Abstract
Background:
Maintaining the patient awake and not intubated during the venovenous extracorporeal membrane oxygenation (VV ECMO) reduces the risk of ventilation-induced lung injury in patients with ARDS. Currently, there is a lack of data on outcomes and complications associated with the awake ECMO approach.
Objectives:
To evaluate outcomes and the occurrence of complications of awake ECMO approach guided by local safety protocol comprising ultrasound-guided cannulation, argatroban-based anticoagulation, respiratory support, and routine sedation targeted to reduce respiratory effort and keeping nurse-to-patient ratio of 1:1.
Design:
A single-center retrospective case series analysis.
Methods:
Consecutive patients with COVID-19-related acute respiratory distress syndrome (ARDS) (CARDS) treated by full awake VV ECMO approach from April 2019 to December 2023 were eligible.
Results:
Our center treated 10 patients (mean age 54.7 ± 11.6 years) with CARDS with an awake ECMO approach. The reasons for awake ECMO included the presence of barotrauma in six patients, a team consensus to prefer awake ECMO instead of mechanical ventilation in three patients, and the patient’s refusal to be intubated in one case. Before ECMO, patients were severely hypoxemic, with a mean value of Horowitz index of 48.9 ± 9.1 mmHg and a mean respiratory rate of 28.8 ± 7.3 breaths per minute on high-flow nasal cannula or noninvasive ventilation support. The mean duration of awake VV ECMO was 558.0 ± 173.6 h. Seven patients (70%) were successfully disconnected from ECMO and fully recovered. Intubation from respiratory causes was needed in three patients (30%), all of whom died eventually. In total, three episodes of delirium, two episodes of significant bleeding, one pneumothorax requiring chest tube insertion, and one oxygenator acute exchange occurred throughout the 5580 h of awake ECMO. No complications related to cannula displacement or malposition occurred.
Conclusion:
The awake ECMO strategy guided by safety protocol appears to be a safe approach in conscious, severely hypoxemic, non-intubated patients with COVID-19-related ARDS.
Plain language summary
Why Was the Study Done? Extracorporeal membrane oxygenation (ECMO) represents a life-saving therapeutic approach that ensures appropriate gas exchange in patients with the most severe form of respiratory failure – acute respiratory distress syndrome (ARDS). Typically, patients are connected to ECMO when already deeply sedated and mechanically ventilated. The awake ECMO approach (keeping the patient awake, not intubated, and breathing spontaneously during ECMO support) minimizes the risks associated with mechanical ventilation and provides several relevant physiological benefits. However, the awake ECMO approach is also associated with several significant risks, including delirium, bleeding, and cannula displacement. Published papers have reported relatively frequent complications and method failures. What Did the Researchers Do? To address safety concerns regarding the awake ECMO approach, we present a single-center retrospective analysis of ten COVID-19-related ARDS patients treated with the awake ECMO approach, guided by the local safety protocol. What Did the Researchers Find? The awake ECMO approach yielded success (i.e., the patient was not intubated for respiratory causes, was successfully disconnected from ECMO, and fully recovered in seven patients (70.0%), outperforming previously published efficacy ranges. Three patients were intubated due to the progression of respiratory failure and eventually died. The incidence of adverse events during the 5,580 hours of awake ECMO was considered low. No cannula displacement or malposition occurred despite routine active physiotherapy, including walking during ECMO treatment in three patients. What Do the Findings Mean? The general applicability of the study is limited by the low number of patients and the retrospective monocentric design. However, the presented data illustrate real-life clinical scenarios and could aid clinicians in managing severely hypoxemic but still conscious and cooperative patients.
