Abstract
Weaning is the most significant event in the life of pigs and is always related with intestinal disruption. Although it is well known that zinc oxide (ZnO) exerts beneficial effects on the intestinal barrier, the mechanisms underlying these effects have not yet been fully elucidated. We examined whether ZnO protects the intestinal barrier via mitogen-activated protein kinases and TGF-β1 signaling pathways. Twelve barrows weaned at 21 d of age were randomly assigned to two treatments (0 verus 2200 mg Zn/kg from ZnO) for 1 wk. The results showed that supplementation with ZnO increased daily gain and feed intake, and decreased postweaning scour scores. ZnO improved intestinal morphology, as indicated by increased villus height and villus height:crypt depth ratio, and intestinal barrier function, indicated by increased transepithelial electrical resistance and decreased mucosal-to-serosal permeability to 4-ku FITC dextran. ZnO decreased the ratios of the phosphorylated to total JNK and p38 (p-JNK/JNK and p-p38/p38), while it increased the ratio of ERK (p-ERK/ERK). Supplementation with ZnO increased intestinal TGF-β1 expression. The results indicate that supplementation with ZnO activates ERK ½, and inhibits JNK and p38 signaling pathways, and increases intestinal TGF-β1 expression in weaned pigs.
Keywords
Introduction
Because of their immature immune and digestive systems, young animals are susceptible to immunologic challenge. 1 Pigs subjected to early weaning stress exhibit more severe diarrhea and intestinal disruption compared with late-weaned pigs. 2 This results mainly from the activation of the mucosal immune system, initiated by bacteria, endotoxins and antigens. 2 The activated immune systems up-regulates the expression of pro-inflammatory cytokines in the intestine, such as TNF-α, IL-6, IL-1β and INF-γ.3,4 The inflammatory response and subsequent overproduction of pro-inflammatory cytokines induces a disrupted intestinal barrier function.1,4
It is well known that zinc exerts anti-inflammatory and anti-diarrheal effects, as well as intestinal barrier protection in animal models and clinical trials. 5 The beneficial role of zinc in the epithelial barrier has been shown both in vitro and in vivo.6,7 In particular, zinc oxide (ZnO) appears to exert a strong effect in resistance to intestinal diseases. 6 Early weaned piglets frequently experience intestinal barrier dysfunction, post-weaning diarrhea and growth retardation. 4 Supplementation with a high level of ZnO (between 2000 and 4000 mg/kg of zinc) in weanling piglets ameliorates diarrhea and improves piglet growth performance.8,9 Although ZnO is reported to maintain mucosal epithelial integrity and the normal function of intestine in weanling piglets,8–10 the molecular mechanisms have not yet been elucidated.
MAPK cascades transduce signals from a diverse array of extracellular stimuli and form a highly integrated network to regulate cell growth and cell differentiation, especially stress responses. 11 Our recent study showed that weaning stress activated MAPK signaling pathways in the intestine, which may be an important mechanism of weaning-associated enteric disorders of piglets. 4 Furthermore, TGF-β1 is also believed to play an important regulatory role in the post-weaning adaptation process in the intestine of the pig. 12 The canonical TGF-β signaling effectors are the SMAD family proteins, which transduce signals from the cell surface directly to the nucleus to regulate target gene transcription. 13 However, research on zinc-regulated MAPK signaling and TGF-β1 expression has only so far been reported in in vitro cell models.13,14 Few data are available regarding the effect of ZnO in vivo. This study was conducted to assess the impact of ZnO supplementation on intestinal barrier function and to explore whether ZnO supplementation could improve intestinal barrier function through regulation of the MAPK and TGF-β1 signaling pathways.
Materials and methods
Experimental design and sample collection
Composition of the basal diet (on an as-fed basis).
