Abstract
The development of acute respiratory distress syndrome (ARDS) carries significant risk of morbidity and mortality. To date, pharmacological therapy has been largely ineffective for patients with ARDS. We present our personal review aimed at outlining current and future directions for the pharmacological prevention of ARDS.
Several available risk-stratification or prediction score strategies for identification of patients at risk of ARDS have been reported. Although not ready for clinical everyday use, they are and will be instrumental in the ongoing and future trials of pharmacoprevention of ARDS.
Several systemic medications established the potential role in ARDS prevention based on the preclinical studies and observational data. Due to potential for systemic adverse effects to neutralize any pharmacological benefits of systemic therapy, inhaled medications appear particularly attractive candidates for ARDS prevention. This is because of their direct delivery to the site of proposed action (lungs), while the pulmonary epithelial surface is still functional.
We postulate that overall morbidity and mortality rates from ARDS in the future will be contingent upon decreasing the overall incidence of ARDS through effective identification of those at risk and early application of proven supportive care and pharmacological interventions.
Introduction
Acute respiratory distress syndrome is a clinicopathologic entity defined by acute onset, hypoxemia and characteristic chest radiographic findings [Ranieri et al. 2012]. Despite the heterogeneous array of systemic and pulmonary insults that can serve as triggers to this devastating condition, ARDS is considered a distinct condition on the basis of a final common pathway in pathogenesis [Matthay et al. 2012] and corresponding histopathologic changes [Thompson, 2014]. Despite decades of research in the treatment of ARDS, not a single effective pharmacological intervention emerged. Several common ARDS precipitants were elucidated, including ventilator-induced lung injury, septic shock, transfusion-related lung injury and ventilator-associated pneumonia (VAP) [Estenssoro et al. 2002], hence management of ARDS has focused on supportive care measures. These include improvements in recognition and timely management of severe sepsis and septic shock [Rivers et al. 2001; Yealy et al. 2014], lung protective ventilation strategies [Acute Respiratory Distress Syndrome Network, 2000], restrictive Packed Red Blood Cell (PRBC), platelet and resh Frozen Plasma (FFP) transfusion strategies [Gajic et al. 2007; Khan et al. 2007; Toy et al. 2012], the adoption of standardized VAP prevention ‘bundles’ [Rosenthal et al. 2012] and other institutional protocols such as the proposed Checklist for Lung Injury Prevention (CLIP, Table 1) [Kor et al. 2012]. With these improvements in the delivery of supportive care, rates of nosocomial ARDS have been on the decline [Li et al. 2011]. However, mortality associated with ARDS has remained at the similar unsettlingly high rate of around 40% for the past approximately 20 years [Phua et al. 2009]. These stagnant mortality data are certainly not reflective of a lack of interest in finding effective pharmacological approaches to the treatment of ARDS. The cavalcade of investigational therapies showing promise in preclinical trials failed to demonstrate any meaningful clinical efficacy [Cepkova and Matthay, 2006; Bosma et al. 2010] when applied after the development of fully established ARDS. As such, a paradigm shift is occurring in how we approach the burden of ARDS, away from treatment and towards prevention of the fully established syndrome [Ortiz-Diaz et al. 2013; Beitler et al. 2014]. Parallels can be drawn to the other critical illness syndromes, of which sepsis has been the most studied and with the best resulting improvement in outcomes following adoption of early identification and goal-directed treatment principles. Since most of the ARDS cases develop 2–5 days after hospital admission [Gajic et al. 2011] there is a window of opportunity where early identification of patients at risk and improvements in supportive care, coupled with effective pharmacological interventions could potentially prevent or ameliorate ARDS in patients at risk. Hence, the key question: can pharmacological intervention administered prior to the fully established ARDS prevent disease development and enhance outcomes in patients at risk? In order to answer this key question, we first have to find answers to the following: who are the patients at risk, when to apply the intervention, and what intervention? The first two questions were answered by the Lung Injury Prediction Study (LIPS), to a certain degree. The final and perhaps the most important subquestion remains: what are the effective interventions? In order to identify a safe pharmacological treatment as an effective intervention, the investigators ought to apply systematic translational research directed towards proven mechanistic pathways of ARDS pathophysiology.
