Abstract
Background
Lymphopenia is a marker of immunosuppression after severe coronavirus disease-2019 (COVID-19) which is characterized by acute respiratory distress syndrome (ARDS). This study aimed to evaluate the relationships between persistent lymphopenia and ARDS.
Methods
A retrospective cohort study of 125 patients with COVID-19 admitted to government-designated treatment center between 14 January 2020, and 20 March 2020 was conducted. We recorded all complete blood cell counts during the day 0th, 3rd, and 7th following the diagnosis of COVID-19. Patients were grouped based on the depression of the lymphocyte cell count, their return, or their failure to normal. The primary outcome was the occurrence of ARDS, and secondary outcomes included developing vital organ dysfunction and hospital lengths of stay.
Results
17.6% (22/125) patients developed ARDS. The lymphocyte counts with ARDS and non-ARDS were 0.94 × 109/L, 1.20 × 109/L at admission, respectively (p = 0.02). On the 3rd and 7th day, the median of lymphocyte count in ARDS was significantly lower than that of non-ARDS. Multivariable logistic regression, which was adjusting for potentially confounding factors (including age, comorbidities, and APACHE II score), showed that persistent lymphopenia within the 7th day was independently associated with ARDS (OR, 3.94 [95% CI, 1.26–12.33, p = 0.018). Further, patients with persistent lymphopenia had longer hospital lengths of stay (p < 0.001).
Conclusion
The results showed persistent lymphopenia predicted ARDS after COVID-19. Further studies are needed to investigate whether immunostimulation of lymphocytes within 1 week can reduce ARDS occurrence in patients with COVID-19.
Introduction
Coronavirus disease-2019 (COVID-19) is a systemic infectious disease mainly caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). SARS-CoV-2 can enter host cells through angiotensin-converting enzyme 2 (ACE2) mediated SARS-CoV-2 and lead the host severe inflammatory response. 1 Immune and inflammation are critically involved in the process of SARS-CoV-2 infection. In many cases, dysregulated host response results in prolonged immunosuppression periods with sepsis.2,3 In previous reports, the mortality was 62% among critically ill patients with COVID-19 and 81% among those requiring mechanical ventilation in Wuhan. 4 Dysregulation of innate and adaptive immunity induced by COVID-19 can prevent pathogen clearance and predispose to secondary infections, evidenced by autopsy studies that had demonstrated ongoing foci of infections and functional defects in immune effector cells in patients who have died of COVID-19. 5 COVID-19 damages not only the lungs but also immune systems. Immune dysfunction is a primary cause of late mortality in sepsis.6,7
Clinical studies have demonstrated that prolonged lymphopenia is a candidate marker of persistent immunosuppression in patients with sepsis. 8 Wu et al. 9 also reported that lymphocyte counts at admission were associated with the development of acute respiratory distress syndrome (ARDS) (HR0.37, p < 0.001), as they did not dynamically observe the relationship between lymphocyte counts and ARDS, and the relationship between them is not fully elucidated. Absolute lymphocyte counts are easily measured during routine care. Therefore, we aimed to identify the pattern of lymphocyte response to COVID-19 and determine whether persistent lymphopenia after admission was associated with ARDS development, and further providing clinical evidence for the timing of the immunoregulation in patients with COVID-19.
Materials and methods
Study design, setting and participants
This retrospective cohort study was designed by the investigators and reported its results following the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines and performed at the Third People’s Hospital (municipal designated hospital for COVID-19) of Shenzhen between 14 January 2020, and 10 March 2020. The data cutoff for the study was 25 March 2020. All patients with COVID-19 were screened. It was estimated that the incidence of ARDS in patients with or without lymphopenia were 40% and 10%, respectively, based on literature. 9 Let α = 0.05 and β = 0.2, at least 58 cases were needed by PASS software. Finally, data were obtained from 149 patients with COVID-19 hospitalized at the Department of Critical Care Medicine and Infection Third Ward during the study dates. A confirmed case of COVID-19 was defined as a positive result on real-time reverse-transcriptase–polymerase-chain-reaction (RT-PCR) assay of pharyngeal swab specimens by the Shenzhen center for disease prevention and control (CDC). The discharge criterion was negative two times 24-h interval result on RT-PCR assay of pharyngeal swab specimens by Shenzhen CDC. The study analyzed de-identified data from the hospital’s healthcare informatics group, supervised by the Shenzhen Municipal Health Commission. The study protocol was approved by the Second People’s Hospital of Shenzhen and First Affiliated Hospital of Shenzhen University (institutional review board number 202003009004).