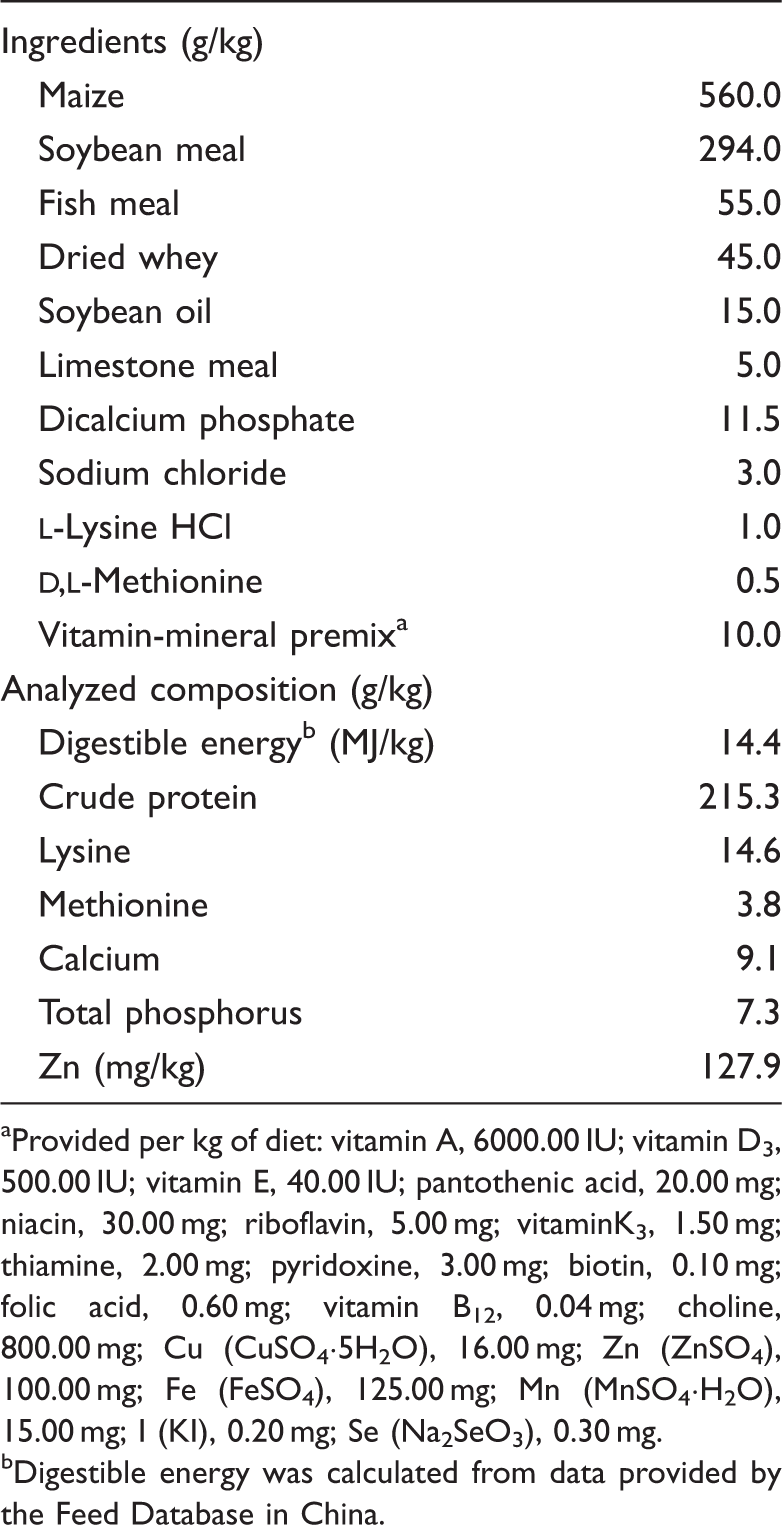
Provided per kg of diet: vitamin A, 6000.00 IU; vitamin D3, 500.00 IU; vitamin E, 40.00 IU; pantothenic acid, 20.00 mg; niacin, 30.00 mg; riboflavin, 5.00 mg; vitaminK3, 1.50 mg; thiamine, 2.00 mg; pyridoxine, 3.00 mg; biotin, 0.10 mg; folic acid, 0.60 mg; vitamin B12, 0.04 mg; choline, 800.00 mg; Cu (CuSO4·5H2O), 16.00 mg; Zn (ZnSO4), 100.00 mg; Fe (FeSO4), 125.00 mg; Mn (MnSO4·H2O), 15.00 mg; I (KI), 0.20 mg; Se (Na2SeO3), 0.30 mg.
Digestible energy was calculated from data provided by the Feed Database in China.
The animals’ feces were observed every day, and the scour score, according to Hu et al.,8,9 for each pig was recorded according. The fecal scoring scale was from 1 to 5, where 1 represented no diarrhea, hard feces; 2 represented no diarrhea, normal consistency of feces; 3 represented mild diarrhea, soft and partially formed feces; 4 represented moderate diarrhea, semi-liquid feces; and 5 represented severe diarrhea, watery feces.
After the feeding trial (d 8 post-weaning), all the piglets were killed and the gastrointestinal tract quickly removed. Intestinal segments measuring 10 cm in length were excised from the proximal jejunum, immediately placed in Ringer’s solution and mounted in Ussing chambers, as described by Hu et al. 9 The specimens from the mid-jejunum were excised and fixed in 10% formalin. The mucosal samples from the mid-jejunum were harvested by scraping with a glass slide, rapidly frozen in liquid nitrogen and stored at −80℃ for further analysis.