Keywords
Introduction
The COVID-19 pandemic represented a challenge for intensivists due to specific clinical conditions arising from the newly recognized disease. Structural and functional damage to carotid bodies caused by coronavirus led to an impairment of the peripheral signaling of arterial oxygen levels; consequently, patients often suffered from severe hypoxemia without a sensation of dyspnea—the condition termed “silent hypoxemia” (SH). 1 However, the central biochemical input determined by inflammatory and mechanical signals from the injured lungs may still cause excessive respiratory effort and expose the lungs to the risk of further aggravation of lung injury induced by vigorous spontaneous ventilation—known as patient self-inflicted lung injury (P-SILI).2,3 The subsequent development of dyspnea should be considered a warning symptom that could trigger the decision to escalate the respiratory support. 4 The most severe cases of P-SILI are characterized by a disruption of the lung parenchyma, causing spontaneous barotrauma (pneumothorax, pneumomediastinum, pneumopericardium, subcutaneous emphysema), which is associated with increased mortality. In patients with COVID-19-related acute respiratory distress syndrome (ARDS) (CARDS), the reported incidence of spontaneous barotrauma was unusually high.5–7 The initiation of mechanical ventilation in such patients further increases the risk of barotrauma progression, including life-threatening tension pneumothorax.
When facing a conscious, non-intubated patient with severe dyspnea and hypoxemia, for whom invasive positive pressure mechanical ventilation (IPPV) would be associated with a high risk of complications, keeping the patient awake, not intubated, and breathing spontaneously on venovenous extracorporeal membrane oxygenation (VV ECMO) support (the so-called “full awake VV ECMO approach”) provides a valuable opportunity to minimize the risks arising from IPPV. The awake ECMO approach also offers other benefits: the diaphragm contractions during inspiration optimize ventilation-perfusion matching while maintaining respiratory muscle tone preserves functional residual capacity, and prevents atelectasis. Negative intrathoracic pressure during inspiration enhances venous return, leading to subsequent optimization of cardiac preload. Furthermore, awake ECMO allows the patient to interact with staff and relatives and to participate in active physiotherapy, reducing the risk of ICU-acquired weakness. The patient can also eat a regular diet, which facilitates meeting nutritional targets.8,9
However, the awake ECMO approach is also associated with several significant risks. Despite adequate oxygenation and carbon dioxide removal by ECMO, vigorous respiratory effort can persist in some patients. 10 Such patients are then at risk for further deterioration of lung injury. Intensive spontaneous movement (mainly due to anxiety and hyperactive delirium) can aggravate bleeding from the cannula insertion site and pose a risk of cannula displacement, with potentially immediate fatal consequences. These risks can be, however, mitigated by implementing safety measures and protocols.
We report a retrospective, single-center series of 10 conscious, spontaneously breathing, but severely hypoxemic CARDS patients who presented with developed barotrauma or were considered at high risk of barotrauma at the time when mechanical support of the respiratory system became imperative. We aim to analyze the occurrence of complications in patients treated in our department with awake ECMO targeted at preventing delirium, excessive respiratory effort, bleeding, thrombotic events, and cannula malposition or decannulation.
Materials and methods
Study design
A retrospective case series analysis.
Patients
All adult patients (age > 18 years) with CARDS treated with a full awake venovenous extracorporeal membrane oxygenation (VV ECMO) approach (i.e., ECMO started in a non-intubated patient) at a single ECMO center (Department of Anesthesiology and Intensive Care Medicine, University Hospital Ostrava, Czech Republic) from March 1, 2020, to December 31, 2023, were included in the analysis.
Decision-making process
The awake ECMO approach was considered for all patients with severe CARDS who were fully awake and conscious when mechanical support of respiratory function became imperative. The decision to implement an awake ECMO strategy was made by the ECMO team based on the patient’s medical history, current clinical condition (consciousness, oxygenation levels, respiratory rate, hemodynamic status, presence of apparent barotrauma), and radiological findings. The risks and benefits of both VV ECMO and mechanical ventilation were discussed with the patient. Before being connected to ECMO, all patients were fully conscious and cooperative, and informed consent for the awake ECMO procedure was obtained from all of them.
