a higher aggressive potential.
Preoperative prediction of VPI in malignant SPNs touching the pleural surface
This study proposed a preoperative predictive model for VPI in malignant SPNs touching the pleural surface. First, preoperative variables that were statistically significant in the univariate analysis were selected for inclusion in the multivariate logistic regression analysis. As shown in Figure 3(a), age (odds ratio (OR) 1.37, 95% CI (1.02–1.84), p = 0.031), air bronchogram sign (OR 2.23, 95% CI (1.29–3.83), p = 0.004), diameter >2 cm (OR 14.07, 95% CI (5.95–33.27), p < 0.001), and a maximum CT value >200 Hu (OR 3.89, 95% CI 1.86–8.55, p < 0.001) were independent risk factors for the presence of VPI.

The construction and validation of a predictive nomogram for VPI based on preoperative variables. (a) Multivariate logistic regression analysis of preoperative risk factors for VPI among peripheral lung adenocarcinoma that appears touching the pleural surface. (b) The construction of a nomogram for VPI prediction based on the significant preoperative variables from the multivariate logistic regression analysis. (c) A ROC curve for the discriminative validation of the nomogram with a C-index of 0.736 (95% CI (0.662–0.790). (d) A calibration curve for the estimation of agreement between the predicted VPI of the nomogram and actual VPI.
The above four significant variables from the multivariate analysis were selected to construct a nomogram for the preoperative prediction of VPI. As shown in Figure 3(b), a tumor diameter >2 cm was defined as the most dominant risk factor for VPI of malignant SPNs that touched the pleural surface, followed by a maximum CT value >200 Hu, air bronchogram sign, and age. Based on the nomogram proposed in this study, the probability of VPI could be conveniently predicted among patients with malignant SPNs adjacent to the pleura. The nomogram model showed favorable discrimination, with a C-index of 0.786 (95% CI (0.754–0.819); Figure 3(c)). Furthermore, this study implemented the bootstrapping method for internal validation and obtained a corrected AUC value of the model. By setting the number of iterations to 1000, this predictive model calculated a corrected AUC of 0.738 (95% CI (0.737–0.739)). Regarding consistent validation, the calibration curve also showed favorable agreement between the predicted VPI of the nomogram and the actual occurrence of VPI (Figure 3(d)).
Overall independent risk factors for the occurrence of VPI in malignant SPNs adjacent to the pleura
This study also integrated clinical variables, preoperative imaging features, and postoperative pathological manifestations to explore overall independent risk factors and their different contributions to the occurrence of VPI. As shown in Figure 4(a), the air bronchogram sign (OR 1.81, 95% CI (0.99–3.89), p = 0.048), a maximum diameter >2 cm (OR 24.48, 95% CI (8.43–71.07), p < 0.001), pathological type (OR 5.01, 95% CI (2.61–9.64), p < 0.001), and Ki-67 > 30% (OR 2.95, 95% CI (1.40–6.21), p = 0.004) were independent risk factors for VPI.

Overall independent risk factors analysis for the occurrence of VPI in malignant SPN touching the pleural surface. (a) Multivariate logistic regression analysis of overall risk factors for VPI. (b) Four categorical variables were included in the model and demonstrated different contributions to the probability of VPI. (c) A ROC curve for the discriminative validation of the nomogram with a C-index of 0.836 (95% CI (0.794–0.878). (d) The calibration curve for the estimation of agreement between the predicted VPI of the nomogram and actual VPI.
Then, a nomogram was developed to assess the different contributions of the four independent categorical variables to the probability of VPI. As shown in Figure 4(b), the pathological type was defined as the predominant risk factor for VPI of malignant SPNs touching the pleural surface, followed by a maximum diameter >2 cm, Ki-67 >30%, and air bronchogram sign. Additionally, as shown in Figure 4(c), the nomogram showed favorable discrimination, with a C-index of 0.836 (95% CI 0.794–0.878). The calibration curve of the nomogram (Figure 4(d)) also demonstrated favorable consistency.
Impact of VPI on postoperative PFS and OS
We collected prognostic data from patients who underwent a follow-up after surgery (n = 285). The postoperative therapeutic intervention for all patients adhered to the National Comprehensive Cancer Network guidelines. Selecting postoperative PFS and OS as the primary outcome endpoints, we compared the individual proportions in the VPI (+) and the VPI (−) groups. As shown in Figure 5, VPI decreased the rate of PFS (HR 3.20, 95% CI (1.80–5.68), p < 0.001) and OS (HR 10.67, 95% CI (2.93–38.90), p < 0.001).