Checklist for Lung Injury Prevention (CLIP).

ARDS, acute respiratory distress syndrome; FACCT, fluid and catheters treatment trial; PEEP, positive end expiratory pressure.
Preventive strategies
Who and when?
The first critical step in instituting an effective primary prevention program was to identify the population most likely to glean benefit from the prophylactic intervention. LIPS [Gajic et al. 2011] is currently the best available tool for risk stratification of patients presenting to the hospital without ARDS. To evaluate this prediction model, LIPS investigators prospectively enrolled 5584 patients admitted for sepsis, shock, pancreatitis, pneumonia, aspiration, high-risk trauma or high-risk surgery and included comorbid conditions such as alcohol abuse, diabetes mellitus, obesity, physical exam and laboratory findings (Table 2). Using a cutoff LIPS score of at least 4, the authors found an 18% positive predictive value and 97% negative predictive value with an overall incidence of acute lung injury (ALI) or ARDS of 6.8%. The patients destined to develop ARDS in the hospital did so most frequently beyond 48 h of hospitalization so any future intervention should be applied within the first 24 h after the admission to allow it sufficient time to exhibit its protective effect.
Lung Injury Prediction Score calculation.

Definition of abbreviations: BMI = body mass index; LIPS = Lung Injury Prediction Score; RR = respiratory rate; Spo2 = oxygen saturation by pulse oximetry.
Add 1.5 points if emergency surgery.
Only if sepsis.
A number of other scoring systems for risk stratification with regard to development of ARDS have been proposed. Secondary analysis of the LIPS data have revealed that oxygen saturation by pulse oximetry (SpO2)/ fraction of inspired oxygen (FiO2) ratio, a single (composite) variable from LIPS, is an independent risk factor for progression to ARDS [Festic et al. 2013a]. Levitt and colleagues reported an Early Acute Lung Injury score, based on the oxygenation impairment, radiographic abnormalities and presence of concomitant immunosuppression [Levitt et al. 2009]. This risk score may be more applicable to hospitalized patients already developing early lung injury. Specific to the postoperative population, the Surgical Lung Injury Prediction model (SLIP) and refined SLIP-2, have shown similar utility in predicting which surgical patients are at the highest risk for development of ARDS [Kor et al. 2011b, 2014]. While none of these prediction models appear ready for everyday use in clinical practice, they represent useful tools for enrollment in clinical trials investigating pharmacological strategies for ARDS prevention.
What interventions?
Key components in the common final pathogenic pathway to ARDS include dysregulated inflammation, maladaptive platelet activation, dysfunction of the coagulation cascade, endothelial and epithelial barrier dysfunction, and failure of T-cell/macrophage-mediated clearance of activated neutrophils and necrotic debris (Figure 1) [Matthay et al. 2012]. As we have begun to elucidate the individual components leading to the full-blown ARDS syndrome, several potential therapeutic targets have emerged (Table 3).

Interrupting the Progression to ARDS. ARDS, acute respiratory distress syndrome; RAAS, renin–angiotensin–aldosterone system; VAP, ventilator-associated pneumonia; PPAR, peroxisome proliferator-activated receptor.
Emerging therapies for the prevention of acute respiratory distress syndrome (ARDS) and previous studies.