Exclusion criteria included without lymphocyte count for three consecutive days on day 0, day 3, and day 7 and without lymphocyte count for two consecutive days on day 3 and day 7.
Data collection and definitions
Patient identification—a confirmed case of COVID-19 was defined as a positive result on real-time RT-PCR assay of pharyngeal swab specimens by Shenzhen center for disease control and prevention (CDC) during the study period. The discharge criterion was negative two times 24-h interval result on RT-PCR assay of pharyngeal swab specimens by Shenzhen CDC.
Leukocyte counts—The data for blood tests collected from the labs were drawn as a standard of care. Every complete blood cell count (CBC) (including the white blood cell count (WBC), neutrophil count, lymphocyte count, and monocyte count) that was obtained for day 0, day 3, day 7, and day 14 from admission on these patients was extracted from the medical record. If multiple leukocyte counts were collected within any 24-h period, the nadir value was documented for that period.
Definitions—Lymphopenia was defined as an absolute lymphocyte count less than 1.0 × 109/L, which is lower than the limit of normal (1.1 × 109/L) at our institution. The absolute lymphocyte count was segregated into three groups. One consisted of patients who did not develop lymphopenia within the first 7 days after admission. Another consisted of patients who developed lymphopenia, which never returned to normal within the first 7 days after admission. The other is to develop a decreased lymphocyte count which returns to normal within the first 7 days after admission. Persistent lymphopenia was defined as whose lymphocyte count did not return to normal within the first 7 days after admission. Acute respiratory distress syndrome was defined according to the Berlin Definition. 10 Acute kidney injury (AKI) was defined based on Kidney Disease: Improving Global Outcomes Clinical Practice Guidelines (KDIGO). 11 Acute cardiac dysfunction was defined as the clinical syndrome characterized by typical symptoms that may be accompanied by signs or elevated biomarkers of myocardial injury. Acute hepatic injury was defined as a state in which the patient’s blood laboratory results met at least one of three criteria: serum total bilirubin (TBil) of 3.0 mg/dL or greater; aspartate aminotransferase (AST) of 41 IU/L or greater; and alanine aminotransferase (ALT) of 41 IU/L or greater; the patients who met none of these criteria were classified as the “normal liver function” group. 12 Hubei exposure was defined as having been to Hubei province, China, in the past 14 days, including short stays.
Baseline characteristics—Detailed chart review was then performed to exclude patients based on inclusion and exclusion criteria and collect demographic and outcome data. For every patient, all routinely collected vital signs and symptoms and laboratory values were extracted from the electronic health records. Data included, but were not limited to, demographic data (e.g., age, gender, and body mass index (BMI)), biochemical parameters (e.g., blood cell count, liver function, kidney function, coagulation function, and blood gas analysis), mechanic ventilation, and comorbidities including hypertension, diabetes, cerebrovascular disease, chronic obstructive pulmonary disease (COPD), and malignancy for the severity of illness. We calculated the Acute Physiology and Chronic Health Evaluation (APACHE) II score 13 within the first 24 h of hospitalization. The study analyzed de-identified data from the hospital’s healthcare informatics group, which was supervised by the Shenzhen Municipal Health Commission.
Main measures and outcomes
The primary outcome was ARDS, and the secondary outcomes included the development of vital organ dysfunction and hospital lengths of stay.