Jejunal morphology and barrier function
The method for making hematoxylin and eosin slices of jejunal tissue, and measurements of villus height and crypt depth, was performed according to the procedures of Hu et al. 8 The transepithelial electrical resistance (TER) and mucosal-to-serosal permeability to 4-ku FITC dextran (FD4; Sigma-Aldrich, St. Louis, MO, USA) were determined in vitro in the Ussing chamber, according to the procedures outlined by Hu et al.4,9 Briefly, the jejunum segments were mounted in the EasyMount Ussing chamber system connected to Acquire and Analyse software (VCC MC6; Physiologic Instruments, San Diego, CA, USA). The TER (Ω·cm2) was recorded at 15-min intervals over a 2-h period and averaged to obtain the TER values for a given pig. Meanwhile, mucosal-to-serosal flux of FD4 (ng/cm2/h) was determined. The FD4 was added on the mucosal side, and the concentration of FD4 in the serosal side was analyzed using a fluorescence microplate reader (FLx800; Bio-Tek Instruments, Winooski, VT, USA).
mRNA expression analysis by RT-PCR
Primer sequences used for RT-PCR.

bp: base pair.
Western blot analysis
The Western blot analysis was performed according to the procedures outlined by Hu et al. 4 The following primary Abs were used: MAPK Abs, rabbit mAb; phospho-p38 (Thr180/Tyr182) rabbit mAb; phospho-ERK 1/2 (Thr202/Tyr204) rabbit mAb; phospho-JNK (Thr183/Tyr185) rabbit mAb; TGF-β1 rabbit mAb (all from Cell Signaling Technology, Danvers, MA, USA). The secondary Ab was HRP-conjugated anti-rabbit antibody (Cell Signaling Technology). Western blot was detected with an enhanced chemiluminescence detection kit (Amersham, Arlington Heights, IL, USA), photographed by a ChemiScope 3400 (Clinx Science Instruments, Shanghai, China) and analyzed using Quantity One software. β-Actin was used as an internal control, and exhibited no difference between the groups. The relative expression of TGF-β1 was expressed as the ratio of TGF-β1 and β-actin. The MAPK signal pathway activation was calculated as the ratios of the phosphorylation levels (p-JNK, p-p38, p-ERK) and the total levels of MAPKs (JNK, p38, ERK 1/2).
Statistical analysis
Data were analyzed using the SAS statistical package (SAS Institute, Cary, NC, USA), 19 with each animal considered an experimental unit. Results are expressed as mean ± SD. The fecal score data were tested using the Mann–Whitney U-test. Differences between the other means were analyzed with an unpaired t-test. Differences were considered significant at P < 0.05.
Results
Growth performance and diarrhea index
Effects of ZnO on growth performance and post-weaning diarrhea of pigs. a

Values are means ± SD (n = 6).
Differences were considered significant at P < 0.05.
Jejunal morphology and barrier function
Effect of ZnO on jejunal morphology and barrier function of weaning piglets. a
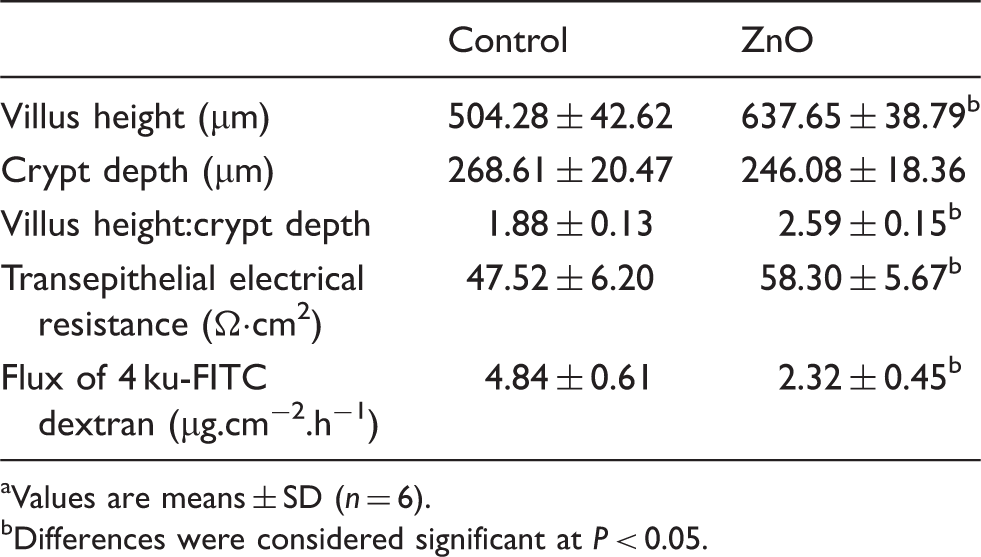
Values are means ± SD (n = 6).