Definitions
The presence of COVID-19 infection was determined by polymerase chain reaction. ARDS was defined according to the Berlin criteria. 11 For hypoxemia evaluation, the arterial partial pressure of oxygen (PaO2) to the fraction of inspired oxygen ratio (PaO2/FiO2 ratio; Horowitz index) was calculated; in patients without an arterial line, peripheral pulse oximetric oxygen saturation (SpO2) was used as a surrogate for the calculation of arterial partial pressure of oxygen according to Brown’s non-linear equation. 12 The available data from the last 6 h before ECMO initiation were used for the evaluation of PaO2/FiO2, as well as the initial (before ECMO) respiratory rate (RR) and heart rate (HR). Values of RR and HR from the first 6 h after ECMO initiation were also used for comparison.
Definitions used for complications
Only complications that arose during the awake ECMO approach were evaluated. Major bleeding was defined as bleeding requiring red blood cell transfusion or invasive surgical intervention. A thrombotic event was defined as an episode of ischemic stroke or pulmonary embolism. Delirium was defined as documented cognitive dysfunction or psychomotor agitation (Richmond Agitation-Sedation Scale (RASS) > 0) requiring additional pharmacological management.
Adverse events related to ECMO were defined as a sudden deterioration of oxygenator function, decannulation, malposition of the cannula, or cessation of blood flow through the circuit requiring urgent circuit exchange. Intubation due to worsening respiratory function (subjectively intolerable dyspnea, development of hypoxemia or hypercapnia despite normal VV ECMO flow and oxygenator function, ineffective shallow breathing, or exhaustion) was considered a failure of awake ECMO.
Management protocol
Patients were treated according to the local protocol of care for awake ECMO patients, addressing the following points:
Ultrasound-guided cannulation
ECMO settings (blood flow, sweep gas flow, oxygen fraction) aimed at maintaining generally accepted gas exchange targets (PaO2 > 8.0 kPa and PaCO2 < 6.0 kPa) 13
Mild sedation targeting a RASS score of 0 to −2 (enabling daytime activities and induction of sleep during the night; all patients received a combination of an opiate and dexmedetomidine from the start)
Controlling the respiratory rate < 20 and relieving dyspnea by a combination of sedation and high-flow nasal cannula (HFNC) support of 45–60 L/min and FiO2 < 0.6
Daily active physiotherapy
Anticoagulation with argatroban targeted to an anti-IIa activity of 0.4–0.6 µg/mL; simultaneously, maintaining fibrinogen blood levels > 2 g/L and platelet counts >100,000/μL
Daily checks of the partial pressure of oxygen in the oxygenator outflow and daily visual evaluation for blood clots in the oxygenator
Control of the cannulation site and cannula position at least four times a day
A nurse-to-patient ratio of strictly 1:1 for awake ECMO patients
For weaning from ECMO, adapted criteria from the Extracorporeal Life Support Organization (ELSO) guidelines were used. 14
Statistics
A Shapiro-Wilk test was used to test the normality of the distributions. Normally distributed variables were reported as means with standard deviations and compared using Student’s t-test. Non-normally distributed continuous data were reported as medians with 95% confidence intervals (CI) and analyzed using the Mann–Whitney U-test. A p-value of < 0.05 was considered statistically significant for all comparisons. Stata software version 18 (StataCorp. 2018. Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC.) was used for all statistical analyses.
Data collection and reporting
VV, MB, TR, and HS collected data retrospectively from the electronic database and patients’ datasheets; PS and MF checked the data.
The manuscript was prepared in adherence to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for cohort studies. 15
Results
Demographic data
We identified 80 patients with COVID-19-related ARDS treated in our center with VV ECMO. Four patients died within 6 h of VV ECMO initiation. Of the remaining 76 patients, ten were treated with the full awake VV ECMO strategy. The mean age of the awake ECMO group was 54.7 ± 11.6 years, and the mean body mass index (BMI) was 31.8 ± 9.1; eight of the ten patients were men. The mean Sequential Organ Failure Assesment (SOFA) score at the time of ECMO initiation was 5.7 ± 0.9.
Parameters before ECMO initiation
The median total time spent on high-flow nasal cannula oxygen therapy (HFNC) and noninvasive ventilation (NIV) before ECMO was 58 h (95% CI: 14.8–135.2). An awake prone position was used in two patients before admission to the ECMO center. Six patients were admitted from other hospitals, two from our hospital’s emergency room and two from our hospital’s COVID-19 general ward.