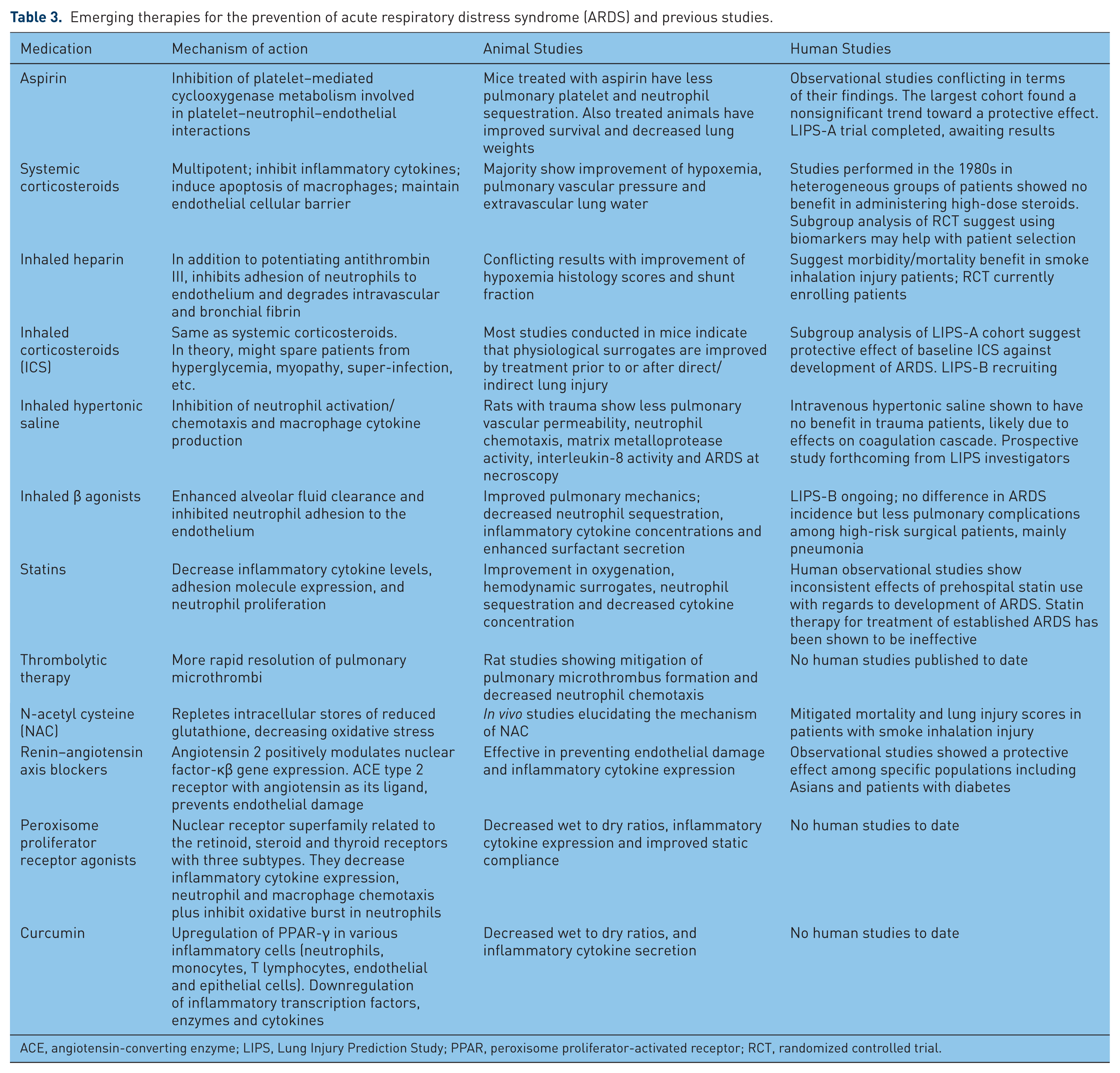
ACE, angiotensin-converting enzyme; LIPS, Lung Injury Prediction Study; PPAR, peroxisome proliferator-activated receptor; RCT, randomized controlled trial.
Systemic delivery
Aspirin
Several observations regarding the role of activated platelets as ‘instigators’ of endothelial dysfunction [Yiming et al. 2008] and neutrophil chemotaxis and degranulation [Zarbock and Ley, 2009] in animal models of ARDS have made aspirin an attractive potential agent. Furthermore, animal models specifically investigating the use of aspirin have been promising. In a ‘two-hit’ murine model of ARDS that included transfusion related ALI and endotoxinemia, control mice were compared with platelet-depleted and aspirin-treated mice [Looney et al. 2009]. In this study, the platelet-depleted and aspirin-treated mice had similarly significant improvements in progression to ARDS and in mortality versus controls. Song and colleagues demonstrated amelioration of the thromboxane-mediated pulmonary hemodynamic changes in aspirin-treated mice with endotoxin-induced ARDS [Song et al. 2004]. And in an acid-induced ‘aspiration’ murine ARDS model, Zarbock and colleagues demonstrated that aspirin decreased neutrophil chemotaxis across pulmonary endothelium, decreased the degree of pulmonary edema and improved oxygenation in mice [Zarbock et al. 2006].