Differences were considered significant at P < 0.05.
MAPK signaling pathways
Figure 1 shows the effects of ZnO on the three MAPK signaling pathways (JNK, p38, ERK). There was no difference in the total protein levels for the MAPKs between the two groups. However, compared with the control, supplemental ZnO significantly decreased (P < 0.05) the ratios of the phosphorylated forms and the total levels of JNK and p38 (p-JNK/JNK and p-p38/p38), while significantly increasing (P < 0.05) the ratio of the phosphorylated forms and the total levels of ERK (p-ERK/ERK).
Effects of ZnO on MAPK signal pathways in the jejunal mucosa of piglets. The three MAPKs are JNK, p38, and ERKs 1 and 2. The bands are representative blots from one of six pigs. The values are calculated as the ratios of their phosphorylation levels (p-JNK, p-p38, p-ERK) and the total levels of MAPK. Data are means ± SD (n = 6). *Differences were considered significant at P < 0.05.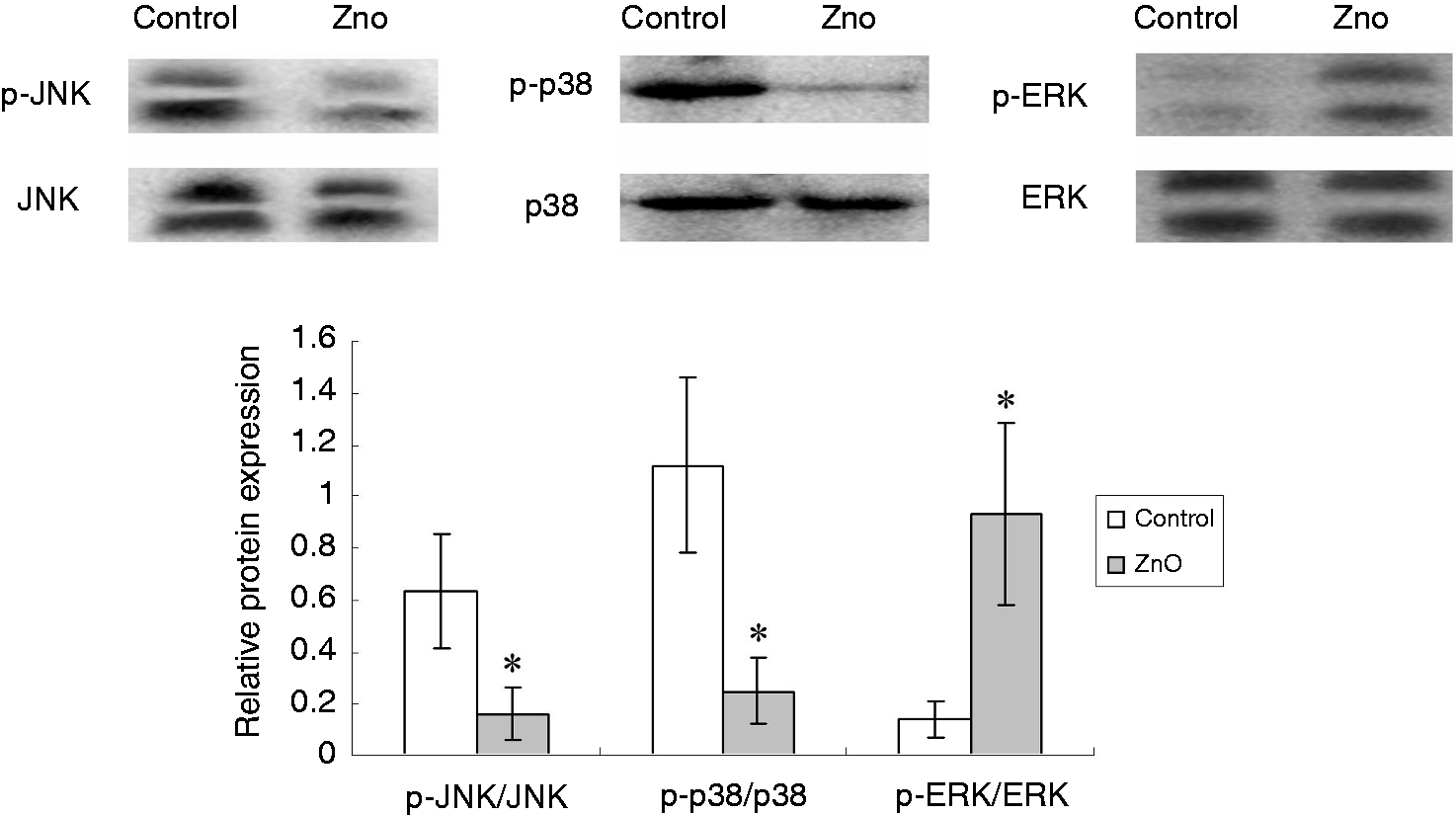