The main reasons for choosing the awake ECMO strategy included:
Existing barotrauma in six cases
A team consensus preferred awake ECMO over mechanical ventilation in three severely hypoxemic but conscious and cooperative patients
A patient’s refusal to be intubated in one case.
One patient was placed on ECMO in another hospital by our mobile team before transport to our department; seven patients were placed on ECMO immediately after admission (<2 h), and one patient was placed on ECMO 22 hours from admission (Table 1).
Demographic data and clinical parameters before ECMO initiation.

CT before ECMO not performed.
Placed on ECMO in another hospital by our mobile team.
BMI, body mass index; ECMO, extracorporeal membrane oxygenation; F, female; HFNO, high-flow nasal cannula oxygenotherapy; M, male; NA, not applicable; NIV, noninvasive ventilation; OH, other hospital; PNM, pneumomediastinum; PNX, pneumothorax.
At the time of decision-making, patients suffered from severe dyspnea, mild tachycardia with a mean HR of 94.5 ± 11.8 beats per minute, tachypnea defined by a mean RR of 28.8 ± 7.3 breaths per minute, and severe hypoxemia with a calculated mean Horowitz index of 48.9 ± 9.1 mmHg on HFNC or NIV support. Two patients had a record of recent hemoptysis. Ultrasound-guided femoral-jugular cannulation was used in all cases, including four patients with subcutaneous truncal emphysema.
Effect of ECMO on physiological parameters
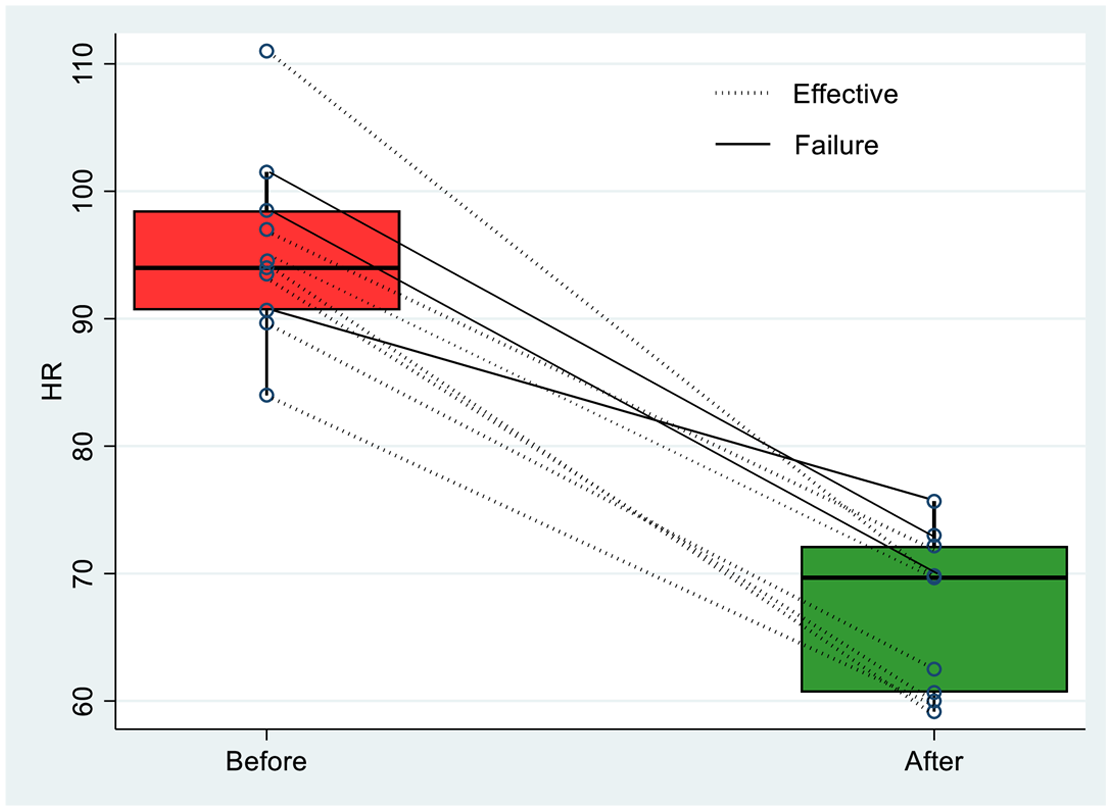
After the slow initial titration, ECMO settings ensuring appropriate gas exchange (PaO2 > 8.0 kPa and PaCO2 < 6.0 kPa) were as follows: mean blood flow of 4.6 ± 0.4 L/min, mean oxygenator sweep gas flow of 5.9 ± 1.1 L/min, with an oxygen fraction of 1.0. Starting ECMO support was associated with substantial subjective relief of dyspnea in nine patients (90.0%); one patient (10.0%) reported only mild improvement. The mean RR decreased from 28.8 ± 7.3 to 15.3 ± 5.4 breaths per minute. The difference in overall RR before ECMO and after ECMO initiation was statistically significant (p < 0.001) (Figure 1). The mean HR also decreased significantly from 94.5 ± 11.8 to 67.0 ± 7.8 beats per minute (p < 0.001) (Figure 2).
Respiratory rate before connection to ECMO (left box) and after ECMO starting (right box). Individual changes are represented by lines (dotted lines—successful awake ECMO treatment; solid lines—awake ECMO failure).

Heart rate before connection to ECMO (left box) and after ECMO starting (right box). Individual changes are represented by lines (dotted lines—successful awake ECMO treatment; solid lines—awake ECMO failure).