Statistical Analysis
The categorical data were summarized as numbers and percentages, and inter-group comparisons were performed using χ2 tests or Fisher’s exact test. Continuous variables were expressed as the arithmetic mean and standard deviation (SD) or as the median and interquartile range, depending on whether or not they showed a Gaussian distribution. Continuous data with Gaussian distribution were compared with the Student’s t-test or one-way ANOVA and those with a non-Gaussian distribution, with the Wilcoxon rank-sum test. To determine the independent effect of persistent lymphopenia on ARDS after accounting for significant confounders, multivariable logistic regression with a forced entry method was used with crude model and fully adjusted model: OR (odds ratio) and 95% confidence interval levels (95% CI). The predictive ability of lymphocyte count on day 0, day 3, and day 7 were assessed using the area under the receiver operating characteristic (AU-ROC) curve method. Youden’s index determined the optimal cutoff value. Statistical analysis was performed using the statistical package SAS 9.4 (Windows, SAS Institute, Cary, North Carolina) and the R software (version 3.6.2). p values (two-tailed) below 0.05 were considered statistically significant.
Results
Demographics and baseline characteristics associated COVID-19
Baseline characteristics of ARDS- and non-ARDS-associated COVID-19.

BMI: body mass index; bpm: beats per minute; IQR: interquartile range; COPD: chronic obstructive pulmonary disease; PCT: procalcitonin; CRP: C-reaction protein; WBC: white blood cell; RBC: red blood cell; HGB: hemoglobin; NLR: neutrophil lymphocyte ratio; PLR: platelets lymphocyte ratio; LMR: lymphocyte monocytes ratio; TBIL: total bilirubin; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BUN: blood urea nitrogen; PT: prothrombin time; APTT: activated partial thromboplastin time; FIB: fibrinogen; APACHE II: Acute Physiology and Chronic Health Evaluation.