The results of two observational studies in humans have thus far shown conflicting results with a possible trend towards a benefit of aspirin therapy for the prevention of ARDS. The charts of patients admitted to the medical intensive care unit (ICU) at the Mayo Clinic in Rochester, Minnesota, USA in 2006 with at least one known risk factor for ALI (ARDS) were reviewed. Prehospital use of antiplatelet medication was found in this series to be associated with a lower hazard ratio of ARDS development [hazard ratio 0.34, 95% confidence interval (CI) 0.13–0.88) [Erlich et al. 2011]. A secondary analysis of 3814 patients (after excluding surgical patients) from the LIPS cohort showed a trend towards a protective effect from aspirin which did not achieve statistical significance after propensity score matching [odds ratio (OR) for development of ARDS 0.7, 95% CI 0.48–1.03, p = 0.07] [Kor et al. 2011a]. In another recently published propensity score analysis of 1149 patients admitted to the surgical or medical ICU at a tertiary care facility, Chen and colleagues found prehospital aspirin use to be associated with a significantly lower risk of developing ARDS versus matched controls even after adjusting for propensity scores for prehospital aspirin use [Chen et al. 2014].
The need for prospective, randomized double-blind human data with regards to aspirin in prevention of ARDS is already being addressed. LIPS with aspirin (LIPS-A) has been completed [ClinicalTrials.gov identifier: NCT01504867] with the results forthcoming. This was a phase II double-blind, placebo-controlled, multicenter trial comparing aspirin (325 mg x 1 day followed by 81 mg x 6 days) with identically appearing placebo. Eligible enrollees were hospitalized adults with a LIPS score of at least 4 at the time of admission. Development of ARDS is the primary outcome and all enrolled patients received CLIP-based support for the standardization of care and avoidance of known nosocomial secondary hits.
Systemic steroids
Systemic steroids have been shown to possess pleiotropic anti-inflammatory properties: they reduce proinflammatory cytokines, chemokines, adhesion molecules and receptors [Frieri, 1999]; they increase anti-inflammatory mediators [Kovalovsky et al. 2000]; they increase activated protein C [Seam et al. 2012]; they reduce inducible nitric oxide expression [Yu et al. 2009]; and they inhibit fibroblast proliferation and collagen deposition [Thompson, 2003]. The last characteristic specifically may play role in tertiary prevention of excessive fibrosis as complication of fully established ARDS. The main limitation of use of systemic steroids in ARDS is their association with adverse effects, which potentially might offset their beneficial anti-inflammatory effects. Furthermore, studies that looked into the role of steroids in the prevention of ARDS were conducted prior to standardization of the lung protective mechanical ventilation; they used different durations of therapy and employed different definitions of ARDS, which further complicated the already heterogeneous characteristics of patients with ARDS. Therefore, it is not surprising that these studies showed conflicting results, hence the equipoise for systemic steroid use in ARDS prevention continues [Brian et al. 2012]. Future studies will have to evaluate which groups of patients at risk and at what point in the course of the disease will favorably respond to systemic steroids. Perhaps, biomarkers will play the important role in attempts to answer these questions. For example, Steinberg and colleagues, in a subgroup analysis of a randomized controlled trial, showed that patients randomized to steroids with higher levels of procollagen peptide III, a marker of fibroproliferation, had significantly better survival [Steinberg et al. 2006].
Statins
It has long been appreciated that the biologic effects of statins extend beyond their ability to lower low-density lipoprotein levels. These so-called pleiotropic effects include ischemic stroke prevention, decreased vascular smooth muscle proliferation and reduced reactive oxygen species production; these effects are thought to be mediated by the inhibition of several isoprenylated proteins or isoprenoids [Liao, 2002; Arnaud et al. 2005; Greenwood and Mason, 2007]. Murine models have demonstrated a role for statins in the prevention of ARDS [Jacobson et al. 2005]. Prospective observational human studies have revealed conflicting results, with some suggesting that prehospital statin use is protective against ALI/ARDS in patients with sepsis [Kruger et al. 2006; Falagas et al. 2008; O’Neal et al. 2011] and others showing no clear benefit [Bajwa et al. 2012]. A recently completed large, multicenter, randomized, double-blinded prospective clinical trial comparing rosuvastatin with placebo in patients with sepsis-associated ARDS failed to show any meaningful benefit of statin therapy in fully established ARDS [Truwit et al. 2014].
