Abstract
Background:
Pulmonary arterial hypertension (PAH) is a life-threatening, progressive disease often diagnosed late in its course.
Objectives:
To present patient-reported data that were captured within a large, multinational, point-in-time survey of PAH-treating physicians and their patients to better understand the diagnostic journey.
Design:
Cross-sectional survey conducted in five European countries (EU5), Japan and the USA.
Methods:
PAH-treating pulmonologists, cardiologists, rheumatologists or internists (USA only) completed a patient record form (PRF) for the next four consecutive adult PAH patients they saw; these patients filled in a patient self-completion (PSC) form on an anonymous, voluntary basis. Our report focuses on patient data; data are from PSC forms unless stated otherwise.
Results:
Physician-reported PRFs and self-completed PSC forms were obtained for 1152 and 572 patients, respectively. Patients’ mean (SD) age was 59.1 (14.0) years, 55.6% were female, and 57.3% had idiopathic PAH. Patient-reported data showed an average delay of 17.0 months between symptom onset and PAH diagnosis. This is longer than physicians estimated (13.8 months): this disparity may be partly due to the time taken by patients to consult a physician about their symptoms [9.6 months overall, longest in the USA (15.3 months)]. Most patients (71.6%) initially consulted primary care physicians about their symptoms and 76.4% of patients were referred to a specialist. Misdiagnoses occurred in 40.9% of patients [most frequent in the USA (51.3%), least common in Japan (27.6%)] and they saw an average of 2.9 physicians overall (3.5 in EU5 versus 2.0 in Japan/USA) before being diagnosed. Diagnosis was most often made by cardiologists (50.4%) or pulmonologists (49.3%).
Conclusion:
Our data suggest that diagnostic delay in PAH results from patient- and physician-related factors, which differ across regions and include lack of awareness of PAH on both sides. Development of better screening strategies may help address this barrier to timely PAH diagnosis.
Background
Pulmonary arterial hypertension (PAH) is a rare, life-threatening and incurable condition that follows a progressive clinical course. 1 PAH is a subgroup of pulmonary hypertension (PH) and is hemodynamically defined by the 2022 European Society of Cardiology/European Respiratory Society guidelines with a resting mean pulmonary arterial pressure of >20 mmHg, a pulmonary artery wedge pressure of ⩽15 mmHg and a pulmonary vascular resistance of >2 Wood units. 2 A recent systematic literature review estimated the prevalence of PAH to be between 47.6 and 54.7 cases per million adults. 3 Multiple medical therapies have been approved for PAH over the past two decades 4 and prognosis has improved substantially, with 1-year survival increasing from 65% in the 1980s to up to 90% in the present day; over the same time frame, median survival has improved from 2.8 to 6 years. 5
While there has been considerable therapeutic progress in PAH, the diagnosis of this condition remains problematic. 6 The REVEAL registry in the USA reported that more than one-fifth of patients waited longer than 2 years from first symptoms to diagnosis, 7 while a German cohort study identified a mean diagnostic delay of 2.3 years. 8 Reasons for this diagnostic delay include the non-specific nature of the symptoms,2,6 the rarity of the disease and the consequent lack of awareness of PH among patients and primary care physicians (PCPs). 9 Transthoracic echocardiography is recommended for all patients with suspected PH; this can indicate the likely presence of PH and other cardiac abnormalities that may give an indication of the PH type in PH cases. However, it is not sufficient in isolation to support therapeutic decision-making, 2 and is a labour-intensive procedure that does not reliably identify PH patients for further testing.10–12 Definitive PAH diagnosis requires the performance of right heart catheterization (RHC), which is an invasive procedure and is often conducted by a specialist in a dedicated PH centre. 2 The importance of understanding and addressing this diagnostic delay is underscored by the adverse effect of diagnostic delay on patient outcomes. 13 For example, a study of idiopathic PAH (IPAH) patients found that patients deteriorated by one functional class during the delay period, which the authors estimated would result in a 20% increase in mortality over 2 years. 14
Further investigation of the PAH diagnostic journey may help to identify areas where improvements can be made. One important source of information is the patient, yet few studies have explored diagnostic delay in PAH using information obtained directly from patients.9,14,15 These studies analysed a relatively small group of patients from a single country, therefore additional data from a larger, more representative cohort are required. Our study focuses on patient-reported data that were captured as part of a large, multinational, point-in-time survey 16 – the Adelphi Disease Specific Programme™ – to understand the PAH patients’ perspective on the diagnostic process.