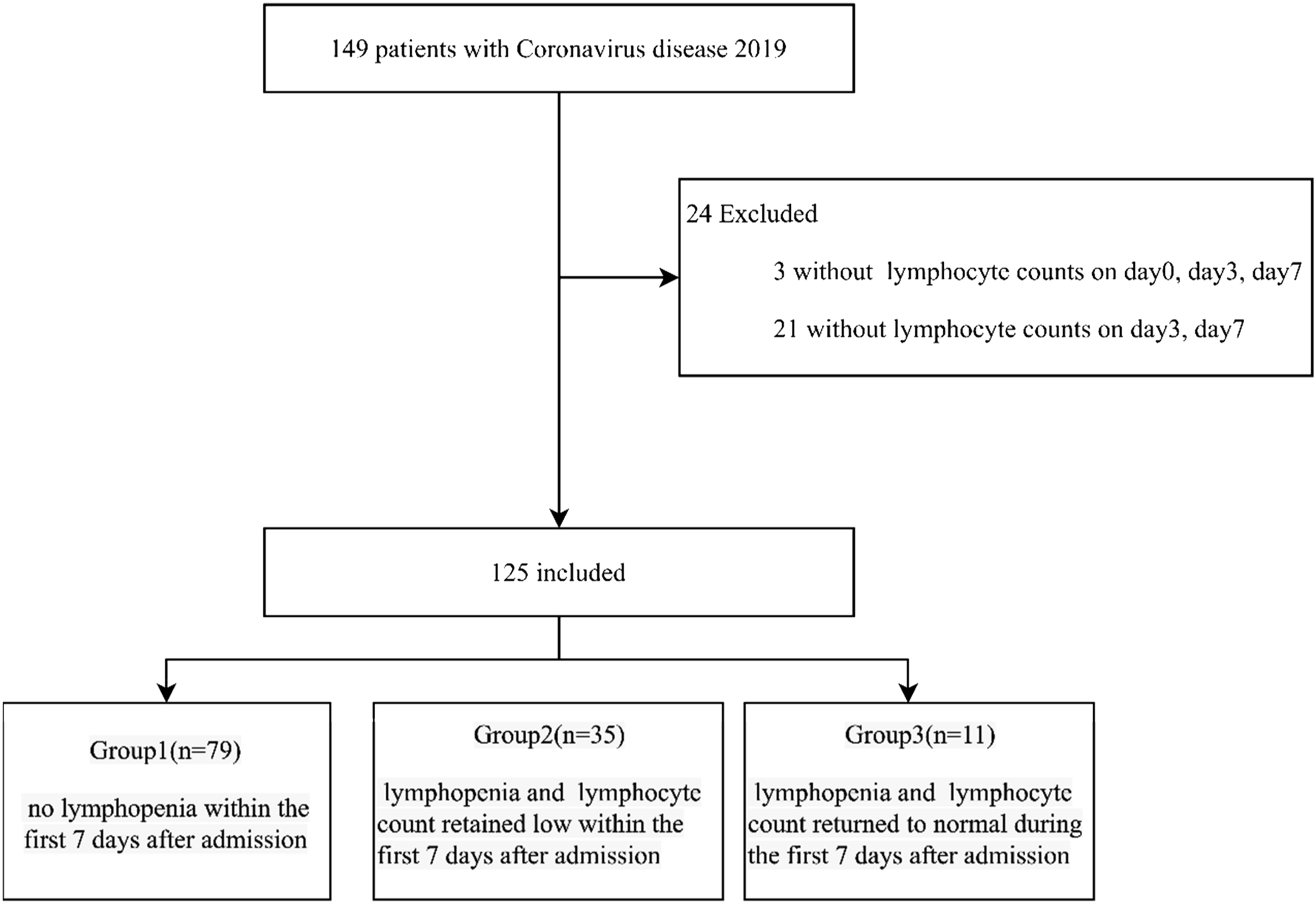
Flow diagram of study subjects.
Characteristics and outcomes of patients stratified lymphocyte count within the first 7 days
Characteristics and outcomes of patients stratified lymphocyte count within the first 7 days after admission.

bpm: beats per minute; IQR: interquartile range; COPD: chronic obstructive pulmonary disease; APACHE II: Acute Physiology and Chronic Health Evaluation; ICU: intensive care unit; ARDS: acute respiratory distress syndrome; AKI: acute kidney injury; AHI: acute hepatic injury; ACD: acute cardiac dysfunction; ICU: intensive care unit; LOS: length of stay; IQR: interquartile range.
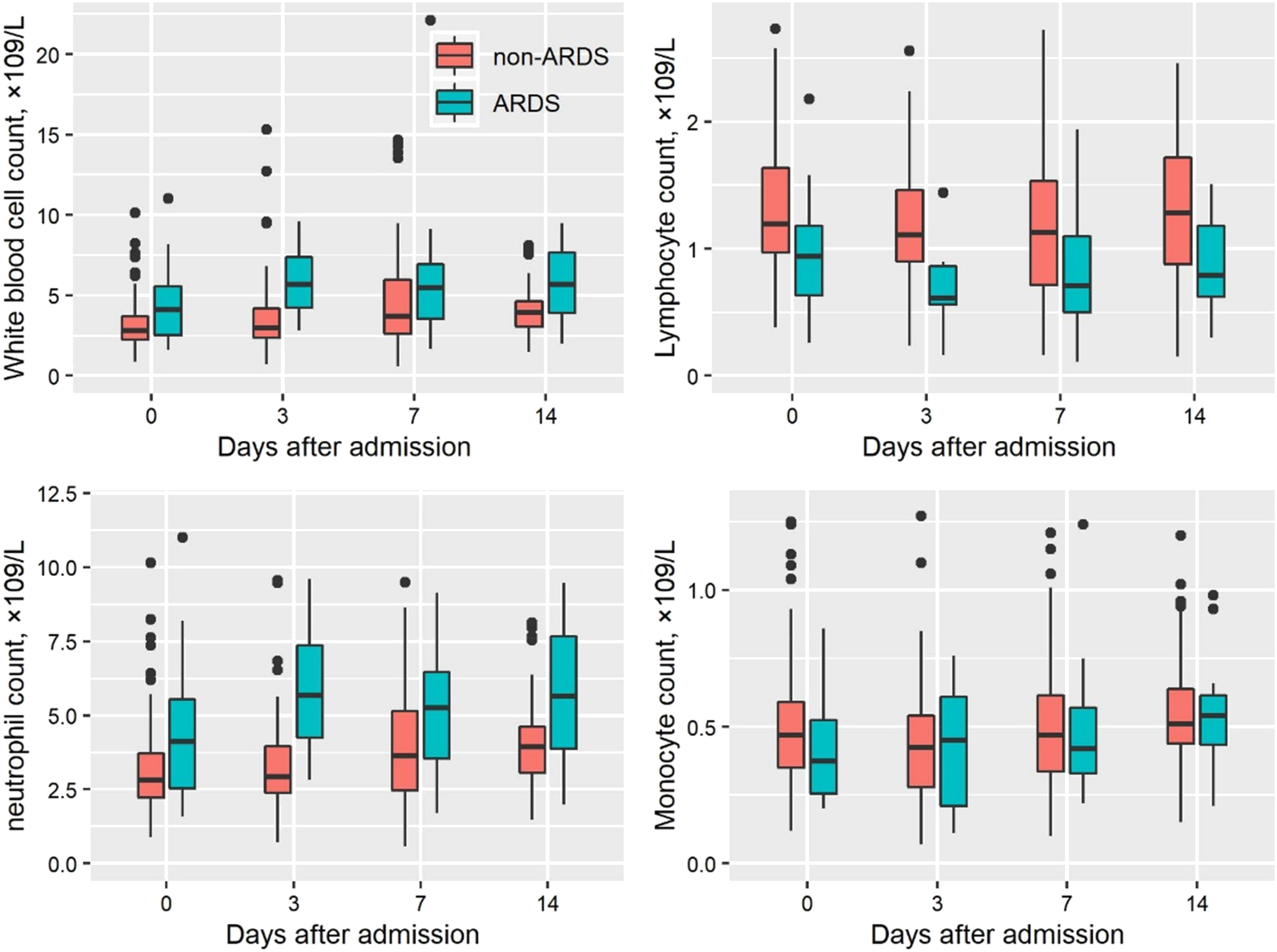
Box plots of leukocyte counts in ARDS and non-ARDS during the first 7 days following admission. ARDS: acute respiratory distress syndrome.
Clinical features and area under the curve of lymphopenia associated ARDS with COVID-19
Multivariable analysis of clinical features associated with ARDS.

APACHE II: Acute Physiology and Chronic Health Evaluation.
Cutoff and ROC curve of lymphocyte count predicting ARDS with COVID-19.

AUC: area under the curve: ARDS: acute respiratory distress syndrome.

Cutoff and ROC curve of lymphocyte count predicting ARDS with COVID-19. ARDS: acute respiratory distress syndrome.
Discussion