The discordance between prehospital statin use and a trend toward benefit and the lack of benefit seen with initiation of statins in established ARDS is likely multifactorial. Whether initiation of statins at the time of hospitalization for those at high risk but before the development of ARDS would be beneficial merits further research.
Renin–angiotensin–aldosterone system blockers
There is compelling evidence to suggest that a prominent role in the development of ARDS is played by local pulmonary vascular angiotensin-converting enzyme (ACE) activity. For example, high levels of ACE have been isolated from broncheoalveolar lavage fluid in subjects with ARDS versus non-ARDS controls [Idell et al. 1987]. Additionally, homozygosity for the polymorphism in the gene encoding ACE associated with higher enzyme activity levels has been shown to be an independent risk factor for the development of ARDS [Marshall et al. 2002]. Elegant in vitro studies have linked angiotensin II, the downstream product of ACE activity, to human lung fibroblast collagen production in a bleomycin-toxicity model. This was attenuated by ACE inhibitor (ACEI) or angiotensin receptor blocker (ARB) therapy [Marshall et al. 2004]. As such, inhibition of the renin–angiotensin–aldosterone axis has been of interest as a potential target for prevention of ARDS, and there have been encouraging preclinical data in this regard.
For example, in a rat model of chemically induced ARDS, captopril therapy markedly improved several surrogate markers of severity of ARDS including circulating endothelial cells, arterial blood oxygen tension, (PaO2), and wet to dry lung weight ratio [Liu and Zhao, 2002]. In another ARDS model using rats subjected to ventilator-induced lung injury, captopril significantly reduced markers of inflammation and apoptosis versus untreated controls [Wosten-van Asperen et al. 2008]. In a similarly designed study, Yao and colleagues showed losartan to be effective at mitigating ventilator-induced lung injury in rats [Yao et al. 2008].
Human data are somewhat conflicting but suggest a protective effect of ACEI or ARB therapy in selected populations. In a large prospective trial, chronic ACEI use was associated with lower rates of pneumonia versus placebo; however this benefit was limited to the Asian population [Ohkubo et al. 2004]. In her review of the interplay between diabetes, hyperglycemia and therapies commonly prescribed for patients with diabetes, Honiden and Gong noted that the decreased rate of mortality from community-acquired pneumonia noted among white people chronically taking ACEI/ARB therapy only extended to patients with diabetes [Honiden and Gong, 2009]. Interestingly, patients with diabetes have been shown to be at decreased risk of developing ARDS compared with those without diabetes [Yu et al. 2013]. A retrospective review of 1423 patients admitted to the ICU in Rochester showed a statistically significant reduction in the risk of developing ARDS in patients taking ACEI/ARB therapy chronically prior to hospitalization, with an OR of 0.49, 95% CI 0.25–0.94 [Trillo-Alvarez et al. 2009]. However, the LIPS cohort study did not find any statistically significant decrease in the incidence of ARDS among patients at high risk for developing ARDS taking an ACEI/ARB prior to hospitalization. On the contrary, the data suggested a trend towards worsening mortality outcomes for those patients taking ACEI/ARB therapy who developed ARDS [Watkins et al. 2011].
These disparate findings perhaps highlight the interplay between genetic and environmental factors in the pathogenesis of ARDS and the need for ongoing prospective research investigating which patients could benefit from the use of ACEI/ARB therapy for the prevention of ARDS, if any.