Active in-bed physiotherapy was initiated in all patients within 48 h after being connected to ECMO. Three patients were able to stand and walk while on ECMO support. No complications related to physiotherapy were reported.
Outcomes and complications
Seven patients (70%) were successfully disconnected from ECMO, fully recovered, and discharged from the hospital. The awake ECMO strategy failed in three patients (30.0%) who required intubation due to respiratory causes. After intubation, the static compliances of their respiratory systems (9, 10, and 12 mL/cm H2O, respectively) indicated severe lung injury. In all three patients, imminent bronchoscopy was performed, revealing small blood clots in the peripheral bronchial structures of two of these patients (both of whom had a record of hemoptysis before ECMO initiation); however, evacuation of the clots did not result in a change in compliance. All three patients later died from multiorgan failure.
One patient was intubated due to a non-respiratory cause (intracerebral hemorrhage requiring surgery). This patient was not considered a failure of awake ECMO because the intubation was performed for a neurological cause. However, this event was considered a complication of ECMO rather than its failure. Still, the patient was successfully disconnected from mechanical ventilation while on ECMO and fully recovered, achieving a Cerebral Performance Category ranking of 1. Besides this patient, an episode of major bleeding occurred in only one other patient (hemothorax requiring chest tube insertion). In total, therefore, major bleeding occurred in 20.0% of patients, with only a single episode in each of them. No thrombotic events occurred.
Mean awake ECMO treatment lasted for 558.0 ± 173.6 h. In patients who were intubated due to respiratory causes, the mean awake ECMO time until intubation was 527.0 ± 128.5 h. Eighteen oxygenators were used throughout the 5580 h of awake ECMO (i.e., a mean oxygenator lifespan of 310.0 h, including the successful disconnection from the functional oxygenator in seven patients). Acute oxygenator exchange was required in only one case due to the sudden deterioration of the patient’s oxygenation. No cannula malposition or displacement occurred.
Three patients (30.0%) developed a hyperactive form of delirium (a single episode in each of them), requiring additional pharmacotherapy. Two of the patients who developed delirium were later intubated due to respiratory causes and died from multiorgan failure. One patient (10.0%) developed pneumothorax requiring chest tube insertion (this patient had massive subcutaneous emphysema on admission, and a pre-ECMO CT scan was not performed) (Table 2).
Outcomes and complications.

