Abstract
Background:
Acute respiratory distress syndrome (ARDS) is a severe complication among patients with severe acute pancreatitis (SAP), which may be associated with increased mortality in hospitalized patients. Thus, an effective model to predict ARDS in patients with SAP is urgently required.
Methods:
We retrospectively analyzed the data from the patients with SAP who recruited in Xiangya Hospital between April 2017 and May 2021. Patients meeting the Berlin definition of ARDS were categorized into the ARDS group. Logistic regression models and a nomogram were utilized in the study. Descriptive statistics, logistic regression models, and a nomogram were used in the current study.
Results:
Comorbidity of ARDS occurred in 109 (46.58%) of 234 patients with SAP. The SAP patients with ARDS group had a higher 60-day mortality rate, an increased demand for invasive mechanical ventilation, and a longer intensive care unit (ICU) stay than those without ARDS (
Conclusion:
The prediction nomogram for ARDS in patients with SAP can be applied using clinical common variables after the diagnosis of SAP. Future studies would be warranted to verify the potential clinical benefits of this model.
Introduction
Acute pancreatitis (AP) is an inflammatory disease of pancreas with varying severity and progression. 1 The global incidence rate of AP is 33.74 per 100,000 person-years, and morbidity is rising gradually. 2 Around 10–20% of patients with AP have a complicated systemic inflammatory response syndrome (SIRS), and multiple organ dysfunction syndrome that can lead to the development of severe AP (SAP) with a 10–15% mortality rate. 3
Acute respiratory distress syndrome (ARDS) is the most prevalent form of organ failure in patients with SAP and remains a major cause of high in-hospital mortality. 4 Moreover, patients with SAP-associated ARDS are typically admitted to the intensive care unit (ICU) for an extended period, thereby increasing healthcare costs. Although an increasing number of studies have identified multiple possible causes of SAP-associated ARDS, including pancreatic necrosis, bacteremia, intestinal barrier failure, activation of inflammatory cascades, and diffuse alveolar damage, the pathological mechanisms are largely unknown. 5 Undoubtedly, the treatment of SAP-associated ARDS is extremely challenging. 6 It is a certainty, however, that appropriate intervention(s) in the early phase of ARDS could help improve clinical prognosis. A prediction model for SAP-associated ARDS using baseline clinical characteristics would aid in the early identification and classification of patients at high risk for SAP-associated ARDS and may provide an opportunity for early therapeutic intervention(s) prior to deteriorating development.
However, several scoring systems, such as the Systemic Inflammatory Response Syndrome (SIRS) score, Bedside Index of Severity in Acute Pancreatitis (BISAP), Modified Marshall (MMF) scoring system, and Sequential Organ Failure Assessment (SOFA) score, have good predictive capabilities for disease severity (mild, moderately severe, and severe according to the revised Atlanta classification) and mortality; however, few, if any, tools work well in predicting ARDS in SAP patients using the available data. As such, through multivariable logistic regression analysis and the development of a nomogram model, we aimed to construct an efficient ARDS prediction model using baseline clinical characteristics that could help to screen for patients who are likely to develop ARDS among those with SAP.
Methods
Study design and participants
We retrospectively collected and analyzed the data from doctor-diagnosed SAP admitted to Department of Biliary-Pancreatic Surgery and Department of Gastroenterology and ICU in Xiangya Hospital of Central South University (Changsha, China) between April 2017 and May 2021. Information of the patients and clinical data were obtained from the institutional electronic medical records by three independent investigators using a spreadsheet (Excel, Microsoft Corporation, Redmond, WA, USA), followed by data anonymization. The study protocol was approved by the Ethics Commission of Xiangya Hospital of Central South University (No. 201912477), and requirements for written informed consent were waived because patient data and analysis were anonymized.
Inclusion criteria were as follows: at least 18 years of age and diagnosed with SAP within 48 h of admission according to the Revised Atlanta classification; any organ dysfunction with ⩾grade 2 severity persisting for >48 h according to the modified Marshall grading in AP was considered SAP. 7 Individuals with a history of previous AP or chronic pancreatitis, unavailability of key data, hospital stay <48 h, malignant tumors, active tuberculosis, malignancy, and chronic obstructive pulmonary disease were excluded. Finally, 234 patients with SAP were included for analysis in the current study. In addition, if the included SAP patients met the Berlin definition for ARDS within 14 days after admission, they would be categorized into the ARDS group. 8
Measurements
Clinical information for each patient, including demographic data, preexisting condition(s), etiology, clinical signs, arterial blood gas analysis results, laboratory findings, scores (SIRS, BISAP, MMF, and SOFA), and outcome, were collected. The etiology of SAP was classified as ‘hypertriglyceridemia’, ‘biliary’, ‘alcohol’, or ‘mixed type’. The SIRS, BISAP, MMF, and SOFA scores were extracted from the admissions database. Clinical variables and outcomes including the incidence of ARDS, 14-day mortality, 30-day mortality, 60-day mortality, use of invasive mechanical ventilation, and ICU length of stay were evaluated among patients with SAP.
Statistical analysis
Data normality of the continuous variables was tested using the Shapiro–Wilk test. Normally distributed data are expressed as mean ± standard deviation, variables with non-normal distribution are reported as median [interquartile range (IQR)], and categorical variables are summarized as frequencies and percentages. Continuous data with normal distribution were compared using the Student’s
Results
Clinical characteristics
In total, 234 patients with SAP were included in the analysis: 109 (46.58%) with ARDS and 125 (53.42%) without (Figure 1). Demographic data, preexisting condition(s), clinical signs, arterial blood gas analysis results, laboratory findings, and scores on admission were compared between SAP patients with and without ARDS.