Peroxisome proliferator-activated receptor agonists
Peroxisome proliferator-activated receptors (PPARs) are a family of intracellular receptors which translocate to the nucleus and modulate gene expression in response to ligand binding. These are well known for their effects with regard to glucose and lipid metabolism but are also important mediators of cellular proliferation and inflammation and are expressed in vascular endothelium, smooth muscle cells and various leukocytes [Brown and Plutzky, 2007]. Animal models have shown PPARγ to be implicated in the inflammation associated with asthma, chronic obstructive pulmonary disease and pulmonary fibrosis [Belvisi and Hele, 2008]. As such, PPAR agonists, particularly the thiazolidinediones, have gained attention as having potential utility in the prevention of ARDS. There are ample animal data to suggest a role may exist for thiazolidinedione therapy in the prevention of ARDS. Rosiglitazone has been shown to protect lung tissue in bleomycin toxicity, endotoxemia, pancreatitis and hyperoxia [Honiden and Gong, 2009].
To date, no prospective human studies involving the use of PPAR agonists have been performed. The association of thiazolidinedione with worsening congestive heart failure in patients with diabetes might have resulted in less interest in investigating this class of drugs.
Inhaled delivery
The appeal of aerosol or nebulized delivery systems for medications lies in the ability to potentially deliver effective doses of agent while minimizing or wholly avoiding systemic side effects. Parallels can be drawn to inhaled medications already available for an array of chronic pulmonary disorders or bronchopulmonary infections.
Inhaled corticosteroids
Investigators have examined the preventative and therapeutic roles of inhaled corticosteroids (ICS) in animal models of ALI through direct and indirect pulmonary insults [Forsgren et al. 1990; Walther et al. 1992, 1993; Wang et al. 2002, 2004; Jansson et al. 2005]. Despite heterogeneity in their methodology, these investigators have reported remarkably uniform observations with regard to the ability of ICS to attenuate lung injury. Related to this, ICS are commonly used medications with an excellent safety profile in humans; however, to date no investigators have performed randomized studies of ICS for the prevention or early treatment of ARDS in human subjects. In a secondary analysis of the LIPS cohort, prehospitalization ICS exhibited raw protective signal towards ARDS development. Even though signal weakened after comprehensive adjustment with propensity matched analysis, the estimate of effect remained consistent, suggesting clinical equipoise and implications for further investigations in the role of ICS in ALI/ARDS prevention [Festic et al. 2013b].
β agonists
Besides dysregulated inflammation, another hallmark of ARDS involves accumulation of protein-rich exudative edema fluid in the airspaces of the lung. β agonists increase the rate of vectorial transport of salt and water across normal epithelium as well as in animal models of ALI [Matthay et al. 2002]. As such, β agonists may be effective as a preventative therapy due to their ability to enhance resolution of pulmonary edema fluid and maintain endothelial barrier functions under baseline conditions [Matthay et al. 2002]. The first ARDS prevention randomized controlled study of β agonists have been recently reported. Perkins and colleagues studied 362 patients undergoing esophagectomy in 12 centers in the United Kingdom over a 3-year period [Perkins et. al, 2014]. The incidence of ARDS did not differ between salmeterol and placebo groups; however, postoperative adverse events (primarily pneumonia) were significantly less frequent in the intervention group. Moreover, in a substudy of 53 patients with available plasma samples, salmeterol reduced several biomarkers of alveolar inflammation and epithelial injury [Perkins et al. 2014].
Combined ICS and β agonists
Given the bronchodilating as well as anti-inflammatory properties of β agonists, it has been proposed that they may act in synergy when used in combination with ICS. Seventy percent of patients on ICS in the secondary analysis of LIPS cohort [Festic et al. 2013b] were concomitantly receiving inhaled β agonists. Based on this experience, investigators initiated a multicenter, double-blind, placebo-controlled, randomized trial evaluating the combined effect of early administered budesonide and formoterol on lung function in patients at risk of ARDS [ClinicalTrials.gov identifier: NCT01783821]. This trial has been designed to test the previous observational study results and provide essential data for a future multicenter phase III trial.
Heparin/thrombolytics
The interplay between local alveolar inflammation and dysregulated coagulation with fibrin deposition/accumulation is integral to the pathogenesis of ARDS. This makes agents targeting the coagulation cascade attractive potential therapeutic options. Although systemic administration of anticoagulant medications would possess the same proposed effect, it carries significant risk of bleeding complications, leading to investigation into the use of inhaled anticoagulant medications.