ECMO, extracorporeal membrane oxygenation; ICH, intracerebral hemorrhage; PNM, pneumomediastinum; PNX, pneumothorax.
Discussion
We present a single-center, retrospective data analysis from 10 severely hypoxemic CARDS patients treated with the awake ECMO approach. The main reasons for the awake ECMO support were barotrauma or the risk of barotrauma in spontaneously breathing, conscious patients with CARDS. The initiation of ECMO support was associated with subjective relief from dyspnea and decreased respiratory and HRs. Despite the long period spent awake on ECMO support (mean duration of approximately 23 days) and early active physiotherapy (including ambulation in three patients), we identified a relatively small number of serious adverse events. The awake ECMO approach resulted in success (i.e., patients not intubated for respiratory causes, successfully disconnected from ECMO, fully recovered, and discharged from the hospital) in seven patients (70.0%). Three patients were intubated due to the progression of respiratory failure and eventually died.
Before the COVID-19 pandemic, only a few studies reported using venovenous ECMO in awake, non-intubated patients with ARDS.16,17 During the COVID-19 pandemic, interest in this method renewed due to the large number of patients who presented with severe hypoxemia but were fully conscious, cooperative, and spontaneously breathing when the need to start mechanical support of the respiratory system became imperative. The effectiveness of the awake ECMO approach in patients with CARDS has been reported in the range of 22.2% to 57.1% in small-sample cohort studies published recently, suggesting unfavorable clinical outcomes for patients who were intubated after failing awake ECMO.18–21 In our sample, all three patients who had to be intubated due to respiratory causes died later from multiorgan failure. These data emphasize the crucial role of identifying patients who may benefit from the awake ECMO approach instead of intubation and mechanical ventilation.
Because IPPV increases the risk of barotrauma progression (including life-threatening tension pneumothorax), we indicated awake ECMO in six patients with apparent barotrauma. Still, no patient required chest tube insertion before ECMO started. Pneumothorax requiring chest tube insertion developed only in one patient with pre-ECMO massive subcutaneous emphysema (pre-ECMO CT not available) on day 4 of awake ECMO. The reported results are consistent with data from seven CARDS patients at high risk of barotrauma (defined by subtle collections of air contiguous to the bronchovascular sheath on lung parenchyma on chest computed tomography scans—Macklin lines) who underwent awake ECMO treatment without progression of pneumothorax or other types of barotrauma.21,22 These limited data suggest that keeping patients breathing spontaneously on ECMO support without the application of IPPV can be a feasible therapeutic strategy for treating patients at risk of life-threatening progression of barotrauma.
ECMO does not provide a direct therapeutic effect but rather represents organ support, allowing the lungs to heal. In mechanically ventilated patients, ultraprotective or near-apneic ventilation minimizes the risk of ventilation-induced lung injury during the ECMO run.14,23 In patients breathing spontaneously, maintaining arterial normoxia and normocapnia, and correcting respiratory acidosis decrease the chemosensory input of respiratory drive, resulting in reduced respiratory effort.3,24 However, non-responder patients may continue to exert vigorous respiratory effort, inducing excessive transpulmonary pressures and other mechanisms associated with patient self-inflicted lung injury (P-SILI), further aggravating existing lung injury.
The initiation of ECMO therapy was associated with a significant decrease in the mean RR in our patients. During awake ECMO, all patients were supported by HFNC support with a flow of 45–60 L/min, titrated to an RR of <20 breaths per minute, and a subjective relief of dyspnea. HFNC provides a flow-dependent improvement in lung mechanics and homogeneity, reducing RR and respiratory effort.25–27 Compared to noninvasive ventilation (NIV), HFNC reduces the risk of barotrauma in patients with COVID-19-related acute respiratory failure. 28 Nevertheless, despite the maximal support provided by ECMO and HFNC, a significant increase in RR and worsening respiratory mechanics leading to exhaustion occurred in three patients in our group. After the intubation of all these patients, catastrophic respiratory system compliance was apparent, suggesting that maintaining arterial normoxia and normocapnia with ECMO and HFNC support was insufficient to prevent P-SILI completely. Bronchoscopy was performed immediately after intubation in all cases, and blood clots were found in two patients who had a history of hemoptysis before being connected to ECMO. However, evacuating the clots from bronchial structures did not significantly improve respiratory system compliance. The “solid-like” structure of lung tissue apparent after intubation calls for identifying early parameters that define the situation when spontaneous breathing turns into deleterious P-SILI.