Flow diagram of the study population.
SAP patients with ARDS group had a higher heart rate and respiration rate, lower pH value, and lower PaO2:FiO2 ratio than those patients without ARDS (
Baseline characteristics of SAP patients with or without ARDS.

ARDS, acute respiratory distress syndrome; BISAP, bedside index of severity in acute pancreatitis; FiO2, fraction of inspired oxygen; MAP, mean artery pressure; MMF, modified Marshall scoring system; PaO2, partial pressure of oxygen; PCO2, partial pressure of carbon dioxide; PH, hydrogen ion concentration; SAP, severe acute pancreatitis; SIRS, systemic inflammatory response syndrome score; SOFA, Sequential Organ Failure Assessment score.
Data are presented as mean ± standard deviation, medians (interquartile range) and n (%).Values of
A
Clinical outcomes
In patients with SAP, the 60-day mortality was commonly used to assess the clinical outcome, and they were 37.62% and 6.40% among SAP patients with or without ARDS, respectively (

Kaplan–Meier curves of mortality for SAP patients with or without ARDS. Patients discharged home considered alive at 60 days. A log-rank test was used to evaluate differences between groups.
Model development
In the preliminary analysis, we revealed that higher heart rate, higher respiration rate, lower pH value, lower PaO2:FiO2 ratio, lower platelet count, and elevated LDH, elevated blood urea nitrogen, elevated creatinine, and elevated procalcitonin levels were potential predictors of ARDS development among patients with SAP using univariate analyses (
Logistic Regression Model for predicting ARDS in SAP patients.