Heparin is unique in its ability to bind not only antithrombin III, but dozens of other so-called ‘heparin-binding proteins’ such as complement proteins, interferons, fibroblast growth factor and other cytokines [Young, 2008]. These biologic actions likely mediate the previously documented but not completely understood anti-inflammatory effects of heparinoids. Miller and colleagues recently published a review of available human and animal data relating to the use of inhaled heparin and heparin-related compounds in smoke inhalation injury. Collectively these data show decreased morbidity and may suggest potential mortality benefit for the use of inhaled heparin in smoke inhalation injury without increasing markers of coagulopathy or increasing the risk of bleeding complications [Miller et al. 2014]. Whether these data will have generalizable applications to the at-risk ARDS population remains to be seen, and further investigation into the utility of routine use of inhaled heparin in smoke inhalation injury is merited and already registered [ClinicalTrials.gov identifier: NCT014548690].
In a murine model of hemorrhagic shock, it has been shown that microthrombus formation precedes the subsequent accumulation of leukocytes and is likely due to sluggish blood flow in the low-pressure pulmonary circulation [Conhaim et al. 2010]. As such, inhaled thrombolytic therapy has been of interest for prevention of ARDS and has been studied in rat models showing mitigation of microthrombus formation and subsequent leukocyte accumulation [Conhaim et al. 2014].
Hypertonic saline
Hypertonic saline has been established as a safe and effective tool in the management of cystic fibrosis [Elkins et al. 2006], and non-cystic fibrosis bronchiectasis [Kellett and Robert, 2011]. It is postulated that in addition to its effects on sputum viscosity, the efficacy of nebulized hypertonic saline is due at least in part to its ability to inhibit neutrophil and macrophage-mediated cytokine production [Cuschieri et al. 2002; Zallen et al. 2000; Deitch et al. 2003]. In a rat model of traumatic shock, Wohlauer and colleagues demonstrated a convincing protective effect of inhaled hypertonic saline against pulmonary vascular permeability, neutrophil chemotaxis, matrix metalloprotease activity, interleukin-8 activity and histologic changes consistent with ARDS [Wohlauer et al. 2012]. A recent preclinical study demonstrated the cytoprotective effect of extracellular hypertonicity induced by aerosolized hypertonic saline on alveolar resident cells, resulting in their resistance to injury [Brian et al.]. Although routine use of intravenous hypertonic saline for resuscitation of hypovolemic shock in trauma was shown not to improve mortality [Bulger et al. 2011] owing to disturbance of coagulation cascade, inhaled hypertonic saline is unlikely to have this systemic effect. A prospective study is being planned by the LIPS investigators.
N-Acetyl cysteine
N-Acetyl cysteine (NAC) is known for its ability to reduce the burden of oxidative stress by maintaining intracellular stores of reduced glutathione [Lauterburg et al. 1983]. There is considerable evidence that oxidative stress stemming from depleted glutathione stores contributes to the pathogenesis of ARDS [Soltan-Sharifi et al. 2007]. In one small prospective clinical trial enrolling patients with smoke inhalation injury, the combination of inhaled NAC, heparin and albuterol was superior to albuterol and placebo with respect to mortality and lung injury scores [Miller et al. 2009]. Two additional clinical trials enrolling patients with active ARDS or undergoing esophagectomy (high-risk surgery) have shown promise for intravenous NAC with regards to surrogate markers of pulmonary morbidity [Zingg et al. 2007; Soltan-Sharifi et al. 2007]. Owing to its well established safety profile and promising preliminary clinical data, NAC should be of particular interest moving forward.
On the horizon
Myriad additional agents are at various stages of preclinical or clinical investigation with regard to their potential use in the prevention of ARDS. Focus of our attention on these agents is not all inclusive as this area of research is rapidly evolving.
In a recently published, industry-sponsored phase I/II trial, interferon β-1a (FP-1201) showed promising, significant results with regard to improving oxygenation and 28-day mortality (OR 0.19, 95% CI 0.03–0.72) in 37 patients receiving treatment versus 59 matched controls with established ARDS. The proposed mechanism is via decreased vascular leakage through upregulation of pulmonary capillary CD73 [Bellingan et al. 2014].