Severe COVID-19 is a life-threatening multi-organ functional injury, which was caused by a dysregulated host response to SARS-CoV-2 and characterized refractory hypoxemia by ARDS. Although some drugs or ways may be useful in treating COVID-19,14,15 human immune factors also play a significant role. Early evaluation of the patient’s immune function is essential for immune-stimulating therapy of severe COVID-19. Information regarding the immune phenotype in COVID-19 patients is vital before any consideration of immune-stimulating interventions occurs. The current study provides new findings in this field and offers potential insight into the areas for further research.
The phenomenon of lymphocyte depletion observed as evidenced by autopsy studies that had demonstrated ongoing fewer bone marrow lymphocytes, smaller splenic lymph nodes, and functional defects in immune effector cells in patients who have died of COVID-19. 5 Many patients with COVID-19 develop persistent immunosuppression before death. 4 In the current study, no one patient died before the 7 days after confirmed COVID-19, and 17.6% (21/125) patients had been confirmed ARDS. This study demonstrated that a persistently low level of lymphocytes on the seventh day following confirmed COVID-19 independently predicts ARDS and may serve as a biomarker for COVID-19-induced immunosuppression.
Similar to the prior study,16,17 absolute lymphocyte counts are easily measured in most clinical laboratories and are already frequently obtained in COVID-19 patients. A previous study had demonstrated persistently decreased levels of absolute lymphocyte count with non-survivors, while survivors experience lymphocyte recovery with sepsis. 18 They theorized that lymphocytes recruited and lymphocyte apoptosis with the development of sepsis. 18 It is a routine test that could be used both clinically and in future trials of COVID-19 therapies to identify the patients at highest risk for immunosuppression.