Due to the low number of patients, our study cannot aim to identify parameters that define the situation when spontaneous breathing induces deleterious P-SILI, and no robust evidence is available in this field. Scoring systems based on simple noninvasive signs of excessive respiratory effort (such as RR, nasal flaring, sternocleidomastoid muscle phasic activity, and abdominal muscle use), and the ratio of oxygen saturation to fraction of inspired oxygen have been shown to be predictive of the necessity for intubation in patients with respiratory failure.29–31 Of semi-invasive and minimally invasive methods, direct monitoring of esophageal pressure and nasal pressure swings were shown to correlate with transpulmonary pressure and respiratory effort in awake ARDS patients.32,33 However, there is a lack of evidence regarding the applicability of the parameters mentioned above in patients treated with the awake ECMO strategy.
Pain, anxiety, and delirium are frequent in ARDS patients, and all can stimulate respiratory drive through multiple physiologic mechanisms, while the use of sedatives and opioids might decrease neural respiratory drive. 3 Moreover, hyperactive delirium in patients on ECMO increases the risk of catastrophic, life-threatening complications such as decannulation, cannula malposition, and bleeding from the site of cannula insertion. The reported prevalence of delirium among hospitalized adults with COVID-19 ranges from 10.17% to 80.22%, with illness severity necessitating ICU admission showing a strong association with delirium. 34 In a study of 18 adult patients treated with awake ECMO due to CARDS, the authors reported a high number (14/18, 78%) of patients requiring intubation. Patients in their sample were initially devoid of sedatives, with delirium and the patients’ explicit wish to be sedated being the main reasons for switching from awake ECMO to intubation and mechanical ventilation. 20
In our sample, all patients received sedatives (a combination of opioids and dexmedetomidine) from the beginning of ECMO to relieve the stress and anxiety caused by the ICU environment and concerns about their health conditions, and to prevent delirium-induced exaggerated movements. Sedation was targeted to simultaneously achieve a RASS score from 0 to −2 (to prevent delirium and induce sleep at night) and an RR of <20 breaths per minute (to reduce respiratory effort). Awake ECMO was generally well-tolerated; patients reported a subjective improvement and relief of dyspnea after the initiation of the procedure. Patients were able to eat a regular diet and communicate with staff and relatives. All patients underwent early (<48 h) active physiotherapy, including ambulation while on ECMO for three patients. Three patients developed delirium (one episode in each; that is, three episodes of delirium occurred throughout the 5580 h of awake ECMO). All episodes were classified as a hyperactive form requiring additional sedation to prevent impending life-threatening complications (such as cannula malposition or decannulation) caused by undesirable motor activity. Two of the patients who developed delirium were later intubated due to respiratory causes and died from multiorgan failure. It remains unclear whether delirium was an early symptom arising from the progression of lung injury or an independent condition not associated with worsening respiratory function.
ECMO cannula malposition and decannulation are catastrophic complications that can lead to immediate death from exsanguination or cessation of blood flow through the circuit, and the duration of ECMO support is a factor associated with life-threatening mechanical complications.35,36 Despite the long duration of awake ECMO in our patients (accompanied by spontaneous movement in bed and daily physiotherapy, including ambulation in three patients), no decannulation or malposition events were recorded. Alongside ultrasound-guided cannulation, appropriate cannula fixation, and regular checks on the insertion site several times a day, we strictly maintain a nurse-to-patient ratio of 1:1 in awake ECMO patients to detect subtle changes in mood and undesirable motor activity.