ARDS, acute respiratory distress syndrome; FiO2, fraction of inspired oxygen; PH, hydrogen ion concentration; PaO2, Partial pressure of oxygen; SAP, severe acute pancreatitis.
A
To further confirm the role of the aforementioned covariates in the predictive ability of ARDS among patients with SAP, receiver operating characteristic (ROC) curve analysis was performed (Table 3, Figure 3(a)). The area under the ROC curve (AUROC) for model combination (PaO2:FiO2 < 200; platelets < 125 × 109/L; lactate dehydrogenase > 250 U/L; creatinine > 111 mg/dL; and procalcitonin > 0.5 ng/mL) was 0.814, respectively.
Receiver operating characteristic curves for predicting ARDS in SAP patients.
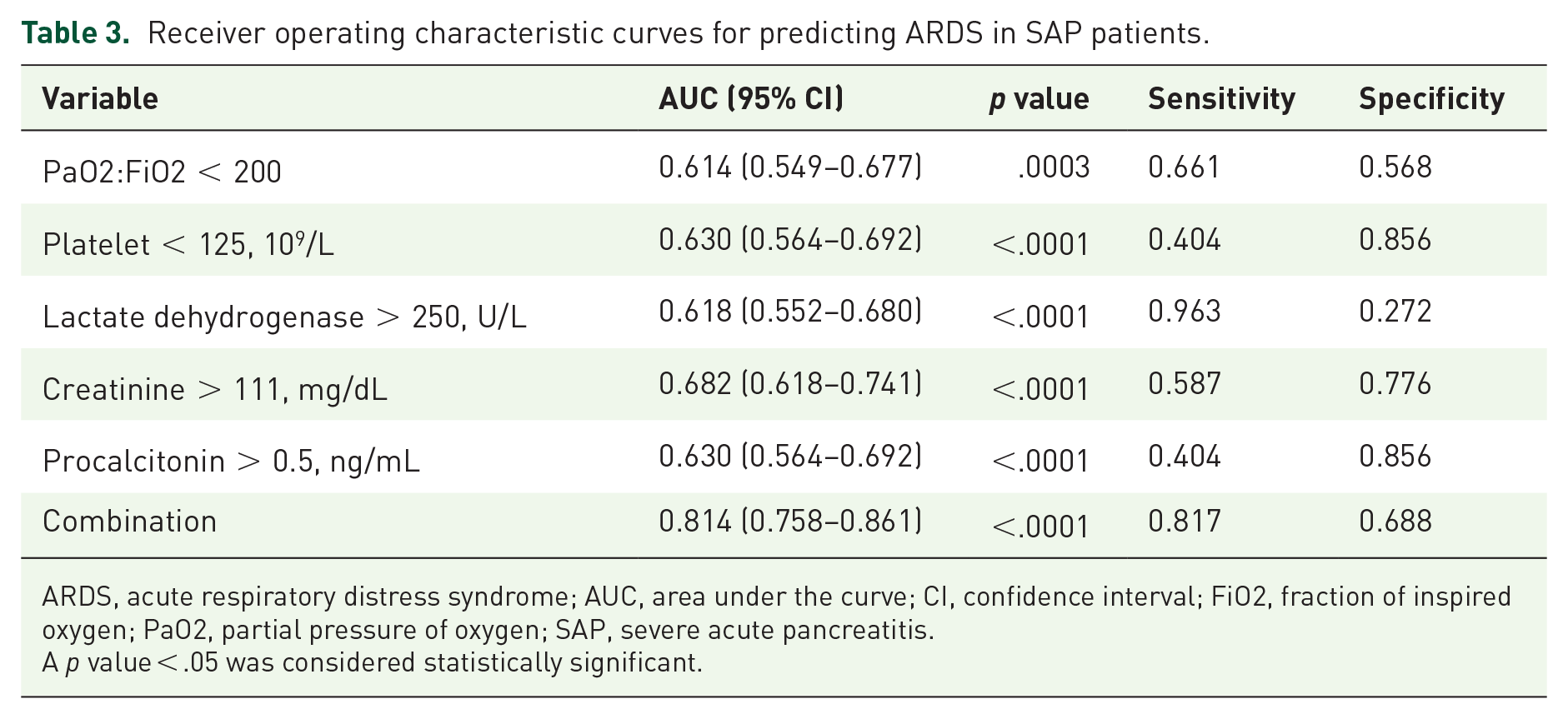
ARDS, acute respiratory distress syndrome; AUC, area under the curve; CI, confidence interval; FiO2, fraction of inspired oxygen; PaO2, partial pressure of oxygen; SAP, severe acute pancreatitis.
A

The ROC curves in prediction of ARDS in patients with SAP. (a) ROC curves of present prediction model. (b) ROC curves of SIRS, BISAP, MMF, SOFA, and nomogram.
Model visualization and performance
A nomogram incorporating the ARDS prediction model incorporating the above independent predictors was constructed (Figure 4(a)), yielding a value of 0.355, indicating that the model fit was acceptable. The median C-index for the prediction nomogram was 0.814 (0.758–0.861) for the clinical data set. The calibration curve demonstrated good agreement between the estimations with the nomogram for predicting the risk for developing ARDS in patients with SAP and actual observations (Figure 4(b)). Area under the curve (AUC) comparison was based on the DeLong method to compare the AUC of the prediction model with the AUC of other clinical scoring systems. As shown in Figure 3(b) and Table 4, the AUC of the prediction model was greater than those using variables including SIRS (Z statistic, 4.521;

(a) Developed ARDS prediction nomogram in patients with SAP. (b) Calibration curves of the ARDS nomogram prediction in the SAP trial.
Predictive performance of different models.