Methods
Study design and participants
Full methodology of the Disease Specific Programme has been published previously. 16 The study was conducted as a single point-in-time survey between July 2019 and June 2020 in five European countries (EU5: France, Germany, Italy, Spain, the United Kingdom), Japan and the USA. Physician eligibility for participation was assessed using an online screening procedure (Figure 1). Eligible physicians were pulmonologists, cardiologists, rheumatologists or internists (USA only) who were personally responsible for the management of patients with PAH and saw a minimum of two such patients per month. Physicians completed a patient record form (PRF) for the next four consecutive patients with a PAH diagnosis that they saw, using patient medical records. The only inclusion criteria for patients were that they must be more than or equal to 18 years of age, have a confirmed diagnosis of PAH and not be participating in a clinical trial. The same four patients from each physician were asked to fill in a patient self-completed (PSC) form on an anonymous, voluntary basis (Supplemental Figure S1); not all patients agreed to participate. PSC responses were not influenced by, or shown to, the treating physician. Patients were able to skip questions in the PSC forms. The analyses reported within this article primarily focus on data obtained from the PSC, unless otherwise specified. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (see Supplemental Appendix for completed STROBE checklist). 17
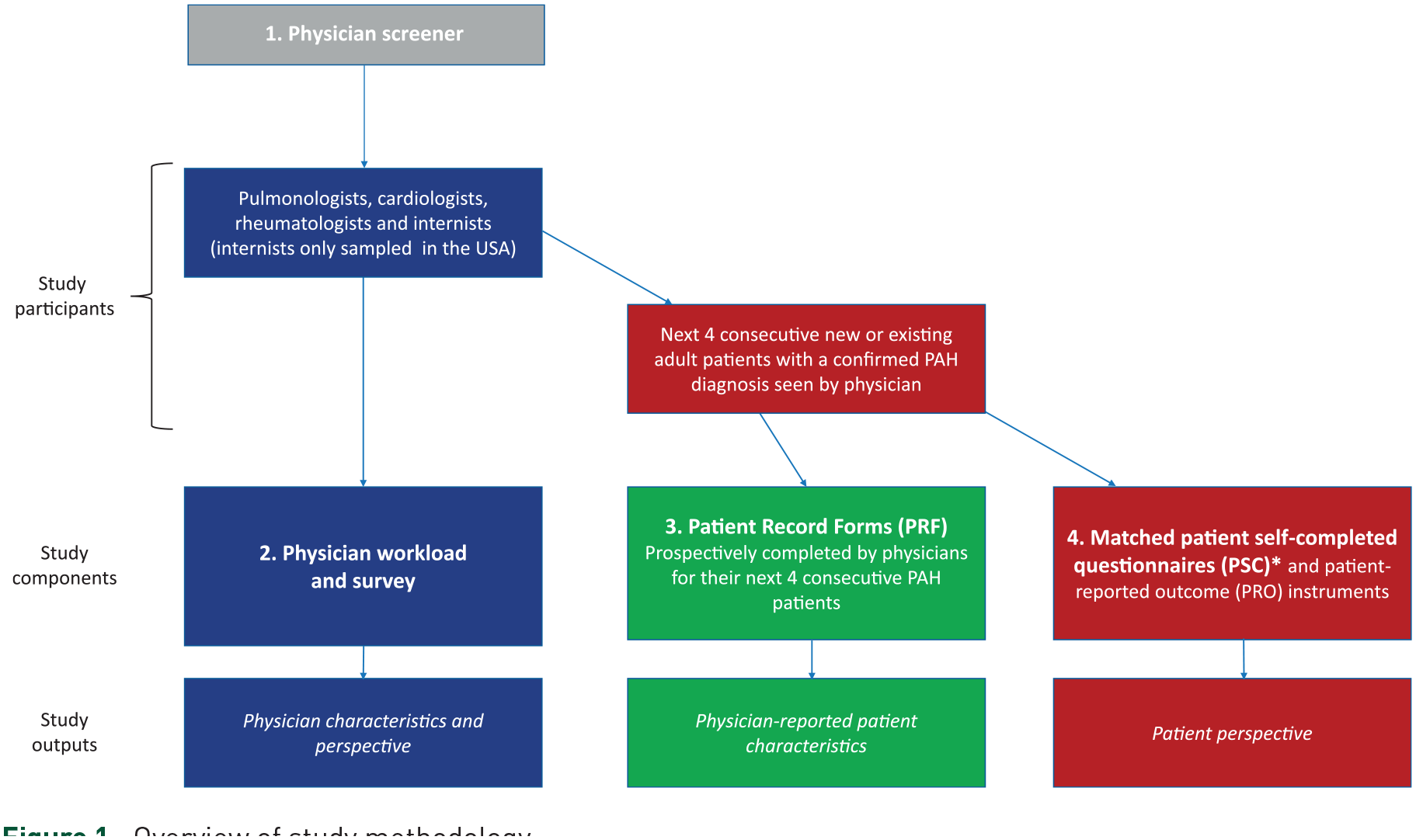
Overview of study methodology.