TGF-β1 expression and its canonical SMAD signaling pathway
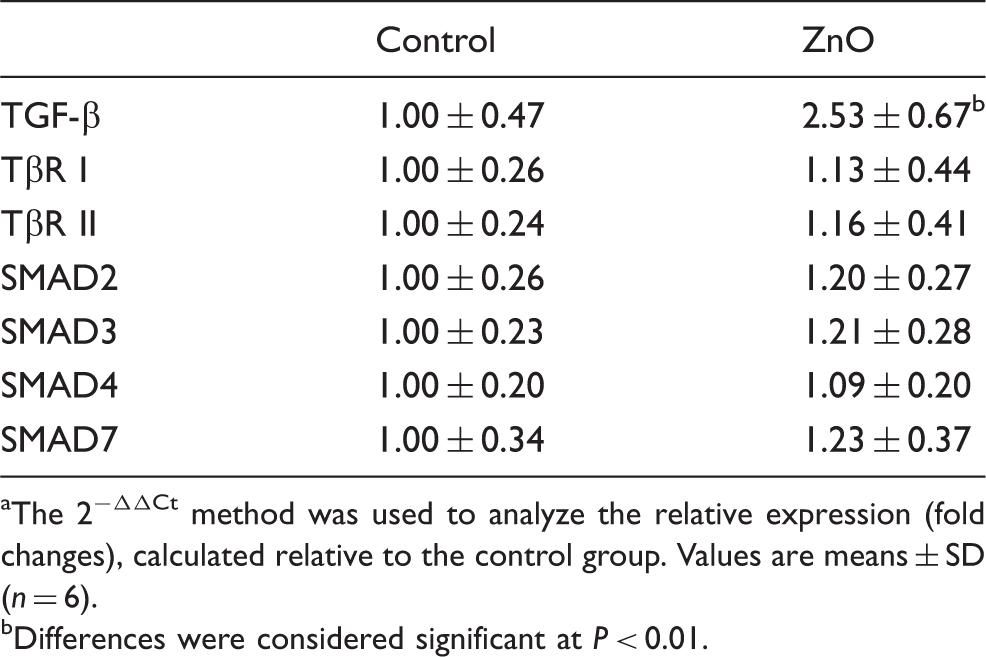
The mRNA level (Table 4) and expression (Figure 2) of TGF-β1 in jejunal mucosa was significantly increased (P < 0.01) in piglets supplemented with ZnO compared with the control pigs. However, the mRNA levels of the TβRs (TβR, TβR II) and the downstream signals (SMAD 2, SMAD 3, SMAD7) showed no difference between the two groups (Table 5).
Effects of ZnO on the expression of TGF-β1 in the jejunal mucosa of weanling piglets. The bands are representative blots from one of six pigs. The relative expression of TGF-β1 was expressed as the ratio of TGF-β1 and β-actin. Data are means ± SD (n = 6). *Differences were considered significant at P < 0.05. Effects of ZnO on the mRNA expression of TGF-β1, TβR and their downstream signals in weanling pigs.