AUC, area under the curve; BISAP, bedside index of severity in acute pancreatitis; MMF, modified Marshall scoring system; SIRS, systemic inflammatory response syndrome score; SOFA, Sequential Organ Failure Assessment score.
A
Discussion
In the current study, we observed a significantly higher mortality in SAP patients with ARDS. In addition, lower PaO2:FiO2 ratio, platelet counts, higher lactate dehydrogenase, creatinine, and procalcitonin on admission had been found to be correlated with higher risk of developing ARDS in patients with SAP. Finally, the predictive model based on above crucial variables had high sensitivity and specificity to identify high-risk population for developing ARDS in patients with SAP, which would help take early interventions to prevent ARDS progression in SAP and improve clinical outcomes.
In our study, the occurrence of ARDS in SAP was 46.58%, which was between the 15% and 60% reported in prior studies.19–22 In pneumonia-caused ARDS, increasing age and comorbidities were associated with high risk of developing ARDS. Surprisingly, there were no statistically significant differences in age and comorbidities among SAP patients with or without ARDS in the present study, which was consistent with previous studies among patients with SAP23,24 and AP,16,25 indicating that age and comorbidities is not as much of a risk factor in SAP-related ARDS as in pneumonia-initiated ARDS. Although hypertriglyceridemia-induced AP has been reported to be associated with risk of lung failure compared with other etiologies,16,26 the etiologies of SAP have no significant differences between the groups with or without ARDS. Further studies on underlying mechanism would be warranted to investigate the associations between SAP etiologies and ARDS.
In addition, our findings demonstrated the 60-day mortality rate of 37.62% and 6.40% among SAP patients with or without ARDS, respectively, confirming the critical role of ARDS in SAP prognosis. In prior studies with ARDS, the mortality rate of ARDS commonly ranged from 36% to 50%,27–29 and the mortality rate of SAP-initiated ARDS in the study fell within this range. Although ARDS has been well known as a leading cause of mortality in SAP, identifying early risk factors of ARDS in SAP is challenging. Previous studies proposed the utility of lung ultrasonography and cytokines in predicting ARDS among patients with SAP; however, individual indicator had difficulty in reflecting whole situation of the body. Besides, lung ultrasonography has not been widely used in most hospitals, particularly in primary healthcare centers. To the best of our knowledge, very few studies focused on multiple indicators using prediction score and/or a nomogram model to predict ARDS development among patients with SAP.
Herein, we developed an effective nomogram model based on several clinical indicators to predict the development of ARDS in SAP patients at the early stage of the disease. These five indicators included in our model, respiratory indexes (PaO2:FiO2), blood routine (platelet counts), blood biochemical (creatinine and LDH), and inflammatory markers (procalcitonin), were clinically meaningful and could reflect pathophysiology of SAP-initiated ARDS from several aspects. The lower PaO2:FiO2 is an indicator for hypoxemic situation, the decrease of PaO2:FiO2 may indicate a forewarning stage of ARDS development initiated by SAP.8,30 In our study, the decreased platelets were found to be an independent risk of SAP-related ARDS, suggesting that platelet activation and consumption may be involved in pathogenesis of this process.31,32 Consistently, previous studies have found lower count of platelets was not only associated with increased risk for ARDS but also with poor prognosis and hemorrhage in AP patients with ARDS.13,33–35 Increased LDH and serum creatinine levels are common indicators of multiple organ injury,14,16,36 and increased procalcitonin is a marker of systematic bacterial infection, which suggest the development of multiple organ dysfunction including ARDS in SAP. 37 Combination of procalcitonin and LDH was applied in a diagnostic model for the disease severity of AP as well. 38 Furthermore, LDH, creatinine, and procalcitonin were reported to be independent factors associated with the severity of SAP, which had more likelihood of inducing the comorbidity of ARDS.14,16,39
Collectively, our findings demonstrate that combining above clinical variables including PaO2:FiO2, platelet count, LDH, creatinine, and procalcitonin levels have a better predictive effect in predict ARDS among SAP patients than BISAP, MMF, SIRS, and SOFA scores, which have been widely used to predict mortality and organ failure in patients with SAP,40–42 thus providing a more simple, quantitative and practical predicting tool for ARDS development in SAP patients. Furthermore, a visualized nomogram would help clinician had more ability to early identify subpopulation with high ARDS risk among individual patients with SAP.
Strengths and limitations
Given the strength that we develop a model of SAP-initiated ARDS prediction that can help to take early interventions to prevent ARDS progression in SAP and improve clinical outcomes, some limitations should not be ignored. First, it was a retrospective, single-center study; as such, it may have been susceptible to the inherent limitations of retrospective analyses. Second, even though our model was proved useful for decision-making to early predict ARDS in patients with SAP, scoring tools should not replace clinical judgment. Third, the sample size of the study was relatively small, thus a larger population-based cohort study would be warranted to verify our findings in the future.
Conclusion
Lower PaO2:FiO2 ratio and platelet counts, higher LDH, creatinine, and procalcitonin levels were identified as independent risk factors for the development of ARDS in patients with SAP, and an easy-to-use nomogram was developed to predict SAP-induced ARDS. If validated, our findings could aid in establishing the appropriate level of care and guiding anticipatory management based on the prediction of ARDS.