Data collection
Physician surveys collected demographic information about physicians, including their practice setting. The following data were collected from physician-completed PRFs: patient demographics and characteristics and physician-reported time from symptom onset to PAH diagnosis. These parameters were only reported for those patients who had completed a PSC form (PSC-matched PRF data). Matching of PSC forms to PRFs was done as follows: (i) a number was assigned to each completed PRF and linked with the corresponding PSC form and (ii) upon uploading to the database, the numbers are re-anonymized and an overall PRF-PSC match variable is created. With the exception of these two parameters, all other reported data are from the PSC forms. Information obtained using the PSC included: patient’s age at first PAH symptoms, first symptoms of PAH, time from first symptoms to first consultation and from first consultation to diagnosis, reasons for initial consultation, misdiagnoses received, number and specialty of physicians seen and specialty of the diagnosing physician.
Statistical analyses
Three subgroup analyses were conducted: one reporting the above-listed parameters for patients sub-grouped by patient-reported time from first symptoms to PAH diagnosis (<6 months; 6 to <12 months; ⩾12 months), and another two reporting time to first medical consultation and first PAH diagnosis for patients sub-grouped according to (i) their New York Heart Association (NYHA) functional classification (FC) 18 before receiving treatment (physician-reported NYHA FC) and (ii) their PAH aetiology [associated PAH (APAH), familial PAH (FPAH) and IPAH]. No formal statistical testing of hypotheses was performed; data were summarized using descriptive statistics only.
Ethics approval and consent to participate
The Disease Specific Programme was conducted as a survey adhering to European Pharmaceutical Market Research Association guidelines, International Chamber of Commerce (ICC)/ESOMAR International code on observational research and Health Insurance Portability and Accountability Act (HIPAA) guidelines. In addition, the survey was submitted to the Western Institutional Review Board (WIRB), a central institutional review board (IRB), where ethical exemption determination was granted on 25 June, 2000 for the USA (WIRB work order #1-1196332-1) and 7 January, 2020 for EU5 and Japan (WIRB work order #1-1251144-1). Informed consent was obtained from all participants prior to their involvement in the study.
Results
Physician characteristics
A total of 378 physicians participated in the study (Supplemental Table S1), comprising 150 pulmonologists, 147 cardiologists, 51 rheumatologists (ex-USA only) and 30 internists (USA only). Approximately half of the physicians (51.1%) were affiliated with a specialist PAH centre or program and a similar proportion (52.5%) spent more than half of their time in a university or teaching hospital. In total, PRFs were completed by physicians for 1152 patients; PSCs were received for 572 of these (49.7%; Supplemental Table S1).
Patient characteristics
Table 1 shows the demographic characteristics for the cohort of patients who submitted PSCs. Patients had a mean [standard deviation (SD)] age of 59.1 (14.0) years and a mean (SD) body mass index (BMI) of 26.4 (12.6) kg/m2. Overall, 55.6% of patients were female and 47.2% were current or former smokers. Mean (SD) time since diagnosis of PAH (n = 484) was 3.6 (4.7) years. The predominant PAH classification (n = 328) was IPAH (57.3%); of the patients with associated PAH (39.3%), the primary cause was given as connective tissue disease (44.0%).
Patient demographics for patients who submitted PSC forms.