COVID-19-associated coagulopathy is characterized by complex interactions between the innate immune response, the coagulation/fibrinolytic pathways, and the activated vascular endothelium. 37 The application of VV ECMO introduces an additional interaction between the already impaired coagulation cascade and artificial materials, creating a “double hit” scenario that further increases the risk of catastrophic thrombotic and bleeding complications. In a meta-analysis of 42 studies that included 2037 COVID-19 patients supported with VV ECMO, the pooled incidence rate of bleeding requiring transfusion was 39%. 38 A relatively high number of thrombotic and bleeding events was also reported in a meta-analysis of 23 studies comprising 6878 COVID-19-related ARDS subjects requiring ECMO treatment. The overall pooled estimate for circuit thrombosis was 21.5%, and 37.4% of the patients experienced significant bleeding, respectively. 39 Recently published data on 25 CARDS patients treated with the awake ECMO approach reported the incidence of bleeding and thrombosis in 52% of all initially awake ECMO patients. 19 In our sample, clinically significant bleeding developed in two patients (one episode in each), and no thrombotic events occurred. Considering the duration that the patients were on ECMO and taking into account that they were moving spontaneously and performing physiotherapy daily, the incidence of severe thrombotic and bleeding adverse events is considered low. Importantly, in the patient with cerebral hemorrhage, the event was detected without delay, as the patient was on awake ECMO, and any changes in consciousness were immediately apparent. The anticoagulation protocol managed by argatroban targeted directly to anti-II activity in the range of 0.4–0.6 µg/mL (aPTT was measured alongside anti-II activity for control and comparison) while simultaneously maintaining fibrinogen levels > 2 g/L and platelet counts above 100,000/µL provided reasonable safety in the prevention of severe thrombosis or significant bleeding.
The presented paper has several limitations. As all patients treated using this approach (i.e., the entire available population) were included retrospectively and no comparison to another group was performed, power analysis was not applicable to this study. Considering the low number of patients and the retrospective monocentric design, this study provides more hypotheses and subtle signals than robust evidence in the field of the awake ECMO approach. All procedures and interventions were based on clinical conditions and local standards of care. Pre-ECMO CT scans were available for only four patients, and peripheral oxygen saturation was used to define hypoxemia instead of arterial partial pressure of oxygen. Moreover, as all patients in our group suffered from CARDS, the applicability of the presented data to patients with ARDS from other causes must be verified in future studies. Further research should also focus on identifying patients who may benefit most from the awake ECMO approach, defining the clinical criteria associated with deleterious respiratory effort, and objectively determining the efficacy of awake ECMO in comparison with traditional ECMO strategies (i.e., in intubated patients) for ARDS patients under mechanical ventilation. Despite the shortcomings mentioned above, the presented data illustrate real-life clinical scenarios and could be helpful in decision-making when invasive mechanical support for respiratory function is urgently required.
Conclusion
The high risk of barotrauma-related life-threatening complications, as well as the patient’s preference not to be sedated and mechanically ventilated, constitute the main clinical reasons for the awake ECMO approach in ARDS patients. According to our data, a strategy based on ultrasound-guided cannulation, argatroban-based anticoagulation, respiratory support, and routine sedation targeted at both reducing respiratory effort and preventing delirium and maintaining a nurse-to-patient ratio of 1:1 appears to be a safe approach for treating CARDS patients who are awake and cooperative at the time when the need for mechanical support of the respiratory system becomes imperative. Although the effectiveness of the awake ECMO strategy in comparison to conventional methods needs to be evaluated in future studies, the outcomes of our study (seven out of 10 CARDS patients treated using awake VV ECMO were successfully weaned from ECMO) appear promising.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241282590 – Supplemental material for Optimizing the safety and efficacy of the awake venovenous extracorporeal membrane oxygenation in patients with COVID-19-related ARDS
Supplemental material, sj-docx-1-tar-10.1177_17534666241282590 for Optimizing the safety and efficacy of the awake venovenous extracorporeal membrane oxygenation in patients with COVID-19-related ARDS by Peter Sklienka, Filip Burša, Michal Frelich, Jan Máca, Vojtech Vodička, Hana Straková, Markéta Bílená, Tereza Romanová and Hana Tomášková in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