a
The 2−ΔΔCt method was used to analyze the relative expression (fold changes), calculated relative to the control group. Values are means ± SD (n = 6). Differences were considered significant at P < 0.01.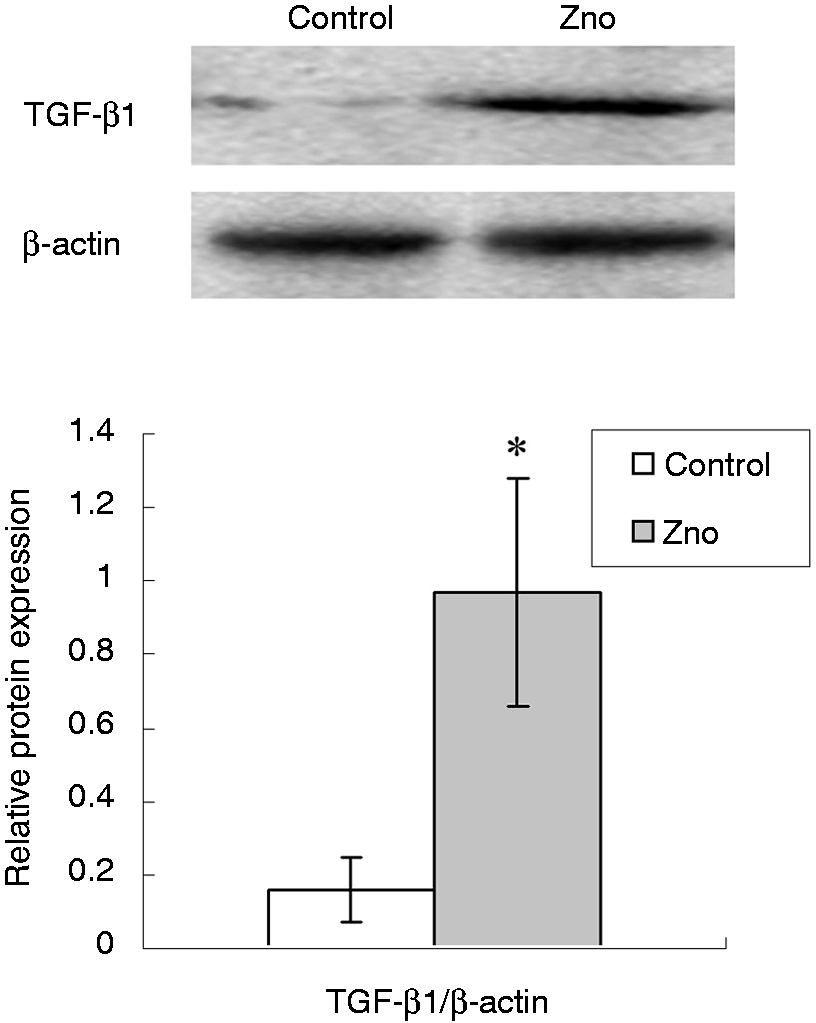
Discussion
Weaning is one of the most stressful events in the life of piglets as they are abruptly forced to experience combined stressors, such as shifting from a milk-based diet to a cereal-based one, removal from the sow and littermates, fighting and establishment of the social hierarchy. 4 Post-weaning diarrhea and growth retardation are frequent occurrences in early-weaned piglets.4,10 The duration and severity of diarrhea can be controlled by feeding pharmacological levels of ZnO (between 2000 and 4000 mg/kg of Zn) to the weanling piglets.8–10 In this study, in line with previous research,8–10 supplementing with ZnO to provide 2200 mg Zn/kg in the weanling pig diet improved growth performance and ameliorated weaning-associated diarrhea.
The gastrointestinal tract is fundamental to the uptake of nutrients, but it also acts as a physical barrier preventing harmful microorganisms and antigens from entry into deeper tissues. The intestinal epithelium barrier is formed from a single layer of epithelial cells and intercellular junctions. The tight junctions, the most apical of the intercellular junctions, work as the rate-limiting step in the paracellular pathway and form a selectively permeable barrier.4,9 The disruption of tight junctions leads to increased intestinal permeability, which contributes to the initiation and development of many diseases, such as Crohn’s disease and ulcerative colitis. Early weaning has been reported to impair the intestinal barrier function of piglets.4,20 As reviewed by Wijtten et al., 21 the intestinal barrier dysfunction induced by weaning is characterized by shortened villus height, increased mucosal permeability, disturbed active and passive absorption, and stimulated pro-inflammatory cytokine production. In the present study, the addition of ZnO to the diet increased villus height and the TER value, and decreased FD4 flux across the jejunal mucosa. These findings demonstrate that feeding high levels of ZnO to weanling piglets ameliorates weaning-associated intestinal injury and improves intestinal barrier function, which is consistent with recent research.9,10 There is abundance of research on the modulation of zinc on intestinal mucosal repair. A high level of ZnO prevented the increase of paracellular permeability induced by enterotoxigenic Escherichia coli in human Caco-2 enterocytes. 6 Rodriguez et al. 22 showed that malnutrition in guinea pigs was associated with a decreased number of tight junctional strands, and pharmacological doses of zinc prevented such an abnormality of tight junction morphology. High-dose zinc alleviated tight junction injury in the intestine of rats with acute colitis caused by dinitro-benzene-sulfonic acid. 7 However, the mechanisms of ZnO on mucosal integrity are still not well elucidated. Some researchers think that ZnO prevents diarrhea owing to its antibacterial activity. In vitro studies show that ZnO inhibits the growth of Staphylococcus aureus and E. coli. 23 In piglets, high doses of ZnO reduce bacterial translocation from the small intestine to the ileal mesenteric lymph node and decrease the viable Clostridium count in the intestine.24,25 On the contrary, Jensen-Waern et al. 26 found that supplementation with 2500 mg/kg in the form of ZnO did not decrease E. coli or Enterococci in feces of weaned piglets. 26 Roselli et al. 6 reported that ZnO protected cells from enterotoxigenic E. coli-induced damage by inhibiting bacterial adhesion and internalization, preventing the disruption of barrier integrity and modulating cytokine expression, without a direct antibacterial effect.