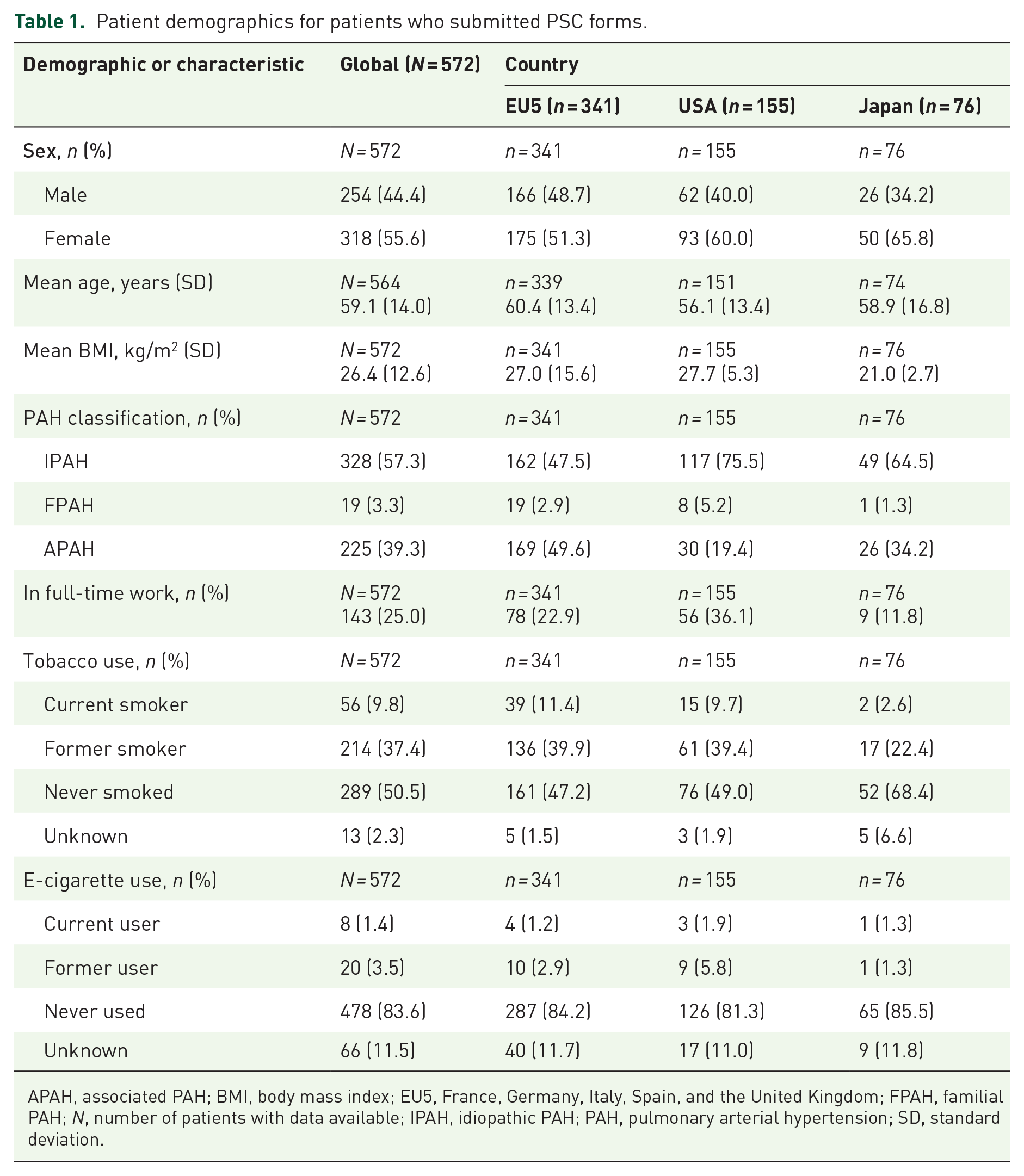
APAH, associated PAH; BMI, body mass index; EU5, France, Germany, Italy, Spain, and the United Kingdom; FPAH, familial PAH; N, number of patients with data available; IPAH, idiopathic PAH; PAH, pulmonary arterial hypertension; SD, standard deviation.
Symptom onset and first medical contact
Patients first reported symptoms of PAH at a mean age of 53.3 years (55.0 years EU5, 54.2 years Japan, 49.1 years USA). The mean number of symptoms reported by an individual patient was 3.1, and the most frequently reported symptoms first noticed by patients were shortness of breath (91.1%) and fatigue (62.8%; Figure 2). Overall, the mean time from the initial appearance of symptoms to consultation with a physician was 9.6 months (Table 2), and disease severity was worst when patients waited longer to see a doctor (Supplementary Figure S1). Japanese patients waited the shortest time to consult a physician (mean 5.2 months) and patients in the USA waited the longest (mean 15.3 months; Table 2).

Symptoms of PAH first noticed by patients.
Time to diagnosis of PAH, as reported by patients and physicians.

Calculated by adding together the patient-reported time from symptoms to initial consult and the patient-reported time from initial consult to PAH diagnosis (in days, and then converting total to months), where both scores are available.
PRF-derived information for patients who submitted PSC forms.