Kaplan–Meier survival analysis for the progression-free survival and overall survival proportion between the VPI (+) and VPI (−) groups. (a) VPI decreased the proportion of Progression-Free Survival (HR 3.20, 95% CI (1.80–5.68), p < 0.001). (b) VPI decreased the proportion of Overall Survival (HR 10.67, 95% CI (2.93–38.90), p < 0.001).
Discussion
To the best of our knowledge, this is the first study to focus on the early preoperative prediction of VPI among malignant SPNs that appear touching the pleural surface. Recently, a study has demonstrated that the distance between the pleura and the tumor constitutes an independent risk factor for VPI. 19 Another study found that lung tumors in contact with the pleura were more likely to develop VPI than those with a specific distance between the tumor and the pleura. 20 Based on the high consistency of the tumor–pleura relationship among the present study subjects, this study is significant and persuasive.
Age has been identified as a significant adverse prognostic factor for patients with NSCLC. 21 Our study also found age to be an independent risk factor for VPI in lung adenocarcinoma adjacent to the pleura. Gender as a risk factor for VPI among lung adenocarcinoma patients remains controversial. One study indicated that gender is an independent risk factor for VPI among malignant SPNs. 13 However, our data showed that gender was not associated with VPI, which is consistent with some studies demonstrating that gender was not a risk factor for VPI in lung adenocarcinoma.2,12
Tumor size was demonstrated to be an important predictive risk factor for VPI12,22 by numerous studies. According to the 8th edition of the TNM staging system, T staging was only determined by the size of the tumor’s invasive component without the epidermal component. 23 Interestingly, a study found that total tumor size was not related to the size of the infiltrative component of the pathological subtype. 3 The current study demonstrated that the tumor size was an independent predictor for VPI in lung adenocarcinoma adjacent to the pleura. This might imply that a stronger correlation exists between total tumor size and the infiltrative component in lung adenocarcinomas adjacent to the pleura compared with those distant from the pleura. More studies are necessary to explore whether there are differences in the proportion of tumor infiltration between subgroups with different distances between malignant SPNs and the pleura.
Similar to a study conducted by Zhai et al., 13 we found that air bronchogram sign was an independent risk factor for VPI among malignant SPNs adjacent to the pleura. Some studies demonstrated that tumor spread through air spaces constituted an essential invasive modality in lung adenocarcinoma. 24 One possible explanation is that malignant SPNs presenting with the air bronchial sign are more prone to invade the lung parenchyma via tumor spread through the air spaces pattern. Despite one study revealing that lobulation was also encompassed in the predictive model for VPI in lung adenocarcinoma, 13 our study revealed no external morphological features of the tumor as independent risk factors for VPI in tumors close to the pleura. This might be attributed to the fact that we studied lung cancer adjacent to the pleura, thereby causing the partial disparity in the findings.
Lymph node metastases and VPI are important predictors of patient prognosis. However, controversy exists regarding the relationship between the occurrence of VPI and lymph node metastases in patients with lung cancer. The present study indicated that lymph node metastasis was more prone to occur in lung adenocarcinoma with VPI. In contrast to the above finding, a recent study found that pathology-reported VPI was not a risk factor for occult lymph node metastasis. 20 In addition, another study demonstrated that lymph node metastasis was an independent predictor for VPI in peripheral NSCLC that was not adjacent to the pleura. 15
Interestingly, our study revealed a positive correlation between the maximum CT value and the likelihood of VPI occurrence in lung adenocarcinoma adjacent to the pleura. This finding underscores the importance of investigating whether CT value quantification can serve as a reliable predictor of tumor invasion, as has been explored in recent studies.15,25 Additionally, some studies found that the quantitative CT values of lung cancer were associated with their pathological subtypes.26,27 Given that pathological subtypes were strongly correlated with the invasive manifestation of lung cancer, the maximum CT value might represent the pathological component and thus affect the occurrence of VPI in lung adenocarcinoma adjacent to the pleura.
Finally, this study has some limitations. First, this is a single-center study, and multicenter studies are needed to validate the proposed nomogram model. Second, since this study mainly focused on the construction of a clinical–radiological predictive model of malignant SPNs adjacent to the pleura, data based on long-term follow-up to determine differences in survival rate between the VPI (+) and VPI (−) groups were lacking.
Conclusion
In conclusion, we investigated a wide spectrum of clinical, radiological, and pathological features of malignant SPNs touching the pleural surface with VPI. Our findings contribute to a better understanding of the clinical nature and independent risk factors for the occurrence of VPI in malignant SPNs. Additionally, we proposed a preoperative clinical–radiological nomogram for VPI prediction among malignant SPNs touching the pleural surface to facilitate informed decision-making regarding surgical approaches and treatment protocols. Considering the relatively small sample size in this research, further prospective multicenter studies are needed to verify the nomogram model proposed in this study.
Supplemental Material
sj-doc-1-tar-10.1177_17534666241285606 – Supplemental material for Risk analysis of visceral pleural invasion in malignant solitary pulmonary nodules that appear touching the pleural surface
Supplemental material, sj-doc-1-tar-10.1177_17534666241285606 for Risk analysis of visceral pleural invasion in malignant solitary pulmonary nodules that appear touching the pleural surface by Ziwen Zhu, Weizhen Jiang, Danhong Zhou, Weidong Zhu and Cheng Chen in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
We would like to thank all the nurses and the clinical staff who provided care for the patient. We would like to thank our friend James Golby for helping us polish the manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