To better elucidate the molecular mechanisms by which ZnO supplementation supports epithelial barrier integrity, we determined, for the first time, whether supplementation with ZnO influenced MAPK signaling pathways in weaned pigs. MAPKs are a family of serine/threonine protein kinases widely conserved among eukaryotes and convert extracellular stimuli into a wide range of cellular responses. The MAPK pathways consist of three major groupings and numerous related proteins that constitute interrelated signal transduction cascades. 11 The three major groupings are the ERK, JNK and p38 cascades. Using a porcine model of intestinal ischemia, Shifflett et al. 11 demonstrated that inhibition of JNK resulted in enhanced recovery of barrier function, whereas inhibition of ERK1/2 and p38 MAPK had a deleterious effect on mucosal recovery from ischemia. A recent study showed that weaning stress activated p38, JNK and ERK 1/2 signaling pathways in the intestine, which may be an important mechanism of weaning-associated enteric disorders of piglets. 4 So far, only a few studies in in vitro cell models have investigated the effects of zinc on MAPK signaling pathways. Liang et al. 14 found that zinc inhibited hydrogen peroxide-induced apoptosis in MC3T3-E1 cells via activation of ERK pathways and inhibition of p38 and JNK pathways. In the present study, we demonstrated, for the first time, that dietary ZnO supplementation decreases the relative proteins levels for phosphorylated p38 and JNK, while increasing phosphorylated ERK 1/2, indicating that ZnO inhibits the JNK and p38 signaling pathways while activating ERK 1/2 signaling in weaned pigs. The JNK and p38 cascades are activated by inflammatory cytokines and by a wide variety of cellular stresses. 27 Early weaning has been reported to up-regulate the expression of pro-inflammatory cytokines in the intestine. 4 A recent study showed that ZnO supplementation down-regulated the expression of pro-inflammatory cytokines in weaned pigs.9,28 The down-regulation of inflammatory cytokines is in line with the beneficial effects of oral IFN-α in piglets at weaning.29,30 Oral IFN-α modulated the inflammatory response to early weaning and increased the daily mean mass gain of pigs. 30 The inhibition of ZnO on p38 and JNK signaling pathways might be caused by the down-regulation of pro-inflammatory cytokine levels. The ERK 1/2 cascade is activated by growth factors, and preferentially regulates cell growth and differentiation. 27 Liang et al. 31 reported that zinc increased osteogenic function in mouse MC3T3-E1 osteoblasts through the stimulation of ERK signaling. 31 Wang et al. 32 reported that supplementation with 3000 mg Zn/kg from ZnO increased the A-Raf-1 expression in the jejunum of weaning piglets. Because Raf is a member of the Ras/Raf/MEK/ERK signaling pathway, the activation of ERK in ZnO-supplemented piglets may be expected. The present study demonstrated, for the first time, that dietary ZnO supplementation activated ERK 1/2 signaling in weaned pigs. In a follow-up study, the specific inhibitors of MAPKs may be used to investigate whether inhibition of MAPKs can affect the beneficial role of ZnO in intestinal barrier of weaned piglets.
TGF-β1 plays an important regulatory role in mucosal immune reactions and intestinal barrier restoration. During injury or disease, TGF-β stimulates epithelial cell migration, increases extracellular matrix and integrin production, promotes intestinal epithelial restitution, and improves intestinal mucosa integrity.33,34 IFN-γ has been reported to be the main permeability increasing cytokine in the gut. 35 TGF-β1 preserves intestinal epithelial barrier function after exposure to IFN-γ. TGF-β plays an important role in the post-weaning adaptation process in the intestine of the pig. Mei and Xu 12 showed that the expression intensity of TGF-β1 at the intestinal villus epithelium decreased significantly 4 d after weaning. The transient decline in the TGF-β1 level of the intestinal villus following weaning contributed to post-weaning intestinal villus atrophy. 12 The research on ZnO regulation of TGF-β1 expression has so far only been reported in vitro. Zinc markedly increased the concentration of TGF-β1 in culture medium secreted by osteoblastic MC3T3-E1 cells. 36 In femoral tissues of newborn rats, and bone tissues with fracture healing, zinc has a stimulatory effect on the production of TGF-β1.37,38 In human Caco-2 enterocytes, Roselli et al. 6 showed that the enterotoxigenic E. coli K88 infection reduced TGF-β1 expression compared with uninfected cells, and ZnO up-regulated TGF-β mRNA in E. coli K88-infected cells. However, owing to post-transcriptional regulatory actions, the increased expression of TGF-β mRNA did not indicate the increased expression of TGF-β protein. 35 Therefore, in the current study, we analyzed the expression of both the mRNA and the protein. We showed, for the first time, that ZnO supplementation increased the intestinal expression of TGF-β1 in weaned pigs. The beneficial role of ZnO in intestinal barrier function might be related to the increase in intestinal TGF-β1 expression.