EU5, France, Germany, Italy, Spain, the United Kingdom; PAH, pulmonary arterial hypertension; N, number of patients with data available.
The majority of patients (71.6%) initially consulted PCPs about their PAH symptoms; pulmonologists and cardiologists were the first point of contact for approximately 15% of patients each (Table 3). In Japan, just 44.7% of patients had their first consultation with a PCP, while the proportions of patients initially seeing a pulmonologist or cardiologist were 23.7% and 26.3%, respectively (Table 3). Patients most often made their first visit to a physician as a result of their own concerns regarding symptoms (67.4%) and almost a quarter (24.4%) were encouraged to see a doctor by family or friends (Table 3). More patients consulted a physician due to their own concerns in the EU5 countries (73.1%) than in the USA (58.4%) or Japan (60.5%; Table 3).
Physicians consulted by patients with PAH.

EU5, France, Germany, Italy, Spain, the United Kingdom; N, number of patients with data available; PAH, pulmonary arterial hypertension; PCP, primary care physician; SD, standard deviation.
Referrals
Most patients (76.4%) were referred to a specialist physician before diagnosis of PAH (Table 3). In the USA and Europe, large percentages of specialist referrals were to pulmonologists (58.1% in Europe and 68.7% in USA) and cardiologists (55.9% and 47.8%), whereas in Japan 73.7% of referrals were to cardiologists and only 15.8% of referrals were to pulmonologists. The mean number of physicians seen prior to a diagnosis of PAH was 2.9; this number was higher in the EU5 countries (3.5) than in Japan and the USA (both 2.0; Table 3).
Misdiagnoses
Overall, 40.9% of patients received a misdiagnosis before being correctly diagnosed with PAH; misdiagnosis was most frequent in the USA (51.3%) and least frequent in Japan (27.6%; Table 4). The mean number of misdiagnoses received was 1.7. Chronic obstructive pulmonary disease (COPD; 10.1%), asthma (8.3%) and heart failure (5.3%) were the most frequently received misdiagnoses overall (Table 4). However, COPD was not reported by any patients from Japan, where the rate of misdiagnosis was generally low; the most common alternative diagnosis in Japan was pneumonia (6 patients, 7.9%).
Misdiagnoses prior to diagnosis of PAH.

Percentages calculated using the total number of patients who reported whether or not they received a misdiagnosis (e.g. 567 patients overall) as the denominator.
CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; EU5, France, Germany, Italy, Spain, and the United Kingdom; N, number of patients with data available; PAH, pulmonary arterial hypertension.
Diagnosis of PAH
The mean time from first consultation to diagnosis of PAH was 7.7 months (range 0.2, 155.9) (Table 2). The interval was shortest in Japan (5.5 months) and longest in the USA (8.6 months). Time to diagnosis in patients whose initial consultation was with a cardiologist (12.2 months; n = 32) was longer than in those who first consulted an internist (7.8 months; n = 28) or a pulmonologist (9.3 months; n = 32). Overall, a similar proportion of patients were diagnosed with PAH by a cardiologist or a pulmonologist (50.4% or 49.3%, respectively) (Table 3). In the USA, pulmonologists made the diagnosis in 64.3% of patients, whereas in Japan, 67.6% of patients were diagnosed by cardiologists (Table 3). Combining patient-reported data for time to first consultation and time from first consultation to diagnosis (for patients who had both scores available) revealed a mean total duration from first symptoms of PAH to diagnosis of 17.0 months (Table 2). Patients with APAH had a shorter time to PAH diagnosis than those with IPAH [mean of 14.8 versus 18.6 months (patient reported) and 12.8 versus 13.4 months (physician reported); Supplemental Table S2].
Physician reports of time from first symptoms to diagnosis were slightly shorter than those from the corresponding patients (mean 13.8 months); this was consistent across all countries, with the largest discrepancy between patients and physicians seen in the USA (5.8 months; Table 2). These physician estimates were based on PRF data for those patients who also submitted PSC forms (PSC-matched PRF data). Of these 572 patients who had matching PRF and PSC data, 77.4% underwent RHC (74.5% Europe, 83.2% USA and 78.9% Japan).
Subgroup analysis according to time to diagnosis
Stratifying patients according to the patient-reported time from first symptoms to PAH diagnosis revealed that 212 patients had an interval of <6 months, 108 patients took 6 to <12 months to be diagnosed and 192 patients waited ⩾12 months. Demographics of these patient subgroups are shown in Supplemental Table S3. The mean age and BMI of patients were similar across the three subgroups, but there were more males in the group taking ⩾12 months versus <6 months to receive a diagnosis (48.4% versus 42.5%, respectively) and more patients receiving a diagnosis of IPAH (63.5% versus 53.3%, respectively).