For this study, we focused on day seven absolute lymphocyte counts based on the mean hospital lengths of stay about 2 weeks, indicating that lymphocyte counts on this day would be a therapeutic window watershed. For those who were discharged, the time from onset to ARDS was 8.0 days. 19 The time point at which patients with COVID-19 is likely extremely variable because of pathogen and host immune. Therefore, we also analyzed days 0, 3, and 7 lymphocyte counts in the subset of patients who had complete blood counts measured to determine whether persistent lymphopenia at an earlier time point could also predict ARDS. We found that day 0, 3, and 7 absolute lymphocyte counts were associated with ARDS after accounting for other covariates, and the AUC were 0.72, 0.82, and 0.70, and the cutoff were 1.2, 0.9, and 0.94 (×109/L) with a sensitivity of 86%, 88%, and 71% and specificity 49%, 74%, and 62%, respectively. Our previous study showed that treatment with thymosin α1 (an immunomodulator) can markedly attenuate acute lung injury or ARDS in critical type COVID-19 patients. 20 Therefore, immune-stimulating therapy was administrated third day at the earliest and seventh day at the latest. Despite this, determining the time at which continued lymphopenia clinically requires consideration of the effects of drugs, such as ribavirin and corticosteroids et al.
Lymphopenia was defined as an absolute lymphocyte count of less than 1.0 × 109/L. Younger adults with persistent unexplained moderate to severe lymphopenia should consider infection, especially viral (including influenza, HIV, and hepatitis) and in those with unexplained moderate to severe lymphopenia (lymphocyte count <1 × 109/L) offer HIV testing. 21 The lymphopenia was also conformed in non-survivors with COVID-19, and the lymphocyte counts continued to decrease until death occurred. 18 Similarly, our study showed severe COVID-19 patients had lymphopenia. Lymphoproliferative disorders can also be associated with the reduction of immunoglobulin levels. 21 Administrated a specific isotype (such as IgG, IgA, and IgM) may help patients with COVID-19 patients. The research showed that early administration (admission ≤7 days) of intravenous immunoglobulin with high dose improves the prognosis of critical type patients with COVID-19. 22 In the future, larger studies stratifying patients by age, comorbidities, or APACHE II score may clarify whether different absolute lymphocyte cutoff values would be more useful in specific subpopulations of patients with COVID-19. Multiple logistic regression results showed that only persistent lymphopenia (OR 3.94, 95% CI 1.26–12.33, p = 0.018) was obviously associated with ARDS induced by COVID-19, indicating that the proportion of ARDS with persistent lymphopenia (<1 × 109/L) was 3.94 times that of non-persistent lymphopenia induced by COVID-19. Therefore, using persistent lymphopenia can identify potential ARDS patients with COVID-19.
Limitations
This study also has limitations. Twenty-four patients were not given blood tests because early symptoms were not obvious, resulting in missing data, and were excluded. Besides, the study does not allow us to conclude whether persistent lymphopenia directly contributes to ARDS induced by COVID-19 or whether it is simply a biomarker of immunosuppression or immunodepletion. We are also not able to presume that immune-stimulatory therapy aimed at reversing lymphopenia would reduce the incidence of ARDS.
Conclusion
In summary, persistent lymphopenia for 1 week predicts ARDS associated with COVID-19. Immunostimulation of lymphocytes within 1 week in patients may be the key target for reducing ARDS occurrence in patients with COVID-19.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part, by grants from Sanming Project of Medicine in Shenzhen (SZSM20162011). Shenzhen Science and Technology Innovation Commission (No. JCYJ20170306091335008, JCYJ20190806163603504). And Clinical Research Project of Health and Family Planning Commission of Shenzhen Municipality (SZLY2017007).
Ethical approval
The Research Ethics Committee of the Second People’s Hospital of Shenzhen and First Affiliated Hospital of Shenzhen University approved the study protocol (institutional review board number 202003009004).
Informed consent
Written informed consent was waived by the Ethics Commission.