The canonical TGF-β signaling pathway is mediated by SMAD family proteins. SMAD proteins—the only substrates of TβR kinase—are critical mediators of the TGF-β signaling transducer. SMAD 2 and SMAD3 are receptor-regulated SMADS (R-SMADS). Following stimulation by TGF-β, SMAD 2 and SMAD 3 become phosphorylated. Phosphorylated SMAD 2/3 can complex with SMAD4 (the common-mediator SMAD), and then translocate to the nucleus and regulate gene expression. 13 To determine whether ZnO supplementation affected SMAD signaling pathways in weaned pigs, mucosa scraped from jejunum was analyzed for the expression of TβR (TβR, TβR II) and the downstream signals (SMAD 2, SMAD 3, SMAD 7). The results showed that ZnO supplementation had no effect on the relative mRNA levels for TβR levels (TβR I, TβR II) or the downstream signals (SMAD 2, SMAD 3, SMAD 7). We also analyzed phosphorylated SMAD 2/3, but we did not find a suitable Ab for pig. Using Ab for humans, we failed to detect the SMADs proteins by Western blotting. Therefore, we analyzed the changes in the mRNA level. However, several studies have also shown that the mRNA expression of SMADs can also be affected in some situations. In further studies, using other methods, we will try to detect the phosphorylation or other post-translational modifications of SMADs.
It is well-established that the beneficial role of TGF-β1 in the epithelial barrier is exerted mainly as a result of SMAD-independent signaling pathways. 39 Howe et al. 33 reported that TGF-β1-induced epithelial barrier enhancement was mediated by ERK 1/2 and SMAD 2/3 signaling pathways in human T84 colonic epithelial cells. TGF-β1 is known to activate the ERK 1/2 pathway in multiple cell types. 33 There is some evidence that the activation of the ERK 1/2 signaling is linked to the TGF-β1 induced modulation of tight junction permeability. 40 In cultured rat intestinal epithelial cells (IEC-6), TGF-β1 increases wound closures via ERK activation. 41 TGF-β1 stimulates re-epithelialization in human corneal epithelial cells through the Ras/MEK/ERK signaling pathway. 42 In mesenchymal cells, TGF-β1 induced ERK activation through the TGF-β/phosphotidylinositol 3-kinase (PI3K)/p21-activated kinase2 (Pak2)/Raf/MEK/ ERK pathway. 43 TGF-β1 also activates ERK signaling through direct phosphorylation of ShcA. 44 ERK was reported to regulate the integrity of tight junction and the barrier function of the epithelium. 45 In Caco-2 cells, phospho-ERK directly interacts with occludin and prevents hydrogen peroxide-induced disruption of tight junctions. 45 The activation of ERK 1/2 leads to increased expression of claudin-4 in MDCK cells. 46 In the present study, supplementation with ZnO increased the intestinal expression of TGF-β1 and activated the ERK 1/2 signaling pathway. The results indicated that the activation of ERK 1/2 signaling might be linked to the improvement of the intestinal barrier induced by supplementation with ZnO.
In summary, dietary addition of ZnO improved intestinal barrier function, activated the ERK 1/2 signaling pathway, while inhibiting the JNK and p38 signaling pathways, and increased intestinal expression of TGF-β1 in weaned piglets.
Footnotes
Funding
This research was jointly supported by National Natural Science Foundation of China (TGF-β1-mediated Smads and MAPKs signaling pathways on restoration of intestinal epithelial barrier in weanling piglets), the Special Fund for Agro-scientific Research in the Public Interest (201403047), Zhejiang Provincial Qianjiang Talent Project (2013R10036), Fundamental Research Funds for the Central Universities (2014FZA6020).
Acknowledgements
Special thanks to Professor J.R. Peng for his constructive suggestions.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