The proportion of patients in NYHA FC III or IV 18 increased with increasing time to diagnosis (<6 months: 45.3%; 6 to <12 months: 48.6%; ⩾12 months: 58.6%; Supplemental Figure S1). Both the time from initial symptoms to physician consultation and the time from first consultation to PAH diagnosis increased as the overall time to diagnosis increased (Supplemental Table S4). Although there was no difference in the number of doctors seen prior to diagnosis, more patients in the subgroup taking ⩾12 months to receive a diagnosis had initially seen a PCP compared with those receiving a diagnosis in <6 months (78.6% versus 64.2%, respectively) and fewer had consulted a doctor because of their own concerns (63.9% versus 72.4%, respectively; Table 5).
Physicians consulted by patients with PAH according to total time to PAH diagnosis.

PAH, pulmonary arterial hypertension; PCP, primary care physician; SD, standard deviation.
The proportion of patients receiving at least one misdiagnosis increased with time to diagnosis (<6 months: 29.7%; 6–<12 months: 41.1%; ⩾12 months: 56.8%), as did the mean number of misdiagnoses (1.2, 1.4 and 2.1, respectively; Supplemental Table S5). This trend of a longer diagnostic interval with higher incidence of misdiagnosis was seen across most of the alternative diagnoses investigated. The greatest difference was observed for asthma, with 15.6% of patients in the ⩾12 months’ time-to-diagnosis subgroup receiving this misdiagnosis versus 3.3% of those in the <6 months group.
Discussion
This large, multinational, point-in-time survey captured real-world patient-level information on the diagnostic journey in PAH, obtained from the patient’s perspective. The mean time from first appearance of PAH symptoms to diagnosis was 17 months, more than half of which resulted from a delay in patients consulting a physician (mean of 9.6 months). Once physician contact was made, further delays resulted from misdiagnoses, which were received by 40.9% of patients, and multiple referrals, with patients seeing an average of 2.9 physicians before a PAH diagnosis.
The majority of published data on diagnostic delay came from physician-derived information. The earliest data, from two 1980s registries in the USA, reported a time from first symptoms to diagnosis of approximately 2 years.19,20 Later registry data from France (2002–2003), Germany (2004–2006) and the USA (2006–2007) revealed that the overall delays were often longer than 2 years.7,8,21 The most recent studies, from Polish (2018) and Australian/New Zealand registries (2004–2017), reported a delay of 12.6 (mean) and 14.4 (median) months, respectively.13,22 Patient-derived data for total time from first symptoms to diagnosis came from a 2007 to 2008 Australian patient survey (mean 47 months) and a 2019 Polish patient survey (median 18 months).14,15 In our study, patient-reported estimates of this interval were shorter (17.0 months). Although there are signs of recent improvement in the timeliness of PAH diagnosis, these data highlight that delayed diagnosis of PAH remains a major problem for the field.
Previous studies have attempted to determine the factors that impact on diagnostic delay in PAH. Age and presence of comorbidities such as sleep apnoea, obstructive airways disease and peripheral vascular disease have been shown to have a significant association.7,13,14 No such analysis was performed in the present study, but data from the subgroup analyses suggest that the overall diagnostic delay is impacted by factors affecting both the time to initial physician consultation and the time from consultation to diagnosis. Misdiagnoses, which are likely to be more common in those with comorbidities, are also a key contributing factor.
Physicians’ estimates of the time between symptom onset and PAH diagnosis were shorter in our survey than patients’ estimates, though the reasons for this finding are unclear. Physicians can only reliably measure symptom onset from a patient’s first clinic visit to discuss the symptoms. Hence, the long delay in patients discussing their symptoms with physicians may be at least partly responsible for the discrepancy between patient- and physician-reported estimates. To our knowledge, this is the first report of ‘matched’ estimates of the length of the diagnostic interval from patient and physician sources.