Mesenchymal stem cell (MSC) therapy has been shown in animal models of sepsis to reduce markers of inflammation and incidence/severity of ARDS [Mei et al. 2010], and decrease histopathologic evidence of ARDS in ex vivo and in vitro studies using human lungs and lung tissue [Lee et al. 2013]. An additional recently published study using human MSCs in a rat ARDS model improved lung compliance, oxygenation, alveolar edema and promoted return to normal lung architecture [Hayes et al. 2014]. MSCs have been shown to lead to more efficient tissue repair, and their use in ARDS is an exciting clinical prospect. One challenge of translating this promising bench research to the clinical setting is the optimal route of delivery of stem cell therapy, which in animal models has shown promise via the intravenous and intratracheal routes. Two clinical trials are ongoing [ClinicalTrials.gov identifiers: NCT01775774, NCT01902082] to assess the optimal doses of intravenous MSC therapy, and an additional phase I trial [ClinicalTrials.gov identifier NCT01632475] is underway assessing the safety of intratracheal administration of MSC in neonates with severe bronchopulmonary dysplasia [Lee et al. 2014].
It has been postulated that at least part of the protective effects of exogenous MSC administration is mediated via their ability to secrete keratinocyte growth factor (KGF) [Zhu et al. 2014]. As such, KGF has been under investigation as a therapeutic agent in and of itself. Pretreatment with KGF prior to pulmonary acid injury in mice decreased markers of inflammation [Yano et al. 1996]. The mechanism by which KGF protects pulmonary epithelial cells from apoptosis and promotes survival and epithelial integrity has been elucidated in exquisite detail [Bao et al. 2005]. KGF therefore may prove to have direct applications in the prevention of ARDS.
Curcumin, a natural phenol found in the spice turmeric, has gained attention for its potential ability to modulate mammalian inflammation. In a murine model of virally induced ARDS, pretreatment with curcumin significantly reduced a number of surrogate severity markers of ARDS [Avasarala et al. 2013]. There has been interest among LIPS investigators to prospectively study curcumin for ARDS prevention (personal communication, Ruxana Sadikot, MD).
Adrenomedullin, a vasoactive peptide hormone initially isolated from pheochromocytoma tissue, has been gaining interest as a potential biomarker for ARDS and may have therapeutic potential. Rat models of ARDS have shown exogenous adrenomedullin to be a potent inhibitor of lung injury in gut ischemia/reperfusion [Dwivedi et al. 2007] as well as endotoxinemia [Itoh et al. 2007]. The potential role of this agent for humans remains to be seen and to our knowledge the only ongoing research using human subjects involves its use as a biomarker in various conditions.
Conclusion
Years of research on pharmacological therapy for established ARDS have not yielded a single effective medication. However, this might not have been in vain. Many of the previously studied pharmacological compounds, as well as new ones, when used early in the disease evolution may prove effective in the prevention of ARDS. We especially favor medications that have been proven to be safe, inexpensive and widely available. As such, these medications should be first studied in smaller phase II trials that are much more feasible.
The completed LIPS-A trial and ongoing Lung Injury Prevention with Budesonide and Beta-Agonist (LIPS-B) trial have already demonstrated the feasibility of early administration of systemic and inhaled medications to patients deemed at risk for ARDS development, frequently started while the patient is still in the emergency room. We postulate that overall morbidity and mortality rates for ARDS in the future will be contingent upon decreasing the overall incidence of ARDS through effective identification of those at risk and early application of proven supportive care and pharmacological interventions.
Investigators, however, need to change the way of conducting randomized clinical trials to discern effective interventions. Some of the proposed strategies should include the following: smaller phase II trials of relatively homogenous patient populations; assessment of outcome variables other than mortality; focus on patient-centered outcomes including longer-term outcomes; pragmatic and adaptive clinical trials, as well as the ‘platform’ trials, which could simultaneously evaluate multiple treatments efficiently.
Footnotes
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
Funding
This study was done in part with the support of NIH 5KL2 TR000136 and UL1 TR000135 grants and the Mayo Clinic Foundation.