In our survey, patients waited an average of 9.6 months before discussing their symptoms with a doctor. The Polish PH registry also reported this interval to be 9.6 months on average, 22 whereas a survey of PAH patients in the United Kingdom reported a mean of 21 months. 9 The primary reason for patients contacting their doctor in our survey was concern about their symptoms. The UK survey found that it was often a ‘critical event’, such as the patient collapsing in the street, that precipitated medical contact. 9 The Polish survey reported worsening of disease, persistence of symptoms and development of new symptoms as factors influencing initiation of physician contact. 15 Notably, fewer patients who waited ⩾12 months versus <6 months for a diagnosis had visited the doctor originally due to their own concerns (63.9% versus 72.4%, respectively), but more of these patients had visited the doctor due to family/friends concern about their symptoms (29.8% versus 18.6%, respectively). This may reflect that this subgroup includes patients who typically try to ignore their symptoms and avoid having to see a doctor if they can. Our survey did not capture reasons for the patients’ delay in reporting symptoms, but other studies suggest that patients ‘dismiss’ their symptoms until they become more serious. The Polish survey found that patients delayed seeing a doctor because their symptoms had little impact on their physical activity, that they expected remission of symptoms, and that they had ascribed the symptoms to other conditions. 15 The UK survey reported that patients attributed their symptoms to other reasons such as increasing age or poor physical fitness. 9 These data demonstrate the importance of efficient investigation of dyspnoea once patients do visit a doctor, and also suggest that efforts to encourage patients to visit a doctor as soon as they notice symptoms (such as making it easier to book an appointment or offering telephone consults) may help address diagnostic delay in PAH.
The greatest diagnostic delay was reported by patients and physicians from the USA, and this appears to be driven largely by the delay in patients reporting symptoms (15.3 months in USA versus 7.8 months in Europe and 5.1 months in Japan). This finding may be related to the lack of a universal healthcare system in the USA, with other studies highlighting that cost is a key factor for people in the USA not seeking medical care when they notice symptoms,23,24 or to patients having to travel further to see a specialist, though we do not have the data to investigate whether this was the case. Compared with their EU5 or USA counterparts, patients in Japan waited less time to make their first physician contact, saw fewer physicians overall (versus EU5 only), were misdiagnosed less often and were ultimately diagnosed quicker. The current Japanese Circulatory Society practice guidelines for PH are based on the 2015 ESC/ERS guidelines, 25 suggesting that diagnostic pathways should be similar across Europe and Japan. However, their healthcare systems are different and, in Japan, patients can directly access secondary care without PCP referral. 26 This is likely the reason that a lower percentage of patients in Japan saw a PCP compared with the other regions, where a PCP was most often the first point of medical contact, consistent with findings from the Polish patient study. 15 While the present survey did not capture reasons for delays in diagnosis, it is logical to assume that the differences between the participating country’s healthcare systems are a major contributor to the regional variation reported for time to PAH diagnosis. Differences in population demographics and characteristics are another potential key contributor. In terms of disease characteristics, a lower percentage of patients from the USA had APAH compared with the other regions. This is notable since patients with APAH had a shorter diagnostic interval than those with IPAH, likely as a result of greater opportunities to detect PAH in patients with an associated condition. For example, patients with systemic sclerosis are recommended to be screened annually for PAH. 2 Other differences between patient populations could also have contributed to the regional variation.
The lack of awareness of PAH among physicians, even specialists, is reflected in the number of doctors seen before the diagnosis was made (mean of 2.9). German registry data from 2010 indicated that more than half of patients saw three or more physicians before their final diagnosis. 8 The UK patient survey reported that most PH patients saw four or more physicians prior to diagnosis; no overall information is provided on the types of specialist consulted, but one patient had seen a ‘cardiac specialist, respiratory specialist, warfarin specialist, radiographer. . .’. 9 The Australian DELAY study reports even higher numbers, with patients seeing an average of three specialists (after an average of 5.3 PCP consultations) before diagnosis was confirmed at a PH centre. 14 Therefore, it appears that there has been no major change over the past decade in the number of physicians seen by PAH patients before receiving a diagnosis. This suggests that physicians are not linking the symptoms to PAH or considering PAH as a possible cause.
The misdiagnoses reported herein could also be evidence of a lack of PAH awareness, with 40.9% of patients receiving an average of 1.7 misdiagnoses. Receiving one or more misdiagnoses appears to have been a major factor impacting diagnostic delay in our survey, with the proportion of misdiagnoses in patients with a time to diagnosis of ⩾12 months almost double that of the group who were diagnosed in <6 months (56.8% versus 29.7%). The number and nature of misdiagnoses in our survey appear consistent with those reported previously. For example, although numerical data were not reported, the UK patient survey described misdiagnoses as common, including stress, panic attacks, asthma, antiphospholipid syndrome and lupus. 9 Moreover, the Australian patient study reported a mean of 2.1 misdiagnoses, with one-third of patients having received treatment for asthma. 14 The availability of better screening strategies in PAH could reduce this delay caused by sequential exclusion of more common causes of dyspnoea. Echocardiogram is recommended for all patients with chronic unexplained dyspnoea; however, if the operator is not aware of a suspicion of PAH, they are much less likely to measure the relevant parameters and, consequently, they may miss PAH. To address this, several groups are applying artificial intelligence to readily available investigative tests to enhance their ability to detect PAH and other cardiovascular diseases.27,28
Generally, when diagnosis of PAH is finally made, this is done by either a cardiologist or a pulmonologist, according to our data. In the German registry, pulmonologists made the diagnosis most often (47%), followed by cardiologists (35%). 8 In contrast, the Polish patient study reported that cardiologists made the majority of diagnoses (80%). 15 This may reflect referral patterns within those individual countries. In our survey, differences between countries/regions in the type of physician most frequently diagnosing PAH appear consistent with the overall proportions of patients being referred to those specialists, and there is no evidence that diagnostic pathways directed to one specialty over the other are any more effective in detecting PAH.
As the behaviour of patients and physicians affects the diagnostic journey in PAH, appropriate educational programs aimed at both these audiences could affect substantial changes in this process. In a survey of UK patients, the failure of physicians to consider PH as a cause of symptoms emerged as a common theme reported by patients. 9 The authors of that study recommended the development of national practice guidelines to assist in the diagnosis of patients with unexplained dyspnoea; although such guidelines now exist, at the time of writing, they do not include PH as a suggested diagnosis. 29 The UK study also identified a tendency for patients to provide their own explanations for the symptoms they experienced, making self-diagnoses based on information from sources such as the internet. 9 More widespread availability of patient-focused information on PAH could therefore decrease the time before the patients contact a physician about their symptoms.
A key strength of this work is that the data reported here are from a much larger number of patients than have been included in previous studies conducted from the patient’s perspective.9,14,15 The inclusion of patients from multiple countries enabled similarities and differences in the diagnostic journey to be compared globally. Furthermore, analysis of both physician- and patient-derived data, which has not previously been performed in the same study, provided an opportunity to make comparisons between these two different perspectives on diagnostic delay. One potential limitation of the study is asking physicians to report data for the next four patients that they saw, as this may have led to the sample not being fully representative of the PAH population. This type of convenience sampling increases the likelihood of selecting patients who visit their physician more often, for whatever reason. Patients completed PSCs on a voluntary basis; this may have attracted patients who were more motivated to participate, perhaps because they were dissatisfied with some aspect of their diagnostic journey. Patients could also skip certain questions in the PSC, hence, there are some missing data. As there were no inclusion criteria requiring patients to have received their diagnosis within a certain timeframe, some time may have passed since the events leading up to PAH diagnosis. The data collected may therefore be subject to recall bias, and it was not possible to subsequently contact the participants to verify the information provided by patients or physicians. Approximately half of the physicians in this study were associated with a PAH specialist centre. Therefore, this physician cohort may not be representative of all PAH-treating physicians; however, guidelines do recommend that all PAH patients have their diagnosis confirmed with a RHC performed at a specialist centre. As this was a real-world study, not all patients had a confirmed PAH diagnosis; only 77.4% of PAH patients had undergone RHC. Another limitation of the study is that no power calculation was performed for estimation of the sample size, therefore the statistical significance of these data cannot be determined.
Conclusion
In conclusion, this is the largest study to date to illustrate a diagnostic delay in PAH from the perspective of the patient. Results indicate that both patient and physician factors contribute to the time taken to obtain the correct diagnosis. Increasing awareness of PAH among both physicians and the general public, and developing more widely available, accurate screening tools could play important roles in enhancing diagnostic speed and improving patient outcomes.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231218886 – Supplemental material for The diagnostic journey of pulmonary arterial hypertension patients: results from a multinational real-world survey
Supplemental material, sj-docx-1-tar-10.1177_17534666231218886 for The diagnostic journey of pulmonary arterial hypertension patients: results from a multinational real-world survey by Mark Small, Loïc Perchenet, Alex Bennett and Jörg Linder in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666231218886 – Supplemental material for The diagnostic journey of pulmonary arterial hypertension patients: results from a multinational real-world survey
Supplemental material, sj-docx-2-tar-10.1177_17534666231218886 for The diagnostic journey of pulmonary arterial hypertension patients: results from a multinational real-world survey by Mark Small, Loïc Perchenet, Alex Bennett and Jörg Linder in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
